Gastrulation: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 40: | Line 40: | ||
* '''A single-cell molecular map of {{mouse}} {{gastrulation}} and early organogenesis'''{{#pmid:30787436|PMID30787436}} "Across the animal kingdom, gastrulation represents a key developmental event during which embryonic pluripotent cells diversify into lineage-specific precursors that will generate the adult organism. Here we report the transcriptional profiles of 116,312 single cells from mouse embryos collected at nine sequential time points ranging from {{ME6.5}} to {{ME8.5}} days post-fertilization. We construct a molecular map of cellular differentiation from pluripotency towards all major embryonic lineages, and explore the complex events involved in the convergence of visceral and primitive streak-derived {{endoderm}}. Furthermore, we use single-cell profiling to show that Tal1-/- chimeric embryos display defects in early {{mesoderm}} diversification, and we thus demonstrate how combining temporal and transcriptional information can illuminate gene function." ([https://www.nature.com/articles/s41586-019-1825-8?WT.ec_id=NATURE-201912 See Nature 11 Dec]) | * '''A single-cell molecular map of {{mouse}} {{gastrulation}} and early organogenesis'''{{#pmid:30787436|PMID30787436}} "Across the animal kingdom, gastrulation represents a key developmental event during which embryonic pluripotent cells diversify into lineage-specific precursors that will generate the adult organism. Here we report the transcriptional profiles of 116,312 single cells from mouse embryos collected at nine sequential time points ranging from {{ME6.5}} to {{ME8.5}} days post-fertilization. We construct a molecular map of cellular differentiation from pluripotency towards all major embryonic lineages, and explore the complex events involved in the convergence of visceral and primitive streak-derived {{endoderm}}. Furthermore, we use single-cell profiling to show that Tal1-/- chimeric embryos display defects in early {{mesoderm}} diversification, and we thus demonstrate how combining temporal and transcriptional information can illuminate gene function." ([https://www.nature.com/articles/s41586-019-1825-8?WT.ec_id=NATURE-201912 See Nature 11 Dec]) | ||
|} | |} | ||
{| class="wikitable mw-collapsible mw-collapsed" | {| class="wikitable mw-collapsible mw-collapsed" | ||
| Line 58: | Line 53: | ||
|- | |- | ||
| {{Older papers}} | | {{Older papers}} | ||
* '''Self-organization of a human organizer by combined Wnt and Nodal signalling'''{{#pmid:29795348|PMID29795348}} "In amniotes, the development of the primitive streak and its accompanying 'organizer' define the first stages of gastrulation. Although these structures have been characterized in detail in model organisms, the human primitive streak and organizer remain a mystery. When stimulated with {{BMP}}4, micropatterned colonies of human embryonic stem cells self-organize to generate early embryonic germ layers 1 . Here we show that, in the same type of colonies, Wnt signalling is sufficient to induce a primitive streak, and stimulation with Wnt and Activin is sufficient to induce an organizer, as characterized by embryo-like sharp boundary formation, markers of {{epithelial mesenchymal transition}} and expression of the organizer-specific transcription factor GSC. Moreover, when grafted into chick embryos, human stem cell colonies treated with Wnt and Activin induce and contribute autonomously to a secondary axis while inducing a neural fate in the host." | |||
* '''Molecular regulation of Nodal signaling during mesendoderm formation'''{{#pmid:29206913|PMID29206913}} "One of the most important events during vertebrate embryogenesis is the formation or specification of the three germ layers, endoderm, mesoderm, and ectoderm. After a series of rapid cleavages, embryos form the mesendoderm and ectoderm during late blastulation and early gastrulation. The mesendoderm then further differentiates into the mesoderm and endoderm. Nodal, a member of the transforming growth factor β ({{TGF-beta}}, TGF-β) superfamily, plays a pivotal role in mesendoderm formation by regulating the expression of a number of critical transcription factors, including Mix-like, GATA, Sox, and Fox. Because the Nodal signal transduction pathway is well-characterized, increasing effort has been made to delineate the spatiotemporal modulation of Nodal signaling during embryonic development. In this review, we summarize the recent progress delineating molecular regulation of Nodal signal intensity and duration during mesendoderm formation." {{TGF-beta}} | |||
* '''{{BMP}} and {{FGF}} signaling interact to pattern {{mesoderm}} by controlling basic helix-loop-helix transcription factor activity'''{{#pmid:29877796|PMID29877796}} "The {{mesoderm}}al germ layer is patterned into mediolateral subtypes by signaling factors including {{BMP}} and {{FGF}}. How these pathways are integrated to induce specific mediolateral cell fates is not well understood. We used mesoderm derived from post-gastrulation neuromesodermal progenitors (NMPs), which undergo a binary mediolateral patterning decision, as a simplified model to understand how FGF acts together with BMP to impart mediolateral fate. Using zebrafish and mouse NMPs, we identify an evolutionarily conserved mechanism of BMP and FGF mediated mediolateral mesodermal patterning that occurs through modulation of basic helix-loop-helix (bHLH) transcription factor activity. BMP imparts lateral fate through induction of Id helix loop helix (HLH) proteins, which antagonize bHLH transcription factors, induced by FGF signaling, that specify medial fate. We extend our analysis of zebrafish development to show that bHLH activity is responsible for the mediolateral patterning of the entire mesodermal germ layer." | |||
* '''Review - Molecular specification of germ layers in vertebrate embryos'''{{#pmid:26667903|PMID26667903}} "In order to generate the tissues and organs of a multicellular organism, different cell types have to be generated during embryonic development. The first step in this process of cellular diversification is the formation of the three germ layers: {{ectoderm}}, endoderm and mesoderm. The {{ectoderm}} gives rise to the nervous system, epidermis and various {{neural crest}}-derived tissues, the endoderm goes on to form the gastrointestinal, respiratory and urinary systems as well as many endocrine glands, and the mesoderm will form the notochord, axial skeleton, cartilage, connective tissue, trunk muscles, kidneys and blood. Classic experiments in amphibian embryos revealed the tissue interactions involved in germ layer formation and provided the groundwork for the identification of secreted and intracellular factors involved in this process." | * '''Review - Molecular specification of germ layers in vertebrate embryos'''{{#pmid:26667903|PMID26667903}} "In order to generate the tissues and organs of a multicellular organism, different cell types have to be generated during embryonic development. The first step in this process of cellular diversification is the formation of the three germ layers: {{ectoderm}}, endoderm and mesoderm. The {{ectoderm}} gives rise to the nervous system, epidermis and various {{neural crest}}-derived tissues, the endoderm goes on to form the gastrointestinal, respiratory and urinary systems as well as many endocrine glands, and the mesoderm will form the notochord, axial skeleton, cartilage, connective tissue, trunk muscles, kidneys and blood. Classic experiments in amphibian embryos revealed the tissue interactions involved in germ layer formation and provided the groundwork for the identification of secreted and intracellular factors involved in this process." | ||
| Line 120: | Line 122: | ||
:'''Links:''' [[Carnegie stage 8]] | :'''Links:''' [[Carnegie stage 8]] | ||
==Germ Layer Markers== | ==Molecular== | ||
===Germ Layer Markers=== | |||
Stem cell researchers have used the following markers to identify early differentiation of cells in the three germ layer in embryoid bodies.{{#pmid:20369364|PMID20369364}} | Stem cell researchers have used the following markers to identify early differentiation of cells in the three germ layer in embryoid bodies.{{#pmid:20369364|PMID20369364}} | ||
| Line 128: | Line 131: | ||
* {{endoderm}} - [[Alpha-Fetoprotein]] | * {{endoderm}} - [[Alpha-Fetoprotein]] | ||
===Signaling - BMP and FGF=== | |||
* '''{{BMP}} and {{FGF}} signaling interact to pattern {{mesoderm}} by controlling basic helix-loop-helix transcription factor activity'''{{#pmid:29877796|PMID29877796}} "The {{mesoderm}}al germ layer is patterned into mediolateral subtypes by signaling factors including {{BMP}} and {{FGF}}. How these pathways are integrated to induce specific mediolateral cell fates is not well understood. We used mesoderm derived from post-gastrulation neuromesodermal progenitors (NMPs), which undergo a binary mediolateral patterning decision, as a simplified model to understand how FGF acts together with BMP to impart mediolateral fate. Using zebrafish and mouse NMPs, we identify an evolutionarily conserved mechanism of BMP and FGF mediated mediolateral mesodermal patterning that occurs through modulation of basic helix-loop-helix (bHLH) transcription factor activity. BMP imparts lateral fate through induction of Id helix loop helix (HLH) proteins, which antagonize bHLH transcription factors, induced by FGF signaling, that specify medial fate. We extend our analysis of zebrafish development to show that bHLH activity is responsible for the mediolateral patterning of the entire mesodermal germ layer." | |||
:'''Links:''' [[Stem_Cells_-_Induced|Induced Stem Cells]] | [[Alpha-Fetoprotein]] | :'''Links:''' [[Stem_Cells_-_Induced|Induced Stem Cells]] | [[Alpha-Fetoprotein]] | ||
Latest revision as of 13:55, 1 December 2021
| Embryology - 14 Jun 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
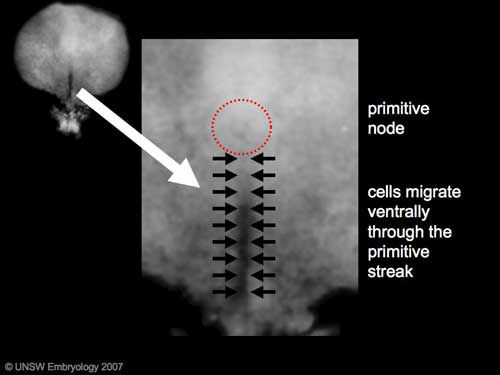
The term gastrulation means the formation of gut (Greek, gastrula = belly), but has now a more broad sense to to describe the formation of the trilaminar embryo. The epiblast layer, consisting of totipotential cells, derives all 3 embryo layers: ectoderm, mesoderm and endoderm. The primitive streak is the visible feature which represents the site of cell migration to form the additional layers.
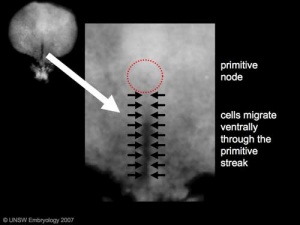
- primitive node - region in the middle of the early embryonic disc epiblast from which the primitive streak extends caudally (tail)
- nodal cilia establish the embryo left/right axis
- axial process extends from the nodal epiblast
- primitive streak - region of cell migration from the epiblast layer forming sequentially the two germ cell layers (endoderm and mesoderm)
| Historic Embryology |
|
Hans Spemann (1869 - 1941) identified this region in amphibia, also called the "Spemann's organiser". The same region in birds it is known as "Hensen's node" named for Victor Hensen (1835 – 1924) and is also known generally as the primitive node or knot. In humans, it is proposed that similar mechanisms regulate gastrulation to those found in other vertebrates. Currently, the molecular and physical mechanisms that regulate patterning and migration during this key event are being investigated in several different animal models. |
Signaling Pathway: BMP4 -> Wnt -> Activin–Nodal
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Inhibitors - Wnt inhibitor IWP2; Nodal inhibitor (SB431542 drug TGF-beta/Smad inhibitor)
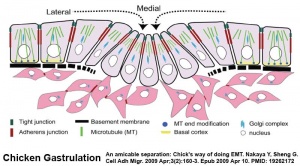
Epithelial to Mesenchymal Transition
Epithelial cells (organised cellular layer) which loose their organisation and migrate/proliferate as a mesenchymal cells (disorganised cellular layers) are said to have undergone an Epithelial Mesenchymal Transition (EMT). Mesenchymal cells have an embryonic connective tissue-like cellular arrangement, that have undergone this process may at a later time and under specific signaling conditions undergo the opposite process, mesenchyme to epithelia. In development, this process can be repeated several times during tissue differentiation.
This process occurs at the primitive streak where epiblast cells undergo an epithelial to mesenchymal transition in order to delaminate and migrate.
| Links: gastrulation | Lecture - Week 3 | Week 3 | Carnegie stage 7 | Carnegie 8 | endoderm | mesoderm | ectoderm | Epithelial Mesenchymal Transition | notochord | ||
|
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Gastrulation |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Gastrulation Movies
|
|
|
|
| |||||||||||||||
| mesoderm spread | mouse cilia | quail extracellular matrix | Planar cell movement | Mesoderm migration |
Human Gastrulation
The site of gastrulation, the primitive streak is visible during week 3 on the epiblast dorsal surface of the embryonic disc.
Stage 7
- Links: Carnegie stage 7
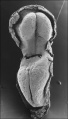
Stage 8
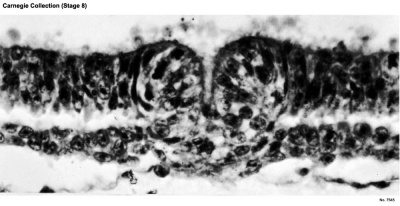
Primitive pit |
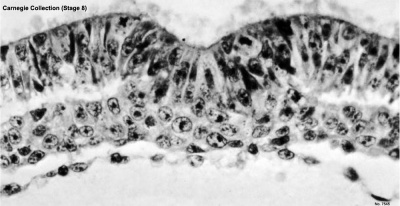
Primitive groove and primitive streak |
- Links: Carnegie stage 8
Molecular
Germ Layer Markers
Stem cell researchers have used the following markers to identify early differentiation of cells in the three germ layer in embryoid bodies.[12]
- ectoderm - SOX1
- mesoderm - Brachyury T
- endoderm - Alpha-Fetoprotein
Signaling - BMP and FGF
- BMP and FGF signaling interact to pattern mesoderm by controlling basic helix-loop-helix transcription factor activity[6] "The mesodermal germ layer is patterned into mediolateral subtypes by signaling factors including BMP and FGF. How these pathways are integrated to induce specific mediolateral cell fates is not well understood. We used mesoderm derived from post-gastrulation neuromesodermal progenitors (NMPs), which undergo a binary mediolateral patterning decision, as a simplified model to understand how FGF acts together with BMP to impart mediolateral fate. Using zebrafish and mouse NMPs, we identify an evolutionarily conserved mechanism of BMP and FGF mediated mediolateral mesodermal patterning that occurs through modulation of basic helix-loop-helix (bHLH) transcription factor activity. BMP imparts lateral fate through induction of Id helix loop helix (HLH) proteins, which antagonize bHLH transcription factors, induced by FGF signaling, that specify medial fate. We extend our analysis of zebrafish development to show that bHLH activity is responsible for the mediolateral patterning of the entire mesodermal germ layer."
- Links: Induced Stem Cells | Alpha-Fetoprotein
Animal Models
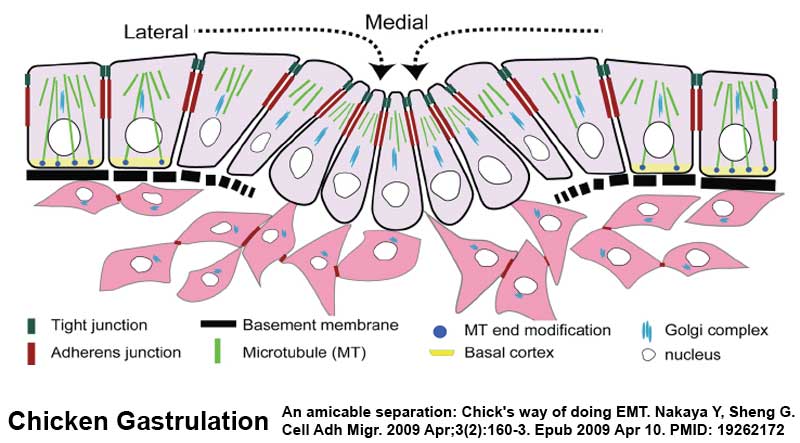
Chicken Gastrulation
- Links: chicken
Frog Gastrulation
|
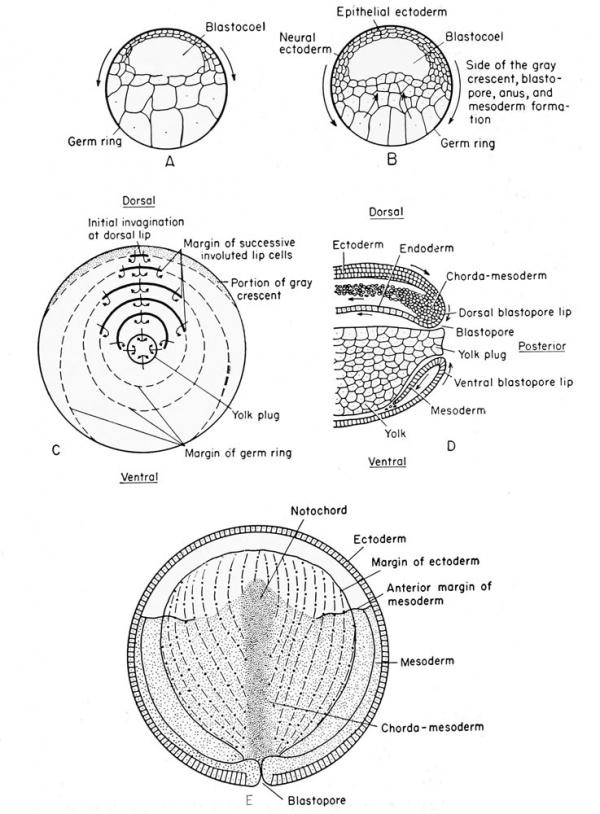
Frog Gastrulation[13]
A The blastula stage, prior to any gastrulation movement. B Movement of the blastula cells preliminary to gastrulation.
D Lateral view of sagittal section during late gastrulation showing the origin of the mesial notochord, and the lateral mesoderm from the proliferated chorda-mesoderm cells at the dorsal lip.
|
- Links: frog
Zebrafish Gastrulation
Kupffer's vesicle (ciliated organ of asymmetry, primitive node) a transient epithelial fluid-filled sac located midventrally posterior to the yolk cell or its extension. The vesicle has been described as equivalent to the primitive node for establishing embryo left-right (L-R) axis.[14]
- Links: zebrafish
Drosophila Gastrulation
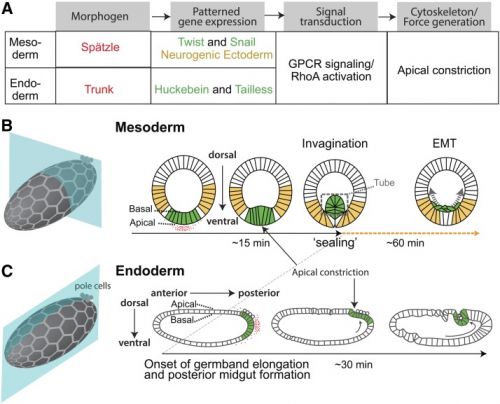
| Drosophila gastrulation[2] | |
|
A Flow chart showing the regulation of cell shape changes that accompany Drosophila gastrulation. Coloured text matches the colours in (B and C).
|
- Links: Drosophila
Additional Images
References
- ↑ Tyser RCV, Mahammadov E, Nakanoh S, Vallier L, Scialdone A & Srinivas S. (2021). Single-cell transcriptomic characterization of a gastrulating human embryo. Nature , , . PMID: 34789876 DOI.
- ↑ 2.0 2.1 Martin AC. (2020). The Physical Mechanisms of Drosophila Gastrulation: Mesoderm and Endoderm Invagination. Genetics , 214, 543-560. PMID: 32132154 DOI.
- ↑ Pijuan-Sala B, Griffiths JA, Guibentif C, Hiscock TW, Jawaid W, Calero-Nieto FJ, Mulas C, Ibarra-Soria X, Tyser RCV, Ho DLL, Reik W, Srinivas S, Simons BD, Nichols J, Marioni JC & Göttgens B. (2019). A single-cell molecular map of mouse gastrulation and early organogenesis. Nature , 566, 490-495. PMID: 30787436 DOI.
- ↑ Martyn I, Kanno TY, Ruzo A, Siggia ED & Brivanlou AH. (2018). Self-organization of a human organizer by combined Wnt and Nodal signalling. Nature , 558, 132-135. PMID: 29795348 DOI.
- ↑ Wei S & Wang Q. (2018). Molecular regulation of Nodal signaling during mesendoderm formation. Acta Biochim. Biophys. Sin. (Shanghai) , 50, 74-81. PMID: 29206913 DOI.
- ↑ 6.0 6.1 Row RH, Pegg A, Kinney B, Farr GH, Maves L, Lowell S, Wilson V & Martin BL. (2018). BMP and FGF signaling interact to pattern mesoderm by controlling basic helix-loop-helix transcription factor activity. Elife , 7, . PMID: 29877796 DOI.
- ↑ Kiecker C, Bates T & Bell E. (2016). Molecular specification of germ layers in vertebrate embryos. Cell. Mol. Life Sci. , 73, 923-47. PMID: 26667903 DOI.
- ↑ Stankova V, Tsikolia N & Viebahn C. (2015). Rho kinase activity controls directional cell movements during primitive streak formation in the rabbit embryo. Development , 142, 92-8. PMID: 25516971 DOI.
- ↑ Poh YC, Chen J, Hong Y, Yi H, Zhang S, Chen J, Wu DC, Wang L, Jia Q, Singh R, Yao W, Tan Y, Tajik A, Tanaka TS & Wang N. (2014). Generation of organized germ layers from a single mouse embryonic stem cell. Nat Commun , 5, 4000. PMID: 24873804 DOI.
- ↑ Xiao Z, Patrakka J, Nukui M, Chi L, Niu D, Betsholtz C, Pikkarainen T, Pikkarainan T, Vainio S & Tryggvason K. (2011). Deficiency in Crumbs homolog 2 (Crb2) affects gastrulation and results in embryonic lethality in mice. Dev. Dyn. , 240, 2646-56. PMID: 22072575 DOI.
- ↑ Seebald JL & Szeto DP. (2011). Zebrafish eve1 regulates the lateral and ventral fates of mesodermal progenitor cells at the onset of gastrulation. Dev. Biol. , 349, 78-89. PMID: 20950598 DOI.
- ↑ Pekkanen-Mattila M, Pelto-Huikko M, Kujala V, Suuronen R, Skottman H, Aalto-Setälä K & Kerkelä E. (2010). Spatial and temporal expression pattern of germ layer markers during human embryonic stem cell differentiation in embryoid bodies. Histochem. Cell Biol. , 133, 595-606. PMID: 20369364 DOI.
- ↑ Rugh R. Book - The Frog Its Reproduction and Development. (1951) The Blakiston Company.
- ↑ Gao W, Xu L, Guan R, Liu X, Han Y, Wu Q, Xiao Y, Qi F, Zhu Z, Lin S & Zhang B. (2011). Wdr18 is required for Kupffer's vesicle formation and regulation of body asymmetry in zebrafish. PLoS ONE , 6, e23386. PMID: 21876750 DOI.
Reviews
Tseng WC, Munisha M, Gutierrez JB & Dougan ST. (2017). Establishment of the Vertebrate Germ Layers. Adv. Exp. Med. Biol. , 953, 307-381. PMID: 27975275 DOI.
Kiecker C, Bates T & Bell E. (2016). Molecular specification of germ layers in vertebrate embryos. Cell. Mol. Life Sci. , 73, 923-47. PMID: 26667903 DOI.
Nowotschin S & Hadjantonakis AK. (2010). Cellular dynamics in the early mouse embryo: from axis formation to gastrulation. Curr. Opin. Genet. Dev. , 20, 420-7. PMID: 20566281 DOI.
Chenoweth JG, McKay RD & Tesar PJ. (2010). Epiblast stem cells contribute new insight into pluripotency and gastrulation. Dev. Growth Differ. , 52, 293-301. PMID: 20298258 DOI.
Thiery JP, Acloque H, Huang RY & Nieto MA. (2009). Epithelial-mesenchymal transitions in development and disease. Cell , 139, 871-90. PMID: 19945376 DOI.
Roszko I, Sawada A & Solnica-Krezel L. (2009). Regulation of convergence and extension movements during vertebrate gastrulation by the Wnt/PCP pathway. Semin. Cell Dev. Biol. , 20, 986-97. PMID: 19761865 DOI.
Rohrschneider MR & Nance J. (2009). Polarity and cell fate specification in the control of Caenorhabditis elegans gastrulation. Dev. Dyn. , 238, 789-96. PMID: 19253398 DOI.
Heisenberg CP & Solnica-Krezel L. (2008). Back and forth between cell fate specification and movement during vertebrate gastrulation. Curr. Opin. Genet. Dev. , 18, 311-6. PMID: 18721878 DOI.
Watters C. (2005). Video views and reviews: gastrulation and the fashioning of animal embryos. Cell Biol Educ , 4, 273-8. PMID: 16344860 DOI.
Viebahn C. (2001). Hensen's node. Genesis , 29, 96-103. PMID: 11170350
Leptin M. (1999). Gastrulation in Drosophila: the logic and the cellular mechanisms. EMBO J. , 18, 3187-92. PMID: 10369659 DOI.
Articles
Kim YY, Moon JS, Kwon MC, Shin J, Im SK, Kim HA, Han JK & Kong YY. (2014). Meteorin regulates mesendoderm development by enhancing nodal expression. PLoS ONE , 9, e88811. PMID: 24558432 DOI.
Chuai M, Hughes D & Weijer CJ. (2012). Collective epithelial and mesenchymal cell migration during gastrulation. Curr. Genomics , 13, 267-77. PMID: 23204916 DOI.
Halacheva V, Fuchs M, Dönitz J, Reupke T, Püschel B & Viebahn C. (2011). Planar cell movements and oriented cell division during early primitive streak formation in the mammalian embryo. Dev. Dyn. , 240, 1905-16. PMID: 21761476 DOI.
Vasiev B, Balter A, Chaplain M, Glazier JA & Weijer CJ. (2010). Modeling gastrulation in the chick embryo: formation of the primitive streak. PLoS ONE , 5, e10571. PMID: 20485500 DOI.
Books
Search Pubmed
Search Pubmed Now: gastrulation
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- Gastrulation: From Cells to Embryo Supplemental Data and Figures
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, June 14) Embryology Gastrulation. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Gastrulation
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G