Abnormal Development - Drugs: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 74: | Line 74: | ||
* First trimester use of prescription medications has increased by more than 60% | * First trimester use of prescription medications has increased by more than 60% | ||
* Use of 4 or more medications in the first trimester has tripled (9.9% to 27.6%) | * Use of 4 or more medications in the first trimester has tripled (9.9% to 27.6%) | ||
The USA has also recently (2015) replaced the drug classification with the "FDA Pregnancy and Lactation Labeling Rule" (PLLR).{{#pmid:27904304|PMID27904304}} | |||
:'''Links:''' [[USA Drug Categories]] | |||
===Europe=== | ===Europe=== | ||
Revision as of 09:38, 16 February 2018
| Embryology - 18 Jun 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
| Educational Use Only - Embryology is an educational resource for learning concepts in embryological development, no clinical information is provided and content should not be used for any other purpose. |
Introduction
This page introduces the possible effects of maternal use of some selected legal drugs (therapeutic chemicals/agents) on development.[1] In some cases these drugs are prescribed to treat pre-existing or pregnancy related maternal medical conditions. This is not a comprehensive drug list and includes some known teratogens as well as data from early studies that require further confirmation. In all cases, a discussion with a medical practioner should be had prior to any reproductive decision.
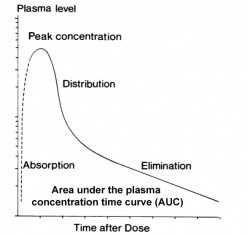
The placenta and fetal tissues may deal with drugs differently from adult target tissues. In particular, drugs are "cleared", metabolised and excreted, at a different rate in both the fetus and in newborn infants. In general there is a much lower rate of clearance.
Legal drugs are classified, usually by each country's appropriate regulatory body, on the safety of drugs during pregnancy. In Australia, the Therapeutic Goods Authority has classes (A, B1, B2, B3, C, D and X) to define their safety. Historically in the USA, before 2015 drugs were classified by the Food and Drug Administration (FDA) into labelling classes (A, B, C, D, and X) to define their safety. Since 2015 this drug classification has been replaced with the "FDA Pregnancy and Lactation Labeling Rule" (PLLR).[2]
Note that pregnant and breastfeeding women are generally excluded from drug clinical trials[3] and most data relies on animal studies.
There are also a growing range of herbal drugs which may not have undergone this type of study and classification.
The importance of careful evaluation of drugs and differences between species{{#pmid3104022|PMID3104022}} can be historically demonstrated with the teratogenic effects of thalidomide, a drug given to treat "morning sickness" in the first trimester of pregnancy, which affected musculoskeletal development. This current page also gives examples of some other current drugs which have been shown to impact on development.
Currently, there is a USA population specific abuse of the prescription drug Oxycodone, commercial name "OxyContin".{{#pmid27513641|PMID27513641}} Oxycodone is a strong opioid analgesic, semisynthetic opioid synthesized from thebaine, that binds μ- and κ-opioid receptors.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Drug teratogenicity <pubmed limit=5>Drug teratogenicity</pubmed> |
Drug Use During Pregnancy
USA
A 2011 study of USA data from 1976-2008 has shown:[6]
- 6 million pregnancies every year
- 50% of pregnant women reported taking at least one medication
- Pregnant women take an average of 2.6 medications at any time during pregnancy
- First trimester use of prescription medications has increased by more than 60%
- Use of 4 or more medications in the first trimester has tripled (9.9% to 27.6%)
The USA has also recently (2015) replaced the drug classification with the "FDA Pregnancy and Lactation Labeling Rule" (PLLR).[2]
- Links: USA Drug Categories
Europe
A 2017 study of European data from 15 European countries from October 2011 to February 2012 has shown:[7]
- 69% of women used medications classified as safe
- 28% used medications classified as risky
- 3% used medications with no classification available
- Socio-demographic and medical factors were associated with use of risky medications during pregnancy
- Having a chronic disorder was the factor with the strongest association with the use of risky medications during pregnancy
Australia
A 2017 study of pregnancy-related calls to Australian national medicines call centre covering 8 years has shown:[8]
- 1166 calls with stage of pregnancy available concerned safety
- 34% of questions related to medication classified as 'safe' during pregnancy
- After antidepressants, most calls were made about over-the-counter (OTC) medicines
- paracetamol, dexchlorpheniramine, codeine
- Safe treatment for everyday conditions was of increasing concern as pregnancy progressed
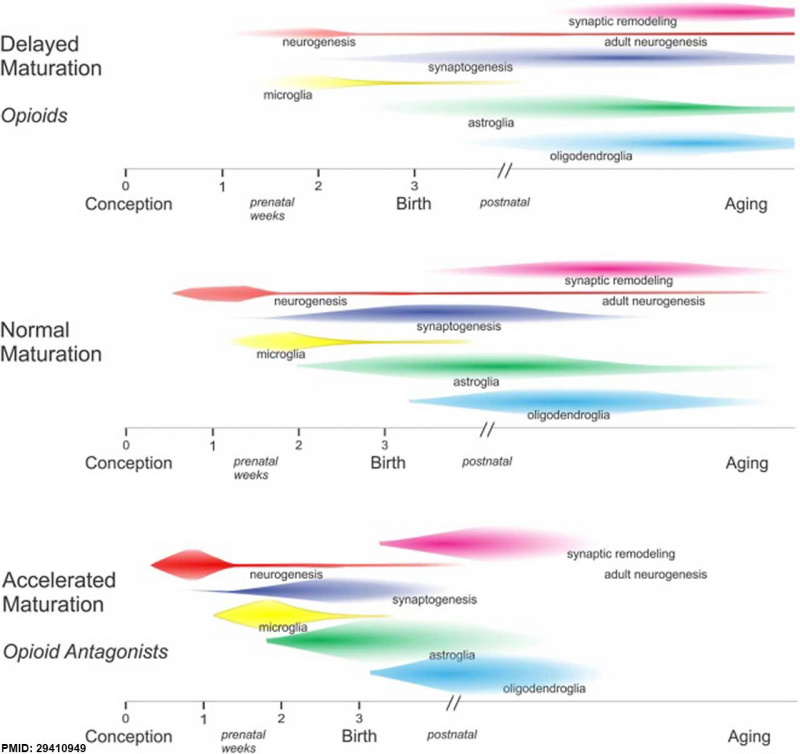
Neural Development
Opioids and neural development timeline effects.{{#pmid29410949|PMID29410949}}
Australia - Advisory Committee on Prescription Medicines
The Australian Drug Evaluation Committee (ADEC) was established in 1963 following the thalidomide experience and in 2010 this committee was replaced by the Advisory Committee on Prescription Medicines (ACPM). The new ACPM advises and makes recommendations to the Therapeutic Goods Administration (TGA) on prescription medicines listed on the Australian Register of Therapeutic Goods (ARTG), established under the Therapeutic Goods Act 1989. There were approximately 54,000 products on the Australian Register of Therapeutic Goods as at 23 May 2008.
Advisory Committee on Prescription Medicines
- inclusion of a prescription medicine on the Australian Register of Therapeutic Goods (the Register)
- changes to an entry of a prescription medicine on the Register
- removal or retention of a prescription medicine on the Register
- Links: Advisory Committee on Prescription Medicines | A guide to labelling drugs and poisons (2007) | Labelling Guide PDF
Australian Drug Categories
| Australian Drug Categories |
|---|
| Legal drugs are classified, usually by each country's appropriate regulatory body, on the safety of drugs during pregnancy. In Australia, the Therapeutic Goods Authority has classes (A, B1, B2, B3, C, D and X) to define their safety. In the USA, drugs are classified by the Food and Drug Administration (FDA) into classes (A, B, C, D, and X) to define their safety. (More? Australian Drug Categories)
|
Legal drugs are classified, usually by each country's appropriate regulatory body, on the safety of drugs during pregnancy. In Australia, the Therapeutic Goods Authority has classes (A, B1, B2, B3, C, D and X) to define their safety. In the USA, drugs are classified by the Food and Drug Administration (FDA) into classes (A, B, C, D, and X) to define their safety. (More? Australian Drug Categories)
Pregnancy Category A
Have been taken by a large number of pregnant women and women of childbearing age without an increase in the frequency of malformations or other direct or indirect harmful effects on the fetus having been observed.
Pregnancy Category B1
Drugs which have been taken by only a limited number of pregnant women and women of childbearing age, without an increase in the frequency of malformation or other direct or indirect harmful effects on the human fetus having been observed. Studies in animals have not shown evidence of an increased occurrence of fetal damage.
Pregnancy Category B2
Have been taken by only a limited number of pregnant women and women of childbearing age, without an increase in the frequency of malformation or other direct or indirect harmful effects on the human fetus having been observed. Studies in animals are inadequate or may be lacking, but available data show no evidence of an increased occurrence of fetal damage.
Pregnancy Category B3
Have been taken by only a limited number of pregnant women and women of childbearing age, without an increase in the frequency of malformation or other direct or indirect harmful effects on the human fetus having been observed. Studies in animals have shown evidence of an increased occurrence of fetal damage, the significance of which is considered uncertain in humans.
Pregnancy Category C
Have caused or may be suspected of causing, harmful effects on the human fetus or neonate without causing malformations. These effects may be reversible.
Pregnancy Category D
Have caused, are suspected to have caused or may be expected to cause, an increased incidence of human fetal malformations or irreversible damage. These drugs may also have adverse pharmacological effects.
Pregnancy Category X
Have such a high risk of causing permanent damage to the fetus that they should NOT be used in pregnancy or when there is a possibility of pregnancy.
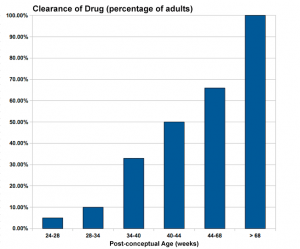
Infant Drug Clearance
The drug clearance data below are only approximate calculated rates for the fetus and infant from NZ Drug Safety in Lactation
| Post-conceptual Age (weeks) | Clearance of Drug (percentage of adults) |
| 24-28 | 5% |
| 28-34 | 10% |
| 34-40 | 33% |
| 40-44 | 50% |
| 44-68 | 66% |
| > 68 | 100% |
Drug Testing
| Drug Testing |
|---|
| Typical testing of new drug compound today involves a lengthy series of animal and human studies.
Animal studies Usually tested in at least two mammalian species (rats and guinea pigs) using both single and repeated doses. For determining reproductive effects, tests on both male and female animals with dosing begins 4 weeks prior to mating are conducted to determine effects on fertility in both sexes, on embryogenesis, and on fetal malformation. Human Clinical trials Following animal studies to determine dose, efficacy and apparent safety, human studies can commence. Clinical trials are carried out under very strict conditions, set by international regulatory bodies in agreement with the principles espoused in the Declaration of Helsinki. There are four phases to the trials.
|
| After phase I to III the pharmaceutical company compiles all study data for independent assessment by government regulatory authorities in each country.
Regulatory Authorities: FDA in the USA, Therapeutic Goods Administration (TGA) in Australia, Medsafe in New Zealand, Medicines & Healthcare products Regulatory Agency (MHRA) in the UK, and Health Products and Food Branch (HPFB) in Canada. |
| Declaration of Helsinki |
| The Declaration of Helsinki was developed by The World Medical Association (WMA) as a statement of ethical principles for medical research involving human subjects, including research on identifiable human material and data. The Declaration is intended to be read as a whole and each of its constituent paragraphs should not be applied without consideration of all other relevant paragraphs. It is widely regarded as the cornerstone document on human research ethics. It is named after the location of its initial adoption in Helsinki, Finland, in June 1964. |
Typical testing of new drug compound today involves a lengthy series of animal and human studies.
Animal studies
Usually tested in at least two mammalian species (rats and guinea pigs) using both single and repeated doses. For determining reproductive effects, tests on both male and female animals with dosing begins 4 weeks prior to mating are conducted to determine effects on fertility in both sexes, on embryogenesis, and on fetal malformation.
Human Clinical trials
Following animal studies to determine dose, efficacy and apparent safety, human studies can commence. Clinical trials are carried out under very strict conditions, set by international regulatory bodies in agreement with the principles espoused in the Declaration of Helsinki. There are four phases to the trials.
- Phase I trials - conducted in small groups of 10 to 20 healthy young male volunteers. Designed to examine how the drug is absorbed, distributed, metabolised and excreted by the body and to establish the safe dose for phase II trials.
- Phase II trials - conducted in 50 to 100 patients with the disease rather than healthy volunteers as in phase I. Designed to examine what effect the drug has on the body (heart rate, blood pressure and cognitive effects) depending on the disease the drug is being developed to treat.
- Phase III trials - conducted in 100’s of patients (larger numbers) with a particular disease or condition and are generally randomised comparative double-blinded studies. Using a comparator of either placebo, another active drug already used, or both. Several phase III trials are usually required by the regulatory authorities. Note that even these studies may not identify uncommon adverse effects, until used widely in the community.
- Phase IV trials - (post-registration) conducted in 1000’s of patients over several years, these trials are randomised controlled trials undertaken after the drug has been registered.
After phase I to III the pharmaceutical company compiles all study data for independent assessment by government regulatory authorities in each country.
- Regulatory Authorities: USA FDA | Australia Therapeutic Goods Administration (TGA) | New Zealand Medsafe | UK Medicines & Healthcare products Regulatory Agency (MHRA) | Canada Health Products and Food Branch (HPFB)
Declaration of Helsinki
The Declaration of Helsinki was developed by The World Medical Association (WMA) as a statement of ethical principles for medical research involving human subjects, including research on identifiable human material and data. The Declaration is intended to be read as a whole and each of its constituent paragraphs should not be applied without consideration of all other relevant paragraphs. It is widely regarded as the cornerstone document on human research ethics. It is named after the location of its initial adoption in Helsinki, Finland, in June 1964.
Teratology
Now consider how different environmental effects during pregnancy may influence developmental outcomes. The terms listed below are often used to describe these environmental effects
- Teratogen (Greek, teraton = monster) any agent that causes a structural abnormality (congenital abnormalities) following fetal exposure during pregnancy. The overall effect depends on dosage and time of exposure. (More? Critical Periods of Development)
- Absolute risk the rate of occurrence of an abnormal phenotype among individuals exposed to the agent. (e.g. fetal alcohol syndrome)
- Relative risk the ratio of the rate of the condition among the exposed and the nonexposed. (e.g. smokers risk of having a low birth weight baby compared to non-smokers) A high relative risk may indicate a low absolute risk if the condition is rare.
- Mutagen a chemical or agent that can cause permanent damage to the deoxyribonucleic acid (DNA) in a cell. DNA damage in the human egg or sperm may lead to reduced fertility, spontaneous abortion (miscarriage), birth defects and heritable diseases.
- Fetotoxicant is a chemical that adversely affects the developing fetus, resulting in low birth weight, symptoms of poisoning at birth or stillbirth (fetus dies before it is born).
- Synergism when the combined effect of exposure to more than one chemical at one time, or to a chemical in combination with other hazards (heat, radiation, infection) results in effects of such exposure to be greater than the sum of the individual effects of each hazard by itself.
- Toxicogenomics the interaction between the genome, chemicals in the environment, and disease. Cells exposed to a stress, drug or toxicant respond by altering the pattern of expression of genes within their chromosomes. Based on new genetic and microarray technologies.
Thalidomide
Thalidomide is a drug that was introduced on to the market on October 1, 1957 in West Germany. Thalidomide soon became a drug prescribed to pregnant women to combat symptoms associated with morning sickness. When taken during the first trimester of pregnancy, thalidomide prevented the proper growth of the fetus resulting in horrific birth defects in thousands of children around the world.
It was the linking of newborn abnormalities with the taking of thalidomide by an Australian clinician, William McBride, that identified it as a teratogenic agent causing a "thalidomide embryopathy".{{#pmid3054428|PMID331548}}
Not all species embryos are affected by the drug in the same way, with human and rabbit being most susceptible to the teratogenic effects. In addition, the effect on human development is also dependent upon the time and dose of the drug exposure, the "critical periods".
Antiepileptic Drugs
This class of drugs are prescribed for a range of neurological conditions and are generally in a class of medications called anticonvulsants. There has been a recent review of information about the teratogenicity of antiepileptic medications.{{#pmid18777479|PMID18777479}} Infant development affected by these drugs has also been called "fetal anticonvulsant syndrome", only a few drug examples are shown below.
Valproic Acid
[[File:Fetal valproate syndrome 01.jpg|thumb|alt=Fetal Valproate Syndrome facial dysmorphism|Fetal Valproate Syndrome facial dysmorphism{{#pmid25400349|PMID25400349}} (Divalproex sodium, Valproate sodium, VPA) Fetal Valproate Syndrome (FVS) results from prenatal exposure to valproic acid.
Valproic acid may also have direct effects on the placenta, altering the expression of transporters required for maternal thyroid hormone to cross to the fetus.{{#pmid27142887|PMID27142887}} (More? Maternal Thyroid)
There has been recently identified in a Danish study is a risk of autism spectrum disorders and childhood autism.{{#pmid23613074|PMID23613074}}
There is a risk of neural tube defects (NTDs) associated with this drug apparently due to a common polymorphism (677C→T) in humans for the methylene tetrahydrofolate reductase gene (MHTFR), that affects folate metabolism. Both genotypes (homozygote and heterozygote) show an increased risk with maternal heterozygotes greater.{{#pmid10563481|PMID10563481}}
Increased risk of heart defects, craniofacial abnormalities, skeletal and limb defects. A recent study suggests that changes in cellular reactive oxygen species (ROS) levels may also lead to increased apoptosis during development.{{#pmid21868484|PMID21868484}}
There have been several case studies of the limb development described with this drug.{{#pmid10826630|PMID10826630}}
| Species | Neural tube defects | Skeletal defects | Route |
| Man | 30 | 20-30 | Oral |
| Monkey | not observed | 150 | oral |
| Rabbit | not observed | 150 | oral |
| Rat | not observed | 150 | oral |
| Hamster | 300 | not investigated | ip |
| Mouse | 200 | 200, 250, 400 | ip, sc, oral |
Carbamazepine
Carbamazepine is an anticonvulsant and mood stabilizing drug used primarily in the treatment of epilepsy and bipolar disorder.
Phenytoin
Antithyroid Drugs
Graves' disease (GD) is the most common cause of hyperthyroidism during pregnancy (estimated 1 in 500 to 1,000 women) and has been treated with the antithyroid drugs propylthiouracil (PTU) and methimazole (MMI).
Propylthiouracil
Propylthiouracil (PTU) is an antithyroid drug used to treat maternal hyperthyroidism, commonly Graves' disease, during pregnancy. A recent study showed cranial neural tube defects in the mouse developmental model.{{#pmid22529993|PMID22529993}}
Danish Study
The following information is from a recent Danish nationwide register-based cohort study of birth defects after early pregnancy use of antithyroid drugs.{{#pmid24151287|PMID24151287}} (See also a recent Japanese study.{{#pmid22547422|PMID22547422}})
Objective: Our objective was to determine to which degree the use of methimazole (MMI)/carbimazole (CMZ) and propylthiouracil (PTU) in early pregnancy is associated with an increased prevalence of birth defects.
Methods: This Danish nationwide register-based cohort study included 817 093 children live-born from 1996 to 2008.
Results: The prevalence of birth defects was high in children exposed to ATD in early pregnancy (PTU, 8.0%; MMI/CMZ, 9.1%; MMI/CMZ and PTU, 10.1%; no ATD, 5.4%; nonexposed, 5.7%; P < .001). Both maternal use of MMI/CMZ (adjusted OR = 1.66 [95% CI 1.35-2.04]) and PTU (1.41 [1.03-1.92]) and maternal shift between MMI/CMZ and PTU in early pregnancy (1.82 [1.08-3.07]) were associated with an increased OR of birth defects. MMI/CMZ and PTU were associated with urinary system malformation, and PTU with malformations in the face and neck region. Choanal atresia, esophageal atresia, omphalocele, omphalomesenteric duct anomalies, and aplasia cutis were common in MMI/CMZ-exposed children (combined, adjusted OR = 21.8 [13.4-35.4]).
Conclusions: Both MMI/CMZ and PTU were associated with birth defects, but the spectrum of malformations differed. More studies are needed to corroborate results in regard to early pregnancy shift from MMI/CMZ to PTU. New ATD with fewer side effects should be developed.
(above text edited from abstract)
- Links: Neural Abnormalities | Thyroid Development
Pain Relief
There has been some recent interest in drugs used for maternal pain relief during pregnancy. Like all data from new research, these findings require more detailed and additional studies to confirm any finding related to pain relief during pregnancy. This section is included to show how today all drugs today can come under a more complex research spotlight, compared to earlier times.
Oxycodone
Currently, there is a USA population specific abuse of the prescription drug Oxycodone, commercial name "OxyContin".{{#pmid27513641|PMID27513641}} Oxycodone is a strong opioid analgesic, semisynthetic opioid synthesized from thebaine, that binds μ- and κ-opioid receptors. Animal studies appear to show no teratogenic effects at clinical doses (TGA 2010 report), though the drug can also pass into breast milk and there may also be untested polysubstance abuse by maternal abusers.
Acetaminophen
A single recent 2014 retrospective study of the Danish National Birth Cohort (1996 - 2002) 64 322 of live-born children using a phone interview has suggested a possible linkage between acetaminophen (paracetamol) and attention-deficit/hyperactivity disorder (ADHD).[9] More than half of all mothers reported acetaminophen use while pregnant.
A second earlier 2013 published study of the Norwegian Mother and Child Cohort (1999 - 2008) of 48 631 children on maternal use of paracetamol at gestational weeks GA 17 and 30 and at 6 months found: "Children exposed prenatally to short-term use of paracetamol (1-27 days) also had poorer gross motor outcomes, but the effects were smaller than with long-term use. Ibuprofen exposure was not associated with neurodevelopmental outcomes."{{#pmid24163279|PMID24163279}}
- Links: MedLine Plus | MedLine Plus Overdose information | Search PubMed - acetaminophen+pregnancy | Search PubMed - ibuprofen+pregnancy
Oral Contraceptives
A recent 2016 Danish study{{#pmid26738512|PMID26738512}} ofbirths from Danish registries between 1997 and 2011 identified that "Oral contraceptive exposure just before or during pregnancy does not appear to be associated with an increased risk of major birth defects."
- Links: Menstrual Cycle
Herbal Drugs
The following herbal drugs have been used for a number of different maternal conditions: Ginkgo Biloba, Kava (Piper methysticum), St. John's wort (Hypericum perforatum), Tian Ma (Gastrodia elata), Valerian (Valeriana officinalis). In some cases very little is known about the potential teratogenic effects of these drugs (More? Herbal Drugs).
HSTAT St. John's Wort | Appendix II: Side Effects, Adverse Effects, Precautions, and Warnings "The safety of using hypericum during pregnancy or lactation has not been proven so it should be avoided." "St. John's wort induces the CYP 450 3A4 metabolic pathway which is also used by many prescription drugs used to prevent conditions (transplant rejection or pregnancy oral contraceptives), health care providers should alert patients about these potential drug interactions."
Anaesthesia
"Under usual circumstances, surgery is only conducted during pregnancy when it is absolutely necessary for the wellbeing of the mother, fetus, or both."{{#pmid22156272|PMID22156272}} Maternal conditions requiring surgery, either related or not related to a pregnancy, may require anaesthesia and all general anaesthetic drugs cross the placenta. Teratogenic effects have not been identified with anaesthesia drugs, though there are suggestions of some impact on neurodevelopment.{{#pmid22405978|PMID22405978}}
Opioids
Neonatal abstinence syndrome(NAS) describes neonatal affects of abrupt discontinuation at birth of opioids exposure and fetal dependence during development in the uterus. Prenatal opioid exposure can occur through prescription or illegal drug use. Prescription opioids include: codeine, dihydrocodeine, fentanyl, hydrocodone, hydromorphone, levorphanol, meperidine, methadone, morphine, oxycodone, oxymorphone, pentazocin, propoxyphene, buprenorphine, tapentadol, and tramadol.
A USA clinical study{{#pmid22546608|PMID22546608}} has identified a trend increase in NAS live birth incidence (2000, 1.2/1,000; 2009 3.39/1000). Animal models have identified neural development abnormalities associated with prenatal opioid exposure.
References
- ↑ Bologa-Campeanu M, Koren G, Rieder M & McGuigan M. (1988). Prenatal adverse effects of various drugs and chemicals. A review of substances of frequent concern to mothers in the community. Med Toxicol Adverse Drug Exp , 3, 307-23. PMID: 3054428
- ↑ 2.0 2.1 Pernia S & DeMaagd G. (2016). The New Pregnancy and Lactation Labeling Rule. P T , 41, 713-715. PMID: 27904304
- ↑ Illamola SM, Bucci-Rechtweg C, Costantine MM, Tsilou E, Sherwin CM & Zajicek A. (2018). Inclusion of pregnant and breastfeeding women in research - efforts and initiatives. Br J Clin Pharmacol , 84, 215-222. PMID: 28925019 DOI.
- ↑ AIHW 2016. Poisoning in children and young people 2012–13. Injury research and statistics series no. 97. Cat. no. INJCAT 173. Canberra: AIHW.
- ↑ Janneke Jentink, Helen Dolk, Maria A Loane, Joan K Morris, Diana Wellesley, Ester Garne, Lolkje de Jong-van den Berg, for the EUROCAT Antiepileptic Study Working Group Intrauterine exposure to carbamazepine and specific congenital malformations: systematic review and case-control study BMJ 2010; 341:c6581 BMJ
- ↑ Mitchell AA, Gilboa SM, Werler MM, Kelley KE, Louik C & Hernández-Díaz S. (2011). Medication use during pregnancy, with particular focus on prescription drugs: 1976-2008. Am. J. Obstet. Gynecol. , 205, 51.e1-8. PMID: 21514558 DOI.
- ↑ Trønnes JN, Lupattelli A & Nordeng H. (2017). Safety profile of medication used during pregnancy: results of a multinational European study. Pharmacoepidemiol Drug Saf , 26, 802-811. PMID: 28449197 DOI.
- ↑ Pijpers EL, Kreijkamp-Kaspers S, McGuire TM, Deckx L, Brodribb W & van Driel ML. (2017). Women's questions about medicines in pregnancy - An analysis of calls to an Australian national medicines call centre. Aust N Z J Obstet Gynaecol , 57, 334-341. PMID: 27624748 DOI.
- ↑ Liew Z, Ritz B, Rebordosa C, Lee P, Olsen J. Acetaminophen Use During Pregnancy, Behavioral Problems, and Hyperkinetic Disorders. JAMA Pediatr. 2014;():. doi:10.1001/jamapediatrics.2013.4914.
Reviews
{{#pmid2228814}}
{{#pmid19104791}}
{{#pmid19012189}}
Articles
{{#pmid1290446}}
Search Pubmed
June 2010 "infant drug clearance rates" All (168) Review (22) Free Full Text (45)
Search Pubmed: infant drug clearance rates | thalidomide teratogenicity |
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- Australia Advisory Committee on Prescription Medicines (ACPM)
- United Nations International Drug Control Programme
- USA TOXNET - Databases on toxicology, hazardous chemicals, environmental health, and toxic releases. mobile
- Centre for Education and Information on Drugs and Alcohol (CEIDA) (Australia)
- Australian Congenital Anomalies Monitoring System (ACAMS)
- Australian Drug Foundation (ADF)
- Child Health and Safety (Australia)
- NIDA (USA)- Consequences of Prenatal Drug Exposure
- Australian Medicines Handbook (no electronic version yet)
- Australian Institute of Health and Welfare (AIHW)
- DrugBank Canadian database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information.
- The Teratogen Information System (TERIS) a computerized database designed to assist physicians or other health care professionals in assessing the risks of possible teratogenic exposures in pregnant women. (subscription only, information is not freely available).
Terms
| Drug Terms | ||
|---|---|---|
| ||
|
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, June 18) Embryology Abnormal Development - Drugs. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Abnormal_Development_-_Drugs
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G