Paper - A human embryo of two to three pairs of somites (1945)
| Embryology - 1 May 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Shaner RF. A human embryo of two to three pairs of somites. (1945) Canad. J. Res. 23: 235-243.
| Online Editor | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| This historic 1945 paper by Shaner describes a Carnegie Stage 9 human embryo of two to three pairs of somites.
Ingalls NW. A human embryo at the beginning of segmentation, with special reference to the vascular system. (1920) Contrib. Embryol., Carnegie Inst. Wash. Publ. 274, 11: 61-90. Bartelmez GW. The origin of the otic and optic primordia in man. (1922) J. Comp. Neural., 34: 201-232.
|
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
A Human Embryo of Two to Three Pairs of Somites
By Ralph F. Shaner
Contribution from the Department of Anatomy, University of Alberta, Edmonton, Alta. Professor of Anatomy.
- Manuscript received Jnty 28, 1945.
Abstract
A description is given of a human embryo with two to three pairs of somites and about 25 days old. In general the embryo tallies with the few other human embryos of the same stage hitherto described. It has a well developed head fold, but no tail fold. Its neural groove is everywhere open. There is a minute neurenteric canal, a long primitive streak, and long cloacal membrane. The last is degenerate in its caudal part. The optic primordium is faint, but a clear otic placode is present. Between the two extends a neural crest, the earliest on record. A slight fore-gut is present, ending in an oral membrane. There is a long tubular allantois. The notochord is strap-like, issuing from a Henson's node and ending in a prechordal plate. Two well developed pairs of somites appear with the beginning of a third. The mesoderm is split to contain two body cavities, which join beneath the fore-gut. The yolk sac is partly covered with blood islands. There are two definite umbilical arteries and traces of umbilical veins. Solid strands of angioblastic tissue beneath the fore-gut are the only signs of a heart. Scattered indications of body vessels also occur.
Introduction
The recent discovery of several human embryos less than two weeks old has attracted so much interest as to make one forget that embryos from the rest of the first month are still few in number. A short description of one embryo with two to three pairs of somites and a comparison of it with others of the same age may still be of value.
The human embryo, the subject of this paper, was found clinging to the external uterine orifice at a routine professional examination. All relevant clinical data are given in the following note from the physician, Dr. J. Ross Vant:
- “This young woman is in her twenties, pregnant for the first time, and her last period began March 6. Her cycle is approximately 31 days. She aborted April 16. I suppose she should have ovulated on or about the seventeenth day of her cycle, which would make the possible date of conception March 22. The age of the ovum would therefore be 25 +/- 2 days.”
To estimate the age of the embryo, Dr. Vant has followed Rock and Hertig (8), who put the ovulation date on the 14th day preceding the onset of the next period. In this instance ovulation would have taken place on the 17th day of a 31-day cycle.
The specimen as removed from the uterine orifice consisted of an intact chorionic vesicle loosely attached to a fragment of decidua. The vesicle was covered with villi except for one small area. After fixation in 10% formalin the vesicle was opened and a.n embryo found within, anchored by a small amount of magma. The embryo was taken out of the vesicle, several camera lucida sketches made of it, and it was then cut into 1G;u transverse sections, which were stained with haematoxylin and eosin. The chorion was trimmed rather too closely to the umbilical stalk. This and a slight gash in the yolk sac are the only defects in an otherwise excellent series.
General Description
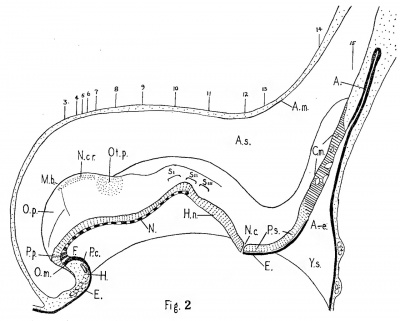
The general layout of the Vant embryo can be seen in Figs. 1 and 2. It has a well developed head fold, but no tail fold. The neural groove is everywhere open, and terminates at a distinct" neurenteric canal. Behind the canal there is a long primitive streak and still longer cloacal membrane. The only specialized parts of the yolk sac are a fore-gut and an allantois. A strap-like notochord is embedded in the roof of the yolk sac from Henson's node to the tip of the fore-gut.
The maximum length of the Vant embryo, measured along the curves of the sagittal reconstruction (Fig. 2) from the brain tip to the end of the primitive streak, is 1.5 mm. Of this the streak is 0.41 mm. or 27%. The straight line length is 1.2 mm. of which the primitive streak is 34%. These measurements are offered with the customary apologies for technical distortion but are nevertheless in accord with those of comparable embryos. The two to three somite embryo of Ingalls (3) has a straight line length of 1.38 mm., of which Bartelmez and Evans (1) allocate 26% to the primitive streak. The two to three somite embryo of Wilson (11) has a straight line length of 1.64 mm. of which the primitive streak is 29%. The two somite embryo of Ludwig (4) has a maximum length of 2 .4 mm. of which one—third is assigned to the primitive streak.
The Vant embryo has a deep concavity instead of the tail fold found in other embryos of the same age. I suspect the concavity is more than an artifact or simple variation. Nearly every section from the brain tip to the neurenteric canal shows at least one mitotic figure in the ectoderm; numerous sections just anterior to the first somite show as many as six figures. Behind the neurenteric canal, only eight sections show mitotic figures, and none more than two. The preservation and staining of the primitive streak and cloacal membrane suggest dying tissue. It is at least plausible that the embryo had ceased to grow in this critical region and had been aborted in consequence.
Fig. 1. Drawing, by Professor William Rowan of a wax model of the Vent embryo. Model X 180, drawing X 80. The dotted lines indicate the positions of the soniites, otic plaeodes, v and the neural crests. For other details compare with Fig. 2.
Fig. 2. Sagittal graphic reconstruction. X 60.
Key to abbreviations used in Figs. 2 to 20.
Nos. 3 to 15, position of sections shown in Figs. 3 to 15; A., allantois; A.e., allantoenteric diverticulum, Arn., amnion; A.s., amniotic sac; C.m., cloacal membrane; E., entoderm; F., fore-gut; H., heart; H.n., Henson’s node; m.b., midbrain bend; N., notochord; N.c., nenrenteric canal; N.cr., neural crest; O.m., oral membrane; O.p., optic primordium; 02!. p., otic placoole; P.c., pericardial cavity; P.p., precltordal plate; P.s., primitive streak; Y.s., yolk sac; S.I., II., III.,. somites; T.s., terminal sinus.
Special Structures Neural Groove
The neural groove is everywhere wide open, as in other embryos of the same age. A casual inspection of the model (Fig. 1) might suggest that closure is imminent at the second somite. Mitotic figures are most numerous just before the first somite, and there is considerable cell division in the ectoderm throughout the somite area. But the lips of the neural folds are still far apart throughout this area (Figs. 10 and 11), and there is really nothing to contradict the general belief that closure of the neural groove begins between the third and sixth s-omites in slightly older embryos.
Eye
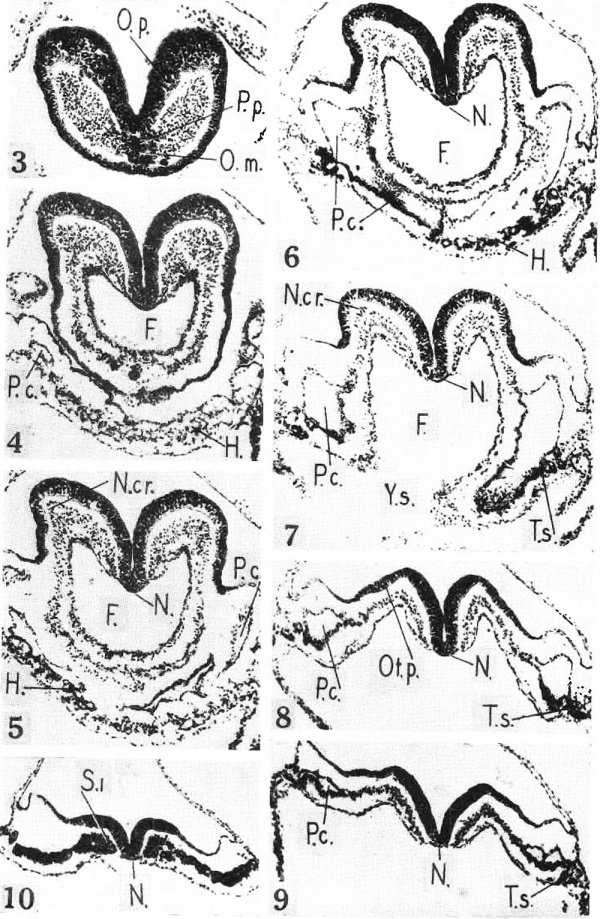
The anterior tips of the neural folds are slightly indented (Fig. 1), and are much thicker in section (Fig. 3) even when the effect of the plane of section is allowed for. These blunted tips are the optic primordia, according to Bartelmez and Evans (1). The left primordium bears a doubtful optic sulcus, about as well developed as in the Ludwig embryo. The optic primordia lie definitely within the limits of the neural tube.
Ear
The otic placode appears as a lenticular thickening just outside the neural folds (Figs. 1 and 8). The placode is about as definite as that of the Ingalls embryo as shown in Fig. 30 of Bartelmez and Evans (1). There is a suggestion of a placode in the Wlilson embryo. No otic placode is found in the comparable embryos of Ludwig (4) and Piersol (7).
Neural Crest
A strand of distinctive cells lies beneath the ectoderm between the optic and otic primordia (Figs. 1 and 2), forming a cap for the underlying mesoderm (Figs. 5 and 7, N. cr.). This I take to be the neural crest. Under low power (Fig. 7, N. cr.), the nuclei of the crest cells remind one of spermatozoa heads~ oval, bead—lil«:e, dark staining nuclei with scanty cytoplasmic processes. Under high power (Fig. 17) the neural crest blends with the mesoderm. The V ant embryo is the youngest human embryo so far reported with a neural crest. Bartelmez and Evans (1) begin their study of the neural crest with the seven somite stage.
Subdivisions of the Nervous System
The identification of the otic placode enables one to establish a few areas of the future brain. The sharp bend in the neural fold halfway between the otic placode and the brain tip marks the midbrain. Between the otic placode and the first somite the neural groove is wider. At this level may be put the rhombomere “C” of Bartelmez and Evans (1). Rhombomere “B” would lie between the two otic placodes. Rhornbomere “A” and the trigeminal area are still indefinite.
Fore-gut
The shallow fore-gut (Figs. 2 and 3 to 7) is tipped with a special clump of cells. The dorsal part of the clump (Figs. 3 and 16, P.,/b.) pertains to the notochord and is the prechordal plate. The ventral part (O.rn.) is fused with the underlying ectoderm to form a very short oral membrane. The dorsal surface of the fore-gut is deeply furrowed to accommodate the neural groove (Figs. 4 to 8). No pharyngeal pouch or thyroid gland can be made out. In general the Vant embryo takes a middle place among known two to three somite embryos. The Ludwig (4) and Fiersol (7) embryos have no oral membrane, pouch, or thyroid. The Wlilson (11) embryo has no oral membrane but does show traces of a first pouch and thyroid. The Ingalls (3) embryo has all three. The slightly older four somite embryos of Sternberg (9) and Orts Llorca (6) have definite first pouches.
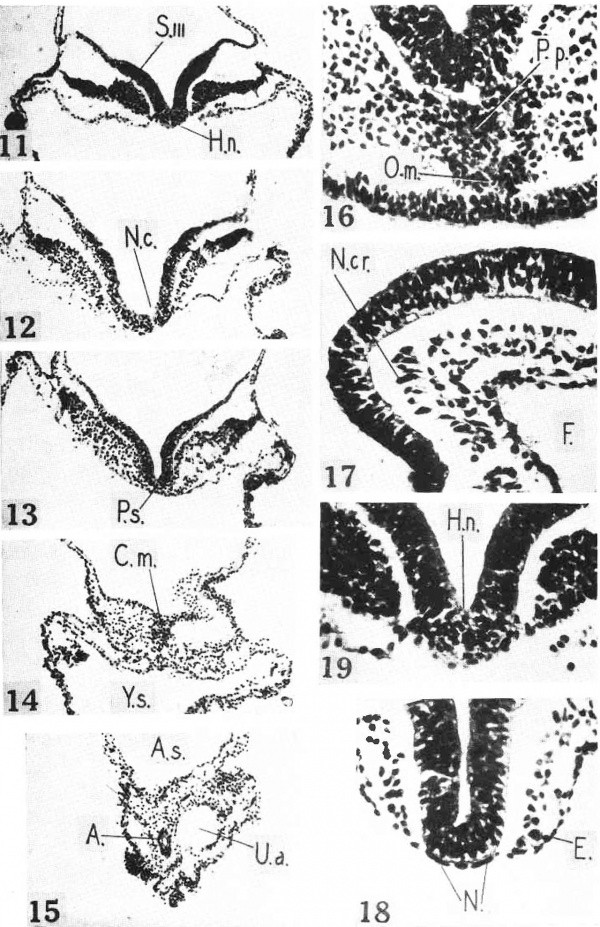
Figs. 3 to 10. Photornicrographs of sections at levels indicated in Fig. 2. Labels as in Fig. 2. X 98. Fig. 3. Section through prechordal plate, oral membrane, and optic prirnordium. Fig. 4. Section through fore—gut andvascular prirnordium of the heart. Fig. 5. Section through fore—gut and heart. Fig. 6. Section through fore—gut, heart, and pericardial cavity. Fig. 7. Section through fore-gut entrance, body cavities, and pre-otic neural crest. Fig. 6’. Section through otic placode. Fig. 9. Section through rhornboniere “C” and tips of body cavities. Fig. 10. Section through first somite.
Hind-gut and Allantois
The Vant embryo has no hind-gut, although one is present in other embryos of the same age. The yolk sac sends a funnel—shaped allantoenteric diverticulum beneath the cloacal membrane 2). From its tip extends a tubular allantois of moderate length. The allantois epithelium is thicker than that of the entoderm elsewhere (Fig. 15).
Henson’s Node, Notoehord, Prechorclal Plate
Between the neurenteric canal and the second somite the yolk sac and the overlying ectoderm are tied together by Henson’s node (Figs. 2, 11, and 19). From the node issues the notochord. The notochord is everywhere a flat ribbon inserted into the entoderm (Figs. 10 to 4). It clings to the overlying neural groove. Although continuous with the entoderm the notochord is easily distinguished from it. The notochord cells have rod—shaped nuclei (Fig. 18) and a cytoplasm that stains a little deeper with eosin. There is no trace of a notochordal canal. The notochord ends anteriorly in a clump of cells that is part oral membrane and part prechordal plate (Figs. 3 and 16). The prechordal plate blends with the head mesoderm.
The relations of Henson’s node, the notochord, and the prechordal plate to one another and to the mesoderm are matters of perennial discussion. My observations on a single human embryo can hardly outweigh the authoritative and exhaustive studies of Streeter (10) on the pig. Streeter derives the notochord entirely from Henson’s node and considers the node to exist for that purpose. In the Vant embryo the notochord certainly appears as if spun out of the node. Streeter is inclined to consider the prechordal plate a specialized entodermal structure and not a part of the notochord. The cells of the Vant prechordal plate are more like those of the entoderm than of the notochord (compare Figs. 16 and 18). Streeter derives the mesoderm almost entirely from the primitive streak and thinks the contact between the mesoderm and the node and notochord to be secondary or accidental, and that any mesodermal production from the prechordal plate is a supplementary one. In the Vant embryo the mesodermal sheets do stick to the node and notochord and nearby entoderm (Figs. 19 and 11 to 7) but there is nothing to indicate that these structures are adding to the mesoderm. On the other hand the blending of the prechordal plate with the head mesoderm is very intimate, so intimate as to make one suspect the mesoderm is receiving additions from this source. Perhaps the supplementary activity of the prechordal plate is greater in man than in the pig.
Neurenteric Canal
The neurenteric canal (Figs. 2 and 12) is included in a single section. The lumen may be plugged with cytoplasm; it is minute at any rate. The dorsal aperture is funnel-shaped and spread over the three following sections.
Primitive Streak
Behind the neurenteric canal lies the primitive streak (Fig. 2). A mid section is shown in Fig. 13. The mesoderm is everywhere streaming out from the under side of the ectoderm. The subjacent entoderm is fairly distinct. As already stated, the streak is not so well preserved and stained as other structures. It has the appearance of post-mortem fixation and staining.
Cloacal Membrama
Next behind the streak follows a long cloacal membrane. It extends the full length of the allantoenteric canal and overlaps the allantois (Fig. 2). It
shows the characteristic fusion of ectoderm and entoderm (Fig. 14), although exclusion of mesoderm is not easy. The anterior third of the membrane is a continuous structure, and poorly preserved like the primitive streak. The caudal two—thirds of the cloacal membrane are perforated in several places (Fig. 2) and show numerous dark staining degeneration granules—signs of an impending shortening of the membrane. A similar degeneration of the caudal part of the cloacal membrane occurs in the four somite embryos of Sternberg (9) and Florian (2). Florian has studied this area in detail and has shown that such a degeneration is normal. Extrophy of the bladder may be traced to some irregularity at this period.
Mesoderm and Somites
The mesoderm of the Vant embryo consists of two great para-axial sheets streaming out of the primitive streak (Fig. 13). At the edge of the embryonic disk each sheet splits to envelop the amnion and yolk sac. The mesoderm extends caudally into the umbilical stalk (Fig. 15). Traced forward the two sheets are loosely attached to Henson’s node (Fig. 11), and the notochord (e.g., Fig. 7). They envelop the fore—gut and fill the head fold (Figs. 6 to 3).
As Streeter states, the only really intimate connection of the mesoderm is with the primitive streak and with the prechordal plate.
There are two definite pairs of somites and the start of a third pair. The first pair lies just rostral to the union of the notochord and Henson’s node (Figs. 2 and 10). The first pair shows typical radial cell arrangement and a central cavity. The second pair lacks the central cavity on the right side. Each somite of the third pair (Fig. 11) is a simple heap of cells.
Body Cavities
Just anterior to the first somite each mesodermal sheet is split to form a pericardial cavity (Figs. 9, 8, 7). The cavities communicate beneath the fore-gut (Figs. 2 and 6) through a tiny transverse passage. Each cavity then extends forward (Figs. 5 and 4) as a blind recess beneath the ectoderm. The general shape of the two pericardial cavities is that of a pair of water Wings. The earlier isolated bits of body cavity have all, or nearly all, been absorbed into the general structure. The splanchnic layer beneath each pericardial cavity is thickened into a cardiogenic plate.
Vascular System and Heart
The vascular system of the Vant embryo has reached an indeterminate stage that baflies reconstruction. About two-thirds of the yolk sac is sprinkled with blood islands (Fig. 20). A large blood island on each side of the mouth of the allantois continues into a well defined umbilical artery (Figs. 20 and 15) that extends the full length of the allantois. Each vessel is a well defined tube containing free blood cells. Around the ends of the arteries are a cluster of irregular spaces that may be umbilical veins.
Fig. 20. Outline of eritire Vaut embryo to Show vascular system. B.i., blood islaud; U.a., umbilical artery; U.e., umbilical vein.
Over the anterior third of the yolk sac blood islands are replaced by a more continuous sheet of angioblastic tissue (Fig. 20), which forms a solid terminal sinus along the margin of the embryo proper (Figs. 20, 9, and 8). Each terminal sinus connects caudally with the blood islands from which the umbilical artery arises. Traced anteriorly the sinuses spread medially beneath the thickened mesoderm and then join beneath the transverse passage between the two pericardial cavities (H in Figs. 20, 6, 5, and 4). The spongy transverse strip of angioblastic tissue with the overlying cardiogenic thickening of the mesoderm constitutes the primordium of the heart.
There are also many traces of an angioblastic plexus inside the embryo, especially anterior to the neurenteric canal. There are spurs from the terminal sinuses and scattered cells that cling to the dorsal side of the entoderm (Figs. 10 to 5). The fore-gut has a collar of such cells, the forerunner of the first arterial arch. Scattered para-axial cells mark the course of the future dorsal aortae.
Taken as a whole, the vascular system of the Vant embryo falls between that of the presomite embryo of M’Intyre (5) and the Ludwig embryo (4). The outstanding feature of the vascular system of the Vant embryo is its sharp division into an anterior part converging on the heart and a posterior part converging on the umbilical arteries, and the decided precocity of the latter. The umbilical arteries are recognizable as such, but of the heart only source materials are present. The curious. precocity of the umbilical. arteries is found in most other human embryos of the same stage of growth.
General Considerations
As the detailed. comparisons have shown, the Vant embryo does not depart greatly from other embryos of the two to three somite stage. The Vant embryo, and those of Ingalls (3), Ludwig (4), Piersol (7), and Wilson (11) corroborate each other and give a definite and consistent picture of the human embryo at their stage of development.
Acknowledgments
I conclude with my great thanks to Dr. J. Ross Vant for the embryo that is the subject of this paper, to Mr. A. G. Fairall who prepared the serial sections of the embryo, and to Prof. W. Rowan who made the splendid drawing of the wax model reconstruction.
References
1. Bartelmez GW. and Evans HM. Development of the human embryo during the period of somite formation, including embryos with 2 to 16 pairs of somites. (1926) Contrib. Embryol., Carnegie Inst. Wash. Publ. 362, 17: 1-67.
2. Florian J. The formation of the connecting stalk and the extension of the amniotic cavity towards the tissue of the connecting stalk in young human embryos. (1930) J. Anat., 64: 454-476.
3. LUDWIG, E. Morphol. Jahrb. 59 :41-104. 1928.
4. M'Intyre D. Title. (1926) Trans. Roy Soc. 55: 77-113.
5. ORTS LLORCA, F. Z. Anat. Entwicklungsgeschichte, 103 : 765-792. 1934.
7. PIERSOL, W. H. Univ. Toronto Studies, Anat. Ser. 8 : 3-26. 1939.
8. Rock J. and Hertig AT. Information regarding the time of human ovulation derived from a study of 3 unfertilized and 11 fertilized ova. (1944) Amer. J. Obstet. Gynecol., 47: 343-356.
9. STERNBERG, H. Z. Anat. Entwicklungsgeschichte, 82 : 142-240. 1927.
10. STREETER, G. L. Carnegie Inst. Wash. Pub. Contrib. Embryol. 19 (100) : 73-92.
11. Wilson JT. Observations upon young human embryos. (1914) J Anat Physiol., 48(3): 315-51 PMID 17233002 PMC1288949
Cite this page: Hill, M.A. (2024, May 1) Embryology Paper - A human embryo of two to three pairs of somites (1945). Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_A_human_embryo_of_two_to_three_pairs_of_somites_(1945)
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G