Neural - Meninges Development: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 51: | Line 51: | ||
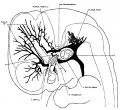
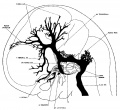
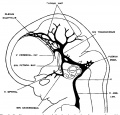
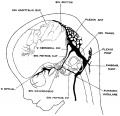
The following figures are from a 1915 study of the venous sinuses of the dura mater in the human embryo.<ref name=Streeter1915>{{Ref-Streeter1915}}</ref> | |||
<gallery> | |||
File:Streeter1915 fig01.jpg|fig 1 embryo 4 mm [[:Category:Carnegie Embryo 588|No. 588]] | |||
File:Streeter1915 fig02.jpg|fig 2 embryo 13.8 mm [[:Category:Carnegie Embryo 940|No. 940]] | |||
File:Streeter1915 fig03.jpg|fig 3 embryo 18 mm [[:Category:Carnegie Embryo 144|No. 144]] | |||
File:Streeter1915 fig04.jpg|fig 4 embryo 21 mm [[:Category:Carnegie Embryo 460|No. 460]] | |||
File:Streeter1915 fig05.jpg|fig 5 embryo 24 mm [[:Category:Carnegie Embryo 632|No. 632]] | |||
File:Streeter1915 fig06.jpg|fig 6 embryo 50 mm [[:Category:Carnegie Embryo 96|No. 96]] | |||
File:Streeter1915 fig07.jpg|fig 7 embryo 13.8 mm [[:Category:Carnegie Embryo 940|No. 940]] | |||
File:Streeter1915 fig08.jpg|fig 8 embryo 20 mm [[:Category:Carnegie Embryo 349|No. 349]] | |||
File:Streeter1915 fig09.jpg|fig 9 fetus 54 mm long No. 458 | |||
</gallery> | |||
==Recent References== | ==Recent References== | ||
Revision as of 11:08, 26 November 2016
| Embryology - 27 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
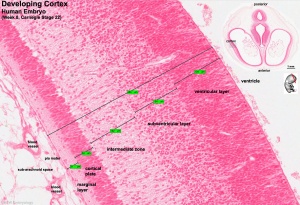
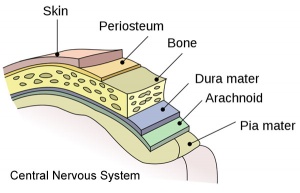
The meninges (singular meninx; Greek, meninx = membrane) are a complex connective tissue surrounding the central nervous system (brain and spinal cord). The 3 layers from the central nervous outward are: pia mater, arachnoid mater, and the dura mater. All three layers form from the meninx primitiva, a meningeal mesenchyme. There have been many theories to the embryonic origins of the three layers that form the meninges, as well as potential differences between species. The safest term would be mesenchymal in origin, but the actual source of this mesenchyme may also differ in the same species at different levels of the central nervous system. The space under the arachnoid layer (subarachnoid space) is filled with cerebrospinal fluid.
Recent studies also suggest that rather than acting as a passive connective tissue "neural container" during development, the meninges may also interact and regulate cranial skull and neural development.
- Draft Page (this notice removed when completed)
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Meninges Development <pubmed limit=5>Meninges Development</pubmed> |
Pia Mater
- A fine connective tissue covering of the central nervous system, forms innermost part of the meningial layers. Lies beneath the arachnoid mater and then tough outer dura mater layer. All three layers form from the meninx primitiva, a meningeal mesenchyme that is mesodermal and neural crest in origin. The space overlying the pia mater (subarachnoid space) is filled with cerebrospinal fluid. The pia mater has close contact with the spinal cord and brain, in the brain it follows down into the sulci and fissures of the cortex. This layer also fuses with the membranous lining of the ventricles (ependyma) forming the choroid plexus.
Arachnoid Mater
- (Greek, arachne = spider + -oeides = form) A meshwork (spider web-like) connective tissue covering of the central nervous system, forms part of the meningial layers. Lies between tough outer dura mater layer and the inner fine pia mater layer. All three layers form from the meninx primitiva, a meningeal mesenchyme that is mesodermal and neural crest in origin. The space underlying the arachnoid mater (subarachnoid space) is filled with cerebrospinal fluid.
Dura Mater
- (Latin, dura mater = hard mother) The outer tough connective tissue meningial coat of the 3 layers that cover the central nervous system of 3 layers (overlays the arachnoid mater middle layer and pia mater inner layer). All three layers form from the meninx primitiva, a meningeal mesenchyme that is mesodermal and neural crest in origin. At the level of the skull, the outer dura layer forms the inner periosteum of the skull and the inner dura layer forms the dural folds (falx and tentorium) that contains the dural sinuses. At the level of the spinal cord, the dura is separated from the periosteum of the vertebral canal by an epidural space.
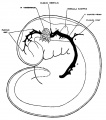
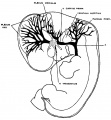
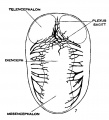
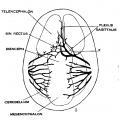
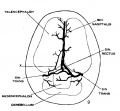
The following figures are from a 1915 study of the venous sinuses of the dura mater in the human embryo.[2]
fig 1 embryo 4 mm No. 588
fig 2 embryo 13.8 mm No. 940
fig 3 embryo 18 mm No. 144
fig 4 embryo 21 mm No. 460
fig 5 embryo 24 mm No. 632
fig 6 embryo 50 mm No. 96
fig 7 embryo 13.8 mm No. 940
fig 8 embryo 20 mm No. 349
Recent References
CoupTFI Interacts with Retinoic Acid Signaling during Cortical Development
PLoS One. 2013;8(3):e58219. doi: 10.1371/journal.pone.0058219. Epub 2013 Mar 5.
Harrison-Uy SJ, Siegenthaler JA, Faedo A, Rubenstein JL, Pleasure SJ. Source Department of Neurology, University of California San Francisco, San Francisco, California, United States of America.
Abstract
We examined the role of the orphan nuclear hormone receptor CoupTFI in mediating cortical development downstream of meningeal retinoic acid signaling. CoupTFI is a regulator of cortical development known to collaborate with retinoic acid (RA) signaling in other systems. To examine the interaction of CoupTFI and cortical RA signaling we utilized Foxc1-mutant mice in which defects in meningeal development lead to alterations in cortical development due to a reduction of RA signaling. By analyzing CoupTFI(-/-);Foxc1(H/L) double mutant mice we provide evidence that CoupTFI is required for RA rescue of the ventricular zone and the neurogenic phenotypes in Foxc1-mutants. We also found that overexpression of CoupTFI in Foxc1-mutants is sufficient to rescue the Foxc1-mutant cortical phenotype in part. These results suggest that CoupTFI collaborates with RA signaling to regulate both cortical ventricular zone progenitor cell behavior and cortical neurogenesis.
PMID 23472160
The cranial dura mater: a review of its history, embryology, and anatomy
Childs Nerv Syst. 2012 Jun;28(6):827-37. doi: 10.1007/s00381-012-1744-6. Epub 2012 Apr 15.
Adeeb N, Mortazavi MM, Tubbs RS, Cohen-Gadol AA. Abstract INTRODUCTION: The dura mater is important to the clinician as a barrier to the internal environment of the brain, and surgically, its anatomy should be well known to the neurosurgeon and clinician who interpret imaging. METHODS: The medical literature was reviewed in regard to the morphology and embryology of specifically, the intracranial dura mater. A historic review of this meningeal layer is also provided. CONCLUSIONS: Knowledge of the cranial dura mater has a rich history. The embryology is complex, and the surgical anatomy of this layer and its specializations are important to the neurosurgeon.
PMID 22526439
A cascade of morphogenic signaling initiated by the meninges controls corpus callosum formation
Neuron. 2012 Feb 23;73(4):698-712. doi: 10.1016/j.neuron.2011.11.036.
Choe Y, Siegenthaler JA, Pleasure SJ. Source Department of Neurology, University of California, San Francisco, San Francisco, CA 94158, USA.
Abstract
The corpus callosum is the most prominent commissural connection between the cortical hemispheres, and numerous neurodevelopmental disorders are associated with callosal agenesis. By using mice either with meningeal overgrowth or selective loss of meninges, we have identified a cascade of morphogenic signals initiated by the meninges that regulates corpus callosum development. The meninges produce BMP7, an inhibitor of callosal axon outgrowth. This activity is overcome by the induction of expression of Wnt3 by the callosal pathfinding neurons, which antagonize the inhibitory effects of BMP7. Wnt3 expression in the cingulate callosal pathfinding axons is developmentally regulated by another BMP family member, GDF5, which is produced by the adjacent Cajal-Retzius neurons and turns on before outgrowth of the callosal axons. The effects of GDF5 are in turn under the control of a soluble GDF5 inhibitor, Dan, made by the meninges. Thus, the meninges and medial neocortex use a cascade of signals to regulate corpus callosum development. Copyright © 2012 Elsevier Inc. All rights reserved.
PMID 22365545
We have got you 'covered': how the meninges control brain development.
Curr Opin Genet Dev. 2011 Jun;21(3):249-55. doi: 10.1016/j.gde.2010.12.005. Epub 2011 Jan 20.
Siegenthaler JA, Pleasure SJ. Source Department of Neurology, Programs in Neuroscience and Developmental Biology, Institute for Regenerative Medicine, University of California, San Francisco, San Francisco, CA 94158, United States.
Abstract
The meninges have traditionally been viewed as specialized membranes surrounding and protecting the adult brain from injury. However, there is increasing evidence that the fetal meninges play important roles during brain development. Through the release of diffusible factors, the meninges influence the proliferative and migratory behaviors of neural progenitors and neurons in the forebrain and hindbrain. Meningeal cells also secrete and organize the pial basement membrane (BM), a critical anchor point for the radially oriented fibers of neuroepithelial stem cells. With its emerging role in brain development, the potential that defects in meningeal development may underlie certain congenital brain abnormalities in humans should be considered. In this review, we will discuss what is known about assembly of the fetal meninges and review the role of meningeal-derived proteins in mouse and human brain development. Copyright © 2011 Elsevier Ltd. All rights reserved.
PMID 21251809
Tissue origins and interactions in the mammalian skull vault
Dev Biol. 2002 Jan 1;241(1):106-16. Jiang X, Iseki S, Maxson RE, Sucov HM, Morriss-Kay GM. Source Institute for Genetic Medicine, University of Southern California Keck School of Medicine, Los Angeles, California 90033, USA.
Abstract
During mammalian evolution, expansion of the cerebral hemispheres was accompanied by expansion of the frontal and parietal bones of the skull vault and deployment of the coronal (fronto-parietal) and sagittal (parietal-parietal) sutures as major growth centres. Using a transgenic mouse with a permanent neural crest cell lineage marker, Wnt1-Cre/R26R, we show that both sutures are formed at a neural crest-mesoderm interface: the frontal bones are neural crest-derived and the parietal bones mesodermal, with a tongue of neural crest between the two parietal bones. By detailed analysis of neural crest migration pathways using X-gal staining, and mesodermal tracing by DiI labelling, we show that the neural crest-mesodermal tissue juxtaposition that later forms the coronal suture is established at E9.5 as the caudal boundary of the frontonasal mesenchyme. As the cerebral hemispheres expand, they extend caudally, passing beneath the neural crest-mesodermal interface within the dermis, carrying with them a layer of neural crest cells that forms their meningeal covering. Exposure of embryos to retinoic acid at E10.0 reduces this meningeal neural crest and inhibits parietal ossification, suggesting that intramembranous ossification of this mesodermal bone requires interaction with neural crest-derived meninges, whereas ossification of the neural crest-derived frontal bone is autonomous. These observations provide new perspectives on skull evolution and on human genetic abnormalities of skull growth and ossification.
PMID 11784098
The meninges in human development
J Neuropathol Exp Neurol. 1986 Sep;45(5):588-608.
O'Rahilly R, Müller F.
Abstract
The brain and cranial meninges were studied in 61 serially sectioned embryos of stages 8-23. Much earlier stages than those examined by previous authors provided a more comprehensive view of meningeal development. As a result, the possible and probable sources of the cranial and spinal meninges are believed to be: (a) prechordal plate, (b) unsegmented paraxial (parachordal) mesoderm, (c) segmented paraxial (somitic) mesoderm, (d) mesectoderm (neural crest), (e) neurilemmal cells (neural crest), and (f) neural tube. Some of these sources (a, b, d) pertain to the cranial meninges, others (c, d, e) to the spinal coverings. The first of the future dural processes to develop is the tentorium cerebelli, which, at the end of the embryonic period proper, differs considerably in shape and composition from the later fetal and postnatal tentorium. The embryonic dural limiting layer (Duragrenzschicht) probably corresponds to the interface layer of the adult meninges. The appropriate literature was reviewed and summarized.
PMID 3746345
References
- ↑ 25428542<pubmed>25428542</pubmed>
- ↑ Streeter GL. The development of the venous sinuses of the dura mater in the human embryo. (1915) Amer. J Anat.18: 145-178.
Reviews
<pubmed></pubmed> <pubmed></pubmed> <pubmed></pubmed> <pubmed></pubmed>
Articles
<pubmed></pubmed> <pubmed></pubmed> <pubmed></pubmed> <pubmed></pubmed>
Search PubMed
Search Pubmed: Development Meninges Development
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Additional Images
Terms
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 27) Embryology Neural - Meninges Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Neural_-_Meninges_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G