BGD Lecture - Endocrine Development
| Embryology - 16 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
BGDB has 2 endocrine related lectures. The first is on endocrine histology (mainly of the HPA axis), the second is endocrine embryology (on this current page).
This lecture covers endocrine development, note that a better understanding can be made if you understand the adult function of each endocrine organ (though this will not be covered in the Lecture). Endocrine development is sometimes divided into neuroendocrine and endocrine and is also generally covered piecemeal in all embryology textbooks, so you may have to look in several different chapters to find supporting textbook information.
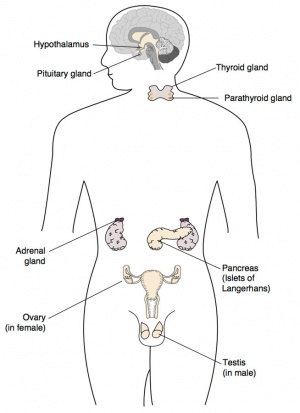
The endocrine system resides within specific endocrine organs and both organs and tissues with other specific functions. Epithelia (ectoderm and endoderm) form the majority of the “ductless” endocrine glands like gastrointestinal and skin associated “ducted” glands. Differentiation of several organs also involves a epithelial/mesenchye interaction, seen in repeated in many differentiation of many different tissues. The endocrine glands produce hormones, which are distributed by the vascular system to the many body tissues, subsequently these organs are richly vascularized.
Hormones “orchestrate” responses in other tissues, including other endocrine organs, and these overall effects can be similar or different in different tissues. These signaling pathways are often described as "axes" the two major types are the: HPA (Hyothalamus-Pituitary-Adrenal) and HPG (Hypothalamus-Pituitary-Gonad). These hormone levels and effects (like music) can be rapid, slow, brief, diurnal, or long-term. Hormone effects can be mimicked, stimulated, and blocked by therapeutic drugs, nutritional and environmental chemicals. Importantly, fetal endocrine development is required for normal fetal growth and differentiation.
| Endocrine in the News |
|---|
|
Papalou O, Kandaraki EA, Papadakis G & Diamanti-Kandarakis E. (2019). Endocrine Disrupting Chemicals: An Occult Mediator of Metabolic Disease. Front Endocrinol (Lausanne) , 10, 112. PMID: 30881345 DOI. "Endocrine disrupting chemicals (EDCs), a heterogeneous group of exogenous chemicals that can interfere with any aspect of endogenous hormones, represent an emerging global threat for human metabolism. There is now considerable evidence that the observed upsurge of metabolic disease cannot be fully attributed to increased caloric intake, physical inactivity, sleep deficit, and ageing. Among environmental factors implicated in the global deterioration of metabolic health, EDCs have drawn the biggest attention of scientific community, and not unjustifiably. EDCs unleash a coordinated attack toward multiple components of human metabolism, including crucial, metabolically-active organs such as hypothalamus, adipose tissue, pancreatic beta cells, skeletal muscle, and liver. Specifically, EDCs' impact during critical developmental windows can promote the disruption of individual or multiple systems involved in metabolism, via inducing epigenetic changes that can permanently alter the epigenome in the germline, enabling changes to be transmitted to the subsequent generations. The clear effect of this multifaceted attack is the manifestation of metabolic disease, clinically expressed as obesity, metabolic syndrome, diabetes mellitus, and non-alcoholic fatty liver disease." |
|
Ghassabian A & Trasande L. (2018). Disruption in Thyroid Signaling Pathway: A Mechanism for the Effect of Endocrine-Disrupting Chemicals on Child Neurodevelopment. Front Endocrinol (Lausanne) , 9, 204. PMID: 29760680 DOI. "Endocrine-disrupting chemicals (EDCs) are natural and synthetic substances with ubiquitous exposure in children and adults including pregnant women. EDCs interfere, temporarily or permanently, with hormonal signaling pathways in the endocrine system by binding to hormone receptors and modifying gene expression." |
|
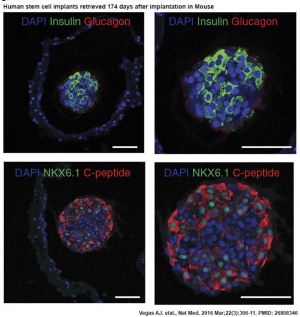
Vegas AJ, Veiseh O, Gürtler M, Millman JR, Pagliuca FW, Bader AR, Doloff JC, Li J, Chen M, Olejnik K, Tam HH, Jhunjhunwala S, Langan E, Aresta-Dasilva S, Gandham S, McGarrigle JJ, Bochenek MA, Hollister-Lock J, Oberholzer J, Greiner DL, Weir GC, Melton DA, Langer R & Anderson DG. (2016). Long-term glycemic control using polymer-encapsulated human stem cell-derived beta cells in immune-competent mice. Nat. Med. , 22, 306-11. PMID: 26808346 DOI. Implanted in the intraperitoneal space of mice treated to chemically induce type 1 diabetes. Implants induced glycemic correction without any immunosuppression until their removal at 174 d after implantation. Human C-peptide concentrations and in vivo glucose responsiveness demonstrated therapeutically relevant glycemic control and retrieved implants contained viable insulin-producing cells.
|
| Oral Contraceptives A recent 2016 Danish study births from Danish registries between 1997 and 2011 identified that:
|
| Ernest Henry Starling (1866-1927)
<html5media>File:Audio - centenary of hormone.mp3</html5media> |
Lecture Objectives
|
|
| Lecture Archive |
|---|
| 2018 PDF | 2017 | 2017 PDF | 2016 | printable version | 2015 Lecture | 2014 Lecture | 2013 Lecture | 2012 Lecture | Endocrine System Development | BGD Lecture - Endocrine Histology | Practical - Virtual Slides |
Endocrine Origins
- Epithelia - (ectoderm) covering embryo, (endoderm) lining gastrointestinal tract, (mesoderm) lining coelomic cavity
- Mesenchyme - (mesoderm) contribution, connective tissue, blood vessels
Hormones
Hormone Types
| Amino acid derivatives | noradrenaline (norepinephrine), adrenalin (epinepherine) , thyroid hormone |
|---|---|
| Proteins, peptides | thyroid stimulating hormone, leutenising hormone, follicle stimulating hormone |
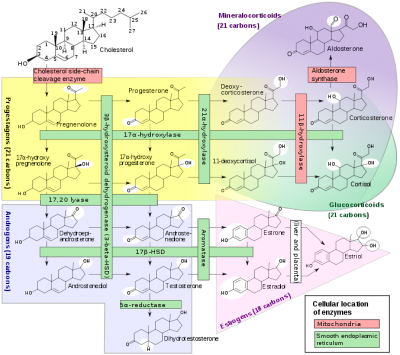
| Steroids (from cholesterol) | androgens, glucocorticoids, mineralocorticoids |
| Signaling Pathways | |
|---|---|
|
|
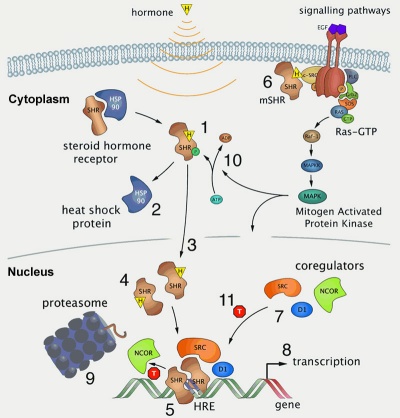
| Steroid biosynthesis pathway | Steroid hormone receptor signaling[1] |
|
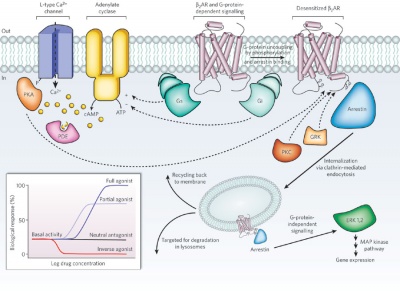
G-protein coupled membrane receptors
|
Hormone Actions
- Autocrine - acts on self (extracellular fluid)
- Paracrine - acts locally (extracellular fluid)
- Endocrine - acts by secretion into blood stream (endocrine organs are richly vascularized)
Hormone Receptors
- Cell surface receptors - modified amino acids, peptides, proteins
- Cytoplasmic/Nuclear Receptors - steroids
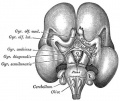
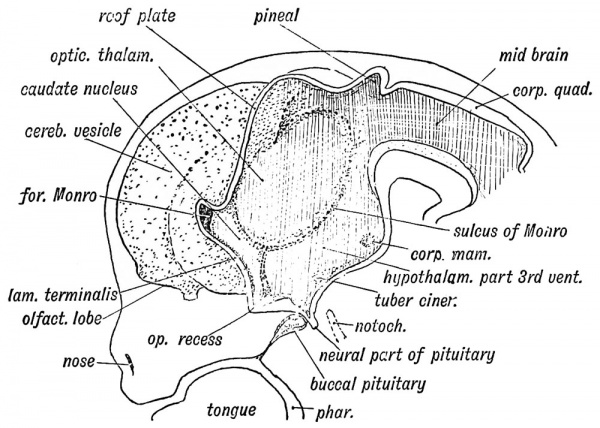
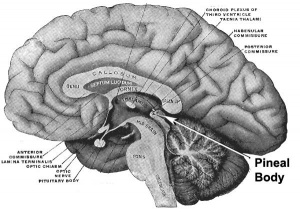
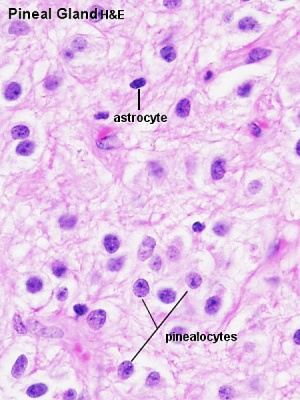
Pineal Gland
|
Pineal Development
|
|

|
| Adult pineal body | Pineal gland position |
|
|
|
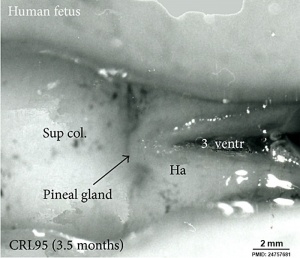
Fetal Pineal Anatomy[2]
|
- Links: pineal | Endocrinology
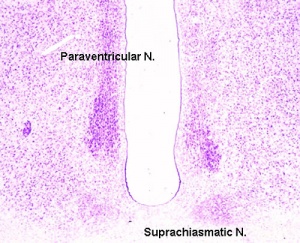
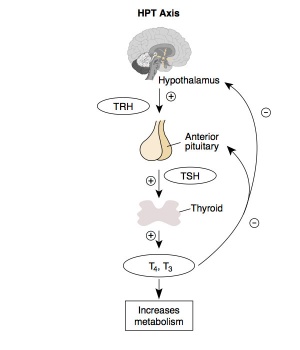
Hypothalamus
Hormones - Corticotrophin releasing hormone (CRH), Thyrotrophin releasing hormone (TRH), Arginine vasopressin (AVP), Gonadotrophin releasing hormone (GnRH), Growth hormone releasing hormone (GHRH), Somatostatin, Prolactin relasing factor (PRF), Dopamine
| Hypothalamic Hormones | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||
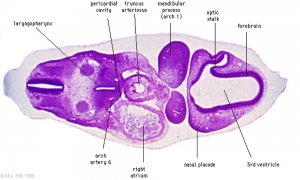
Hypothalamus Development
- Neuroectoderm - prosenecephalon then diencephalon
- ventro-lateral wall intermediate zone proliferation
- Mamillary bodies - form pea-sized swellings ventral wall of hypothalamus
|
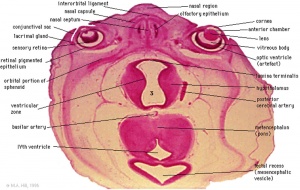
|
| Diencephalon region, shown by optic stalk (Stage 13) |
Late embryonic hypothalamus (Stage 22) |
- Links: hypothalamus
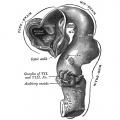
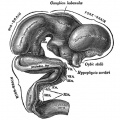
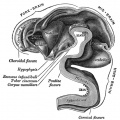
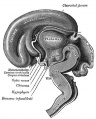
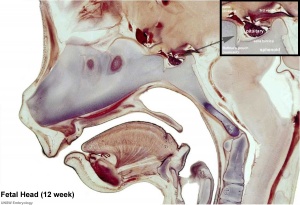
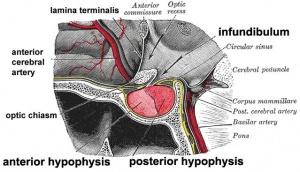
Pituitary

|
The pituitary (hypophysis) sits anatomically within the sella turcica, a space within the sphenoid bone.
Anterior pituitary hormones - Thyroid-stimulating hormone (TSH), Adrenocorticotrophic hormone (ACTH), Luteinizing hormone (LH), Follicle-stimulating hormone (FSH), Somatotrophin/growth hormone (GH), Prolactin (PRL), Melanocyte-stimulating hormone (MSH) Posterior pituitary hormones - Oxytocin, Arginine vasopressin |
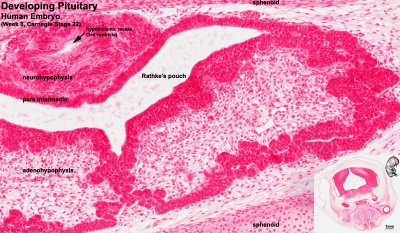
Pituitary Development
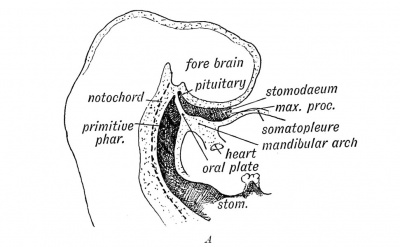
|
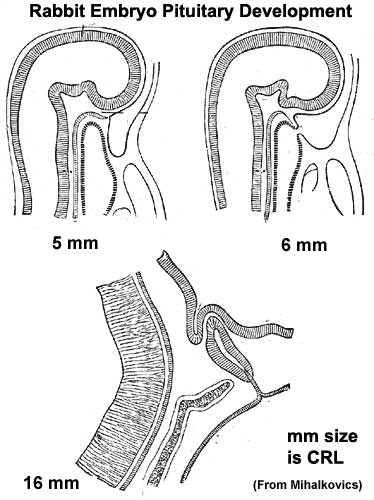
|

|
Blue - neural tube ectoderm
Red - surface ectoderm |
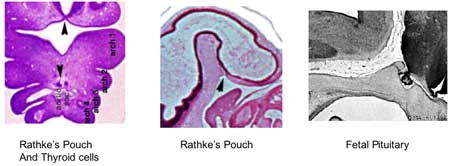
- Dual ectoderm origins
- Ectoderm - ectoderm roof of stomodeum, Rathke's pouch, adenohypophysis
- Neuroectoderm - prosenecephalon then diencephalon, neurohypophysis
Adenohypophysis
- Anterior wall proliferates - pars distalis
- Posterior wall little growth – pars intermedia
- Rostral growth around infundibular stem – pars tuberalis
Neurohypophysis
- Infundibulum – median eminence, infundibulum, pars nervosa
Pituitary Timeline
- Week 4 - hypophysial pouch, Rathke’s pouch, diverticulum from roof
- Week 5 - elongation, contacts infundibulum, diverticulum of diencephalon
- Week 6 - connecting stalk between pouch and oral cavity degenerates
- Week 8 - basophilic staining cells appear
- Week 9 - acidophilic staining cells appear
- Week 10 - growth hormone and ACTH detectable
- Week 16 - adenohypophysis fully differentiated and TSH increases to peak at 22 weeks
- Week 20 to 24 - growth hormone levels peak, then decline
- Birth - second TSH surge and decreases postnatally
- Links: pituitary | Embryo Images - Pituitary | Endocrinology
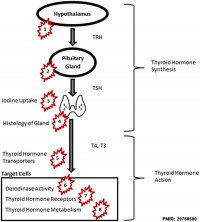
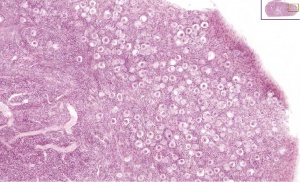
Thyroid
- Maternal thyroid hormone - required for early stages of brain development
- Fetal thyroid - begins function from week10, (GA week 12) required for neural development, stimulates metabolism (protein, carbohydrate, lipid), reduced/absence = cretinism (see abnormalities)
Hormones - TH (amino acid derivatives) Thyroxine (T4), Triiodothyronine (T3)
Thyroid Development
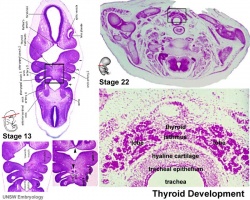
|
|
|
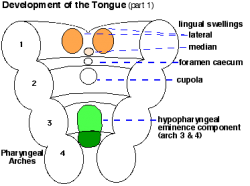
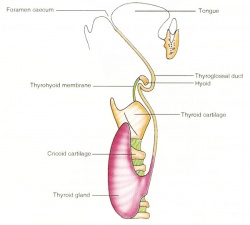
- thyroid median endodermal thickening in the floor of pharynx, outpouch – thyroid diverticulum.
- tongue grows, cells descend in neck.
- thyroglossal duct - proximal end at the foramen caecum of tongue.
- thyroid diverticulum - hollow then solid, right and left lobes, central isthmus.
Thyroid Timeline
- 24 days - thyroid median endodermal thickening in the floor of pharynx, outpouch – thyroid diverticulum
- Week 11 - colloid appearance in thyroid follicles, iodine and thyroid hormone (TH) synthesis. Growth factors (insulin-like, epidermal) stimulates follicular growth.
- Week 16 - 18 - (GA 18-20 weeks) fully functional
Fetal Thyroid Hormone
- Initial secreted biologically inactivated by modification
- serum thyroid hormone levels are relatively low and tissue concentration of thyroid hormone is modified by iodothyronine deiodinases
- Iodine deficiency - during this period, leads to neurological defects (cretinism)
- Late fetal secretion - develops brown fat
- Birth - TSH levels increase, thyroxine (T3) and T4 levels increase to 24 h, then 5-7 days postnatal decline to normal levels
- Post-natal - TH required for bone development
Maternal TH - iodine/thyroid status can affect development.
- recent studies show that both high and low maternal thyroid hormone impact on neural development (PMID 26497402)
- Links: thyroid | Endocrinology |
Parathyroid
Principal cells cords of cells |
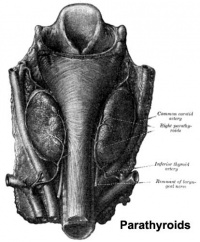
Adult Parathyroid |
Parathyroid Development
|
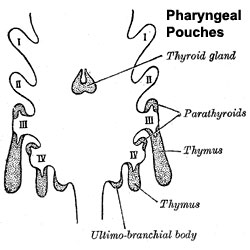
Pharyngeal pouches |
- Links: parathyroid | Endocrinology
Thymus
- Thymus - bone-marrow lymphocyte precursors become thymocytes, and subsequently mature into T lymphocytes (T cells)
- Thymus hormones - thymosins stimulate the development and differentiation of T lymphocytes
Thymus Development
|

Fetal Thymus |
- Links: thymus
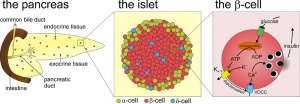
Pancreas
|
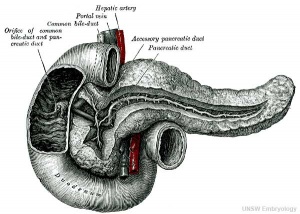
|
Pancreas Development
- Pancreatic buds - duodenal level endoderm, splanchnic mesoderm forms dorsal and ventral mesentery, dorsal bud (larger, first), ventral bud (smaller, later)
- Pancreas Endoderm - pancreas may be opposite of liver
- Heart cells promote/notochord prevents liver formation
- Notochord may promote pancreas formation
- Heart may block pancreas formation
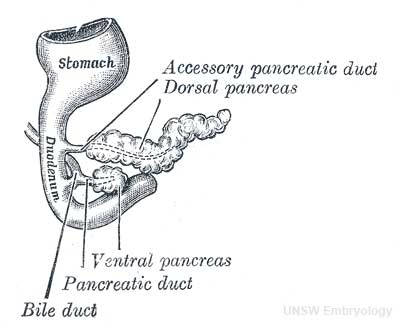
|
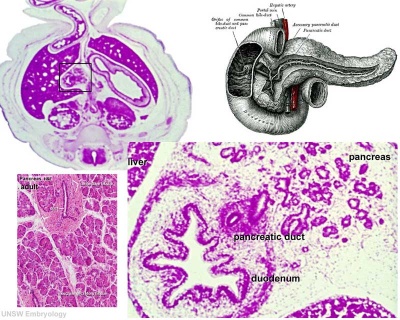
|
| Pancreatic buds and duct developing | Pancreas week 8 (Stage 22) |
- Duodenum growth/rotation - brings ventral and dorsal buds together, fusion of buds See Figure 2.32
- Pancreatic duct - ventral bud duct and distal part of dorsal bud, exocrine function
- Islet cells - cords of endodermal cells form ducts, from which cells bud off to form islets
| Islet |
|---|
| Arterial blood (from splenic, hepatic and superior mesenteric arteries) enters the core of each islet and capillaries then drain outwardly to the periphery where venous blood drains into the splenic and superior mesenteric veins.
Histology image shows blood vessels injected with ink. |
Pancreatic Islets
- Islets of Langerhans - 4 endocrine cell types
- Alpha - glucagon, mobilizes lipid
- Beta - insulin, increase glucose uptake
- Beta cells, stimulate fetal growth, continue to proliferate to postnatal, in infancy most abundant
- Delta - somatostatin, inhibits glucagon, insulin secretion
- F-cells - pancreatic polypeptide
Pancreas Timeline
- Week 7 to 20 - pancreatic hormones secretion increases, small amount maternal insulin
- Week 10 - glucagon (alpha) differentiate first, somatostatin (delta), insulin (beta) cells differentiate, insulin secretion begins
- Week 15 - glucagon detectable in fetal plasma
- Links: [[Pancreas}} | Gastrointestinal Tract - Pancreas Development | Endocrinology
Adrenal
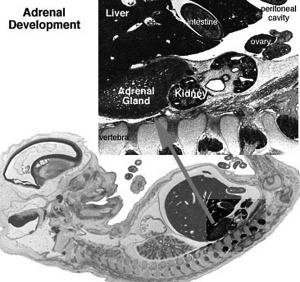
- Richly vascularized - arterioles passing through cortex, capillaries from cortex to medulla, portal-like circulation
- Fetal Cortex - produces a steroid precursor (DHEAS), converted by liver and then placenta into estrogen
- Adult Medulla - produces adrenalin (epinephrine), noradrenaline (norepinephrine)
- Fetal adrenal hormones - influence lung maturation
Adrenal cortical hormones - (steroids) Cortisol, Aldosterone, Dehydroepiandrosterone
- zona glomerulosa - regulated by renin-angiotensin-aldosterone system controlled by the juxtaglomerular apparatus of the kidney.
- zona fasciculata - regulated by hypothalamo-pituitary axis with the release of CRH and ACTH respectively.
Adrenal medullary hormones - (amino acid derivatives) epinephrine, norepinephrine
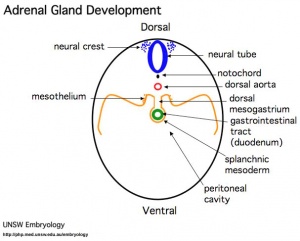
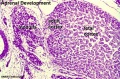
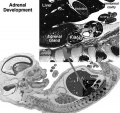
Adrenal Development
- Week 6 - fetal cortex, from mesothelium adjacent to dorsal mesentery; Medulla, neural crest cells from adjacent sympathetic ganglia
- Fetal Adrenals - fetal cortex later replaced by adult cortex
- Adult cortex - mesothelium mesenchyme encloses fetal cortex
Adrenal Cortex
- Late Fetal Period - differentiates to form cortical zones
- Birth - zona glomerulosa, zona fasiculata present
- Year 3 - zona reticularis present
|
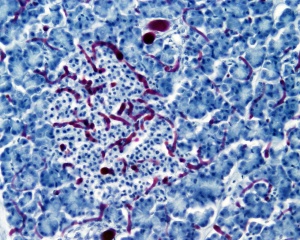
Adrenal Medulla
|
|
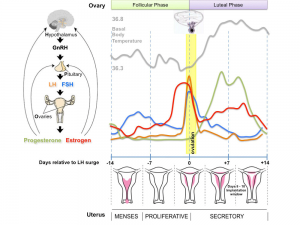
Gonad
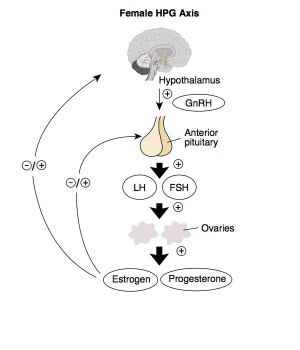
Adult Hypothalamus - Pituitary - Gonad (female) |
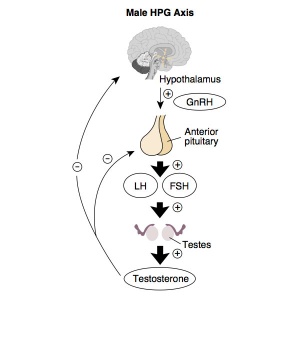
Adult Hypothalamus - Pituitary - Gonad (male) |
HPG Axis - Endocrinology - Simplified diagram of the actions of gonadotrophins
Gonad Development
- mesoderm - mesothelium and underlying mesenchyme
- Gonadal ridge - mesothelium thickening, medial mesonephros
- Primordial Germ cells - yolk sac, to mesentery of hindgut, to genital ridge of developing kidney
Differentiation
- testis-determining factor (TDF) from Y chromosome: presence (testes), absence (ovaries)
Testis
- 8 Weeks - mesenchyme, interstitial cells (of Leydig) secrete testosterone, androstenedione.
- 8 to 12 Weeks - hCG stimulates testosterone production.
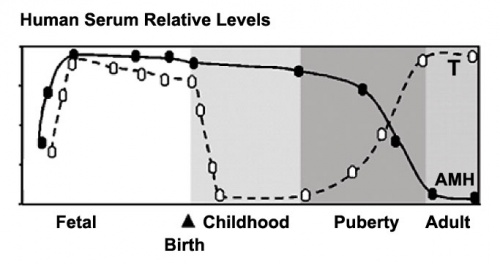
- Sustentacular (Sertoli) cells - produce anti-mullerian hormone (AMH) to puberty.
- AMH - anti-Müllerian hormone (Müllerian inhibiting factor (MIF), Müllerian-inhibiting hormone (MIH), and Müllerian-inhibiting substance (MIS)).
Ovary
- X chromosome genes regulate ovary development
- Hormone levels increase at puberty with follicle development.
I will cover this topic in detail again in sexual differentiation lecture/practical.
Placenta
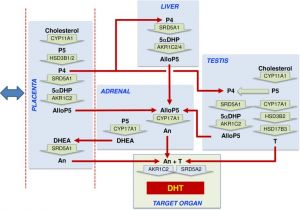
The corpus luteum provides initial support, when it degenerates in the embryonic period, placental estrogen and progesterone production increases exponentially to term.
Placenta - Maternal (decidua) and Fetal (trophoblastic cells, extraembryonic mesoderm) components
|
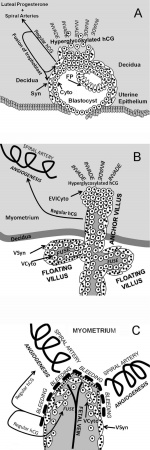
Trophoblast hCG function |
Other Endocrine
Endocrine Heart
- Atrial natriuretic peptide (ANP) - Increase Filtration rate / decrease Na+ reabsorption
- Endothelins - ET-1, ET-2, ET-3, Vasoconstriction / Increase NO
- Nitric oxide (NO) - Vasodilatation
Endocrine Kidney
- Renin - Increase Angiotensin-aldosterone system
- Prostaglandins - decrease Na+ reabsorption
- Erythropoietin - Increase Erythrocyte (rbc) production
- 1,25 (OH)2 vitamin D - calcium homeostasis
- Prekallikreins - Increase Kinin production
GIT Endocrine
Enteric control of digestive function
- Gastrin - Secreted from stomach (G cells), role in control of gastric acid secretion
- Cholecystokinin - small intestine hormone, stimulates secretion of pancreatic enzymes and bile
- Secretin - small intestine hormone (epithelial cells), stimulates secretion of bicarbonate-rich fluids from pancreas and liver
Adipose Tissue
- Leptin - polypeptide hormone produced in adipose and many other tissues with also many different roles
- Adiponectin - regulation of energy homeostasis and glucose and lipid metabolism, as well as acting as an anti-inflammatory on the cellular vascular wall
- Resistin - (for resistance to insulin, RETN) a 108 amino acid polypeptide and the related resistin-like protein-beta (Resistin-like molecule-beta, RELMbeta) stimulate endogenous glucose production
- Links: Endocrine - Other Tissues
Endocrine Functional Changes
- Puberty - Increased activity.
- Menopause - Decreased activity.
- Disease - (diabetes, thyroid, kidney) suggested trends that genetics, health, nutrition, lifestyle may influence time that these events occur.
- Pharmaceutical impact - birth control, steroids, Hormone Replacement Therapy (HRT).
Abnormalities
NIH Genes & Disease Glands and Hormones
Pineal
- hypoplasia - associated with retinal disease.
- tumours - in children are associated with abnormal puberty development.
Pituitary
- craniopharyngeal canal - Rathke's pouch abnormality, from the anterior part of the fossa hypophyseos of the sphenoid bone to the under surface of the skull.
- pituitary tumours (adenomas) - several abnormalities associated with abnormal levels of the hormonal output of the pituitary.
- Growth hormone (GH) adenomas - benign pituitary tumors lead to chronic high GH output levels, that may lead to acromegaly.
- Cushing's disease - caused either by a pituitary adenoma produces excess adrenocorticotropic hormone (ACTH, corticotropin) or due to ectopic tumors secreting ACTH or corticotropin-releasing hormone (CRH).
Thyroid
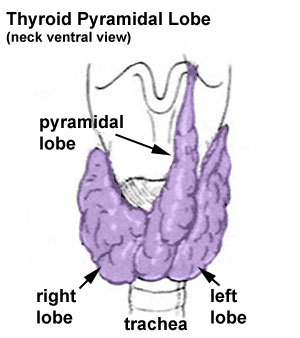
- Pyramidal lobe - from isthmus (50% of people) attached to hyoid bone distal end of thryoglossal duct.
- Congenital hypothyroidism - approximately 1 in 3000 births, associated with neurological abnormalities.
- Lingual thyroid gland - failure of thyroid descent.
- Thyroglossal cyst - persistance of thyroglossal duct. Image - thyroglossal duct
- Thyroglossal fistula - partial degeneration of the thyroglossal duct.
- Abnormal development of the thyroid - incomplete or excessive descent.
- Childhood hypothyroidism delays ossification and bone mineralization.
Iodine Deficiency
- A teaspoon of iodine, total lifetime requirement, cannot be stored for long periods by our body, tiny amounts are needed regularly
- Areas of endemic iodine deficiency, where soil and therefore crops and grazing animals do not provide sufficient dietary iodine to the populace
- food fortification and supplementation - Iodized salt programs and iodized oil supplements are the most common tools in fight against IDD
Parathyroid
- Usually four glands are present (2 on each side), but three to six glands have been found in human.
- Can have displaced parathyroid development with thymus.
- Lower parathyroid glands arise from the third pharyngeal pouch and descend with the thymus. Variable descent can lead to a range of adult locations, from just beneath the mandible to the anterior mediastinum.
Pancreas
- Type 1 Diabetes - juvenile onset diabetes, more severe form of illness, increases risk of blindness, heart disease, kidney failure, neurological disease, T-lymphocyte-dependent autoimmune disease, infiltration and destruction of the islets of Langerhans, Approx 16 million Americans
- Type 2 Diabetes - loosely defined as "adult onset" diabetes, becoming more common cases of type 2 diabetes seen in younger people
- Risk of developing diabetes - environmental factors (food intake and exercise play an important role, either overweight or obese), Inherited factors (genes involved remain poorly defined)
Adrenal
- Congenital Adrenal Hyperplasia (CAH) - family of inherited disorders of adrenal steroidogenesis enzymes which impairs cortisol production by the adrenal cortex. Androgen excess leads newborn females with external genital ambiguity and postnatal progressive virilization in both sexes.
- Enzymes most commonly affected: 21-hydroxylase (21-OH), 11beta-hydroxylase, 3beta-hydroxysteroid dehydrogenase.
- Enzymes less commonly affected: 17alpha-hydroxylase/17,20-lyase and cholesterol desmolase.
- Pheochromocytomas (PCC) - Catecholamine-producing (neuro)endocrine tumor located in the adrenal medulla. Similar catecholamine-producing tumors outside the adrenal gland are called paragangliomas (PGL).
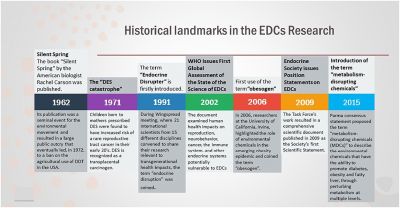
Endocrine Disruptors
Exogenous chemicals that interfere with the function of hormones. There are 3 main mechanisms: mimic, block or interfere.
Mimic - effects of natural hormones by binding receptors
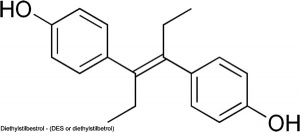
- Diethylstilbestrol - (DES or diethylstilbetrol) a drug prescribed to women from 1938-1971 to prevent miscarriage in high-risk pregnancies. Acts as a potent estrogen (mimics natural hormone) and therefore a potential endocrine disruptor. Female fetus, increased risk abnormal reproductive tract and cancer. Male fetus, abnormal genitalia. Banned by USA FDA in 1979 as a teratogen, previously used as livestock growth promoter.
Block - binding of a hormone to receptor or hormone synthesis
- Finasteride - chemical used to prevent male pattern baldness and enlargement of prostate glands. An anti-androgen (blocks synthesis of dihydrotestosterone) and therefore a potential endocrine disruptor, exposed pregnant women can impact on male fetus genetial development.
- Vinclozolin - a dicarboximide fungicide, perinatal exposure in rats inhibits morphological sex differentiation. In adult rats, shown to cause gonad tumours (Leydig cell) and atrophy. Chemical has androgen-antagonist (antiandrogenic) activity, metabolies compete with natural androgen
Interfere - with hormone transport or elimination
- Polychlorinated biphenyl pollutants - (PCBs) Rats exposed to PCBs have low levels of thyroid hormone. Compete for binding sites of thyroid hormone transport protein. Without being bound to this protein, thyroid hormones are excreted from the body (McKinney et al. 1985; Morse et al. 1996)
- Links: Endocrine Disruptors
References
- ↑ Griekspoor A, Zwart W, Neefjes J & Michalides R. (2007). Visualizing the action of steroid hormone receptors in living cells. Nucl Recept Signal , 5, e003. PMID: 17464358 DOI.
- ↑ Møller M, Phansuwan-Pujito P & Badiu C. (2014). Neuropeptide Y in the adult and fetal human pineal gland. Biomed Res Int , 2014, 868567. PMID: 24757681 DOI.
- ↑ O'Shaughnessy PJ, Antignac JP, Le Bizec B, Morvan ML, Svechnikov K, Söder O, Savchuk I, Monteiro A, Soffientini U, Johnston ZC, Bellingham M, Hough D, Walker N, Filis P & Fowler PA. (2019). Alternative (backdoor) androgen production and masculinization in the human fetus. PLoS Biol. , 17, e3000002. PMID: 30763313 DOI.
- ↑ Raeside JI. (2017). A Brief Account of the Discovery of the Fetal/Placental Unit for Estrogen Production in Equine and Human Pregnancies: Relation to Human Medicine. Yale J Biol Med , 90, 449-461. PMID: 28955183
- ↑ Albrecht ED & Pepe GJ. (2010). Estrogen regulation of placental angiogenesis and fetal ovarian development during primate pregnancy. Int. J. Dev. Biol. , 54, 397-408. PMID: 19876841 DOI.
- Endocrinology: An Integrated Approach Nussey, S.S. and Whitehead, S.A. London:Taylor & Francis; c2001 Major hormone types
- Genes and Disease, Bethesda (MD): National Library of Medicine (US), NCBI Glands and Hormones
Search
- Bookshelf endocrine | pineal gland | hypothalamus | pituitary gland | thyroid gland | parathyroid gland | thymus gland | endocrine pancreas | adrenal gland
- Pubmed endocrine development
Embryonic
Terms
| Endocrine Terms (expand to view) |
|---|
|
| Other Terms Lists |
|---|
| Terms Lists: ART | Birth | Bone | Cardiovascular | Cell Division | Endocrine | Gastrointestinal | Genital | Genetic | Head | Hearing | Heart | Immune | Integumentary | Neonatal | Neural | Oocyte | Palate | Placenta | Radiation | Renal | Respiratory | Spermatozoa | Statistics | Tooth | Ultrasound | Vision | Historic | Drugs | Glossary |
Endocrine Development Interactive Component
| Attempt the Quiz - Endocrine Development | |
|---|---|
Here are a few simple Quiz questions that relate to Endocrine development and abnormalities from the lecture. Some questions may require some additional research.
|
BGDB: Lecture - Gastrointestinal System | Practical - Gastrointestinal System | Lecture - Face and Ear | Practical - Face and Ear | Lecture - Endocrine | Lecture - Sexual Differentiation | Practical - Sexual Differentiation | Tutorial
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 16) Embryology BGD Lecture - Endocrine Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/BGD_Lecture_-_Endocrine_Development
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G