Developmental Signals - Fibroblast Growth Factor: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 1: | Line 1: | ||
{{ | {{Header}} | ||
==Introduction== | ==Introduction== | ||
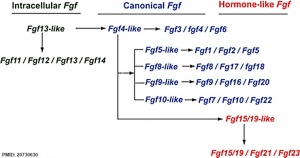
[[File:Fgf_gene_family_evolution.jpg|thumb|:300px|Fgf gene family evolution<ref>Itoh N. '''Hormone-like (endocrine) Fgfs: their evolutionary history and roles in development, metabolism, and disease.''' Cell Tissue Res. 2010 Oct;342(1):1-11. [http://www.ncbi.nlm.nih.gov/pubmed/20730630 PMID20730630]</ref>]] | [[File:Fgf_gene_family_evolution.jpg|thumb|:300px|Fgf gene family evolution<ref>Itoh N. '''Hormone-like (endocrine) Fgfs: their evolutionary history and roles in development, metabolism, and disease.''' Cell Tissue Res. 2010 Oct;342(1):1-11. [http://www.ncbi.nlm.nih.gov/pubmed/20730630 PMID20730630]</ref>]] | ||
| Line 10: | Line 9: | ||
{{Factor_Links}} | |||
==Protein Properties== | ==Protein Properties== | ||
| Line 19: | Line 19: | ||
== Some Recent Findings == | == Some Recent Findings == | ||
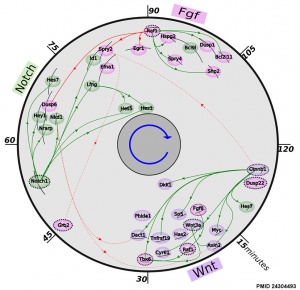
[[File:Mouse somitogenesis genes.jpg|thumb|Mouse somitogenesis genes<ref name=PMID24304493><pubmed>24304493</pubmed>| [http://www.biomedcentral.com/1471-213X/13/42 BMC Dev Biol.]</ref>]] | |||
{| | {| | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* '''The precise timeline of transcriptional regulation reveals causation in mouse somitogenesis network'''<ref name=PMID24304493><pubmed>24304493</pubmed>| [http://www.biomedcentral.com/1471-213X/13/42 BMC Dev Biol.]</ref> "In vertebrate development, the segmental pattern of the body axis is established as somites, masses of mesoderm distributed along the two sides of the neural tube, are formed sequentially in the anterior-posterior axis. This mechanism depends on waves of gene expression associated with the Notch, Fgf and Wnt pathways." | |||
* '''Prolonged FGF signaling is necessary for lung and liver induction in Xenopus'''<ref name=PMID22988910><pubmed>22988910</pubmed></ref> "FGF signaling plays numerous roles during organogenesis of the embryonic gut tube. Mouse explant studies suggest that different thresholds of FGF signaling from the cardiogenic mesoderm induce lung, liver, and pancreas lineages from the ventral foregut progenitor cells. The mechanisms that regulate FGF dose in vivo are unknown. Here we use Xenopus embryos to examine the hypothesis that a prolonged duration of FGF signaling from the mesoderm is required to induce foregut organs....These results suggest that the liver and lungs are specified at progressively later times in development requiring mesoderm contact for different lengths of time. Our data suggest that this is achieved at least in part through prolonged FGF signaling. In addition to providing a foundation for further mechanistic studies on foregut organogenesis using the experimental advantages of the Xenopus system, these data have implications for the directed differentiation of stem cells into foregut lineages." | * '''Prolonged FGF signaling is necessary for lung and liver induction in Xenopus'''<ref name=PMID22988910><pubmed>22988910</pubmed></ref> "FGF signaling plays numerous roles during organogenesis of the embryonic gut tube. Mouse explant studies suggest that different thresholds of FGF signaling from the cardiogenic mesoderm induce lung, liver, and pancreas lineages from the ventral foregut progenitor cells. The mechanisms that regulate FGF dose in vivo are unknown. Here we use Xenopus embryos to examine the hypothesis that a prolonged duration of FGF signaling from the mesoderm is required to induce foregut organs....These results suggest that the liver and lungs are specified at progressively later times in development requiring mesoderm contact for different lengths of time. Our data suggest that this is achieved at least in part through prolonged FGF signaling. In addition to providing a foundation for further mechanistic studies on foregut organogenesis using the experimental advantages of the Xenopus system, these data have implications for the directed differentiation of stem cells into foregut lineages." | ||
* '''FGF-signaling gradient maintains symmetrical proliferative divisions of midbrain neuronal progenitors'''<ref><pubmed>21074523</pubmed></ref> "For the correct development of the central nervous system, the balance between self-renewing and differentiating divisions of the neuronal progenitors must be tightly regulated. To maintain their self-renewing identity, the progenitors need to retain both apical and basal interfaces. However, the identities of fate-determining signals which cells receive via these connections, and the exact mechanism of their action, are poorly understood. The conditional inactivation of Fibroblast growth factor (FGF) receptors 1 and 2 in the embryonic mouse midbrain-hindbrain area results in premature neuronal differentiation. Here, we aim to elucidate the connection between FGF-signaling and neuronal progenitor maintenance. Our results reveal that the loss of FGF-signaling leads to downregulation of Hes1 and upregulation of Ngn2, Dll1, and p57 in the ventricular zone (VZ) cells, and that this increased neurogenesis occurs cell-autonomously. Yet the cell-cycle progression, apico-basal-polarity, cell-cell connections, and the positioning of mitotic spindle in the mutant VZ appear unaltered. Interestingly, FGF8-protein is highly concentrated in the basal lamina. Thus, FGFs may act through basal processes of neuronal progenitors to maintain their progenitor status. Indeed, midbrain neuronal progenitors deprived in vitro of FGFs switched from symmetrical proliferative towards symmetrical neurogenic divisions. We suggest that FGF-signaling in the midbrain VZ is cell-autonomously required for the maintenance of symmetrical proliferative divisions via Hes1-mediated repression of neurogenic genes." | * '''FGF-signaling gradient maintains symmetrical proliferative divisions of midbrain neuronal progenitors'''<ref><pubmed>21074523</pubmed></ref> "For the correct development of the central nervous system, the balance between self-renewing and differentiating divisions of the neuronal progenitors must be tightly regulated. To maintain their self-renewing identity, the progenitors need to retain both apical and basal interfaces. However, the identities of fate-determining signals which cells receive via these connections, and the exact mechanism of their action, are poorly understood. The conditional inactivation of Fibroblast growth factor (FGF) receptors 1 and 2 in the embryonic mouse midbrain-hindbrain area results in premature neuronal differentiation. Here, we aim to elucidate the connection between FGF-signaling and neuronal progenitor maintenance. Our results reveal that the loss of FGF-signaling leads to downregulation of Hes1 and upregulation of Ngn2, Dll1, and p57 in the ventricular zone (VZ) cells, and that this increased neurogenesis occurs cell-autonomously. Yet the cell-cycle progression, apico-basal-polarity, cell-cell connections, and the positioning of mitotic spindle in the mutant VZ appear unaltered. Interestingly, FGF8-protein is highly concentrated in the basal lamina. Thus, FGFs may act through basal processes of neuronal progenitors to maintain their progenitor status. Indeed, midbrain neuronal progenitors deprived in vitro of FGFs switched from symmetrical proliferative towards symmetrical neurogenic divisions. We suggest that FGF-signaling in the midbrain VZ is cell-autonomously required for the maintenance of symmetrical proliferative divisions via Hes1-mediated repression of neurogenic genes." | ||
Revision as of 20:12, 28 December 2013
| Embryology - 14 May 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
Fibroblast Growth Factors (FGF) were originally identified by their ability to stimulate fibroblast cell proliferation but have a role in a growing number of different tissues development and differentiation and continue to have a role in the adult.
The first two identified factors were originally given the nomenclature of acidic or basic. We now know there to be at least 22 different human FGFs (Fgf1–Fgf23). These protein growth factors are bound by 4 different cell membrane receptors (FGFR1-4). FGFRs belong to the tyrosine kinase receptor family.
The mammalian Fgf family can be divided into the intracellular Fgf11/12/13/14 subfamily (iFGFs), the endocrine hormone-like Fgf15/21/23 subfamily (hFGFs), and the paracrine canonical Fgf subfamilies, including Fgf1/2/5, Fgf3/4/6, Fgf7/10/22, Fgf8/17/18, and Fgf9/16/20.
| Factor Links: AMH | hCG | BMP | sonic hedgehog | bHLH | HOX | FGF | FOX | Hippo | LIM | Nanog | NGF | Nodal | Notch | PAX | retinoic acid | SIX | Slit2/Robo1 | SOX | TBX | TGF-beta | VEGF | WNT | Category:Molecular |
Protein Properties
Human FGF
- ~150–300 amino acids
- have a conserved ~120-residue core with ~30–60% identity
Some Recent Findings
|
Recent References | References
Abnormalities
- FGFR1 mutation has been associated with the relatively milder form of Pfeiffer syndrome type 1.
- FGFR2 and FGFR3 have been associated with the Apert, Crouzon and Pfeiffer syndromes.
References
- ↑ Itoh N. Hormone-like (endocrine) Fgfs: their evolutionary history and roles in development, metabolism, and disease. Cell Tissue Res. 2010 Oct;342(1):1-11. PMID20730630
- ↑ 2.0 2.1 <pubmed>24304493</pubmed>| BMC Dev Biol.
- ↑ <pubmed>22988910</pubmed>
- ↑ <pubmed>21074523</pubmed>
- ↑ <pubmed>19741606</pubmed>
Reviews
Articles
<pubmed>20582225</pubmed>
Search Pubmed
Search Bookshelf Fibroblast Growth Factor
Search Pubmed Now: Fibroblast Growth Factor