Developmental Signals - Fibroblast Growth Factor: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 28: | Line 28: | ||
* '''Fgf8 morphogen gradient forms by a source-sink mechanism with freely diffusing molecules'''<ref><pubmed>19741606</pubmed></ref> "It is widely accepted that tissue differentiation and morphogenesis in multicellular organisms are regulated by tightly controlled concentration gradients of morphogens. How exactly these gradients are formed, however, remains unclear. Here we show that Fgf8 morphogen gradients in living zebrafish embryos are established and maintained by two essential factors: fast, free diffusion of single molecules away from the source through extracellular space, and a sink function of the receiving cells, regulated by receptor-mediated endocytosis. Evidence is provided by directly examining single molecules of Fgf8 in living tissue by fluorescence correlation spectroscopy, quantifying their local mobility and concentration with high precision. By changing the degree of uptake of Fgf8 into its target cells, we are able to alter the shape of the Fgf8 gradient. Our results demonstrate that a freely diffusing morphogen can set up concentration gradients in a complex multicellular tissue by a simple source-sink mechanism." | * '''Fgf8 morphogen gradient forms by a source-sink mechanism with freely diffusing molecules'''<ref><pubmed>19741606</pubmed></ref> "It is widely accepted that tissue differentiation and morphogenesis in multicellular organisms are regulated by tightly controlled concentration gradients of morphogens. How exactly these gradients are formed, however, remains unclear. Here we show that Fgf8 morphogen gradients in living zebrafish embryos are established and maintained by two essential factors: fast, free diffusion of single molecules away from the source through extracellular space, and a sink function of the receiving cells, regulated by receptor-mediated endocytosis. Evidence is provided by directly examining single molecules of Fgf8 in living tissue by fluorescence correlation spectroscopy, quantifying their local mobility and concentration with high precision. By changing the degree of uptake of Fgf8 into its target cells, we are able to alter the shape of the Fgf8 gradient. Our results demonstrate that a freely diffusing morphogen can set up concentration gradients in a complex multicellular tissue by a simple source-sink mechanism." | ||
|} | |} | ||
{| class="wikitable collapsible collapsed" | {| class="wikitable mw-collapsible mw-collapsed" | ||
! More recent papers | ! More recent papers | ||
|- | |- | ||
Revision as of 12:19, 31 August 2014
| Embryology - 14 May 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
Fibroblast Growth Factors (FGF) were originally identified by their ability to stimulate fibroblast cell proliferation but have a role in a growing number of different tissues development and differentiation and continue to have a role in the adult.
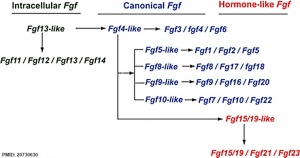
The first two identified factors were originally given the nomenclature of acidic or basic. We now know there to be at least 22 different human FGFs (Fgf1–Fgf23). These protein growth factors are bound by 4 different cell membrane receptors (FGFR1-4). FGFRs belong to the tyrosine kinase receptor family.
The mammalian Fgf family can be divided into the intracellular Fgf11/12/13/14 subfamily (iFGFs), the endocrine hormone-like Fgf15/21/23 subfamily (hFGFs), and the paracrine canonical Fgf subfamilies, including Fgf1/2/5, Fgf3/4/6, Fgf7/10/22, Fgf8/17/18, and Fgf9/16/20.
| Factor Links: AMH | hCG | BMP | sonic hedgehog | bHLH | HOX | FGF | FOX | Hippo | LIM | Nanog | NGF | Nodal | Notch | PAX | retinoic acid | SIX | Slit2/Robo1 | SOX | TBX | TGF-beta | VEGF | WNT | Category:Molecular |
Protein Properties
Human FGF
- ~150–300 amino acids
- have a conserved ~120-residue core with ~30–60% identity
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Fibroblast Growth Factor <pubmed limit=5>Fibroblast Growth Factor</pubmed> |
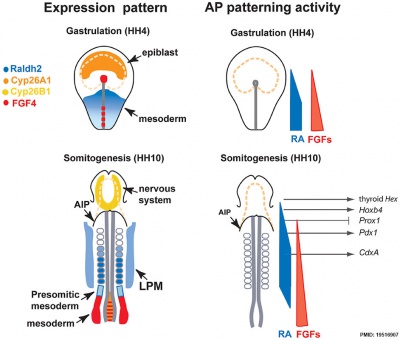
Endoderm
Chicken antero-posterior endoderm patterning[6]
- Links: Endoderm | Chicken Development
Abnormalities
- FGFR1 mutation has been associated with the relatively milder form of Pfeiffer syndrome type 1.
- FGFR2 and FGFR3 have been associated with the Apert, Crouzon and Pfeiffer syndromes.
References
- ↑ Itoh N. Hormone-like (endocrine) Fgfs: their evolutionary history and roles in development, metabolism, and disease. Cell Tissue Res. 2010 Oct;342(1):1-11. PMID20730630
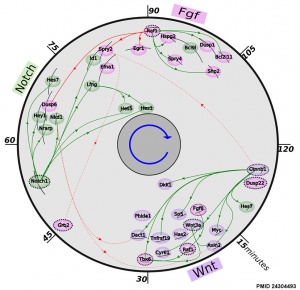
- ↑ 2.0 2.1 <pubmed>24304493</pubmed>| BMC Dev Biol.
- ↑ <pubmed>22988910</pubmed>
- ↑ <pubmed>21074523</pubmed>
- ↑ <pubmed>19741606</pubmed>
- ↑ 19516907</pubmed>| PLoS One.
Reviews
Articles
<pubmed>20582225</pubmed>
Search Pubmed
Search Bookshelf Fibroblast Growth Factor
Search Pubmed Now: Fibroblast Growth Factor