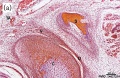
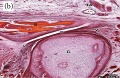
Musculoskeletal System - Joint Development
| Embryology - 23 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
In the adult, the region where two skeletal bones meet and articulate is called a "joint", that are classified based upon their: anatomical structure, mobility and shape.
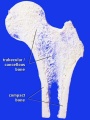
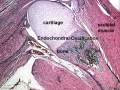
In the embryo, the majority of the vertebrate skeleton is initially formed as a cartilage template, that is later replaced by bone except at the interface between two adjacent bones, leaving in the adult a layer of cartilage in this region. The musculoskeletal system consists of skeletal muscle, bone, and cartilage and is mainly mesoderm in origin with some neural crest contribution.
| Joint Links: joint | synovial joint | temporomandibular joint | musculoskeletal | cartilage | Category:Joint | ||
|
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Joint Development | Joint Morphogenesis | Synovial Joint Development | Temporomandibular Joint Development | Shoulder Joint Development |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Joint Types
Classification
- Fibrous (synarthrodial) - immoveable joints found in cranial vault and teeth
- Cartilagenous (synchondroses and sympheses) - partially moveable joints
- Synovial (diarthrosis) - freely moveable joints are the most common found in the skeleton
Movement
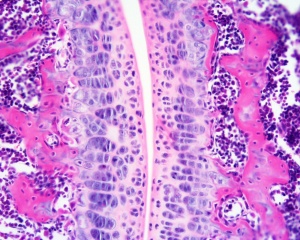
- Hinge - (elbow and knee) Flexion/Extension
- Pivot - (neck, atlas and axis bones) Rotation of one bone around another
- Ball and Socket - (shoulder and hip)
- Saddle - (thumb)
- Condyloid - (wrist joints)
- Gliding - (intercarpal joints) Gliding movements
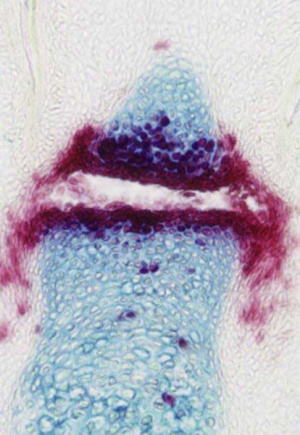
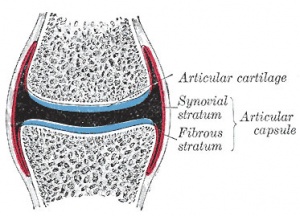
Synovial Joint
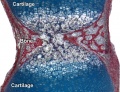
| Skeletal joint cavity development (cavitation) occurs along planes of the future articular surfaces of synovial joints. A number of different markers have been shown to be present in the interzone at the time of cavitation (hyaluronan and hyaluronan synthase, but not chondroitin sulphates).
Fibroblast-like cells (and/or adjacent chondrocytes) with uridine-diphospho glucose dehydrogenase (UDPGD) activity contribute to glycosaminoglycan levels (increases in hyaluronan). These cells are located on the intimal surface of the synovial lining and have been suggested as the possible cavitation mechanism, switching from cellular cohesion to dissociation.[8] |
|
Temporomandibular Joint
The temporomandibular joint (TMJ) is a unique synovial joint. It forms from two blastemata that grow toward each another. The articulating surfaces of the joint are not covered by hyaline cartilage, but a fibrous tissue consisting of both elastic and collagen fibres. Finally, condylar bone formation is different from that seen in other growth plates.
Joint components:
- mandibular condyle
- a fibrocartilagenous disc
- the glenoid fossa of the temporal bone.
The skeletal muscles attached to the joint include the lateral pterygoid and temporalis muscles.
Historic
- 1957 The prenatal development of the human temporomandibular joint[11] that describes development using human embryos from the Carnegie Collection.
- 1962 Embryogenesis of the human temporomandibular joint.[12]
- 1982 Human temporomandibular joint development using human embryos from the Minot Collection.[13]
- 1999 Development of the human temporomandibular joint describes three developmental stages using human embryos from the Madrid Collection. [14]
- first stage (weeks 7-8) - blastematic stage, onset of condyle, articular disc and capsule organization. During week 8 intramembranous ossification of the temporal squamous bone begins.
- second stage (weeks 9-11) - cavitation stage, corresponding to the initial formation of the inferior joint cavity (week 9) and the start condylar chondrogenesis. Week 11 marks the initiation of organization of the superior joint cavity.
- third stage (after week 12) - maturation stage.
- 2009 Development of the mandibular condylar cartilage in human specimens of 10-15 weeks' gestation using human embryos from the Madrid Collection.[15]
Molecular
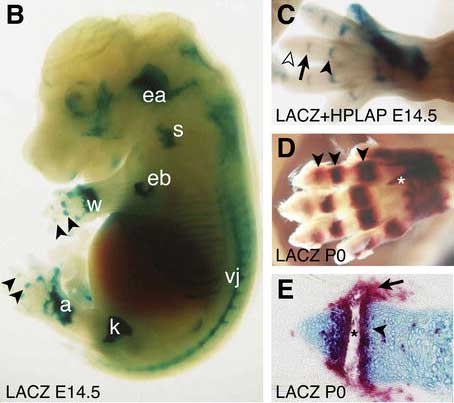
Sprouty
- Spry1 and Spry2 expressed in lateral pterygoid and temporalis muscles.
- inactivation of Spry1 and Spry2 results in overgrowth of these muscles.[16]
- in turn disrupts normal development of the glenoid fossa.
- Sprouty family proteins are evolutionarily conserved inhibitors of tyrosine kinase signaling
- spry protein is an antagonist of fibroblast growth factor (FGF) signaling
- 3 different human homologs
- SPRY1 4q28.1 OMIM - Spry1
- SPRY2 13q31.1 OMIM - Spry2
- SPRY3 Xq28 OMIM - Spry3
Shoulder Joint
This timeline data comes from a recent study of 32 human embryos (Carnegie stages 16-23) and 26 fetuses (9-13 weeks.[4]
| Carnegie Stage | Event |
|---|---|
| 17 | chondrogenic progenitor of the humerus and the medial border of the scapula can be observed. |
| 18 | chondrogenic progenitor for rest of the scapula appears. |
| 19 | glenohumeral joint will begin delaminating and showing a looser central band (interzone). Denser lateral bands will join the humeral head (caput humeri) and the margins of the articular surface of the scapula, thus forming the glenoid labrum (glenoid ligament). |
| 21 | long head of the biceps tendon present |
| 22 | glenoid labrum (glenoid ligament) present |
| 23 | coracohumeral ligament present |
| Week | |
| Fetal Week 10 | osteogenic process begins in the humeral head. Primitive glenohumeral ligament present |
| Fetal Week 11 | osteogenic process begins in the scapula |
| Links: shoulder | joint | limb | timeline Data from human histological study.[4] | |
Knee Joint
In human embryo at week 7 the femur and tibia cartilage template is present (stage 18), by week 8 the posterior cruciate ligament appears (stage 21), and by stage 23 the knee cavity and the anterior cruciate ligament are both also present.[17]
|
Knee Morphogenesis
|
Joint Abnormalities
FGFR-Related Craniosynostosis Syndromes
Pfeiffer syndrome, Apert syndrome, Crouzon syndrome, Beare-Stevenson syndrome, FGFR2-related isolated coronal synostosis, Jackson-Weiss syndrome, Crouzon syndrome with acanthosis nigricans (AN), and Muenke syndrome
- Links: skull
Multiple Epiphyseal Dysplasia
Arthrogryposis
Arthrogryposis (arthrogryposis multiplex congenital, AMC) is a congenital joint contracture occurring in two or more body regions.
Large range of causes including:
- single-gene disorders autosomal recessive, autosomal dominant or X-linked traits.
- part of chromosomal disorders (Trisomy 18, many microdeletions and micro duplications)
- connective tissue disorders
Temporomandibular Disorders
- Links: temporomandibular joint
Osteoarthritis
Clutton's joints
Historic clinical term for a symmetrical joint swelling occurring in patients of both sexes between 5 to 20 years of age with congenital syphilis. Joint swelling is usually in the knees, but can also affect the ankles, elbows, wrists and fingers. Named after Henry Hugh Clutton who first described the condition in 1886.
- Links: syphilis)
References
- ↑ Carrera-Pinzón AF, Márquez-Flórez K, Kraft RH, Ramtani S & Garzón-Alvarado DA. (2019). Computational model of a synovial joint morphogenesis. Biomech Model Mechanobiol , , . PMID: 31863216 DOI.
- ↑ Bechtold TE, Kurio N, Nah HD, Saunders C, Billings PC & Koyama E. (2019). The Roles of Indian Hedgehog Signaling in TMJ Formation. Int J Mol Sci , 20, . PMID: 31847127 DOI.
- ↑ Singh PNP, Shea CA, Sonker SK, Rolfe RA, Ray A, Kumar S, Gupta P, Murphy P & Bandyopadhyay A. (2018). Precise spatial restriction of BMP signaling in developing joints is perturbed upon loss of embryo movement. Development , 145, . PMID: 29467244 DOI.
- ↑ 4.0 4.1 4.2 Hita-Contreras F, Sánchez-Montesinos I, Martínez-Amat A, Cruz-Díaz D, Barranco RJ & Roda O. (2018). Development of the human shoulder joint during the embryonic and early fetal stages: anatomical considerations for clinical practice. J. Anat. , 232, 422-430. PMID: 29193070 DOI.
- ↑ Giorgi M, Carriero A, Shefelbine SJ & Nowlan NC. (2014). Mechanobiological simulations of prenatal joint morphogenesis. J Biomech , 47, 989-95. PMID: 24529755 DOI.
- ↑ Khan IM, Redman SN, Williams R, Dowthwaite GP, Oldfield SF & Archer CW. (2007). The development of synovial joints. Curr. Top. Dev. Biol. , 79, 1-36. PMID: 17498545 DOI.
- ↑ Iwamoto M, Tamamura Y, Koyama E, Komori T, Takeshita N, Williams JA, Nakamura T, Enomoto-Iwamoto M & Pacifici M. (2007). Transcription factor ERG and joint and articular cartilage formation during mouse limb and spine skeletogenesis. Dev. Biol. , 305, 40-51. PMID: 17336282 DOI.
- ↑ Edwards JC, Wilkinson LS, Jones HM, Soothill P, Henderson KJ, Worrall JG & Pitsillides AA. (1994). The formation of human synovial joint cavities: a possible role for hyaluronan and CD44 in altered interzone cohesion. J. Anat. , 185 ( Pt 2), 355-67. PMID: 7525525
- ↑ Wright DM & Moffett BC. (1974). The postnatal development of the human temporomandibular joint. Am. J. Anat. , 141, 235-49. PMID: 4416417 DOI.
- ↑ Mathew AL, Sholapurkar AA & Pai KM. (2011). Condylar Changes and Its Association with Age, TMD, and Dentition Status: A Cross-Sectional Study. Int J Dent , 2011, 413639. PMID: 22114595 DOI.
- ↑ Moffatt BC. The prenatal development of the human temporomandibular joint. (1957) Carnegie Instn. Wash. Publ. 611, Contrib. Embryol., 36: .
- ↑ BAUME LJ. (1962). Embryogenesis of the human temporomandibular joint. Science , 138, 904-5. PMID: 13966992
- ↑ Keith DA. (1982). Development of the human temporomandibular joint. Br J Oral Surg , 20, 217-24. PMID: 6958320 DOI.
- ↑ Mérida-Velasco JR, Rodríguez-Vázquez JF, Mérida-Velasco JA, Sánchez-Montesinos I, Espín-Ferra J & Jiménez-Collado J. (1999). Development of the human temporomandibular joint. Anat. Rec. , 255, 20-33. PMID: 10321990
- ↑ Mérida Velasco JR, Rodríguez Vázquez JF, De la Cuadra Blanco C, Campos López R, Sánchez M & Mérida Velasco JA. (2009). Development of the mandibular condylar cartilage in human specimens of 10-15 weeks' gestation. J. Anat. , 214, 56-64. PMID: 19166473 DOI.
- ↑ Purcell P, Jheon A, Vivero MP, Rahimi H, Joo A & Klein OD. (2012). Spry1 and spry2 are essential for development of the temporomandibular joint. J. Dent. Res. , 91, 387-93. PMID: 22328578 DOI.
- ↑ Mérida-Velasco JA, Sánchez-Montesinos I, Espín-Ferra J, Mérida-Velasco JR, Rodríguez-Vázquez JF & Jiménez-Collado J. (1997). Development of the human knee joint ligaments. Anat. Rec. , 248, 259-68. PMID: 9185992
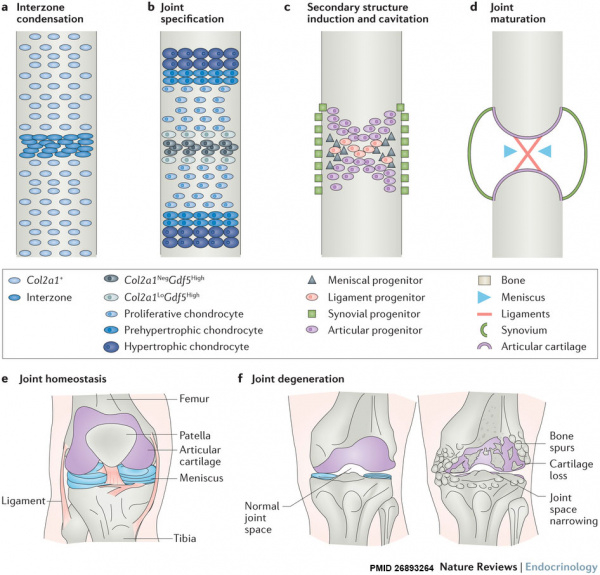
- ↑ Salazar VS, Gamer LW & Rosen V. (2016). BMP signalling in skeletal development, disease and repair. Nat Rev Endocrinol , 12, 203-21. PMID: 26893264 DOI.
Online Textbooks
Developmental Biology Gilbert, Scott F. Sunderland (MA): Sinauer Associates, Inc. ; c2000 Forming the joints
Reviews
Bender ME, Lipin RB & Goudy SL. (2018). Development of the Pediatric Temporomandibular Joint. Oral Maxillofac Surg Clin North Am , 30, 1-9. PMID: 29153232 DOI.
Articles
Purcell P, Jheon A, Vivero MP, Rahimi H, Joo A & Klein OD. (2012). Spry1 and spry2 are essential for development of the temporomandibular joint. J. Dent. Res. , 91, 387-93. PMID: 22328578 DOI.
Rountree RB, Schoor M, Chen H, Marks ME, Harley V, Mishina Y & Kingsley DM. (2004). BMP receptor signaling is required for postnatal maintenance of articular cartilage. PLoS Biol. , 2, e355. PMID: 15492776 DOI.
Mérida-Velasco JA, Sánchez-Montesinos I, Espín-Ferra J, Mérida-Velasco JR, Rodríguez-Vázquez JF & Jiménez-Collado J. (2000). Development of the human elbow joint. Anat. Rec. , 258, 166-75. PMID: 10645964
Koyama E, Leatherman JL, Shimazu A, Nah HD & Pacifici M. (1995). Syndecan-3, tenascin-C, and the development of cartilaginous skeletal elements and joints in chick limbs. Dev. Dyn. , 203, 152-62. PMID: 7544653 DOI.
Edwards JC, Wilkinson LS, Jones HM, Soothill P, Henderson KJ, Worrall JG & Pitsillides AA. (1994). The formation of human synovial joint cavities: a possible role for hyaluronan and CD44 in altered interzone cohesion. J. Anat. , 185 ( Pt 2), 355-67. PMID: 7525525
Search PubMed
Search July 2010 "Joint Development" All (19900) Review (3137) Free Full Text (3325)
Search Pubmed: Joint Development
Additional Images
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 23) Embryology Musculoskeletal System - Joint Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Musculoskeletal_System_-_Joint_Development
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G