Cat Development
| Embryology - 15 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
Cats (Felis catus) are seasonally polyestrous animals that have multiple estrous cycles only during certain periods of the year.
The cat genome was initially sequenced in 2007[1] and has been recently annotated in August 2014.[2]
| Animal Development: axolotl | bat | cat | chicken | cow | dog | dolphin | echidna | fly | frog | goat | grasshopper | guinea pig | hamster | horse | kangaroo | koala | lizard | medaka | mouse | opossum | pig | platypus | rabbit | rat | salamander | sea squirt | sea urchin | sheep | worm | zebrafish | life cycles | development timetable | development models | K12 |
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Cat Development | Cat Embryology | Feline Embryology |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Developmental Timeline
Twenty-two stages have been described for the prenatal development of the domestic cat.[9]
The following data on early development is based upon the time after copulation[10]
oviduct embryo development
- 64 hours - 1 to 4 cells (17 of 20; 85.0%)
- 76 hours - 5 to 8 cells (18 of 28; 64.3% )
- 100 hours - 9 to 16 cells (14 of 24; 58.3%)
- 124 hours - morulae (15 of 21; 71.4% )
uterine embryo development
- 148 hours - compact morulae or early blastocysts
- days 12-14 - implantation occurs
See also this historic paper on early cat development.
- 1924 Cat Development: 1. Ovum of the Cat | 2. Process of Cleavage | 3. Formation of the Blastocyst | 4. Discussion | Plates | cat
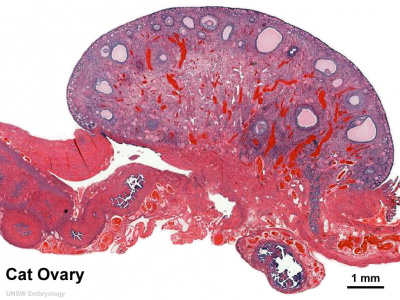
Cat Ovary
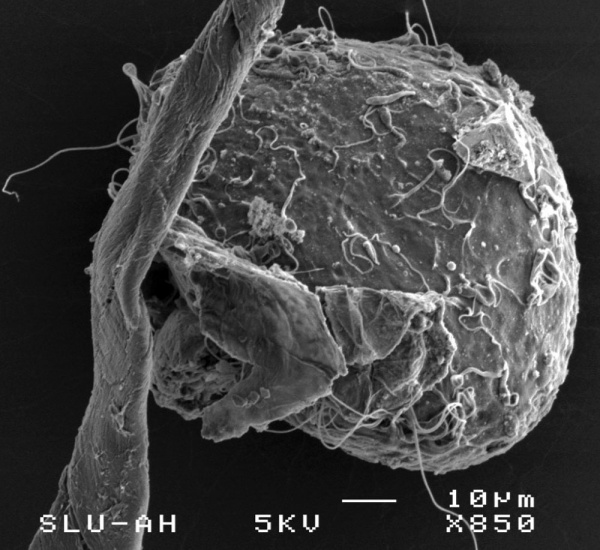
Oocyte and Spermatozoa
The following scanning electron micrographs are from a recent paper on fresh and frozen cat oocytes.[11] Scale bar is 10 microns.
Genetics
Lineage: Eukaryota; Opisthokonta; Metazoa; Eumetazoa; Bilateria; Coelomata; Deuterostomia; Chordata; Craniata; Vertebrata; Gnathostomata; Teleostomi; Euteleostomi; Sarcopterygii; Tetrapoda; Amniota; Mammalia; Theria; Eutheria; Laurasiatheria; Carnivora; Feliformia; Felidae; Felinae; Felis; Felis catus
The cat genome was initially sequenced in 2007[1] and has been recently annotated in August 2014.[12]
- Mitochondria - entire mitochondrial genome 17,009 bp has been sequenced.
- Links: Genome Mitochondrial Genome
Renal Development
The time course data below is based on a recent cat study.[3]
- 15 to 19 days - pronephros is present at early stages in embryos with the presence of pronephro's corpuscles, ducts and tubules.
- 17 and 37 days - mesonephros contains mesonephric ducts, mesonephric tubules, and glomeruli.
- 21 days - metanephros with the presence of glomeruli, proximal and distal contorted tubules
- 37 days - cortex-medullary region is differentiated.
- Links: renal
Placenta
- zonary placenta without cotyledons
- relatively small marginal hematoma
- materno-fetal barrier is endothelial-chorial
- superficially invasive into the endometrium but not into the myometrium
- placental labryrinth has characteristic giant cells
Placental cord
- two pairs of vessels in the cord
- two arteries and two veins
- allantoic duct
- cord average length 2 to 3 cm and 0.3 to 0.5 cm in diameter
- inserts at the margin of the zonary organ
- no spirals, no vitelline duct, and no additional vessels or structures
Additional Images
Historic Images
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
References
- ↑ 1.0 1.1 Pontius JU, Mullikin JC, Smith DR, Lindblad-Toh K, Gnerre S, Clamp M, Chang J, Stephens R, Neelam B, Volfovsky N, Schäffer AA, Agarwala R, Narfström K, Murphy WJ, Giger U, Roca AL, Antunes A, Menotti-Raymond M, Yuhki N, Pecon-Slattery J, Johnson WE, Bourque G, Tesler G & O'Brien SJ. (2007). Initial sequence and comparative analysis of the cat genome. Genome Res. , 17, 1675-89. PMID: 17975172 DOI.
- ↑ Tamazian G, Simonov S, Dobrynin P, Makunin A, Logachev A, Komissarov A, Shevchenko A, Brukhin V, Cherkasov N, Svitin A, Koepfli KP, Pontius J, Driscoll CA, Blackistone K, Barr C, Goldman D, Antunes A, Quilez J, Lorente-Galdos B, Alkan C, Marques-Bonet T, Menotti-Raymond M, David VA, Narfström K & O'Brien SJ. (2014). Annotated features of domestic cat - Felis catus genome. Gigascience , 3, 13. PMID: 25143822 DOI.
- ↑ 3.0 3.1 Mario LC, Morais MP, Borghesi J, Favaron PO, Oliveira FD, Anunciação ARA, Agopian RG, Gomes SA & Miglino MA. (2018). Development of urinary organs in domestic cat during the embryonic and fetal periods. Microsc. Res. Tech. , , . PMID: 30341968 DOI.
- ↑ Johnson AK. (2018). Assisted Reproduction in the Female Cat. Vet. Clin. North Am. Small Anim. Pract. , 48, 523-531. PMID: 29656770 DOI.
- ↑ Okotrub KA, Mokrousova VI, Amstislavsky SY & Surovtsev NV. (2018). Lipid Droplet Phase Transition in Freezing Cat Embryos and Oocytes Probed by Raman Spectroscopy. Biophys. J. , 115, 577-587. PMID: 30099990 DOI.
- ↑ Malandain E, Rault D, Froment E, Baudon S, Desquilbet L, Begon D & Chastant-Maillard S. (2011). Follicular growth monitoring in the female cat during estrus. Theriogenology , 76, 1337-46. PMID: 21798582 DOI.
- ↑ Inomata T, Ariga M, Sakita K, Kashiwazaki N, Ito J, Yokoh K, Ichikawa M, Ninomiya H & Inoue S. (2009). Development of external genitalia in fetal and neonatal domestic cats. J. Vet. Med. Sci. , 71, 139-45. PMID: 19262023
- ↑ Wang C, Swanson WF, Herrick JR, Lee K & Machaty Z. (2009). Analysis of cat oocyte activation methods for the generation of feline disease models by nuclear transfer. Reprod. Biol. Endocrinol. , 7, 148. PMID: 20003339 DOI.
- ↑ Knospe C. (2002). Periods and stages of the prenatal development of the domestic cat. Anat Histol Embryol , 31, 37-51. PMID: 11841356
- ↑ Swanson WF, Roth TL & Wildt DE. (1994). In vivo embryogenesis, embryo migration, and embryonic mortality in the domestic cat. Biol. Reprod. , 51, 452-64. PMID: 7803616
- ↑ Hermansson U, Axnér E & Holst BS. (2007). Application of a zona pellucida binding assay (ZBA) in the domestic cat benefits from the use of in vitro matured oocytes. Acta Vet. Scand. , 49, 28. PMID: 17908298 DOI.
- ↑ Tamazian, G. etal., Annotated features of domestic cat - Felis cats genome. GigaScience 2014, 3:13
Reviews
Glatzle M, Hoops M, Kauffold J, Seeger J & Fietz SA. (2017). Development of Deep and Upper Neuronal Layers in the Domestic Cat, Sheep and Pig Neocortex. Anat Histol Embryol , 46, 397-404. PMID: 28677231 DOI.
Articles
Piras AR, Ariu F, Zedda MT, Paramio MT & Bogliolo L. (2020). Selection of Immature Cat Oocytes with Brilliant Cresyl Blue Stain Improves In Vitro Embryo Production during Non-Breeding Season. Animals (Basel) , 10, . PMID: 32847086 DOI.
Prozorowska E, Ratajczak M & Jackowiak H. (2019). Ultrastructural study of uterine epithelium in the domestic cat during prenatal development. Theriogenology , 130, 49-61. PMID: 30865874 DOI.
Prozorowska E, Jackowiak H & Skieresz-Szewczyk K. (2018). Morphology and topography of internal reproductive organs in the female cat during prenatal and postnatal development: Scanning electron microscope and three-dimensional reconstruction study. J. Morphol. , 279, 1764-1775. PMID: 30443927 DOI.
Inomata T, Ninomiya H, Sakita K, Kashiwazaki N, Ito J, Ariga M & Inoue S. (2009). Developmental changes of Müllerian and Wolffian ducts in domestic cat fetuses. Exp. Anim. , 58, 41-5. PMID: 19151510
Inomata T, Ariga M, Sakita K, Kashiwazaki N, Ito J, Yokoh K, Ichikawa M, Ninomiya H & Inoue S. (2009). Development of external genitalia in fetal and neonatal domestic cats. J. Vet. Med. Sci. , 71, 139-45. PMID: 19262023
Ciani F, Cocchia N, Rizzo M, Ponzio P, Tortora G, Avallone L & Lorizio R. (2008). Sex determining of cat embryo and some feline species. Zygote , 16, 169-77. PMID: 18405438 DOI.
França LR & Godinho CL. (2003). Testis morphometry, seminiferous epithelium cycle length, and daily sperm production in domestic cats (Felis catus). Biol. Reprod. , 68, 1554-61. PMID: 12606460 DOI.
Knospe C. (2002). Periods and stages of the prenatal development of the domestic cat. Anat Histol Embryol , 31, 37-51. PMID: 11841356
Hill JP. and Tribe M. The early development of the cat (Felis domestica). (1924) Quart. J. Microsc. Sci., 68: 513-602.
Search Pubmed
cat development | feline development
| Animal Development: axolotl | bat | cat | chicken | cow | dog | dolphin | echidna | fly | frog | goat | grasshopper | guinea pig | hamster | horse | kangaroo | koala | lizard | medaka | mouse | opossum | pig | platypus | rabbit | rat | salamander | sea squirt | sea urchin | sheep | worm | zebrafish | life cycles | development timetable | development models | K12 |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 15) Embryology Cat Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Cat_Development
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G