Musculoskeletal System - Bone Development: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| (26 intermediate revisions by the same user not shown) | |||
| Line 3: | Line 3: | ||
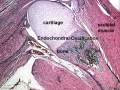
[[File:Endochondral_bone.jpg|thumb|Endochondral bone]] | [[File:Endochondral_bone.jpg|thumb|Endochondral bone]] | ||
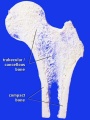
[[File:Bone-femur.jpg|thumb|alt=Bone femur diagram|Bone femur]] | [[File:Bone-femur.jpg|thumb|alt=Bone femur diagram|Bone femur]] | ||
The mesoderm | The skeleton consists of {{bone}} developing from {{mesoderm}}, except within the head where {{neural crest}} also contributes connective tissues. Each tissue ({{cartilage}}, {{bone}}, and {{skeletal muscle}}) goes through many different mechanisms of differentiation. | ||
| Line 9: | Line 9: | ||
Bone is formed through a lengthy process involving ossification of a cartilage formed from mesenchyme. Two main forms of ossification occur in different bones, intramembranous (eg skull) and | Bone is formed through a lengthy process involving ossification of a cartilage formed from mesenchyme. Two main forms of ossification occur in different bones, {{intramembranous ossification}} (eg skull) and {{endochondral ossification}} (eg limb long bones) ossification. Ossification continues postnatally, through puberty until mid 20's. Early ossification occurs at the ends of long bones. A study of 18,713 individuals identified the male/female age at attainment of peak {{bone mineral density}} (BMD).{{#pmid:31760214|PMID31760214}} | ||
| Line 26: | Line 26: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* ''' | |||
* ''' | * '''Review - New Insights Into Cranial Synchondrosis Development'''{{#pmid:32850826|PMID32850826}} "The synchondroses formed via endochondral ossification in the cranial base are an important growth center for the neurocranium. Abnormalities in the synchondroses affect cranial base elongation and the development of adjacent regions, including the craniofacial bones. In the central region of the cranial base, there are two synchondroses present-the intersphenoid synchondrosis and the spheno-occipital synchondrosis. These synchondroses consist of mirror image bipolar growth plates. The cross-talk of several signaling pathways, including the parathyroid hormone-like hormone (PTHLH)/parathyroid hormone-related protein (PTHrP), Indian hedgehog (Ihh), Wnt/β-catenin, and fibroblast growth factor (FGF) pathways, as well as regulation by cilium assembly and the transcription factors encoded by the RUNX2, SIX1, SIX2, SIX4, and TBX1 genes, play critical roles in synchondrosis development. Deletions or activation of these gene products in mice causes the abnormal ossification of cranial synchondrosis and skeletal elements. Gene disruption leads to both similar and markedly different abnormalities in the development of intersphenoid synchondrosis and spheno-occipital synchondrosis, as well as in the phenotypes of synchondroses and skeletal bones. This paper reviews the development of cranial synchondroses, along with its regulation by the signaling pathways and transcription factors, highlighting the differences between intersphenoid synchondrosis and spheno-occipital synchondrosis." | ||
* {{#pmid:32835424|PMID32835424}} '''YAP and TAZ promote periosteal osteoblast precursor expansion and differentiation for fracture repair''' "In response to bone fracture, periosteal progenitor cells proliferate, expand, and differentiate to form cartilage and bone in the fracture callus. These cellular functions require the coordinated activation of multiple transcriptional programs, and the transcriptional regulators Yes-associated protein (YAP) and transcriptional co-activator with PDZ-binding motif (TAZ) regulate osteochondroprogenitor activation during endochondral bone development. However, recent observations raise important distinctions between the signaling mechanisms used to control bone morphogenesis and repair. Here, we tested the hypothesis that YAP and TAZ regulate osteochondroprogenitor activation during endochondral bone fracture healing in mice. Constitutive YAP and/or TAZ deletion from Osterix-expressing cells impaired both cartilage callus formation and subsequent mineralization. However, this could be explained either by direct defects in osteochondroprogenitor differentiation after fracture, or by developmental deficiencies in the progenitor cell pool prior to fracture. Consistent with the second possibility, we found that developmental YAP/TAZ deletion produced long bones with impaired periosteal thickness and cellularity. Therefore, to remove the contributions of developmental history, we next generated adult onset-inducible knockout mice (using Osx-CretetOff ) in which YAP and TAZ were deleted prior to fracture, but after normal development." | |||
* '''SHP2 regulates intramembranous ossification by modifying the TGFβ and BMP2 signaling pathway'''{{#pmid:30471432|PMID30471432}} "SHP2 is a ubiquitously expressed protein tyrosine phosphatase, which is involved in many signaling pathways to regulate the skeletal development. In endochondral ossification, SHP2 is known to modify the osteogenic fate of osteochondroprogenitors and to impair the osteoblastic transdifferentiation of hypertrophic chondrocytes. However, how SHP2 regulates osteoblast differentiation in intramembranous ossification remains incompletely understood. To address this question, we generated a mouse model to ablate SHP2 in the Prrx1-expressing mesenchymal progenitors by using "Cre-loxP"-mediated gene excision and examined the development of calvarial bone, in which the main process of bone formation is intramembranous ossification. Phenotypic characterization showed that SHP2 mutants have severe defects in calvarial bone formation. Cell lineage tracing and in situ hybridization data showed less osteoblast differentiation of mesenchymal cells and reduced osteogenic genes expression, respectively. Further mechanistic studies revealed enhanced TGFβ and suppressed BMP2 signaling in SHP2 ablated mesenchymal progenitors and their derivatives. Our study uncovered the critical role of SHP2 in osteoblast differentiation through intramembranous ossification and might provide a potential target to treat craniofacial skeleton disorders." | |||
|} | |} | ||
{| class="wikitable mw-collapsible mw-collapsed" | {| class="wikitable mw-collapsible mw-collapsed" | ||
! More recent papers | ! More recent papers | ||
|- | |- | ||
| [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | | [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | ||
Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Bone+Embryology ''Bone Embryology''] | Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Bone+Embryology ''Bone Embryology''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Bone+Development ''Bone Development''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=endochondral+ossification ''endochondral ossification''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=intramembranous+ossification ''intramembranous ossification''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=osteoblast ''osteoblast''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=osteoclast ''osteoclast''] | ||
|} | |||
{| class="wikitable mw-collapsible mw-collapsed" | |||
! Older papers | |||
|- | |||
| {{Older papers}} | |||
* '''CT analysis of anatomical distribution of melorheostosis challenges the sclerotome hypothesis'''{{#pmid:30218789|PMID30218789}} "Melorheostosis (MEL) is a rare disease of high bone mass with patchy skeletal distribution affecting the long bones. We recently reported somatic mosaic mutations in MAP2K1 in 8 of 15 patients with the disease. The unique anatomic distribution of melorheostosis is of great interest. The disease remains limited to medial or lateral side of the extremity with proximo-distal progression. This pattern of distribution has historically been attributed to sclerotomes (area of bone which is innervated by a single spinal nerve level). ... This suggests that the mutation occurred after the formation of dorso-ventral plane. Further studies on limb development are needed to better understand the etiology, pathophysiology and pattern of disease distribution in all patients with MEL." | |||
* '''Cell-matrix signals specify bone endothelial cells during developmental osteogenesis'''{{#pmid:28218908|PMID28218908}} “Blood vessels in the mammalian skeletal system control bone formation and support haematopoiesis by generating local niche environments. Here, we report that embryonic and early postnatal long bone contains a specialized endothelial cell subtype, termed type E, which strongly supports osteoblast lineage cells and later gives rise to other endothelial cell subpopulations. The differentiation and functional properties of bone endothelial cells require cell-matrix signalling interactions." [[Cardiovascular System - Blood Vessel Development|Blood Vessel Development]] | | [https://omim.org/entry/135630 INTEGRIN BETA-1] | |||
* '''Disruption of Scube2 impairs endochondral bone formation'''{{#pmid:25639508|PMID25639508}} "SCUBE2 (signal peptide-CUB-EGF domain-containing protein 2) belongs to a secreted and membrane-tethered multi-domain SCUBE protein family composed of 3 members found in vertebrates and mammals. Recent reports suggested that zebrafish scube2 could facilitate sonic hedgehog (Shh) signaling for proper development of slow muscle. However, whether SCUBE2 can regulate the signaling activity of two other hedgehog ligands (Ihh and Dhh), and the developmental relevance of the SCUBE2-induced hedgehog signaling in mammals remain poorly understood. In this study, we first showed that as compared with SCUBE1 or 3, SCUBE2 is the most potent modulator of IHH signaling in vitro. In addition, gain and loss-of-function studies demonstrated that SCUBE2 exerted an osteogenic function by enhancing Ihh-stimulated osteoblast differentiation in the mouse mesenchymal progenitor cells." [[Developmental Signals - Sonic hedgehog|Sonic hedgehog]] | |||
* '''Vascularization of primary and secondary ossification centres in the human growth plate'''{{#pmid:25164565|PMID25164565}} "The switch from cartilage template to bone during endochondral ossification of the growth plate requires a dynamic and close interaction between cartilage and the developing vasculature. Vascular invasion of the primarily avascular hypertrophic chondrocyte zone brings chondroclasts, osteoblast- and endothelial precursor cells into future centres of ossification. ...Vascularization of ossification centres of the growth plate was mediated by sprouting of capillaries coming from the bone collar or by intussusception rather than by de-novo vessel formation involving endothelial progenitor cells. Vascular invasion of the joint anlage was temporally delayed compared to the surrounding joint tissue." | |||
|} | |} | ||
==Adult Human Skeleton== | ==Adult Human Skeleton== | ||
| Line 89: | Line 104: | ||
|} | |} | ||
==Ossification Centres== | ==Ossification Centres== | ||
[[File:Mouse_limb_cartilage_and_bone_E14.5.jpg|thumb|300px|Mouse limb showing primary ossification. | [[File:Mouse_limb_cartilage_and_bone_E14.5.jpg|thumb|300px|Mouse limb showing primary ossification.{{#pmid:20386744|PMID20386744}}]] | ||
===Primary Ossification=== | ===Primary Ossification=== | ||
* Primary ossification centres are the first sites of bone formation and where cartilage has begun to degenerate. In long bones, this lis generally located mid-diaphysis (shaft). In other bones (e.g. base of skull) these are the initial locations of bone formation. | * Primary ossification centres are the first sites of bone formation and where cartilage has begun to degenerate. In long bones, this lis generally located mid-diaphysis (shaft). In other bones (e.g. base of skull) these are the initial locations of bone formation. | ||
| Line 103: | Line 118: | ||
Most of the bony skeleton forms by this process, that replaces a developmental cartilage template with bone. | Most of the bony skeleton forms by this process, that replaces a developmental cartilage template with bone. | ||
{| | {| | ||
| colspan=6|Longitudinal views of endochondral bone formation in mouse limbs. | | colspan=6|Longitudinal views of endochondral bone formation in mouse limbs.{{#pmid:26893264|PMID26893264}} | ||
|- | |- | ||
| colspan=6|[[File:Endochondral bone cartoon.jpg|100%]] | | colspan=6|[[File:Endochondral bone cartoon.jpg|100%]] | ||
| Line 127: | Line 142: | ||
==Intramembranous Ossification== | ==Intramembranous Ossification== | ||
The process of {{intramembranous ossification}} (desmal ossification) occurs with mesenchyme directly ossifying into bone without a pre-existing cartilage template. Vascularised regions of mesenchymal cells{{#pmid:23737393|PMID23737393}} proliferate and differentiate into initially pre-osteoblasts and then osteoblasts, occurs in parts of the skull and the clavicle. | |||
The vascularising endothelial cells promotes mesenchymal intramembranous ossification by {{BMP}} signaling pathway activated through Yap/Taz transcriptional activity.{{#pmid:27273480|PMID27273480}} | |||
See also [[Bone Histology]] | See also [[Bone Histology]] | ||
[[File:Ossification_centre.jpg]] [[File:Intramembranous_ossification_centre.jpg]] | [[File:Ossification_centre.jpg]] [[File:Intramembranous_ossification_centre.jpg]] | ||
| Line 152: | Line 170: | ||
[[File:Fetal_head_section.jpg|600px]] | [[File:Fetal_head_section.jpg|600px]] | ||
==Bone Mineral Density== | |||
Bone mineral density (BMD) is measured postnatally by an {{X-ray}} test and is generally used to detect bone loss associated with osteoporosis. The density of the total hip is a predictor for a hip fracture, while the lumbar spine is the site for monitoring the effect of clinical treatment. | |||
A study of 18,713 individuals, has identified the postnatal male/female age at attainment of peak bone mineral density (BMD).{{#pmid:31760214|PMID31760214}} | |||
{{Peak Bone Mineral Density Age table1}} | |||
There are two main identified forms of osteoporosis: | |||
# Primary osteoporosis - related to bone loss from ageing. | |||
# Secondary osteoporosis - results from specific conditions that may be reversible. | |||
:'''PubMed:''' [https://www.ncbi.nlm.nih.gov/pubmed/?term=Primary+osteoporosis Primary osteoporosis] | [https://www.ncbi.nlm.nih.gov/pubmed/?term=Secondary+osteoporosis Secondary osteoporosis] | [https://www.ncbi.nlm.nih.gov/pubmed/?term=Bone+mineral+density Bone mineral density] | |||
==Bone Structure== | ==Bone Structure== | ||
| Line 173: | Line 205: | ||
===Periosteum=== | ===Periosteum=== | ||
{| | |||
| The embryonic origin of this layer is still controversial. The connective tissue coating covering the surface of bone, except at the articular surfaces, consisting of two distinct main layers with sub-layers.{{#pmid:20049593|PMID20049593}} | |||
# '''outer fibrous layer''' | |||
## outer - portion highly vascularized, cell poor with a predominant collagenous matrix and few elastic fibers. | |||
## inner - portion not highly vascularized, cell poor with a fibroelastic layer, containing many elastic fibers. | |||
# '''cambium layer''' - a highly cellular layer containing mesenchymal progenitor cells, differentiated osteogenic progenitor cells, osteoblasts and fibroblasts in a sparse collagenous matrix. | |||
| [[File:Periosteum.jpg]] | |||
|} | |||
===Endosteum=== | ===Endosteum=== | ||
| Line 197: | Line 236: | ||
* mature bone-forming cells embedded in '''lacunae''' within the bone matrix | * mature bone-forming cells embedded in '''lacunae''' within the bone matrix | ||
* osteoblasts and osteocytes - secrete organic matrix of bone (osteoid), converted into '''osteocytes''' when become embedded in matrix (which calcifies soon after deposition) | * osteoblasts and osteocytes - secrete organic matrix of bone (osteoid), converted into '''osteocytes''' when become embedded in matrix (which calcifies soon after deposition) | ||
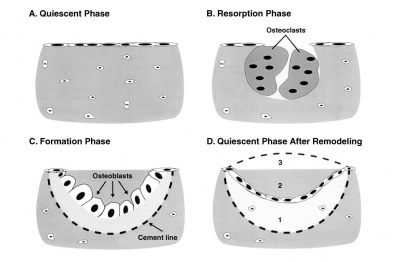
===Osteoclasts=== | ===Osteoclasts=== | ||
[[File:Osteoclast.jpg]] [[File:Bone_remodeling_cycle.jpg|400px]] | [[File:Osteoclast.jpg]] [[File:Bone_remodeling_cycle.jpg|400px]] | ||
Recently, Heterogeneous nuclear ribonucleoprotein K ([https://www.omim.org/entry/600712 hnRNPK]), a DNA/RNA-binding protein, has been shown to be essential for the formation of osteoclast.{{#pmid:31538344PMID31538344}} | |||
* bone-resorbing multinucleated macrophage-like cells | * bone-resorbing multinucleated macrophage-like cells | ||
* | * originate by fusion of monocytes or macrophages, Blood macrophage precursor, Attach to bone matrix | ||
* seal a small segment of extracellular space (between plasma membrane and bone surface), HCl and lysosomes secreted into this space by osteoclasts dissolves calcium phosphate crystals (give bone rigidity and strength) | * seal a small segment of extracellular space (between plasma membrane and bone surface), HCl and lysosomes secreted into this space by osteoclasts dissolves calcium phosphate crystals (give bone rigidity and strength) | ||
** Resorptive bay - (Howship's lacuna) shallow bay lying directly under an osteoclast. | ** Resorptive bay - (Howship's lacuna) shallow bay lying directly under an osteoclast. | ||
* do not mistake for megakaryocytes, found in bone marrow not associated with bone matrix. | * do not mistake for megakaryocytes, found in bone marrow not associated with bone matrix. | ||
** megakaryocytes are also multi-niucleated and form platelets | ** megakaryocytes are also multi-niucleated and form platelets | ||
:'''Links:''' [https://www.omim.org/entry/600712 OMIM - hnRNPK] | |||
===Osteoclastogenesis=== | |||
* Formation of mature osteoclasts involved in bone resorption - the osteoblasts regulate this process through the production of RANKL (Receptor Activator for Nuclear Factor κ B Ligand) which is found on the cell surface of osteoblasts. | |||
* RANKL is a key player in rheumatoid arthritis. | |||
* Osteoclast origin - fusion of monocytes or macrophages, Blood macrophage precursor | |||
* Attach to bone matrix - very large cells containing 15-20 nucleii. | |||
* Lysosomes - released into space between ruffled border and bone matrix, enzymes break down collagen fibres, resorption bays or Howship's lacunae | |||
===Hippo=== | |||
Hippo signaling pathway has recently been identified as a regulator of osteoclast formation (see review{{#pmid:29219182|PMID29219182}} | |||
* Hippo signaling pathway regulatory molecules - RASSF2, NF2, MST1/2, SAV1, LATS1/2, MOB1, YAP, and TAZ. | |||
* osteoclast differentiation - upon activation, MST and LAST, transcriptional co-activators YAP and TAZ bind to the members of the TEA domain (TEAD) family transcription factors | |||
** regulate expression of downstream target genes connective tissue growth factor (CTGF/CCN2) and cysteine-rich protein 61 (CYR61/CCN1). | |||
* RANKL-mediated signaling cascades including NF-κB, MAPKs, AP1, and NFATc1, Hippo-signaling molecules such as YAP/TAZ/TEAD complex, RASSF2, MST2, and Ajuba could also potentially modulate osteoclast differentiation and function. | |||
:'''Links:''' {{Hippo}} | |||
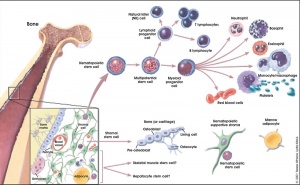
===Bone Marrow=== | ===Bone Marrow=== | ||
| Line 213: | Line 277: | ||
* '''stromal cells''' - all other support cells not involved in haematopoiesis | * '''stromal cells''' - all other support cells not involved in haematopoiesis | ||
:'''Links:''' | |||
:'''Links:''' {{Blood}} | |||
Marrow stroma components: | Marrow stroma components: | ||
| Line 246: | Line 311: | ||
==Molecular== | ==Molecular== | ||
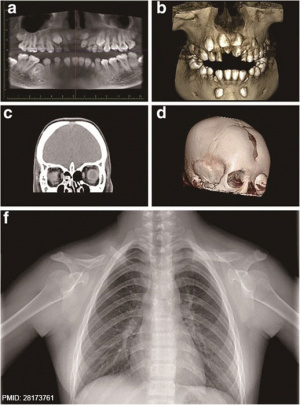
[[File:Cleidocranial dysplasia 01.jpg|thumb|alt=Cleidocranial dysplasia CT and Xray|Cleidocranial dysplasia associated with RUNX2 mutation.{{#pmid:28173761|PMID28173761}}]] | |||
The transcription factors Runx2 and Runx3 are essential for chondrocyte maturation, while Runx2 and Osterix are essential for osteoblast differentiation. | The transcription factors Runx2 and Runx3 are essential for chondrocyte maturation, while Runx2 and Osterix are essential for osteoblast differentiation. | ||
===Osterix=== | ===Osterix=== | ||
| Line 256: | Line 323: | ||
===Osteogenesis Imperfecta=== | ===Osteogenesis Imperfecta=== | ||
Osteogenesis Imperfecta (OI, brittle bone disease) originally described as a collagen 1 gene mutation, but can have several different genetic causes and can be classified into eight different types (I-VIII). | Osteogenesis Imperfecta (OI, brittle bone disease) originally described as a collagen 1 gene mutation, but can have several different genetic causes and can be classified into eight different types (I-VIII).{{#pmid:19907330|PMID19907330}} | ||
* COL1A1 and COL1A2 mutations | * COL1A1 and COL1A2 mutations | ||
| Line 269: | Line 336: | ||
===Reviews=== | ===Reviews=== | ||
32850793 | |||
{{#pmid:28786518}} | |||
{{#pmid:26453494}} | |||
{{#pmid:23794334}} | |||
{{#pmid:23284041}} | |||
{{#pmid:20049593}} | |||
{{#pmid:19883365}} | |||
{{#pmid:17659995}} | |||
===Articles=== | ===Articles=== | ||
===Journals=== | ===Journals=== | ||
| Line 317: | Line 392: | ||
{{Footer}} | {{Footer}} | ||
[[Category:System Development]][[Category:Bone]] | |||
[[Category:System Development]] | |||
Latest revision as of 13:19, 28 August 2020
| Embryology - 15 Jun 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
The skeleton consists of bone developing from mesoderm, except within the head where neural crest also contributes connective tissues. Each tissue (cartilage, bone, and skeletal muscle) goes through many different mechanisms of differentiation.
The 2 key developmental processes are the initial "patterning" of bone location and then the overt "differentiation" of bone through the process of ossification. For details on specific bone differentiation in human development see Bone Development Timeline.
Bone is formed through a lengthy process involving ossification of a cartilage formed from mesenchyme. Two main forms of ossification occur in different bones, intramembranous ossification (eg skull) and endochondral ossification (eg limb long bones) ossification. Ossification continues postnatally, through puberty until mid 20's. Early ossification occurs at the ends of long bones. A study of 18,713 individuals identified the male/female age at attainment of peak bone mineral density (BMD).[1]
The two major parts of the human skeleton are the axial (80 bones in skull, vertebra, ribs, sternum) and appendicular (126 bones in limbs, shoulders, pelvis) skeletons.
Musculoskeletal and limb abnormalities are one of the largest groups of congenital abnormalities.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Bone Embryology | Bone Development | endochondral ossification | intramembranous ossification | osteoblast | osteoclast |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Adult Human Skeleton
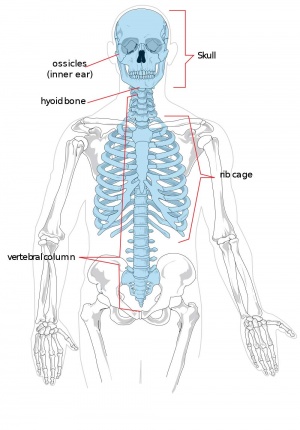
|

|
Textbooks
- The Developing Human: Clinically Oriented Embryology (8th Edition) by Keith L. Moore and T.V.N Persaud - Moore & Persaud Chapter 15 the skeletal system
- Larsen’s Human Embryology by GC. Schoenwolf, SB. Bleyl, PR. Brauer and PH. Francis-West - Chapter 11 Limb Dev (bone not well covered in this textbook)
- Before we Are Born (5th ed.) Moore and Persaud Chapter 16,17: p379-397, 399-405
- Essentials of Human Embryology Larson Chapter 11 p207-228
Objectives
- Identify the components of a somite and the adult derivatives of each component.
- Give examples of sites of (a) endochondral and (b) intramembranous ossification and to compare these two processes.
- Identify the general times (a) of formation of primary and (b) of formation of secondary ossification centres, and (c) of fusion of such centres with each other.
- Briefly summarise the development of the limbs.
- Describe the developmental abnormalities responsible for the following malformations: selected growth plate disorders; congenital dislocation of the hip; scoliosis; arthrogryposis; and limb reduction deformities.
Development Overview
Below is a very brief overview using simple figures of 3 aspects of early musculoskeletal development. More detailed overviews are shown on other notes pages Mesoderm and Somite, Vertebral Column, Limb in combination with serial sections and Carnegie images.
Mesoderm Development
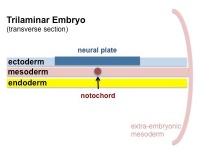
|
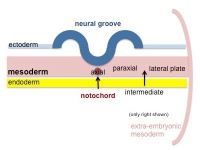
|
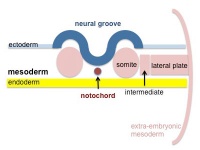
|
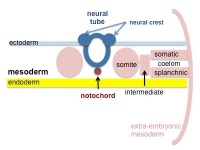
|
| Cells migrate through the primitive streak to form mesodermal layer. Extraembryonic mesoderm lies adjacent to the trilaminar embryo totally enclosing the amnion, yolk sac and forming the connecting stalk. | Paraxial mesoderm accumulates under the neural plate with thinner mesoderm laterally. This forms 2 thickened streaks running the length of the embryonic disc along the rostrocaudal axis. In humans, during the 3rd week, this mesoderm begins to segment. The neural plate folds to form a neural groove and folds. | Segmentation of the paraxial mesoderm into somites continues caudally at 1 somite/90minutes and a cavity (intraembryonic coelom) forms in the lateral plate mesoderm separating somatic and splanchnic mesoderm. Note intraembryonic coelomic cavity communicates with extraembryonic coelom through portals (holes) initially on lateral margin of embryonic disc. | Somites continue to form. The neural groove fuses dorsally to form a tube at the level of the 4th somite and "zips up cranially and caudally and the neural crest migrates into the mesoderm. |
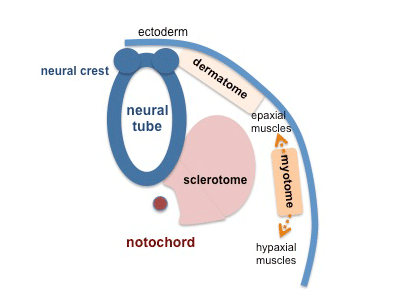
Limb Development
|
Ossification Centres
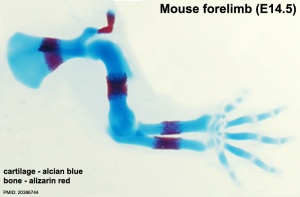
Primary Ossification
- Primary ossification centres are the first sites of bone formation and where cartilage has begun to degenerate. In long bones, this lis generally located mid-diaphysis (shaft). In other bones (e.g. base of skull) these are the initial locations of bone formation.
Secondary Ossification
- Secondary ossification centres develop in the cartilage epiphysis of the long bones.
- No medulary cavity forms in a secondary ossification center.
- Appears late in fetal development.
- Used as a marker for term development if a secondary ossification centre present in either: head of femur, head of tibia, of head of humerus.
- The last secondary centre to appear is the clavical medial epiphysis, that does not develop until 18 or 20 years.
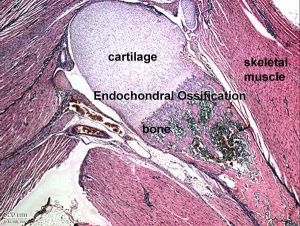
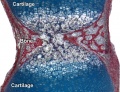
Endochondral Ossification
Most of the bony skeleton forms by this process, that replaces a developmental cartilage template with bone.
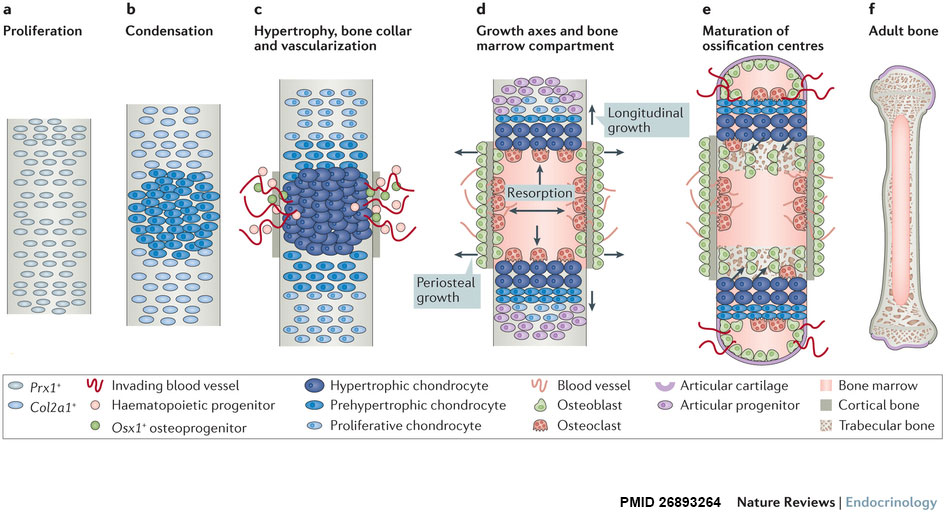
| Longitudinal views of endochondral bone formation in mouse limbs.[10] | |||||
| |||||
| a - Prx1+ progenitors from lateral plate mesoderm proliferate to populate the emerging limb bud. | b - Cells nearest the centre undergo mesenchymal condensation, express Col2a1 as they enter a chondrogenic differentiation program, and deposit a cartilage template. | c to d - Differentiating cells upregulate Col10a1 as they become hypertrophic, which triggers local formation of a bone collar and vascularization of the cartilage template. Invading blood vessels deliver an influx of haematopoietic cells that give rise to osteoclasts which excavate the cartilage template, and Osx1+ osteoblast progenitors and other blood cell types that populate the newly formed marrow cavity. | d - A longitudinal growth axis is established when vascularization and osteoclast-mediated resorption bisect the presumptive skeletal element, producing two growth plates with opposing directionality. A perpendicular growth axis is driven by periosteal osteoblasts and allows the bone to grow in width. | e - Within the remodelled cartilage template, bone-forming osteoblasts are derived from Osx1+ cells arriving with the invading vasculature, as well as hypertrophic Col10a1+ chondroctyes that transdifferentiate as they exit the growth plate into the marrow cavity. As bones grow in length and width, a second wave of vascularization forms the secondary ossification centres. | f - Mature endochondral bone. |
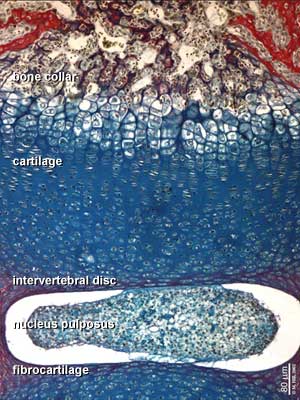
See also Bone Histology
- Links: Blue Histology - endochondral | Dev Biology - endochondral ossification | endochondral ossification animation
Intramembranous Ossification
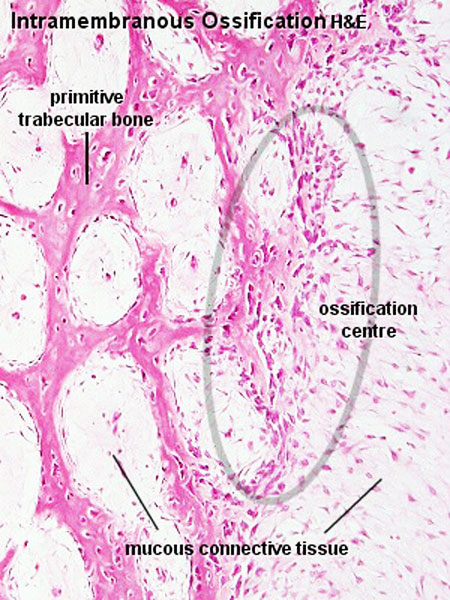
The process of intramembranous ossification (desmal ossification) occurs with mesenchyme directly ossifying into bone without a pre-existing cartilage template. Vascularised regions of mesenchymal cells[11] proliferate and differentiate into initially pre-osteoblasts and then osteoblasts, occurs in parts of the skull and the clavicle.
The vascularising endothelial cells promotes mesenchymal intramembranous ossification by BMP signaling pathway activated through Yap/Taz transcriptional activity.[12]
See also Bone Histology
- Links:
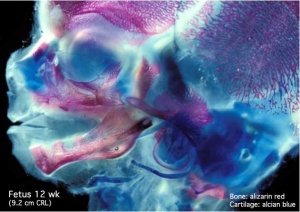
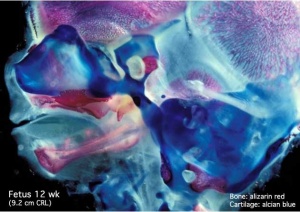
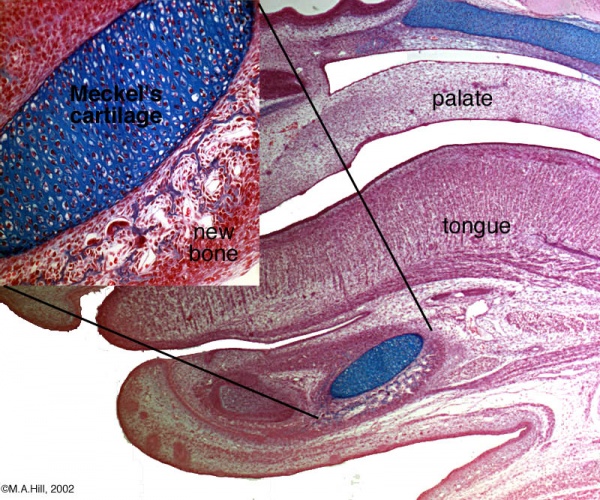
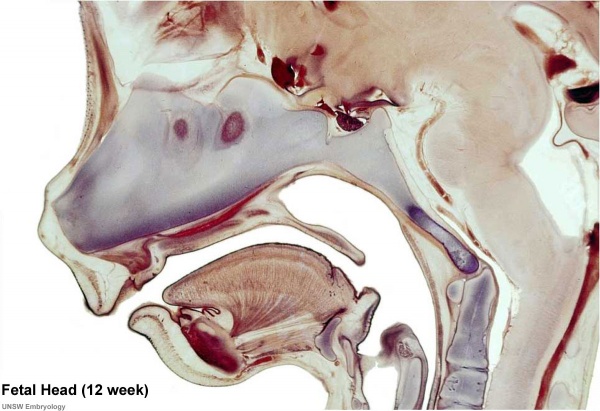
Human Fetal Head (12 week)
Bone Mineral Density
Bone mineral density (BMD) is measured postnatally by an X-ray test and is generally used to detect bone loss associated with osteoporosis. The density of the total hip is a predictor for a hip fracture, while the lumbar spine is the site for monitoring the effect of clinical treatment.
A study of 18,713 individuals, has identified the postnatal male/female age at attainment of peak bone mineral density (BMD).[1]
| Age (years) of Peak Bone Mineral Density (BMD) | |||
|---|---|---|---|
| femoral neck | total hip | lumbar spine | |
| Male | 20.5 | 21.2 | 23 .6 |
| Female | 18.7 | 19.0 | 20.1 |
| Table based on data from[1] {More? bone) | |||
There are two main identified forms of osteoporosis:
- Primary osteoporosis - related to bone loss from ageing.
- Secondary osteoporosis - results from specific conditions that may be reversible.
Bone Structure
Terminology
|

|
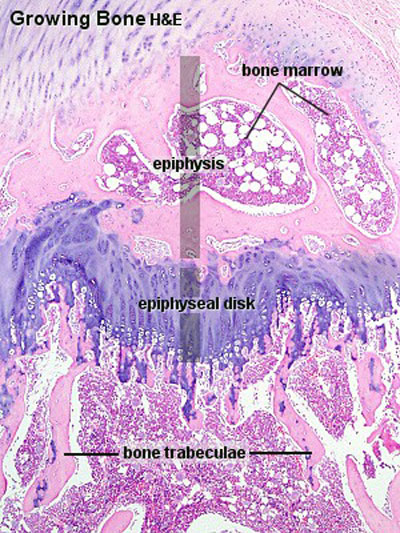
Compact bone
- (dense) no spaces or hollows in the bone matrix visible to the eye.
- forms the thick-walled tube of the shaft (or diaphysis) of long bones, which surrounds the marrow cavity (or medullary cavity). A thin layer of compact bone also covers the epiphyses of long bones.
Trabecular bone
- (cancellous or spongy bone) consists of delicate bars (spicules) and sheets of bone, trabeculae
- branch and intersect to form a sponge-like network
- ends of long bones (or epiphyses) consist mainly of trabecular bone.
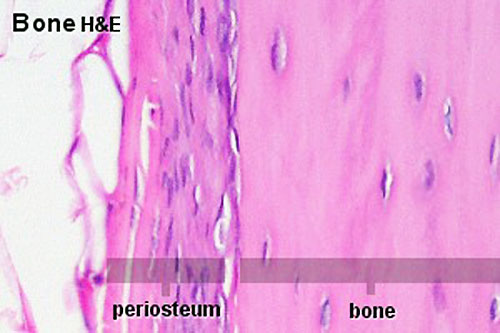
Periosteum
The embryonic origin of this layer is still controversial. The connective tissue coating covering the surface of bone, except at the articular surfaces, consisting of two distinct main layers with sub-layers.[13]
|
|
Endosteum
Connective tissue lining inner surface of bone.
Bone Growth
- Appositional growth occurs at either the periosteum (outer surface), or the endosteum (inner surface).
- Osteoblasts secrete osteoid, a pre-bone material composed mainly of type I collagen that becomes mineralized.
- Early bone matrix deposited in development and during repair is woven rather than lamellar in appearance and structure.
- In development, there are 2 distinct types of bone formation (intramembranous and endochondral)
Bone Cells
Osteoblasts
- derive from osteogenic stem cells the osteoprogenitor cells that differentiate to form pre-osteoblast then osteoblasts maturing to an osteocyte
- osteoprogenitor cells - "resting cell" line the inner and outer surfaces of bone
Osteocytes
- mature bone-forming cells embedded in lacunae within the bone matrix
- osteoblasts and osteocytes - secrete organic matrix of bone (osteoid), converted into osteocytes when become embedded in matrix (which calcifies soon after deposition)
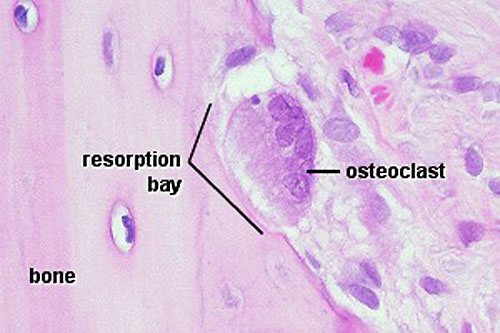
Osteoclasts
Recently, Heterogeneous nuclear ribonucleoprotein K (hnRNPK), a DNA/RNA-binding protein, has been shown to be essential for the formation of osteoclast.PubmedParser error: Invalid PMID, please check. (PMID: 31538344PMID31538344)
- bone-resorbing multinucleated macrophage-like cells
- originate by fusion of monocytes or macrophages, Blood macrophage precursor, Attach to bone matrix
- seal a small segment of extracellular space (between plasma membrane and bone surface), HCl and lysosomes secreted into this space by osteoclasts dissolves calcium phosphate crystals (give bone rigidity and strength)
- Resorptive bay - (Howship's lacuna) shallow bay lying directly under an osteoclast.
- do not mistake for megakaryocytes, found in bone marrow not associated with bone matrix.
- megakaryocytes are also multi-niucleated and form platelets
- Links: OMIM - hnRNPK
Osteoclastogenesis
- Formation of mature osteoclasts involved in bone resorption - the osteoblasts regulate this process through the production of RANKL (Receptor Activator for Nuclear Factor κ B Ligand) which is found on the cell surface of osteoblasts.
- RANKL is a key player in rheumatoid arthritis.
- Osteoclast origin - fusion of monocytes or macrophages, Blood macrophage precursor
- Attach to bone matrix - very large cells containing 15-20 nucleii.
- Lysosomes - released into space between ruffled border and bone matrix, enzymes break down collagen fibres, resorption bays or Howship's lacunae
Hippo
Hippo signaling pathway has recently been identified as a regulator of osteoclast formation (see review[14]
- Hippo signaling pathway regulatory molecules - RASSF2, NF2, MST1/2, SAV1, LATS1/2, MOB1, YAP, and TAZ.
- osteoclast differentiation - upon activation, MST and LAST, transcriptional co-activators YAP and TAZ bind to the members of the TEA domain (TEAD) family transcription factors
- regulate expression of downstream target genes connective tissue growth factor (CTGF/CCN2) and cysteine-rich protein 61 (CYR61/CCN1).
- RANKL-mediated signaling cascades including NF-κB, MAPKs, AP1, and NFATc1, Hippo-signaling molecules such as YAP/TAZ/TEAD complex, RASSF2, MST2, and Ajuba could also potentially modulate osteoclast differentiation and function.
- Links: Hippo
Bone Marrow
- red marrow - mainly haematopoietic (myeloid) tissue, newborn has all red marrow
- yellow marrow - mainly fat cells, found in diaphysis region of long bones
- stromal cells - all other support cells not involved in haematopoiesis
- Links: blood
Marrow stroma components:
- osteoblasts - enclose the marrow compartment in bone tissue.
- endothelial and smooth muscle cells - organized into a complex vascular network composed of arterioles, capillaries, sinusoids, and a large central vein.
- nerves - sensory and sympathetic nerve fibres, glia, and perineural cells that innervate the marrow compartment to form a neural network.
- adipocytes - support metabolic functions of the bone marrow.
- stromal cells - support haematopoiesis and retain skeletal potential.
Bone Matrix
The bone matrix has 2 major components.
- Organic portion composed of mainly collagen Type 1 (about 95%) and amorphous ground substance.
- Inorganic portion (50% dry weight of the matrix) composed of hydroxyapatite crystals, calcium, phosphorus, bicarbonate, nitrate, Mg, K, Na.
- storage calcium and phosphate
- regulate blood calcium levels
Haversian Systems
- also called osteons
- Volkmann's canals - interconnect Haversian systems
Lamellae
- concentric - surrounding each Haversian System
- interstitial - bony plates that fill in between the haversian systems.
- circumferential - layers of bone that underlie the periosteum and endosteum
Cells
- osteocytes extending cytoplasmic processes into canaliculi
- Additional Histology images: low | medium | high
Molecular
The transcription factors Runx2 and Runx3 are essential for chondrocyte maturation, while Runx2 and Osterix are essential for osteoblast differentiation.
Osterix
Osterix (OSX) encodes a transcription factor containing three Cys2-His2 zinc-finger DNA-binding domains at its C terminus that has been shown to be essential for bone formation.
Abnormalities
Osteogenesis Imperfecta
Osteogenesis Imperfecta (OI, brittle bone disease) originally described as a collagen 1 gene mutation, but can have several different genetic causes and can be classified into eight different types (I-VIII).[16]
- COL1A1 and COL1A2 mutations
- CRTAP and LEPRE1 mutations, in severe/lethal and recessively inherited osteogenesis imperfecta
References
- ↑ 1.0 1.1 1.2 Xue S, Kemal O, Lu M, Lix LM, Leslie WD & Yang S. (2020). Age at attainment of peak bone mineral density and its associated factors: The National Health and Nutrition Examination Survey 2005-2014. Bone , 131, 115163. PMID: 31760214 DOI.
- ↑ Funato N. (2020). New Insights Into Cranial Synchondrosis Development: A Mini Review. Front Cell Dev Biol , 8, 706. PMID: 32850826 DOI.
- ↑ Kegelman CD, Nijsure MP, Moharrer Y, Pearson HB, Dawahare JH, Jordan KM, Qin L & Boerckel JD. (2020). YAP and TAZ promote periosteal osteoblast precursor expansion and differentiation for fracture repair. J. Bone Miner. Res. , , . PMID: 32835424 DOI.
- ↑ Wang L, Huang J, Moore DC, Song Y, Ehrlich MG & Yang W. (2019). SHP2 regulates intramembranous ossification by modifying the TGFβ and BMP2 signaling pathway. Bone , 120, 327-335. PMID: 30471432 DOI.
- ↑ Jha S, Laucis N, Kim L, Malayeri A, Dasgupta A, Papadakis GZ, Karantanas A, Torres M & Bhattacharyya T. (2018). CT analysis of anatomical distribution of melorheostosis challenges the sclerotome hypothesis. Bone , 117, 31-36. PMID: 30218789 DOI.
- ↑ Langen UH, Pitulescu ME, Kim JM, Enriquez-Gasca R, Sivaraj KK, Kusumbe AP, Singh A, Di Russo J, Bixel MG, Zhou B, Sorokin L, Vaquerizas JM & Adams RH. (2017). Cell-matrix signals specify bone endothelial cells during developmental osteogenesis. Nat. Cell Biol. , 19, 189-201. PMID: 28218908 DOI.
- ↑ Lin YC, Roffler SR, Yan YT & Yang RB. (2015). Disruption of Scube2 Impairs Endochondral Bone Formation. J. Bone Miner. Res. , 30, 1255-67. PMID: 25639508 DOI.
- ↑ Walzer SM, Cetin E, Grübl-Barabas R, Sulzbacher I, Rueger B, Girsch W, Toegel S, Windhager R & Fischer MB. (2014). Vascularization of primary and secondary ossification centres in the human growth plate. BMC Dev. Biol. , 14, 36. PMID: 25164565 DOI.
- ↑ Galli A, Robay D, Osterwalder M, Bao X, Bénazet JD, Tariq M, Paro R, Mackem S & Zeller R. (2010). Distinct roles of Hand2 in initiating polarity and posterior Shh expression during the onset of mouse limb bud development. PLoS Genet. , 6, e1000901. PMID: 20386744 DOI.
- ↑ Salazar VS, Gamer LW & Rosen V. (2016). BMP signalling in skeletal development, disease and repair. Nat Rev Endocrinol , 12, 203-21. PMID: 26893264 DOI.
- ↑ Percival CJ & Richtsmeier JT. (2013). Angiogenesis and intramembranous osteogenesis. Dev. Dyn. , 242, 909-22. PMID: 23737393 DOI.
- ↑ Uemura M, Nagasawa A & Terai K. (2016). Yap/Taz transcriptional activity in endothelial cells promotes intramembranous ossification via the BMP pathway. Sci Rep , 6, 27473. PMID: 27273480 DOI.
- ↑ Dwek JR. (2010). The periosteum: what is it, where is it, and what mimics it in its absence?. Skeletal Radiol. , 39, 319-23. PMID: 20049593 DOI.
- ↑ Yang W, Han W, Qin A, Wang Z, Xu J & Qian Y. (2018). The emerging role of Hippo signaling pathway in regulating osteoclast formation. J. Cell. Physiol. , 233, 4606-4617. PMID: 29219182 DOI.
- ↑ Xu W, Chen Q, Liu C, Chen J, Xiong F & Wu B. (2017). A novel, complex RUNX2 gene mutation causes cleidocranial dysplasia. BMC Med. Genet. , 18, 13. PMID: 28173761 DOI.
- ↑ Shapiro JR & Sponsellor PD. (2009). Osteogenesis imperfecta: questions and answers. Curr. Opin. Pediatr. , 21, 709-16. PMID: 19907330 DOI.
Reviews
32850793
Katsimbri P. (2017). The biology of normal bone remodelling. Eur J Cancer Care (Engl) , 26, . PMID: 28786518 DOI.
Berendsen AD & Olsen BR. (2015). Bone development. Bone , 80, 14-18. PMID: 26453494 DOI.
Takigawa M. (2013). CCN2: a master regulator of the genesis of bone and cartilage. J Cell Commun Signal , 7, 191-201. PMID: 23794334 DOI.
Long F & Ornitz DM. (2013). Development of the endochondral skeleton. Cold Spring Harb Perspect Biol , 5, a008334. PMID: 23284041 DOI.
Dwek JR. (2010). The periosteum: what is it, where is it, and what mimics it in its absence?. Skeletal Radiol. , 39, 319-23. PMID: 20049593 DOI.
Yang Y. (2009). Skeletal morphogenesis during embryonic development. Crit. Rev. Eukaryot. Gene Expr. , 19, 197-218. PMID: 19883365
Mackie EJ, Ahmed YA, Tatarczuch L, Chen KS & Mirams M. (2008). Endochondral ossification: how cartilage is converted into bone in the developing skeleton. Int. J. Biochem. Cell Biol. , 40, 46-62. PMID: 17659995 DOI.
Articles
Journals
- BONE is an interdisciplinary forum for the rapid publication of original articles and reviews on basic, translational, and clinical aspects of bone and mineral metabolism.
Search PubMed
Search Pubmed: Bone Development | developmental ossification | endochondral ossification | intramembranous ossification
Additional Images
Terms
| Bone Terms | ||
|---|---|---|
Bone Development
| ||
|
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- Virtual Slidebox of Histology (USA) Skeletal system
- e-radiography Ossification
- UWA Blue Histology bone
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, June 15) Embryology Musculoskeletal System - Bone Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Musculoskeletal_System_-_Bone_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G