Endocrine - Hypothalamus Development: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| (3 intermediate revisions by the same user not shown) | |||
| Line 24: | Line 24: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* '''Development of the Basal Hypothalamus through Anisotropic Growth'''{{#pmid:31050853|PMID31050853}} "The adult hypothalamus is subdivided into distinct domains: pre-optic, anterior, tuberal and mammillary. Each domain harbours an array of neurons that act together to regulate homeostasis. The embryonic origins and development of hypothalamic neurons, however, remains enigmatic. Here we summarise recent studies in model organisms that challenge current views of hypothalamic development, which traditionally have attempted to map adult domains to correspondingly-located embryonic domains that expand isotropically. Instead, new studies indicate that hypothalamic neurons arise from progenitor cells that undergo anisotropic growth in different dimensions. Here we describe how a multipotent Shh/ Fgf10-expressing progenitor population gives rise to progenitors that grow anisotropically, expanding to a greater extent than other progenitors and giving rise to cells throughout the basal hypothalamus. Further, Shh/Fgf10+ive -derived progenitors grow sequentially in different directions from the multipotent Shh/ Fgf10 population: first, a subset displaced rostrally give rise to anterior-ventral/tuberal neuronal progenitors, then a subset displaced caudally give rise to mammillary neuronal progenitors; finally, a subset(s) displaced ventrally give rise to tuberal infundibular glial progenitors. As this occurs, stable populations of Shh+ive and Fgf10+ive progenitors form. We describe current understanding of the mechanisms that induce Shh/Fgf10+ive progenitors, and begin to direct their differentiation to anterior-ventral/tuberal neuronal progenitors, mammillary neuronal progenitors and tuberal infundibular progenitors. Together these studies suggest a new model for hypothalamic development that we term the Anisotropic growth model. We discuss the implications of the model for understanding the origins of adult hypothalamic neurons." | |||
* '''Anatomy, development, and plasticity of the neurosecretory hypothalamus in {{zebrafish}}'''{{#pmid:30109407|PMID30109407}} "The paraventricular nucleus (PVN) of the hypothalamus harbors diverse neurosecretory cells with critical physiological roles for the homeostasis. Decades of research in rodents have provided a large amount of information on the anatomy, development, and function of this important hypothalamic nucleus. However, since the hypothalamus lies deep within the brain in mammals and is difficult to access, many questions regarding development and plasticity of this nucleus still remain. In particular, how different environmental conditions, including stress exposure, shape the development of this important nucleus has been difficult to address in animals that develop in utero. To address these open questions, the transparent larval zebrafish with its rapid external development and excellent genetic toolbox offers exciting opportunities. In this review, we summarize recent information on the anatomy and development of the neurosecretory preoptic area (NPO), which represents a similar structure to the mammalian PVN in zebrafish. We will then review recent studies on the development of different cell types in the neurosecretory hypothalamus both in mouse and in fish. Lastly, we discuss stress-induced plasticity of the PVN mainly discussing the data obtained in rodents, but pointing out tools and approaches available in zebrafish for future studies. This review serves as a primer for the currently available information relevant for studying the development and plasticity of this important brain region using zebrafish." | |||
* '''Expression of progesterone receptor, estrogen receptors α and β, and kisspeptin in the hypothalamus during perinatal development of gonad-lacking steroidogenic factor-1 knockout mice'''{{#pmid:30776325|PMID30776325}} "Gonadal hormones contribute to brain sexual differentiation. We analyzed expression of {{progesterone}} receptor (PR), estrogen receptor-α (ERα), ERβ, and {{kisspeptin}}, in the preoptic area (POA) and/or the arcuate nucleus (ARC), in gonad-lacking steroidogenic factor-1 knockout (KO) mice during perinatal development. At postnatal-day (P) 0-P7, POA PR levels were higher in wild-type (WT) males compared with WT females, while those in KO males were lower than in WT males and similar to those in WT and KO females. At P14-P21, PR levels in all groups increased similarly. POA ERα levels were similar in all groups at embryonic-day (E) {{ME15.5}}-P14. Those in WT but not KO males reduced during postnatal development to be significantly lower compared with females at P21. POA ERβ levels were higher in WT males than in WT females, while those in KO males were lower than in WT males and similar to those in WT and KO females at P0-P21. POA kisspeptin expression was female-biased in WT mice, while levels in KO females were lower compared with WT females and similar to those in WT and KO males. ARC kisspeptin levels were equivalent among groups at {{ME15.5}}-P0. At P7-P21, ARC levels in WT but not KO males became lower compared with WT females. Diethylstilbestrol exposure during P0-P6 and P7-P13 increased POA PR and ERβ, and decreased POA ERα and ARC kisspeptin levels at P7 and/or P14 in both sexes of KO mice. These data further understanding of gonadal hormone action on neuronal marker expression during brain sexual development." | |||
* '''Novel insights into the spatial and temporal complexity of hypothalamic organization through precision methods allowing nanoscale resolution'''{{#pmid:30027599|PMID30027599}} "The mammalian hypothalamus contains an astounding heterogeneity of neurons to achieve its role in coordinating central responses to virtually any environmental stressor over the life-span of an individual. ... Mechanistic data encompass cell identities, synaptic connectivity within and outside the hypothalamus to link vegetative and conscious levels of innate behaviours, and context- and circadian rhythm-dependent rules of synaptic neurophysiology to distinguish hypothalamic foci that either tune the body's metabolic set-point or specify behaviours. Consequently, novel insights emerge to explain the evolutionary advantages of non-laminar organization for neuroendocrine circuits coincidently using fast neurotransmitters and neuropeptides." | * '''Novel insights into the spatial and temporal complexity of hypothalamic organization through precision methods allowing nanoscale resolution'''{{#pmid:30027599|PMID30027599}} "The mammalian hypothalamus contains an astounding heterogeneity of neurons to achieve its role in coordinating central responses to virtually any environmental stressor over the life-span of an individual. ... Mechanistic data encompass cell identities, synaptic connectivity within and outside the hypothalamus to link vegetative and conscious levels of innate behaviours, and context- and circadian rhythm-dependent rules of synaptic neurophysiology to distinguish hypothalamic foci that either tune the body's metabolic set-point or specify behaviours. Consequently, novel insights emerge to explain the evolutionary advantages of non-laminar organization for neuroendocrine circuits coincidently using fast neurotransmitters and neuropeptides." | ||
| Line 35: | Line 41: | ||
| [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | | [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | ||
Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Hypothalamus+Embryology ''Hypothalamus Embryology''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Supraoptic+Nucleus ''Supraoptic Nucleus''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Paraventricular+Nucleus ''Paraventricular Nucleus''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Suprachiasmatic+Nucleus ''Suprachiasmatic Nucleus''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Tuberal+Lateral+Nucleus ''Tuberal Lateral Nucleus''] | Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Hypothalamus+Development ''Hypothalamus Development''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Hypothalamus+Embryology ''Hypothalamus Embryology''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Supraoptic+Nucleus ''Supraoptic Nucleus''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Paraventricular+Nucleus ''Paraventricular Nucleus''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Suprachiasmatic+Nucleus ''Suprachiasmatic Nucleus''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Tuberal+Lateral+Nucleus ''Tuberal Lateral Nucleus''] | ||
|} | |} | ||
Revision as of 19:28, 17 May 2019
| Embryology - 26 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
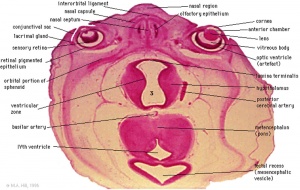
The hypothalamus has a key role in regulating the nearby pituitary and peripheral endocrine organ functions. This neural region therefore is associated with hormonally related behaviour (anger and sexual activity), homeostatic regulation (blood pressure, heart rate, appetite, and temperature) and functions that relate to both (puberty, reproductive cycles, and lactation).
In the early embryo, neuroectoderm of the forebrain (prosenecephalon) primary brain vesicle divides to form two secondary brain vesicles, telencephalon (endbrain, cortex) and diencephalon. The historic description is that the diencephalon ventro-lateral wall, intermediate zone proliferation generates the primordial hypothalamus. A recent "Prosomeric model"[1][2], based on gene expression in the mouse, suggests an overall different origin of the hypothalamus arising from the telencephalon.
Hypothalamus development also occurs differentially in male and female embryos, described as part of neural sexual dimorphism.
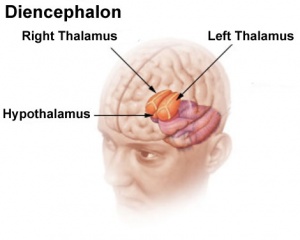
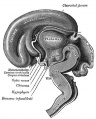
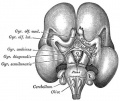
In the adult, large neurosecretory cells of the supraoptic nucleus (SON) and the paraventricular nucleus (PVN) project to the neurohypophysis from the hypothalamus. Other key nuclei within the hypothalamus include the sexually dimorphic nucleus (SDN, intermediate nucleus, INAH-1), suprachiasmatic nucleus (SCN) and tuberal lateral nucleus (NTL). Anatomically surrounded by the anterior commissure and lamina terminalis anteriorly, the mammillary bodies and midbrain posteriorly, and thalamus superiorly.
Category:Hypothalamus | Neural System Development | Lecture - Ectoderm Development | Lecture - Neural Development | Lecture - Head Development
Some Recent Findings
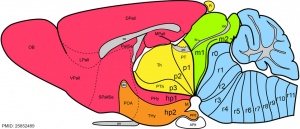
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Hypothalamus Development | Hypothalamus Embryology | Supraoptic Nucleus | Paraventricular Nucleus | Suprachiasmatic Nucleus | Tuberal Lateral Nucleus |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Hypothalamus Endocrine Axes
HPG Axis
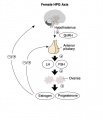
Hypothalamus - Pituitary - Gonad endocrine axis.
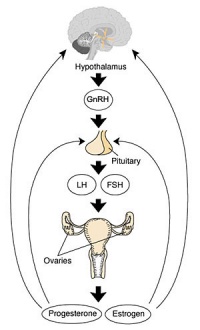
|
This cartoon shows the Hypothalamus - Pituitary - Gonad (female) endocrine axis.
The arrows indicate how each of these endocrine organs interact and regulate each others secretions through endocrine feedback mechanisms. |
Other Endocrine Axes
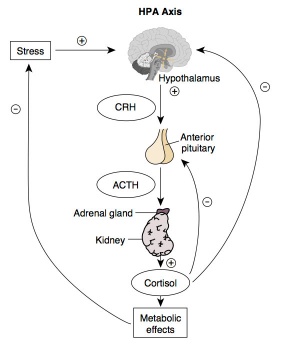
Hypothalamus - Pituitary - Adrenal endocrine axis. |
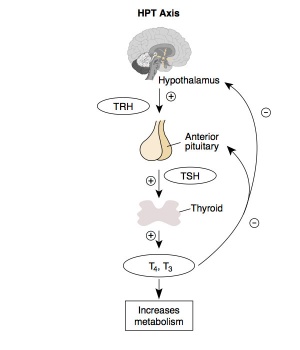
Hypothalamus - Pituitary - Thyroid endocrine axis. |
Development Overview
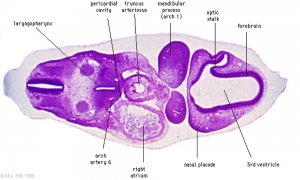
|

|
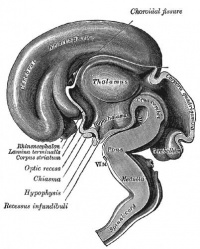
|
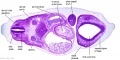
| Diencephalon region, shown by optic stalk (Stage 13) |
Late embryonic hypothalamus (Stage 22) |
Early fetal human brain (3 months, from a model by Wilhelm His) |
- Neuroectoderm - prosenecephalon then rostral diencephalon after induction by the underlying prechordal plate.
- Sonic hedgehog (Shh) - initially expressed in prechordal plate, is essential for inductive process.
- ventro-lateral wall intermediate zone proliferation
- Mamillary bodies - form pea-sized swellings ventral wall of hypothalamus
Week 5
Secondary Brain Vesicles - Stage 13
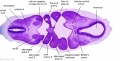
|
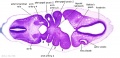
|
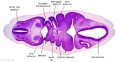
|
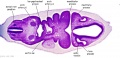
|
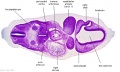
|
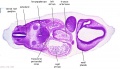
|
|
| B1L | B2L | B3L | B4L | B5L | B6L | B7L |
Week 8
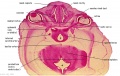
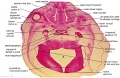
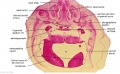
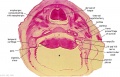
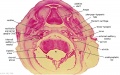
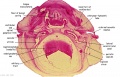
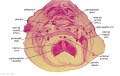
Embryo Brain - Stage 22
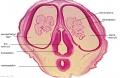
|
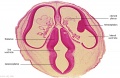
|
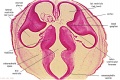
|
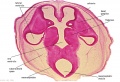
|
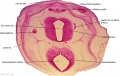
|
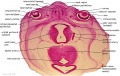
|
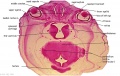
|
| A1L | A2L | A3L | A4L | A5L | A6L | A7L |
|
|
|
|
|
|
|
| B1L | B2L | B3L | B4L | B5L | B6L | B7L |
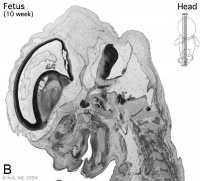
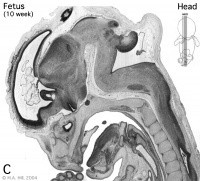
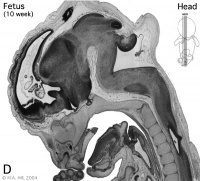
Week 10
Early Fetal Brain
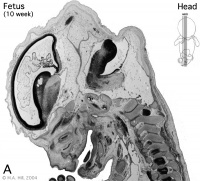
|
|
|
|
Adult Hypothalamus Hormones
Adult Human Hypothalamus[12]
| Secreted hormone | Abbreviation | Produced by | Effect |
|---|---|---|---|
| Thyrotropin-releasing hormone (Prolactin-releasing hormone) |
TRH, TRF, or PRH | Parvocellular neurosecretory neurons | thyroid-stimulating hormone (TSH) release from anterior pituitary (primarily) Stimulate prolactin release from anterior pituitary |
| Dopamine (Prolactin-inhibiting hormone) |
DA or PIH | Dopamine neurons of the arcuate nucleus | Inhibit prolactin release from anterior pituitary |
| Growth hormone-releasing hormone | GHRH | Neuroendocrine neurons of the Arcuate nucleus | Growth hormone (GH) release from anterior pituitary |
| Somatostatin (growth hormone-inhibiting hormone) |
SS, GHIH, or SRIF | Neuroendocrine cells of the Periventricular nucleus | Growth hormone (GH) release from anterior pituitary Inhibit Thyroid-stimulating hormone|thyroid-stimulating hormone (TSH) release from anterior pituitary |
| Gonadotropin-releasing hormone | GnRH or LHRH | Neuroendocrine cells of the Preoptic area | follicle-stimulating hormone (FSH) release from anterior pituitary Stimulate Luteinizing hormone|luteinizing hormone (LH) release from anterior pituitary |
| Corticotropin-releasing hormone | CRH or CRF | Parvocellular neurosecretory neurons | adrenocorticotropic hormone (ACTH) release from anterior pituitary |
| Oxytocin | Magnocellular neurosecretory cells | Lactation (letdown reflex) | |
| Vasopressin (antidiuretic hormone) |
ADH or AVP | Magnocellular neurosecretory neurons | Increases water permeability in the distal convoluted tubule and collecting duct of nephrons, thus promoting water reabsorption and increasing blood volume |
| Melanin-concentrating hormone | MCH | tuberal lateral nucleus (lateral hypothalamic area) | Neuropeptide with appetite stimulant (orexigenic) and sleep-promoting activities by projecting to a variety of brain areas. |
| Links: hypothalamus | |||
Hormones - Thyrotrophin releasing hormone (TRH), Corticotrophin releasing hormone (CRH), Arginine vasopressin (AVP), Gonadotrophin releasing hormone (GnRH), Growth hormone releasing hormone (GHRH), Somatostatin, Prolactin relasing factor (PRF), Dopamine
Growth hormone (GH) secretion from the pituitary is controlled in many different ways, including positive/negative regulation of synthesis and release by the hypothalamus. Hypothalamic GH releasing hormone (GHRH) activates and somatostatin suppresses growth hormone synthesis and release.
Hypothalamic Nuclei
Supraoptic Nucleus
(SON) Arginine vasopressin (AVP) and oxytocin (OT) synthesis and released in the posterior pituitary. These 2 hormones differ only in 2 amino acids in positions 3 and 8.
Arginine vasopressin (AVP) peptide hormone (9aa, nonapeptide)
- synthesized as inactive preprohormone
- regulates blood volume and pressure acting on the kidney and heart
- Increased osmolality, due to blood volume reduction, increases AVP secretion.
Oxytocin (OT) peptide hormone (9aa, nonapeptide)
- mainly produced in hypothalamic magnocellular neurons
- regulates female reproductive pregnancy-associated changes (stimulation of milk ejection, uterine contractions)
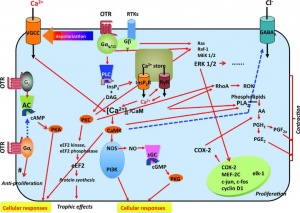
- acts on one type of OT receptor (OTR)
- hormone is a non-glycosylated protein undergoes an initial cleavage by the convertase magnolysin (EC 3.4.24.62) to OT-Gly-Lys-Arg (OT-GKR).
- additional processing produces other OT extended molecules: OT-Gly-Lys (OT-GK) and OT-Gly (OT-G, OT-X).
- OT-G is converted by an α-amidating enzyme to C-amidated nonapeptide which is released into the circulation
- may have a role in fetal heart growth[13] and in vascular tone, regrowth and remodeling.[14]
| About Oxytocin Hormone | |
|---|---|
| Structure | Function |
|
|
| Links: Birth | Hypothalamus | Endocrine | Mammary Gland | Milk | image - Oxytocin receptor pathways | |
Abnormalities: diabetes insipidus (vasopressin deficiency), syndrome of inappropriate antidiuresis (vasopressin excess)
- Links: Birth | Search Bookshelf
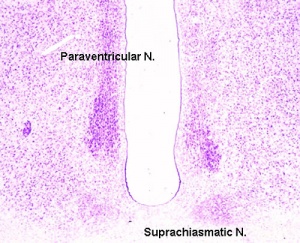
Paraventricular Nucleus
(PVN) Arginine vasopressin (AVP) and oxytocin synthesis and released in the posterior pituitary. These 2 hormones differ only in 2 amino acids in positions 3 and 8.
- magnocellular neurons
- parvocellular neurosecretory neurons project axons to the pituitary median eminence
- interneurons and neurons also project centrally
Arginine vasopressin (AVP) peptide hormone (9aa, nonapeptide)
- synthesized as inactive preprohormone
- regulates blood volume and pressure acting on the kidney and heart
- Increased osmolality, due to blood volume reduction, increases AVP secretion.
Oxytocin (OT) peptide hormone (9aa, nonapeptide)
- mainly produced in hypothalamic magnocellular neurons
- regulates female reproductive pregnancy-associated changes
- central effect on social and other behaviors, release from magnocellular dendrites and axonal projections of parvocellular neurons
- acts on one type of OT receptor (OTR)
- hormone is a non-glycosylated protein undergoes an initial cleavage by the convertase magnolysin (EC 3.4.24.62) to OT-Gly-Lys-Arg (OT-GKR).
- additional processing produces other OT extended molecules: OT-Gly-Lys (OT-GK) and OT-Gly (OT-G, OT-X).
- OT-G is converted by an α-amidating enzyme to C-amidated nonapeptide which is released into the circulation
- may have a role in fetal heart growth[13]
- Links: Birth | Search Bookshelf
Sexually Dimorphic Nucleus
(SDN, intermediate nucleus, INAH-1)
- Twice as large in young male adults as in young females.
- At birth 20% of the adult SDN cell number is present, from then until 2-4 years of age cell numbers increase equally rapidly in both sexes.
- After this age cell numbers start to decrease in girls, creating the sex difference.
The SCN is the clock of the brain and shows circadian and seasonal fluctuations in vasopressin-expressing cell numbers. (SDN and SCN text modified from: Swaab, 1995)
- Links: Search Bookshelf
Suprachiasmatic Nucleus
(SCN) This nucleus is the clock of the brain and shows circadian and seasonal fluctuations in vasopressin-expressing cell numbers.
- Links: Search Bookshelf
Tuberal Lateral Nucleus
- Links: Search Bookshelf
(NTL) This nucleus is involved in feeding behavior and energy metabolism.
Tracts
- retino-hypothalamus tract - (RHT)is the main connection between the eye and the hypothalamus, acts as a direct afferent pathway from the retina going through the optic chiasma.
Molecular
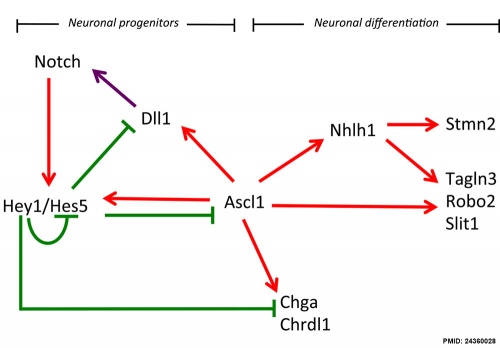
Transcription factor cascade (Sim1, Otp, Sim2, and Brn2) regulate differentiation of the anterior hypothalamus neuroendocrine cells.
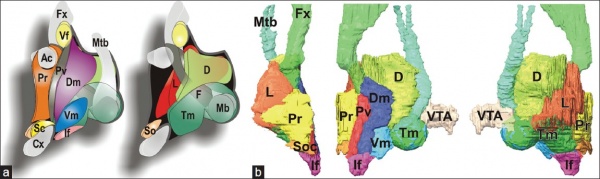
Hypothalamus Development Gene Interaction Model[15]
- Links: Notch
References
- ↑ Rubenstein JL, Martinez S, Shimamura K & Puelles L. (1994). The embryonic vertebrate forebrain: the prosomeric model. Science , 266, 578-80. PMID: 7939711
- ↑ 2.0 2.1 2.2 Puelles L & Rubenstein JL. (2015). A new scenario of hypothalamic organization: rationale of new hypotheses introduced in the updated prosomeric model. Front Neuroanat , 9, 27. PMID: 25852489 DOI.
- ↑ Fu T, Pearson C, Towers M & Placzek M. (2019). Development of the Basal Hypothalamus through Anisotropic Growth. J. Neuroendocrinol. , , e12727. PMID: 31050853 DOI.
- ↑ Nagpal J, Herget U, Choi MK & Ryu S. (2019). Anatomy, development, and plasticity of the neurosecretory hypothalamus in zebrafish. Cell Tissue Res. , 375, 5-22. PMID: 30109407 DOI.
- ↑ Ikeda Y, Kato-Inui T, Tagami A & Maekawa M. (2019). Expression of progesterone receptor, estrogen receptors α and β, and kisspeptin in the hypothalamus during perinatal development of gonad-lacking steroidogenic factor-1 knockout mice. Brain Res. , 1712, 167-179. PMID: 30776325 DOI.
- ↑ Alpár A & Harkany T. (2018). Novel insights into the spatial and temporal complexity of hypothalamic organization through precision methods allowing nanoscale resolution. J. Intern. Med. , , . PMID: 30027599 DOI.
- ↑ Luo SX, Huang J, Li Q, Mohammad H, Lee CY, Krishna K, Kok AM, Tan YL, Lim JY, Li H, Yeow LY, Sun J, He M, Grandjean J, Sajikumar S, Han W & Fu Y. (2018). Regulation of feeding by somatostatin neurons in the tuberal nucleus. Science , 361, 76-81. PMID: 29976824 DOI.
- ↑ Xie Y & Dorsky RI. (2017). Development of the hypothalamus: conservation, modification and innovation. Development , 144, 1588-1599. PMID: 28465334 DOI.
- ↑ Alvarez-Bolado G, Grinevich V & Puelles L. (2015). Editorial: Development of the hypothalamus. Front Neuroanat , 9, 83. PMID: 26157363 DOI.
- ↑ Hughes AT, Guilding C & Piggins HD. (2011). Neuropeptide signaling differentially affects phase maintenance and rhythm generation in SCN and extra-SCN circadian oscillators. PLoS ONE , 6, e18926. PMID: 21559484 DOI.
- ↑ Wen S, Ai W, Alim Z & Boehm U. (2010). Embryonic gonadotropin-releasing hormone signaling is necessary for maturation of the male reproductive axis. Proc. Natl. Acad. Sci. U.S.A. , 107, 16372-7. PMID: 20805495 DOI.
- ↑ Lemaire JJ, Nezzar H, Sakka L, Boirie Y, Fontaine D, Coste A, Coll G, Sontheimer A, Sarret C, Gabrillargues J & De Salles A. (2013). Maps of the adult human hypothalamus. Surg Neurol Int , 4, S156-63. PMID: 23682342 DOI.
- ↑ 13.0 13.1 Jankowski M, Danalache B, Wang D, Bhat P, Hajjar F, Marcinkiewicz M, Paquin J, McCann SM & Gutkowska J. (2004). Oxytocin in cardiac ontogeny. Proc. Natl. Acad. Sci. U.S.A. , 101, 13074-9. PMID: 15316117 DOI.
- ↑ Jankowski M, Wang D, Hajjar F, Mukaddam-Daher S, McCann SM & Gutkowska J. (2000). Oxytocin and its receptors are synthesized in the rat vasculature. Proc. Natl. Acad. Sci. U.S.A. , 97, 6207-11. PMID: 10811917 DOI.
- ↑ Ratié L, Ware M, Barloy-Hubler F, Romé H, Gicquel I, Dubourg C, David V & Dupé V. (2013). Novel genes upregulated when NOTCH signalling is disrupted during hypothalamic development. Neural Dev , 8, 25. PMID: 24360028 DOI.
Reviews
Xie Y & Dorsky RI. (2017). Development of the hypothalamus: conservation, modification and innovation. Development , 144, 1588-1599. PMID: 28465334 DOI.
Clarke IJ. (2015). Hypothalamus as an endocrine organ. Compr Physiol , 5, 217-53. PMID: 25589270 DOI.
Pearson CA & Placzek M. (2013). Development of the medial hypothalamus: forming a functional hypothalamic-neurohypophyseal interface. Curr. Top. Dev. Biol. , 106, 49-88. PMID: 24290347 DOI.
Viero C, Shibuya I, Kitamura N, Verkhratsky A, Fujihara H, Katoh A, Ueta Y, Zingg HH, Chvatal A, Sykova E & Dayanithi G. (2010). REVIEW: Oxytocin: Crossing the bridge between basic science and pharmacotherapy. CNS Neurosci Ther , 16, e138-56. PMID: 20626426 DOI.
Markakis EA. (2002). Development of the neuroendocrine hypothalamus. Front Neuroendocrinol , 23, 257-91. PMID: 12127306
Articles
Watts AG. (2011). Structure and function in the conceptual development of mammalian neuroendocrinology between 1920 and 1965. Brain Res Rev , 66, 174-204. PMID: 20637232 DOI.
Divall SA, Williams TR, Carver SE, Koch L, Brüning JC, Kahn CR, Wondisford F, Radovick S & Wolfe A. (2010). Divergent roles of growth factors in the GnRH regulation of puberty in mice. J. Clin. Invest. , 120, 2900-9. PMID: 20628204 DOI.
Alkemade A, Friesema EC, Unmehopa UA, Fabriek BO, Kuiper GG, Leonard JL, Wiersinga WM, Swaab DF, Visser TJ & Fliers E. (2005). Neuroanatomical pathways for thyroid hormone feedback in the human hypothalamus. J. Clin. Endocrinol. Metab. , 90, 4322-34. PMID: 15840737 DOI.
Books
Endocrinology - An Integrated Approach Stephen Nussey and Saffron Whitehead St. George's Hospital Medical School, London, UK Oxford: BIOS Scientific Publishers; 2001. ISBN-10: 1-85996-252-1
- Hypothalamic control of adrenocortical steroid synthesis - CRH and vasopressin
- Anatomical and functional connections of the hypothalamo-pituitary axis
- Blood supply of the hypothalamo-pituitary axis
- Autonomic functions of the hypothalamus
Search PubMed
Search Pubmed: hypothalamus development
Additional Images
Terms
- 3ß-diol - 5alpha-androstane-3beta, 17beta-diol
- 3α-diol - 5alpha-androstane-3alpha, 17beta-diol
- 5-HT - serotonin
- 5αR - 5-alpha-reductase
- ACTH - adrenocortcotropin releasing hormone
- AR - androgen receptor
- ARN - (AR) Arcuate nucleus
- AVP - arginine vasopressin
- BnST - bed nucleus of the stria terminalis
- BSA - bovine serum albumin
- CBP - CREB binding protein
- CORT - corticosterone
- CRE - cyclic adenosine monophosphate response element
- CREB - CRE binding protein
- CRF - corticotropin releasing factor
- DES - diethylstilbesterol
- DEX - dexamethasone
- DHT - dihydrotestosterone
- DMN - (DM) Dorsomedial hypothalamic nucleus
- DPN - diarylproprionitrile
- ER - estrogen receptor
- ERE - estrogen response element
- FSH - follicle stimulating hormone
- FSL - flinders sensitive line
- GABA - gamma-aminobutyric acid
- GAS - general adaptation syndrome
- GH - growth hormone
- GLP-1 - glucagon-like peptide 1
- GnRH - gonadotropin releasing hormone
- GPER - G-protein coupled estrogen receptor
- GR - glucocorticoid receptor
- HPA - hypothalamo–pituitary–adrenal
- HPG - hypothalamo–pituitary–gonadal
- HRE - hormone respone element
- HSD - hydroxysteroid dehydrogenase
- ICV - intracerebroventricular
- ir - immunoreactive
- ISH - in situ hybridization
- LH - luteinizing hormone
- LHRH - luteinizing hormone releasing hormone
- median eminence - part of the inferior boundary of the hypothalamus
- MPOA - medial preoptic area
- MR - mineralocorticoid receptor
- NTS - nucleus of the solitary tract
- PPT - propylpyrazoletriol
- PR - progesterone receptor
- PVN - (PVA, PVH) Paraventricular nucleus
- RAR - retinoic acid receptors
- SCN - (SC) Suprachiasmatic nucleus
- SON - (SO) Supraoptic nucleus
- SRC - steroid receptor coactivators
- T - testosterone
- THR - thyroid hormone receptors
- TPH - tryptophan hydroxylase
- TRH - thyrotropin releasing hormone
- TSH - thyroid stimulating hormone
- VMN - (VM) Ventromedial nucleus
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 26) Embryology Endocrine - Hypothalamus Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Endocrine_-_Hypothalamus_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G