Zebrafish Development: Difference between revisions
| Line 21: | Line 21: | ||
* '''The zebrafish transcriptome during early development'''<ref><pubmed>21609443</pubmed></ref> "The three earliest developmental stages were similar when comparing highly expressed genes, whereas the 50% epiboly stage differed from the other three stages in the identity of highly expressed genes, number of uniquely expressed genes and enrichment of GO molecular functions. Taken together, these observations indicate a major transition in gene regulation and transcriptional activity taking place between the 512-cell and 50% epiboly stages, in accordance with previous studies." | * '''The zebrafish transcriptome during early development'''<ref><pubmed>21609443</pubmed></ref> "The three earliest developmental stages were similar when comparing highly expressed genes, whereas the 50% epiboly stage differed from the other three stages in the identity of highly expressed genes, number of uniquely expressed genes and enrichment of GO molecular functions. Taken together, these observations indicate a major transition in gene regulation and transcriptional activity taking place between the 512-cell and 50% epiboly stages, in accordance with previous studies." | ||
* '''Genetic analysis of fin development in zebrafish identifies furin and hemicentin1 as potential novel fraser syndrome disease genes'''<ref><pubmed>20419147</pubmed></ref> " Three of them are due to mutations in zebrafish orthologues of FRAS1, FREM1, or FREM2, large basement membrane protein encoding genes that are mutated in mouse bleb mutants and in human patients suffering from Fraser Syndrome, a rare congenital condition characterized by syndactyly and cryptophthalmos. Fin blistering in a fourth group of zebrafish mutants is caused by mutations in Hemicentin1 (Hmcn1), another large extracellular matrix protein the function of which in vertebrates was hitherto unknown. Our mutant and dose-dependent interaction data suggest a potential involvement of Hmcn1 in Fraser complex-dependent basement membrane anchorage. Furthermore, we present biochemical and genetic data suggesting a role for the proprotein convertase FurinA in zebrafish fin development and cell surface shedding of Fras1 and Frem2, thereby allowing proper localization of the proteins within the basement membrane of forming fins." | * '''Genetic analysis of fin development in zebrafish identifies furin and hemicentin1 as potential novel fraser syndrome disease genes'''<ref><pubmed>20419147</pubmed></ref> " Three of them are due to mutations in zebrafish orthologues of FRAS1, FREM1, or FREM2, large basement membrane protein encoding genes that are mutated in mouse bleb mutants and in human patients suffering from Fraser Syndrome, a rare congenital condition characterized by syndactyly and cryptophthalmos. Fin blistering in a fourth group of zebrafish mutants is caused by mutations in Hemicentin1 (Hmcn1), another large extracellular matrix protein the function of which in vertebrates was hitherto unknown. Our mutant and dose-dependent interaction data suggest a potential involvement of Hmcn1 in Fraser complex-dependent basement membrane anchorage. Furthermore, we present biochemical and genetic data suggesting a role for the proprotein convertase FurinA in zebrafish fin development and cell surface shedding of Fras1 and Frem2, thereby allowing proper localization of the proteins within the basement membrane of forming fins." | ||
|} | |} | ||
[[Talk:Zebrafish Development|Recent References]] | [[#References|References]] | |||
==Timeline and Stages of Embryonic Development== | ==Timeline and Stages of Embryonic Development== | ||
Revision as of 09:03, 2 January 2013
Introduction
Zebrafish or zebra danio (danio rerio) are seen as the latest "model' for embryological development studies. These embryos have the great advantage that they develop as "see through" embryos, that is, all internal development can be clearly observed from the outside in the living embryo. Much of the early modern work using this embryo model began with the papers of Kimmel.[1][2]
Several large laboratories in the US are now developing large breeding programs to carry out "knockouts" and to find spontaneous mutants of interest.
| Fish Links: Zebrafish Development | Medaka Development | Salmon Development | Movie - Zebrafish Heart | Student Group Project - Zebrafish | Recent References | Category:Zebrafish | Category:Medaka |
| Animal Development: axolotl | bat | cat | chicken | cow | dog | dolphin | echidna | fly | frog | goat | grasshopper | guinea pig | hamster | horse | kangaroo | koala | lizard | medaka | mouse | opossum | pig | platypus | rabbit | rat | salamander | sea squirt | sea urchin | sheep | worm | zebrafish | life cycles | development timetable | development models | K12 |
Some Recent Findings
|
Recent References | References
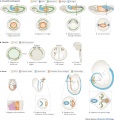
Timeline and Stages of Embryonic Development
| Duration | Period Name | Image |
| 0 - 0.75 hrs | Zygote Period | 
|
| 0.75 - 2.25 hrs | Cleavage Period | 
|
| 2.25 - 5.25 hrs | Blastula Period | 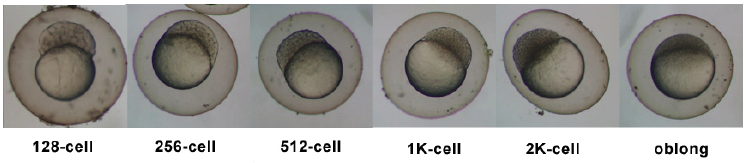
|
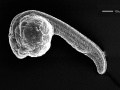
| 5.25 - 10.33 hrs | Gastrula Period | 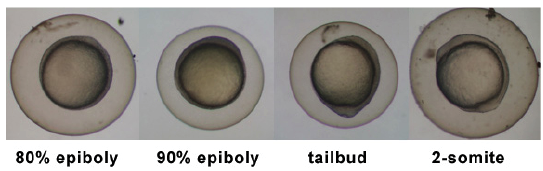
|
| 10.33 - 24 hrs | Segmentation Period | 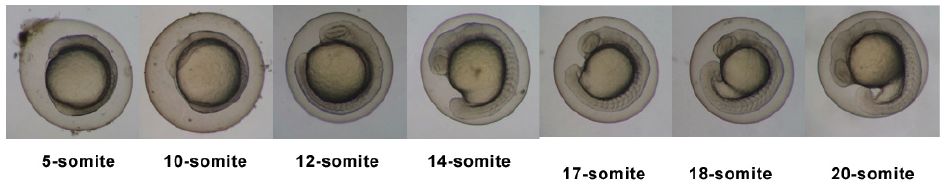
|
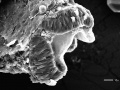
| 24 - 48 hrs | Pharyngula Period | 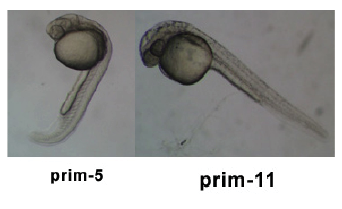
|
| 48-72 hrs | Hatching Period | 
|
| 72 hrs - 30 Days | Larval Period | 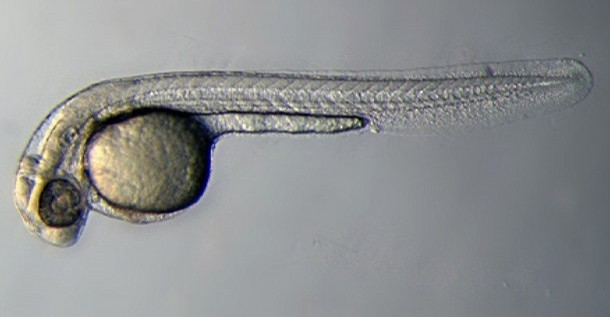
|
Skull

|
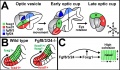
Zebrafish Skull Neural Crest Contribution[6]
|
Molecular
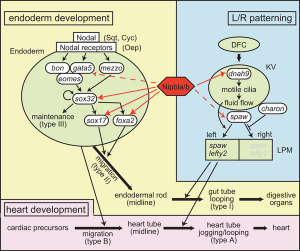
Fibroblast Growth Factor
- Fgf8 and Fgf3 - regulating the segmentation of the pharyngeal endoderm into pouches. [7]
- Fgf24 and Fgf8 - promotes posterior mesodermal development.[8]
- Sox9 - required for cartilage morphogenesis.[9]
References
- ↑ <pubmed>7229136</pubmed>
- ↑ <pubmed>3077108</pubmed>
- ↑ 3.0 3.1 <pubmed>22039349</pubmed>
- ↑ <pubmed>21609443</pubmed>
- ↑ <pubmed>20419147</pubmed>
- ↑ Kague E, Gallagher M, Burke S, Parsons M, Franz-Odendaal T, et al. (2012) Skeletogenic Fate of Zebrafish Cranial and Trunk Neural Crest. PLoS ONE 7(11): e47394. doi:10.1371/journal.pone.0047394 PLoS ONE
- ↑ <pubmed>15509770</pubmed>
- ↑ <pubmed>12925590</pubmed>
- ↑ <pubmed>12397114</pubmed>
Journals
Zebrafish "is the only peer-reviewed journal to focus on the zebrafish, which has numerous valuable features as a model organism for the study of vertebrate development. Due to its prolific reproduction and the external development of the transparent embryo, the zebrafish is a prime model for genetic and developmental studies, as well as research in toxicology and genomics. While genetically more distant from humans, the vertebrate zebrafish nevertheless has comparable organs and tissues, such as heart, kidney, pancreas, bones, and cartilage." [jour PubMed listing]
Reviews
<pubmed>21501748</pubmed> <pubmed>20674361</pubmed> <pubmed>19557689</pubmed> <pubmed>19371733</pubmed> <pubmed>18992377</pubmed>
Articles
Search Pubmed
Search Pubmed: Zebrafish Development
Additional Images
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- NIH NIH Zebrafish Initiative
- ZFIN - The Zebrafish Model Organism Database
- Keller at European Molecular Biology Laboratory, Germany Movies - Reconstruction of zebrafish early embryonic development by scanned light sheet microscopy
| Animal Development: axolotl | bat | cat | chicken | cow | dog | dolphin | echidna | fly | frog | goat | grasshopper | guinea pig | hamster | horse | kangaroo | koala | lizard | medaka | mouse | opossum | pig | platypus | rabbit | rat | salamander | sea squirt | sea urchin | sheep | worm | zebrafish | life cycles | development timetable | development models | K12 |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, May 1) Embryology Zebrafish Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Zebrafish_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G