Developmental Signals - Anti-Mullerian Hormone: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| (32 intermediate revisions by the same user not shown) | |||
| Line 1: | Line 1: | ||
{{Header}} | {{Header}} | ||
==Introduction== | ==Introduction== | ||
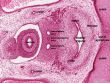
[[File:Stage 22 image 302.jpg|thumb|Male embryo showing the degenerating paramesonephric duct (Mullerian duct)]] | |||
{{Anti-Mullerian Hormone}} ({{AMH}}; Anti-Müllerian Hormone; Mullerian Inhibiting Substance, MIS; Mullerian Inhibiting Factor, MIF) is a secreted glycoprotein factor (transforming growth factor-beta, TGF-beta superfamily) that regulates gonadal and genital tract development. In the male embryo, the Sertoli cell secrete AMH and inhibit paramesonephric (Mullerian) duct development. In postnatal males, AMH increases during the first month, reaching peak level at 6 months of age, and then slowly declines during childhood falling to low levels in puberty. | |||
In reproductive age women, AMH is produced in the ovary by the {{granulosa cell}}s surrounding preantral and small antral follicles and serum levels may reflect the remaining follicle cohort and decrease with age. | |||
<br> | |||
{{Anti-Mullerian Hormone Vignette}} | |||
:'''Links:''' | :'''Links:''' {{AMH}} | {{Male}} | {{uterus}} | {{ovary}} | {{testis}} | {{endocrine}} | ||
<br> | |||
{{Factor Links}} | {{Factor Links}} | ||
| Line 18: | Line 21: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* ''' | * '''Review - AMH and AMHR2 mutations: A spectrum of reproductive phenotypes across vertebrate species'''{{#pmid:31301298|PMID31301298}} "Anti-Müllerian hormone (AMH) is a member of the Transforming Growth Factor-β family of secreted signaling proteins. AMH is expressed in Sertoli cells of the fetal and adult testes and granulosa cells of the postnatal ovary. AMH is required for the regression of the Müllerian ducts in mammalian fetuses during male differentiation. AMH signals through its Type II receptor, AMHR2. AMHR2 is expressed in mesenchyme adjacent to the Müllerian ducts, and in Sertoli, Leydig, and granulosa cells. Although AMH and AMHR2 genes have been identified in numerous vertebrate species, spontaneous or engineered mutations or variants have been found or created in only a few mammals and teleost fishes. AMH or AMHR2 mutations in mammals lead to the development of Persistent Müllerian Duct Syndrome (PMDS), a recessive condition in which affected males are fully virilized but retain Müllerian duct-derived tissues, including a uterus and oviducts, and in human and dog, undescended testes. Amh mutant female mice had accelerated ovarian primordial follicle recruitment, suggesting a role for AMH in regulating germ cells. amh and amhr2 mutations have also been experimentally generated in various teleost fishes. Depending on the fish species, loss of AMH signaling results in infertility, germ cell tumors, or male-to-female sex reversal. Here we compare the spectrum of phenotypes caused by AMH and AMHR2 mutations in a variety of vertebrate species. There are both common and unique phenotypes between species, highlighting the range of biological processes regulated by AMH signaling." | ||
* '''Anti-Müllerian hormone levels in recurrent embryonic miscarriage patients are frequently abnormal, and may affect pregnancy outcomes'''{{#pmid:30917731|PMID30917731}} "This prospective cohort study measured anti-Müllerian hormone (AMH) levels in recurrent miscarriage (RM) patients, compared them to a normal population, and assessed the pregnancy outcomes. The RM patients demonstrated AMH levels that were significantly lower than the normal population, both in women aged ≤35 years, and those aged >35 years. AMH percentiles were found to be significantly lower in the study group of RM patients ≤35 years (p< .004) in the 5th and 50th percentiles, and in all percentiles in women >35 years (p< .03), were compared to women from a normal population. Serum AMH levels may reflect quality, and quantity of the remaining {{oocyte}}s in these patients, and RM patients may have a low ovarian reserve, and a potentially poor oocyte quality, as shown by low circulating AMH. The evaluation of AMH levels in a RM work up may allow realistic counselling and possible {{ART}} referral in RM patients. Impact statement What is already known on this subject? There is some evidence to show that low AMH levels are associated with recurrent miscarriages and this is thought to be due to a decreased oocyte quality. The AMH levels are lower in the patients with endometriosis, and are often significantly higher in the patients with polycystic ovarian syndrome. Both conditions are independently associated with miscarriages. What the results of this study add? Anti-Müllerian hormone (AMH) levels were found to be significantly lower in recurrent miscarriage patients, compared to a normal population. This may be another factor contributing to miscarriages. The spontaneous pregnancy rates in the miscarriage group significantly improved with increasing AMH levels. This may confirm that patients with low AMH levels have poorer quality oocytes, and thus may be considered 'sub-fertile'. It was also found that the utilisation of assisted reproductive technologies (ART) to achieve a pregnancy was significantly reduced in the groups with a higher serum AMH. What the implications are of these findings for clinical practice and/or further research? Serum AMH levels should be offered to all patients as part of a recurrent miscarriage work up. Detecting the low AMH levels and counselling the patients on these findings may allow them the option of accessing ART. ART may have the ability to expedite conception rates, and with pre-implantation genetic analyses, could possibly select the embryos with the greatest chance of survival. Further research is needed to establish how the decreased AMH levels contribute to recurrent miscarriages." | |||
* '''Review - Anti-Müllerian Hormone and Its Predictive Utility in Assisted Reproductive Technologies Outcomes''' "Anti-Müllerian hormone (AMH) has become one of the most informative biochemical markers of the ovary and is considered the earliest and most sensitive marker of reproductive aging. The accuracy of AMH in predicting ovarian response to controlled ovarian stimulation has led to AMH-based prognostication counseling and individualization of assisted reproductive technology (ART) stimulation protocols to optimize ovarian response and minimize hyperstimulation risks. Although AMH is considered a good predictor of quantitative ART outcomes, its correlation with qualitative ART outcomes is still controversial. The aim of this review is to provide an updated overview of the clinical utility of AMH in predicting ART outcomes.{{#pmid:30994481|PMID30994481}} | |||
|} | |} | ||
{| class="wikitable mw-collapsible mw-collapsed" | {| class="wikitable mw-collapsible mw-collapsed" | ||
| Line 29: | Line 35: | ||
Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Anti-Mullerian+Hormone ''Anti-Mullerian Hormone''] | Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Anti-Mullerian+Hormone ''Anti-Mullerian Hormone''] | ||
|} | |} | ||
{| class="wikitable mw-collapsible mw-collapsed" | |||
! Older papers | |||
|- | |||
| {{Older papers}} | |||
* '''Salivary and serum androgens with anti-Müllerian hormone measurement for the diagnosis of polycystic ovary syndrome'''{{#pmid:29491484|PMID29491484}} To determine the predictive value of a raised androgen level with an elevated anti-Müllerian hormone (AMH) for the diagnosis or exclusion of polycystic ovary syndrome (PCOS), a prospective cross-sectional study of 170 women (105 with PCOS type A and 65 normal) was undertaken. AMH was combined with one of, total serum testosterone (T); calculated free androgen index; salivary testosterone (salT); serum androstenedione (A); salivary androstenedione (salA).... Therefore, the combination of an AMH with a cut off of 35 pmol/l combined with a raised T and/or a FAI will confirm PCOS whilst a normal AMH with a normal T and/or FAI will exclude PCOS, thus addressing diagnostic uncertainty." | |||
* '''Review - In-vitro regulation of primordial follicle activation'''{{#pmid:29503209|PMID29503209}} "In vivo, primordial follicles gradually exit the resting pool, whereas when primordial follicles are placed into culture, global activation of these follicles occurs. Therefore, the addition of a factor(s) that can regulate primordial follicle activation in vitro may be beneficial to the development of culture systems for ovarian tissue from cancer patients. Several factors have been observed to inhibit follicle activation, including anti-Müllerian hormone, stromal-derived factor 1 and members of the c-Jun-N-terminal kinase pathway." | |||
|} | |||
==Historic== | |||
[[File:Johannes Muller.jpg|thumb|Johannes Müller]] | |||
Named after Johannes Peter Müller (1801-1858) a German medical scientist who made important contributions to several branches of medicine, including anatomy, physiology, embryology, and pathology. He was the first to describe the duct that contributes the female internal genital tract ({{uterus}}) and named after him, the "Müllerian duct". The historic terminology is still used, though the current descriptive terminology is the "paramesonephric duct". | |||
==Male== | |||
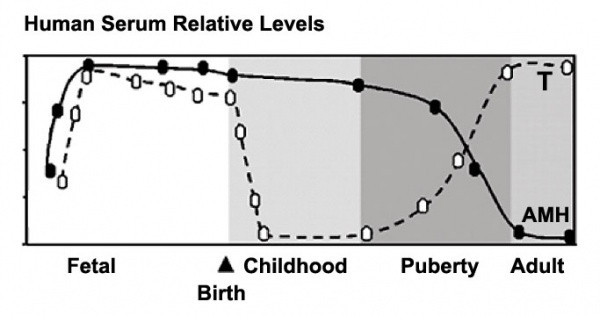
[[File:Male testosterone and AMH level graph.jpg|600px]] | |||
Human Male Testosterone and Anti-Müllerian Hormone (AMH) levels{{#pmid:16544032|PMID16544032}} | |||
==Movies== | ==Movies== | ||
| Line 47: | Line 71: | ||
|} | |} | ||
==Gene== | ==Gene== | ||
* cytogenetic location - | * cytogenetic location - {{Chr19}}p13.3 | ||
* human gene has 5 exons | * human gene has 5 exons | ||
:'''Links:''' [https://www.genenames.org/cgi-bin/gene_symbol_report?match=AMH HGNC] | [ | :'''Links:''' [https://www.genenames.org/cgi-bin/gene_symbol_report?match=AMH HGNC] | [https://www.omim.org/entry/600957 OMIM - AMH] | ||
==Protein== | ==Protein== | ||
* Secreted protein | * Secreted protein | ||
* 560 amino acid polypeptide | |||
* C-terminal domain homology with human transforming growth factor-beta and the beta chain of porcine inhibin | |||
* Testis {{sertoli cell}}s and ovary follicle cells release AMH as a prohormone (proAMH) | |||
* cleaved by subtilisin/kexin-type proprotein convertases or serine proteinases, AMH as active form (AMHN,C) | |||
* In circulation both proAMH and AMHN,C.{{#pmid:26163524|PMID26163524}} | |||
* | 1 mrdlpltsla lvlsalgall gtealraeep avgtsglifr edldwppgip qeplclvalg | ||
61 gdsngssspl rvvgalsaye qaflgavqra rwgprdlatf gvcntgdrqa alpslrrlga | |||
121 wlrdpggqrl vvlhleevtw eptpslrfqe pppggagppe lallvlypgp gpevtvtrag | |||
181 lpgaqslcps rdtrylvlav drpagawrgs glaltlqprg edsrlstarl qallfgddhr | |||
241 cftrmtpall llprsepapl pahgqldtvp fppprpsael eesppsadpf letltrlvra | |||
301 lrvpparasa prlaldpdal agfpqglvnl sdpaalerll dgeeplllll rptaattgdp | |||
361 aplhdptsap watalarrva aelqaaaael rslpglppat apllarllal cpggpgglgd | |||
421 plrallllka lqglrvewrg rdprgpgraq rsagataadg pcalrelsvd lraersvlip | |||
481 etyqanncqg vcgwpqsdrn prygnhvvll lkmqargaal arppccvpta yagkllisls | |||
541 eerisahhvp nmvatecgcr | |||
:'''Links:''' [https://www.ncbi.nlm.nih.gov/protein/EAW69397.1 protein] | [http://www.uniprot.org/uniprot/P03971 UniProt] | |||
==Signaling Pathway== | |||
{| | |||
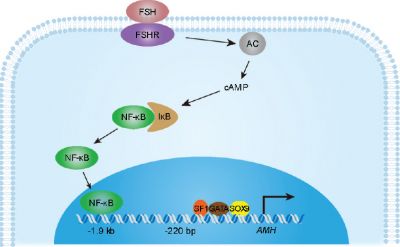
! FSH regulation of AMH transcriptional activation | |||
|- | |||
| [[File:FSH regulation of AMH transcriptional activation.jpg|alt=FSH regulation of AMH transcriptional activation|400px]] | |||
| | |||
* '''{{FSH}}''' - follicle-stimulating hormone | |||
* '''FSHR''' - FSH receptor | |||
* '''AC''' - adenylate cyclase | |||
* '''NF-κB''' - nuclear factor kappa-B | |||
* '''IκB''' - NF-κB inhibitor | |||
* '''SP1''' - transcription factor Sp1 | |||
* '''GATA''' - GATA-binding proteins | |||
* '''{{SOX}}9''' - sex-determining region Y box 9 | |||
* '''AMH''' - anti-Müllerian hormone | |||
* '''cAMP''' - cyclic adenosine monophosphate | |||
|} | |||
===Receptors=== | |||
* 2 type II receptors, transmembrane serine/threonine kinase receptors. | |||
* BMPR1A type I receptor for AMH-induced regression of mullerian ducts. | |||
* Intracellular pathway - similar to bone morphogenetic protein ({{BMP}}) and growth differentiation factor (GDF) ligands. | |||
:'''Links:''' [ | :'''Links:''' [https://www.omim.org/entry/601299 BMPR1A] | ||
==Functions== | ==Functions== | ||
AMH can inhibit the autophosphorylation of the Epidermal Growth Factor Receptor (EGFR) in vitro even though it does not compete with EGF for receptor binding sites. | AMH can inhibit the autophosphorylation of the Epidermal Growth Factor Receptor (EGFR) in vitro even though it does not compete with EGF for receptor binding sites. | ||
==Genital Development== | ==Genital Development== | ||
Male sex differentiation by the fetal testis. | |||
# Anti-Mullerian Hormone (AMH) - by sertoli cells, causes regression of paramesonephric ducts (mullerian ducts) that would differentiate into the uterus and fallopian tubes. | |||
# Testosterone - by Leydig cells, virilizes the external genitalia and promotes prostatic growth. | |||
==Abnormalities== | |||
===Persistent Müllerian Duct Syndrome=== | |||
[[File:Persistent Mullerian duct syndrome 01.jpg|thumb|alt=File:Persistent Mullerian duct syndrome|Uterine remnant present in male during surgery{{#pmid:27464416|PMID27464416}}]] | |||
About 85% of all Persistent Müllerian Duct Syndrome (PMDS) cases are due to gene mutations in anti-Müllerian hormone (AMH) or the receptor (AMHR2) with autosomal recessive transmission. Often incidentally diagnosed during surgical repair of inguinal hernia or cryptorchidism.{{#pmid:27464416|PMID27464416}} | |||
===Polycystic ovary syndrome=== | |||
Polycystic ovary syndrome (PCOS) has been suggested to involve an over-expression of AMH in the PCOS granulosa cells.{{#pmid:28944702|PMID28944702}} | |||
===Ovarian granulosa cell tumours=== | |||
==References== | ==References== | ||
| Line 71: | Line 151: | ||
<references/> | <references/> | ||
=== | ===Reviews=== | ||
{{#pmid:29350886}} | |||
{{#pmid:29175649}} | |||
{{#pmid:28356501}} | |||
{{#pmid:27464416}} | |||
{{#pmid:26163524}} | |||
{{#pmid:11420127}} | |||
===Articles=== | ===Articles=== | ||
| Line 83: | Line 174: | ||
'''Search Bookshelf:''' [http://www.ncbi.nlm.nih.gov/sites/entrez?db=Books&cmd=search&term=Anti-Mullerian+Hormone Anti-Mullerian Hormone] | '''Search Bookshelf:''' [http://www.ncbi.nlm.nih.gov/sites/entrez?db=Books&cmd=search&term=Anti-Mullerian+Hormone Anti-Mullerian Hormone] | ||
==Terms== | |||
{{Genital terms}} | |||
==External Links== | ==External Links== | ||
{{External Links}} | {{External Links}} | ||
* OMIM - [https://www.omim.org/entry/600957 600957 AMH] | |||
* Gene Names - [https://www.genenames.org/cgi-bin/gene_symbol_report?match=AMH HGNC] | |||
{{Glossary}} | {{Glossary}} | ||
{{Footer}} | {{Footer}} | ||
[[Category:Human Embryo]] [[Category:Genital]] [[Category:Male]] [[Category:Molecular]][[Category:AMH]] | |||
[[Category:Human Embryo]] [[Category:Genital]] [[Category:Male]] [[Category:Molecular]] | |||
Latest revision as of 14:59, 8 January 2020
| Embryology - 19 Jun 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
Anti-Mullerian Hormone (AMH; Anti-Müllerian Hormone; Mullerian Inhibiting Substance, MIS; Mullerian Inhibiting Factor, MIF) is a secreted glycoprotein factor (transforming growth factor-beta, TGF-beta superfamily) that regulates gonadal and genital tract development. In the male embryo, the Sertoli cell secrete AMH and inhibit paramesonephric (Mullerian) duct development. In postnatal males, AMH increases during the first month, reaching peak level at 6 months of age, and then slowly declines during childhood falling to low levels in puberty.
In reproductive age women, AMH is produced in the ovary by the granulosa cells surrounding preantral and small antral follicles and serum levels may reflect the remaining follicle cohort and decrease with age.
| Historic Embryology |
| Johannes Peter Müller (1801 - 1858) in 1830 was the first to describe the female genital duct that develops as the uterus and vagina, historically named after him as the "Müllerian duct". The current terminology is the "paramesonephric duct".
Alfred Jost (1916-1991), a French endocrinologist researcher, first discovered in 1947 Anti-Mullerian Hormone he used a rabbit model to identify this hormone as responsible for Müllerian duct (paramesonephric duct) regression during fetal rabbit development. His findings explained several abnormalities of sexual development. This included the "freemartin calf", that acquires AMH from a male twin in utero generating an infertile female with masculinized behavior and non-functioning ovaries. |
| Factor Links: AMH | hCG | BMP | sonic hedgehog | bHLH | HOX | FGF | FOX | Hippo | LIM | Nanog | NGF | Nodal | Notch | PAX | retinoic acid | SIX | Slit2/Robo1 | SOX | TBX | TGF-beta | VEGF | WNT | Category:Molecular |
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Anti-Mullerian Hormone |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Historic
Named after Johannes Peter Müller (1801-1858) a German medical scientist who made important contributions to several branches of medicine, including anatomy, physiology, embryology, and pathology. He was the first to describe the duct that contributes the female internal genital tract (uterus) and named after him, the "Müllerian duct". The historic terminology is still used, though the current descriptive terminology is the "paramesonephric duct".
Male
Human Male Testosterone and Anti-Müllerian Hormone (AMH) levels[6]
Movies
| <html5media height="500" width="200">File:Testis 001.mp4</html5media> |
|
Gene
- cytogenetic location - 19p13.3
- human gene has 5 exons
- Links: HGNC | OMIM - AMH
Protein
- Secreted protein
- 560 amino acid polypeptide
- C-terminal domain homology with human transforming growth factor-beta and the beta chain of porcine inhibin
- Testis sertoli cells and ovary follicle cells release AMH as a prohormone (proAMH)
- cleaved by subtilisin/kexin-type proprotein convertases or serine proteinases, AMH as active form (AMHN,C)
- In circulation both proAMH and AMHN,C.[7]
1 mrdlpltsla lvlsalgall gtealraeep avgtsglifr edldwppgip qeplclvalg
61 gdsngssspl rvvgalsaye qaflgavqra rwgprdlatf gvcntgdrqa alpslrrlga
121 wlrdpggqrl vvlhleevtw eptpslrfqe pppggagppe lallvlypgp gpevtvtrag
181 lpgaqslcps rdtrylvlav drpagawrgs glaltlqprg edsrlstarl qallfgddhr
241 cftrmtpall llprsepapl pahgqldtvp fppprpsael eesppsadpf letltrlvra
301 lrvpparasa prlaldpdal agfpqglvnl sdpaalerll dgeeplllll rptaattgdp
361 aplhdptsap watalarrva aelqaaaael rslpglppat apllarllal cpggpgglgd
421 plrallllka lqglrvewrg rdprgpgraq rsagataadg pcalrelsvd lraersvlip
481 etyqanncqg vcgwpqsdrn prygnhvvll lkmqargaal arppccvpta yagkllisls
541 eerisahhvp nmvatecgcr
Signaling Pathway
| FSH regulation of AMH transcriptional activation | |
|---|---|
|
Receptors
- 2 type II receptors, transmembrane serine/threonine kinase receptors.
- BMPR1A type I receptor for AMH-induced regression of mullerian ducts.
- Intracellular pathway - similar to bone morphogenetic protein (BMP) and growth differentiation factor (GDF) ligands.
- Links: BMPR1A
Functions
AMH can inhibit the autophosphorylation of the Epidermal Growth Factor Receptor (EGFR) in vitro even though it does not compete with EGF for receptor binding sites.
Genital Development
Male sex differentiation by the fetal testis.
- Anti-Mullerian Hormone (AMH) - by sertoli cells, causes regression of paramesonephric ducts (mullerian ducts) that would differentiate into the uterus and fallopian tubes.
- Testosterone - by Leydig cells, virilizes the external genitalia and promotes prostatic growth.
Abnormalities
Persistent Müllerian Duct Syndrome
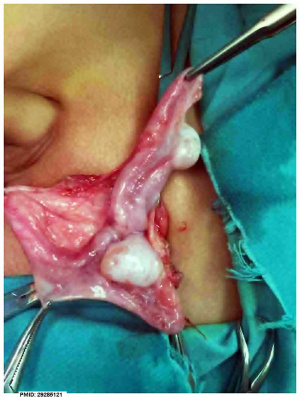
About 85% of all Persistent Müllerian Duct Syndrome (PMDS) cases are due to gene mutations in anti-Müllerian hormone (AMH) or the receptor (AMHR2) with autosomal recessive transmission. Often incidentally diagnosed during surgical repair of inguinal hernia or cryptorchidism.[8]
Polycystic ovary syndrome
Polycystic ovary syndrome (PCOS) has been suggested to involve an over-expression of AMH in the PCOS granulosa cells.[9]
Ovarian granulosa cell tumours
References
- ↑ Mullen RD, Ontiveros AE, Moses MM & Behringer RR. (2019). AMH and AMHR2 mutations: A spectrum of reproductive phenotypes across vertebrate species. Dev. Biol. , 455, 1-9. PMID: 31301298 DOI.
- ↑ McCormack CD, Leemaqz SY, Furness DL, Dekker GA & Roberts CT. (2019). Anti-Müllerian hormone levels in recurrent embryonic miscarriage patients are frequently abnormal, and may affect pregnancy outcomes. J Obstet Gynaecol , 39, 623-627. PMID: 30917731 DOI.
- ↑ Granger E & Tal R. (2019). Anti-Müllerian Hormone and Its Predictive Utility in Assisted Reproductive Technologies Outcomes. Clin Obstet Gynecol , 62, 238-256. PMID: 30994481 DOI.
- ↑ Sathyapalan T, Al-Qaissi A, Kilpatrick ES, Dargham SR, Keevil B & Atkin SL. (2018). Salivary and serum androgens with anti-Müllerian hormone measurement for the diagnosis of polycystic ovary syndrome. Sci Rep , 8, 3795. PMID: 29491484 DOI.
- ↑ Bertoldo MJ, Walters KA, Ledger WL, Gilchrist RB, Mermillod P & Locatelli Y. (2018). In-vitro regulation of primordial follicle activation: challenges for fertility preservation strategies. Reprod. Biomed. Online , , . PMID: 29503209 DOI.
- ↑ Rey R. (2005). Anti-Müllerian hormone in disorders of sex determination and differentiation. Arq Bras Endocrinol Metabol , 49, 26-36. PMID: 16544032 DOI.
- ↑ McLennan IS & Pankhurst MW. (2015). Anti-Müllerian hormone is a gonadal cytokine with two circulating forms and cryptic actions. J. Endocrinol. , 226, R45-57. PMID: 26163524 DOI.
- ↑ 8.0 8.1 Elias-Assad G, Elias M, Kanety H, Pressman A & Tenenbaum-Rakover Y. (2016). Persistent Müllerian Duct Syndrome Caused by a Novel Mutation of an Anti-MüIlerian Hormone Receptor Gene: Case Presentation and Literature Review. Pediatr Endocrinol Rev , 13, 731-40. PMID: 27464416
- ↑ Stracquadanio M, Ciotta L & Palumbo MA. (2018). Relationship between serum anti-Mullerian hormone and intrafollicular AMH levels in PCOS women. Gynecol. Endocrinol. , 34, 223-228. PMID: 28944702 DOI.
Reviews
Roly ZY, Backhouse B, Cutting A, Tan TY, Sinclair AH, Ayers KL, Major AT & Smith CA. (2018). The cell biology and molecular genetics of Müllerian duct development. Wiley Interdiscip Rev Dev Biol , , . PMID: 29350886 DOI.
Shahrokhi SZ, Kazerouni F & Ghaffari F. (2018). Anti-Müllerian Hormone: genetic and environmental effects. Clin. Chim. Acta , 476, 123-129. PMID: 29175649 DOI.
Mossa F, Jimenez-Krassel F, Scheetz D, Weber-Nielsen M, Evans ACO & Ireland JJ. (2017). Anti-Müllerian Hormone (AMH) and fertility management in agricultural species. Reproduction , 154, R1-R11. PMID: 28356501 DOI.
Elias-Assad G, Elias M, Kanety H, Pressman A & Tenenbaum-Rakover Y. (2016). Persistent Müllerian Duct Syndrome Caused by a Novel Mutation of an Anti-MüIlerian Hormone Receptor Gene: Case Presentation and Literature Review. Pediatr Endocrinol Rev , 13, 731-40. PMID: 27464416
McLennan IS & Pankhurst MW. (2015). Anti-Müllerian hormone is a gonadal cytokine with two circulating forms and cryptic actions. J. Endocrinol. , 226, R45-57. PMID: 26163524 DOI.
Josso N, di Clemente N & Gouédard L. (2001). Anti-Müllerian hormone and its receptors. Mol. Cell. Endocrinol. , 179, 25-32. PMID: 11420127
Articles
Kissell KA, Danaher MR, Schisterman EF, Wactawski-Wende J, Ahrens KA, Schliep K, Perkins NJ, Sjaarda L, Weck J & Mumford SL. (2014). Biological variability in serum anti-Müllerian hormone throughout the menstrual cycle in ovulatory and sporadic anovulatory cycles in eumenorrheic women. Hum. Reprod. , 29, 1764-72. PMID: 24925522 DOI.
Search Pubmed
Search Pubmed: Anti-Mullerian Hormone | AMH
Search Bookshelf: Anti-Mullerian Hormone
Terms
| Genital System Terms (expand to view) |
|---|
Note there are additional glossaries associated with spermatozoa, oocyte renal.
|
| Other Terms Lists |
|---|
| Terms Lists: ART | Birth | Bone | Cardiovascular | Cell Division | Endocrine | Gastrointestinal | Genital | Genetic | Head | Hearing | Heart | Immune | Integumentary | Neonatal | Neural | Oocyte | Palate | Placenta | Radiation | Renal | Respiratory | Spermatozoa | Statistics | Tooth | Ultrasound | Vision | Historic | Drugs | Glossary |
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- OMIM - 600957 AMH
- Gene Names - HGNC
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, June 19) Embryology Developmental Signals - Anti-Mullerian Hormone. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Developmental_Signals_-_Anti-Mullerian_Hormone
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G