Frog Development
| Embryology - 24 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction

The frog has been historically been used as an amphibian animal model of development due to the ease of observation from the fertilized egg through to tadpole stage. The later metamorphosis of the tadpole to frog has also been studied for hormonal controls and limb development. There have also been many different species used in these developmental studies.
The frog was historically used by many of the early embryology investigators and currently there are many different molecular mechanisms concerning development of the frog. The 2012 Nobel prize in medicine was recently awarded to John Gurdon for his 1960's experiments involving nuclear transplantation with adult nuclei into frog eggs, these studies were the precursor to current research in stem cells.
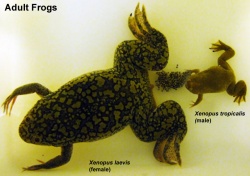
The African clawed frog (Xenopus laevis) has been used in many embryological and electrophysiological studies as well as the basis of a historic pregnancy test. The advantages of this frog is the fertility cycle can be easliy controlled and the eggs develop entirely independently and easily visible to the investigator. You can see an overview of the frog life cycle with links to specific stages as well as movies of the early process of gastrulation. This animal model has also shown that localization of maternal messenger RNA (eg vegetal and review) appears to play a key role in the development of early embryological patterns.
The Leopard frog (Rana pipiens) in 1952 became the first successful nuclear transfer experiment. Nuclear transfer is an embryological technique, and involves removal of the nucleus from an egg and replacement with the nucleus of another donor cell. This experiment paved the way for what we know today as the field of cloning.[1]
In Australia, the cane toad (Bufo marinus) species was introduced in 1935 to control cane insect pests. It has now itself become an introduced pest and has also been studied/used more in order to try and biologically control. The area which they occupy has continued to expand. This toad has a poisonous secretion that is extremely toxic and should be handled with care at all times.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Frog Embryology <pubmed limit=5>Frog Embryology</pubmed> |
| Older papers |
|---|
|
Taxon
Xenopus Laevis
Eukaryotae; mitochondrial eukaryotes; Metazoa; Chordata;Vertebrata; Amphibia; Batrachia; Anura; Mesobatrachia; Pipoidea;Pipidae; Xenopodinae; Xenopus
Rana pipiens
Taxonomy Id: 8404 Preferred common name: northern leopard frog Rank: species
Genetic code: Translation table 1 (Standard) Mitochondrial genetic code: Translation table 2 Lineage( abbreviated ):
Eukaryota; Metazoa; Chordata; Craniata; Vertebrata; Amphibia; Batrachia; Anura; Neobatrachia; Ranoidea; Ranidae; Raninae; Rana
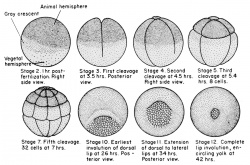
Frog Life Cycle
Development Timeline
|
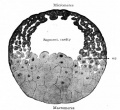
Typical frog development at 18oC from fertilised egg.
|
|
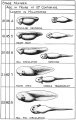
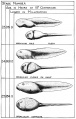
Stages in the Normal Development of Rana pipiens
Oocyte Balbiani body
- spherical cytoplasmic region that forms within the oocyte in early oogenesis and then fragments and disperses in late oogenesis.
- membrane-less structure consisting of mitochondria, endoplasmic reticulum (ER), membranous vesicles and lipid droplets.
Xenopus stage I oocytes
- Balbiani body is ∼40 μm in diameter
- contains half a million mitochondria, with different morphology and metabolism from other cytoplasmic mitochondria
- rich in membranous vesicles, and ER cysternae.
- vegetal apex (METRO region) contains germinal granules and localized RNAs
- Xlsirts[14]
- family of interspersed repeat RNAs that contain from 3 to 13 repeat units (each 79 to 81 nucleotides long) flanked by unique sequences.
- homologous to the mammalian Xist gene involved in X chromosome inactivation
- stage 2 oocytes - appears first in the mitochondrial cloud (Balbiani body)
- stage 3 oocytes - translocated as island-like structures to the vegetal cortex coincident with the localization of the germ plasm.
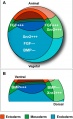
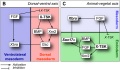
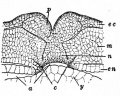
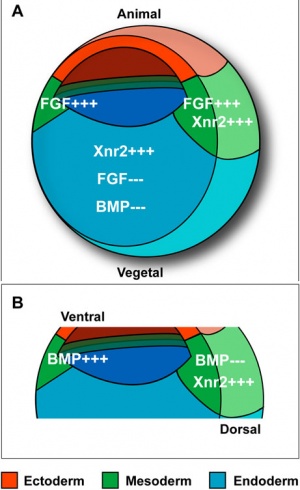
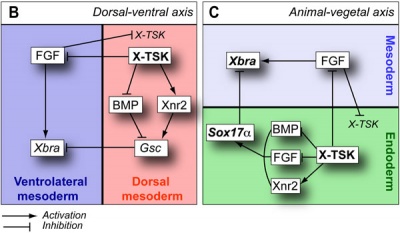
Germ Layers
The following paper cartoons[15] show models of signaling mechanisms that occur during early development of the germ cell layers (ectoderm, mesoderm and endoderm).
|
|
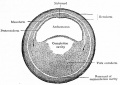
Neural
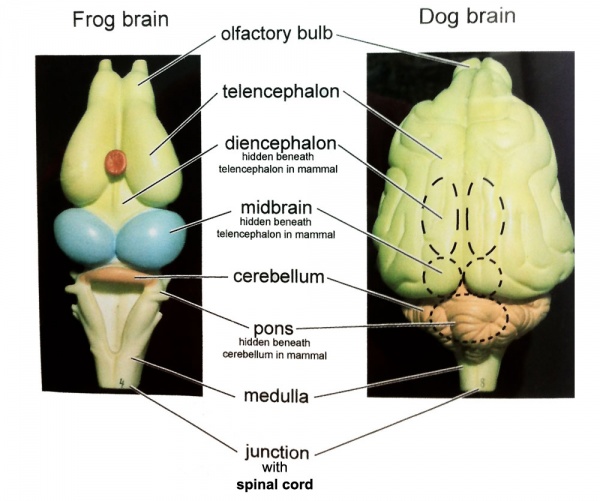
Comparative brain anatomy frog and dog models.
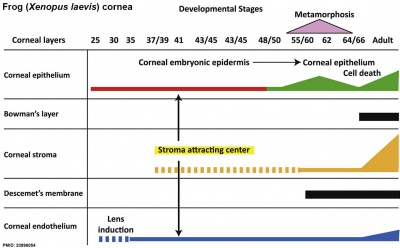
Cornea
| Stage | Event |
|---|---|
| stage 25 | cornea starts from a simple embryonic epidermis overlying developing optic vesicle |
| stage 30 | detachment of lens placode, cranial neural crest cells start to invade the space between the lens and the embryonic epidermis to construct the corneal endothelium. |
| stage 41 | second wave of migratory cells containing presumptive keratocytes invades the matrix, leading to formation of inner cornea and outer cornea. A unique cell mass (stroma attracting center) connects the two layers like the center pole of a tent. |
| stage 48 | secondary stromal keratocytes individually migrate to the center and form the stroma layer. |
| stage 60 | stroma space is filled by collagen lamellae and keratocytes, and the stroma attracting center disappears. At early metamorphosis, embryonic epithelium gradually changes to adult corneal epithelium, covered by microvilli. |
| stage 62 | embryonic epithelium thickens and cell death is observed in the epithelium, coinciding with eyelid opening. |
| After metamorphosis | cornea has attained the adult structure of three cellular layers, epithelium, stroma, and endothelium, and between the cellular layers lie two acellular layers (Bowman's layer and Descemet's membrane) |
| Table data from Xenopus laevis[16] | frog | vision | |
- Links: Cornea Development | Vision Development
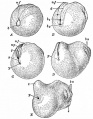
Metamorphosis
Metamorphosis of the frog, Rana catesbiana.
Sequence from left to right, top and bottom:
- tadpole
- tadpole with hind legs only
- tadpole with two pairs of legs
- tadpole with disappearing tail, ready to emerge from water to land
- immature terrestrial frog
- mature frog
Xenbase
Xenbase is a Xenopus model organism computer database with 4 GB of data in many hundreds of tables that has recently (2012) been updated, as described in the abstract of an NAR article.[17]
- "Xenbase (http://www.xenbase.org) is a model organism database that provides genomic, molecular, cellular and developmental biology content to biomedical researchers working with the frog, Xenopus and Xenopus data to workers using other model organisms. As an amphibian Xenopus serves as a useful evolutionary bridge between invertebrates and more complex vertebrates such as birds and mammals. Xenbase content is collated from a variety of external sources using automated and semi-automated pipelines then processed via a combination of automated and manual annotation. A link-matching system allows for the wide variety of synonyms used to describe biological data on unique features, such as a gene or an anatomical entity, to be used by the database in an equivalent manner. Recent updates to the database include the Xenopus laevis genome, a new Xenopus tropicalis genome build, epigenomic data, collections of RNA and protein sequences associated with genes, more powerful gene expression searches, a community and curated wiki, an extensive set of manually annotated gene expression patterns and a new database module that contains data on over 700 antibodies that are useful for exploring Xenopus cell and developmental biology."
Historic Researchers

|

|

|
| Wilhelm Roux (1850 – 1924) | Hans Spemann (1869 - 1941) | John Gurdon (1933 - ) |
| A German zoologist and pioneer of experimental embryology. Experimented by pricking and destroying one of the two blastomeres, to obtain half an embryo from the other. | A German embryologist who worked extensively on amphibian development and was the discoverer of the organiser region (or primitive node) the controller of gastrulation. Received the 1935 Nobel Prize in Physiology or Medicine "for his discovery of the organizer effect in embryonic development". | An English embryologist in 1962 used nuclear transplantation and cloning to show that the nucleus of a differentiated somatic cell retains the totipotency necessary to form a whole organism. Received the 2012 Nobel Prize "for the discovery that mature cells can be reprogrammed to become pluripotent". |
References
- ↑ Briggs R & King TJ. (1952). Transplantation of Living Nuclei From Blastula Cells into Enucleated Frogs' Eggs. Proc. Natl. Acad. Sci. U.S.A. , 38, 455-63. PMID: 16589125
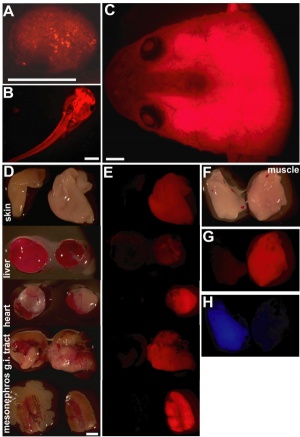
- ↑ Waldner C, Roose M & Ryffel GU. (2009). Red fluorescent Xenopus laevis: a new tool for grafting analysis. BMC Dev. Biol. , 9, 37. PMID: 19549299 DOI.
- ↑ Itoh K, Ossipova O & Sokol SY. (2021). Pinhead antagonizes Admp to promote notochord formation. iScience , 24, 102520. PMID: 34142034 DOI.
- ↑ Bae CJ, Hong CS & Saint-Jeannet JP. (2018). Anosmin-1 is essential for neural crest and cranial placodes formation in Xenopus. Biochem. Biophys. Res. Commun. , 495, 2257-2263. PMID: 29277616 DOI.
- ↑ Sun J, Wang X, Shi Y, Li J, Li C, Shi Z, Chen Y & Mao B. (2018). EphA7 regulates claudin6 and pronephros development in Xenopus. Biochem. Biophys. Res. Commun. , 495, 1580-1587. PMID: 29223398 DOI.
- ↑ Lewis PA, Bradley IC, Pizzey AR, Isaacs HV & Evans GJO. (2017). N1-Src Kinase Is Required for Primary Neurogenesis inXenopus tropicalis. J. Neurosci. , 37, 8477-8485. PMID: 28765332 DOI.
- ↑ Ziková A, Lorenz C, Hoffmann F, Kleiner W, Lutz I, Stöck M & Kloas W. (2017). Endocrine disruption by environmental gestagens in amphibians - A short review supported by new in vitro data using gonads of Xenopus laevis. Chemosphere , 181, 74-82. PMID: 28431277 DOI.
- ↑ Keenan SR & Beck CW. (2016). Xenopus Limb bud morphogenesis. Dev. Dyn. , 245, 233-43. PMID: 26404044 DOI.
- ↑ Sáenz-Ponce N, Mitgutsch C & del Pino EM. (2012). Variation in the schedules of somite and neural development in frogs. Proc. Natl. Acad. Sci. U.S.A. , 109, 20503-7. PMID: 23184997 DOI.
- ↑ Tokmakov AA, Iguchi S, Iwasaki T & Fukami Y. (2011). Unfertilized frog eggs die by apoptosis following meiotic exit. BMC Cell Biol. , 12, 56. PMID: 22195698 DOI.
- ↑ Ploper D, Lee HX & De Robertis EM. (2011). Dorsal-ventral patterning: Crescent is a dorsally secreted Frizzled-related protein that competitively inhibits Tolloid proteases. Dev. Biol. , 352, 317-28. PMID: 21295563 DOI.
- ↑ Kosubek A, Klein-Hitpass L, Rademacher K, Horsthemke B & Ryffel GU. (2010). Aging of Xenopus tropicalis eggs leads to deadenylation of a specific set of maternal mRNAs and loss of developmental potential. PLoS ONE , 5, e13532. PMID: 21042572 DOI.
- ↑ Venkatarama T, Lai F, Luo X, Zhou Y, Newman K & King ML. (2010). Repression of zygotic gene expression in the Xenopus germline. Development , 137, 651-60. PMID: 20110330 DOI.
- ↑ Kloc M, Spohr G & Etkin LD. (1993). Translocation of repetitive RNA sequences with the germ plasm in Xenopus oocytes. Science , 262, 1712-4. PMID: 7505061
- ↑ Morris SA, Almeida AD, Tanaka H, Ohta K & Ohnuma S. (2007). Tsukushi modulates Xnr2, FGF and BMP signaling: regulation of Xenopus germ layer formation. PLoS ONE , 2, e1004. PMID: 17925852 DOI.
- ↑ 16.0 16.1 Hu W, Haamedi N, Lee J, Kinoshita T & Ohnuma S. (2013). The structure and development of Xenopus laevis cornea. Exp. Eye Res. , 116, 109-28. PMID: 23896054 DOI.
- ↑ James-Zorn C, Ponferrada VG, Jarabek CJ, Burns KA, Segerdell EJ, Lee J, Snyder K, Bhattacharyya B, Karpinka JB, Fortriede J, Bowes JB, Zorn AM & Vize PD. (2013). Xenbase: expansion and updates of the Xenopus model organism database. Nucleic Acids Res. , 41, D865-70. PMID: 23125366 DOI.
Books
Rugh, R. The Frog Its Reproduction and Development The Blakiston Company, New York, 1951.
Reviews
Kaneda T & Motoki JY. (2012). Gastrulation and pre-gastrulation morphogenesis, inductions, and gene expression: similarities and dissimilarities between urodelean and anuran embryos. Dev. Biol. , 369, 1-18. PMID: 22634398 DOI.
Jones CM & Smith JC. (2008). An overview of Xenopus development. Methods Mol. Biol. , 461, 385-94. PMID: 19030813 DOI.
del Pino EM, Venegas-Ferrín M, Romero-Carvajal A, Montenegro-Larrea P, Sáenz-Ponce N, Moya IM, Alarcón I, Sudou N, Yamamoto S & Taira M. (2007). A comparative analysis of frog early development. Proc. Natl. Acad. Sci. U.S.A. , 104, 11882-8. PMID: 17606898 DOI.
Articles
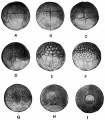
TAYLOR AC & KOLLROS JJ. (1946). Stages in the normal development of Rana pipiens larvae. Anat. Rec. , 94, 7-13. PMID: 21013391
Search Pubmed
Search Pubmed: frog development | xenopus development
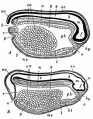
Additional Images
| Historic Images |
|---|
|
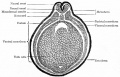
Bailey, F.R. and Miller, A.M. (1921). Text-Book of Embryology. Early development of the frog |
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- Xenbase A database of information pertaining to the cell and developmental biology of the frog, Xenopus
- Xenopus Laboratory List A database of Labs studying Xenopus
- Xenopus Microarrays
- Digital photographs of Xenopus stages (2005)
- Xenopus Cell Biology
- The Xenopus Molecular Marker Resource An electronic library of information on embryonic development of the frog, Xenopus laevis | Index page for all Markers | whole mount staining patterns
- Molecular Markers of Development: cement gland XA, XAG, XCG | early mesoderm - BMP2, BMP4, Chordin, goosecoid, Mix,[Marker_pages/organizer/noggin.html noggin], Xbra, Xnr3, Xwnt-8, XVent1 and XVent2 | endothelial - Xl-fli | germ cells - Xpat | heart - cardiac troponin I , XNKX-2.5, XTin1 (XNKX-2.3) | lateral line - tor70, [Marker_pages/CNS/2G9.html 2G9] | muscle - 5A3, 12/101, cardiac actin, XMyf-5, XMyoD | neural crest - Slug, XTwist , xAP2 | notochord - Xnot, tor70 | pronephros - [Marker_pages/pronephros/3G8.html 3G8 ], Wilms' tumor (xWT1), Xlim-1, Xwnt-4 | pronephric duct - 4A6
- Frogs of Greater Brisbane Region (Australia)
- Developmental Biology- Laurie Iten's Serially Sectioned Frog and Chick Embryos
- Developmental Biology- Jeff Hardin's Amphibian Embryology Tutorial
- NIH- Organisms for biomedical research
- Columbia University Kelley Lab - The natural and unnatural histories of xenopus laevis
| Animal Development: axolotl | bat | cat | chicken | cow | dog | dolphin | echidna | fly | frog | goat | grasshopper | guinea pig | hamster | horse | kangaroo | koala | lizard | medaka | mouse | opossum | pig | platypus | rabbit | rat | salamander | sea squirt | sea urchin | sheep | worm | zebrafish | life cycles | development timetable | development models | K12 |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 24) Embryology Frog Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Frog_Development
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G