ASA Meeting 2013 - Placenta
Placenta Embryology and Circulation
| Australian Sonographers Association (ASA) Annual Conference 2013
Educate Innovate Celebrate
|

|
Abstract
Sonographic analysis of the placenta and uterus and their associated blood flow are key diagnostic prenatal assessments in human development. This review will provide an overview of the basic biology of the placentation process and key events in the developmental timeline. This talk should be of value to those wanting a better understanding of the process of human haemochorial placentation.
Placentation begins at the implantation site in the second week of development (GA week 4) with conceptus trophoblast cells invading the maternal endometrial epithelium and stroma. From that time on the process of placentation involves complex interactions between maternal uterine and fetal tissues. While there are many animal models of this process, none currently exactly match that seen in humans.
Maternally, these changes include modification of the maternal vascular, endocrine and immune response. Fetally, an entire organ is grown from extra-embryonic tissue that has many functions outside of acting as a simple exchange tissue. The main maternal vascular changes include increased vascularity and trophoblast modification of spiral arteries. The fetal vascular bed consists of large cord vessels and an exponentially growing capillary bed consisting of kilometres of villous capillaries. Identification of cord vessel number, size and blood flow, are important indices of normal fetal development. Furthermore extensive remodeled of the capillary bed occurs throughout development, with some villi morphologies influencing the efficiency of diffusional gas exchange. Clinically, abnormalities of placentation site, placental development, function and blood flow can have both maternal and fetal ramifications.
The full presentation and additional information/resources/links is available online (http://php.med.unsw.edu.au/embryology/index.php?title=ASA_Meeting_2013_-_Placenta).
Introduction
This talk is intended to provide a broad introduction to placentation and its associated vascular development. This is such an interesting, and both clinically and diagnostically relevant topic in sonography.
My talk will go through a time-course of development from earliest implantation through to the term placenta, specifically related to vascular development. Given that this process takes about 9 months, I will pick some key events in my 20 minute talk.
I have designed this online resource for ongoing "self-directed" learning after the conference.
As always, I welcome feedback and questions from my readers and encourage you to contact me with any potential educational materials that you would like to share.
--Mark Hill 09:14, 10 May 2013 (EST)
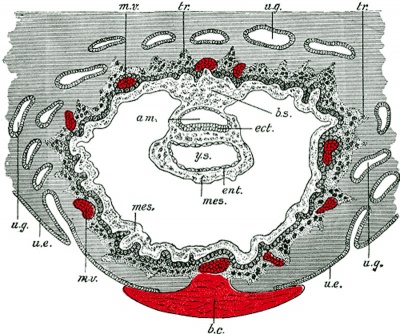
Embryo GA Week 4 (Week 2)
| <html5media height="520" width="360">File:Week2_001.mp4</html5media> |
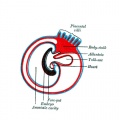
This animation shows the process of implantation, occurring during week 2 of development in humans. The beginning of the animation shows adplantation to the the uterus lining (endometrium epithelium). The hatched blastocyst with a flat outer layer of trophoblast cells (green), the inner cell mass which has formed into the bilaminar embryo (epiblast and hypoblast) and the large fluid-filled space (blastocoel).
|
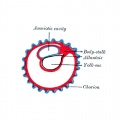
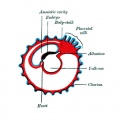
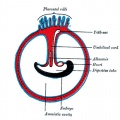
| <html5media height="260" width="220">File:Chorion 001.mp4</html5media> | Animation shows the events following implantation and focuses on changes in the the spaces surrounding the embryonic disc, the extraembryonic coelom.
The blastoceol cavity is converted into two separate spaces: the yolk sac and the chorionic cavity. The third space lies above the epiblast layer of the embryonic disc, the amniotic cavity.
|
Early embryo membrane development cartoons: Image 24 | Image 25 | Image 26 | Image 27 | Image 28
Embryo GA Week 5 (Week 3)
| Week 2 (GA 4) | Week 3 (GA 5) | Week 4 (GA 6) | ||||||
|---|---|---|---|---|---|---|---|---|
|
|
| ||||||
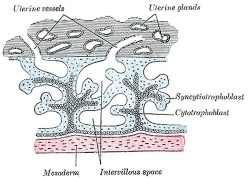
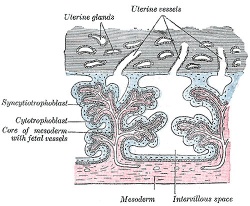
| Primary villi | Secondary villi | Tertiary villi | ||||||
| first stage of chorionic villi development, trophoblastic shell cells (syncitiotrophoblasts and cytotrophoblasts) form finger-like extensions into maternal decidua. | second stage of chorionic villi development, extraembryonic mesoderm grows into villi, covers entire surface of chorionic sac. Basal region will form chorionic plate. | third stage of chorionic villi development, mesenchyme differentiates into blood vessels and cells, forms arteriocapillary network, fuse with placental vessels, developing in connecting stalk. | ||||||
|
|
| Placental Classification |
|---|
|
Classification of placenta is on the basis of histological (microscopic) structural organization and layers between fetal and maternal circulation. Three main groups:
The presence of these three differing types of placenta have also been used to describe the pattern mammalian evolution. |
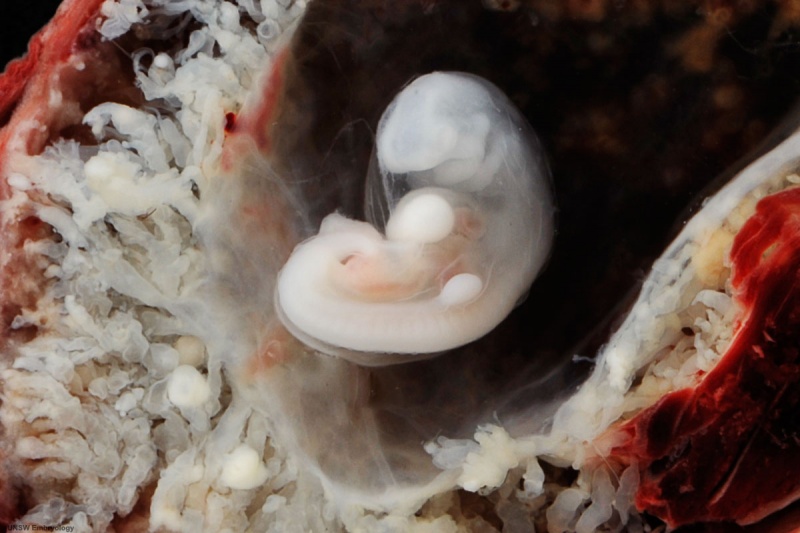
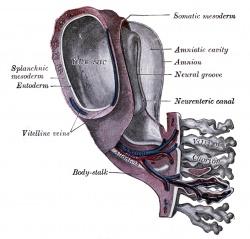
Embryo GA Week 7 (Week 5)
- Embryo Links: Embryo and Placenta | Embryo | Embryo (label) | Embryo (animated) | Embryo (animated large) | Head region | Head region (label) | Body region | Body region (label) | Carnegie stage 13
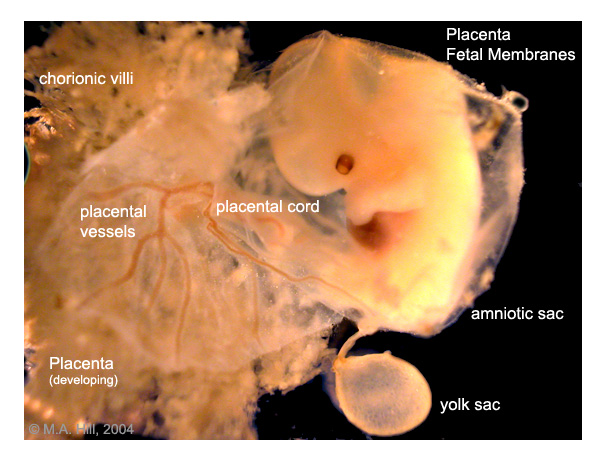
Embryo GA Week 9 (Week 7)
|
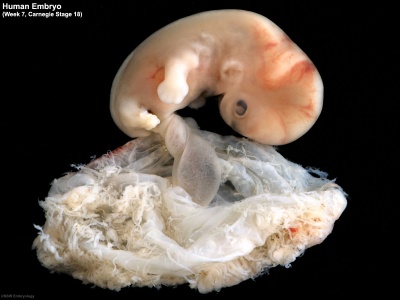
|
| Embryo in gestational sac | Embryo open sac |
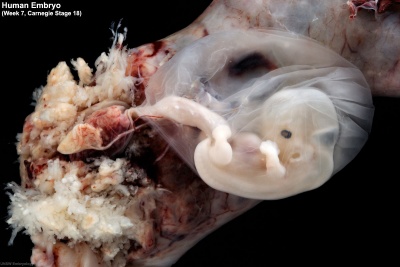
|
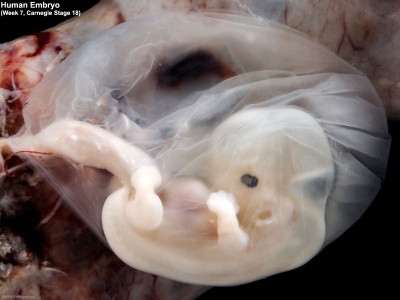
|
| Embryo with placentation (ectopic) | Embryo in amniotic sac |
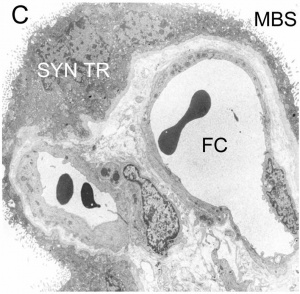
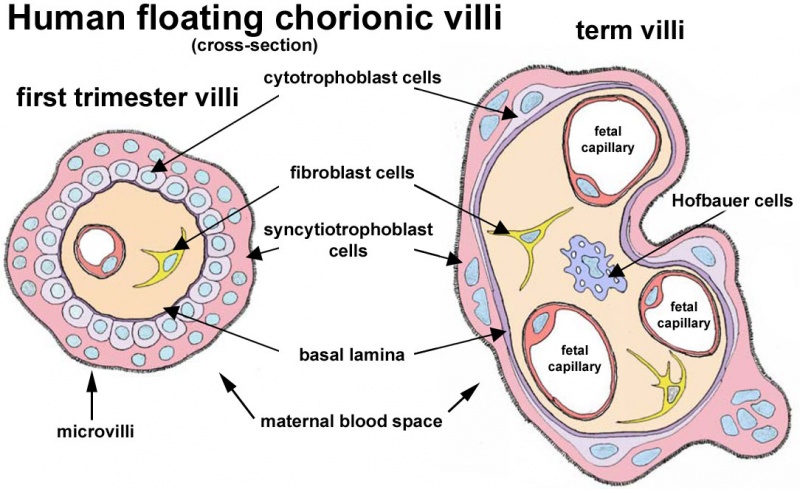
Villi Development
| First Trimester | Third Trimester |
|---|---|
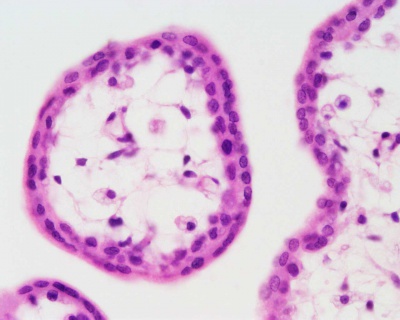
|
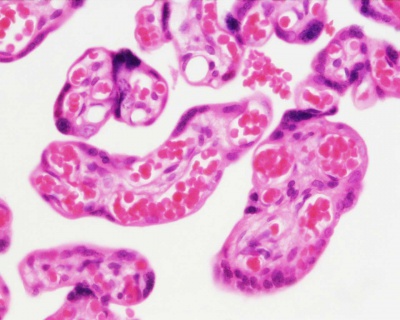
|
| Secondary Villi | Tertiary Villi |
|---|---|
|
|
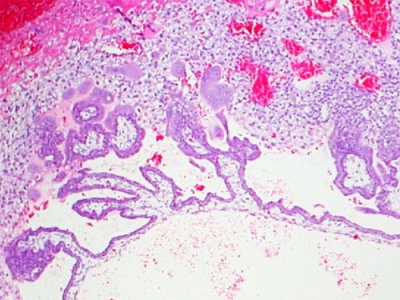
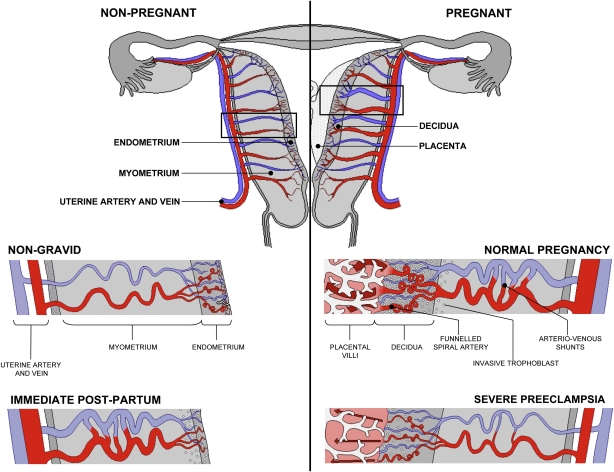
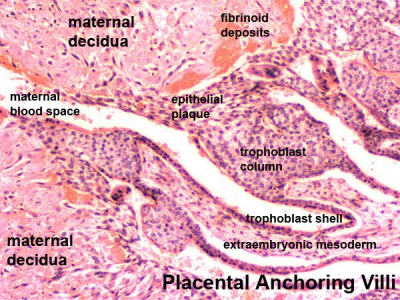
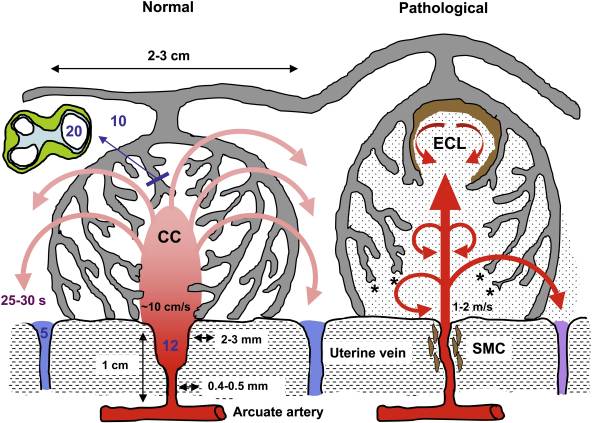
Maternal Blood Vessels
Uterine and Placental Vasculature in Non-pregnant, Pregnant and immediate Post-partum State
Diagrammatic representation of uterine and placental vasculature (red shading = arterial; blue shading = venous) in the non-pregnant, pregnant and immediate post-partum state.
| More figure information |
|---|
|
| Maternal Decidua chances |
|---|
FibrinoidExist as 2 forms of extracellular matrix:
Fibrinoid layer (Nitabuch's layer) is thought to act to prevent excessively deep implantation. DecidualizationProcess of endometrial stromal cells (fibroblast-like) change in morphology (polygonal cells) and protein expression and secretion (specific decidual proteins: prolactin, insulin-like growth factor binding protein-1, tissue factor, interleukin-15, and VEGF).
Artery DilatationDue to extravillous trophoblast cells invading uterine wall and maternal spiral arteries replacing both smooth muscle with fibrinoid material and part of vessel endothelium. There is also a proliferation of maternal blood vessels. Other changes
Preeclampsia
|
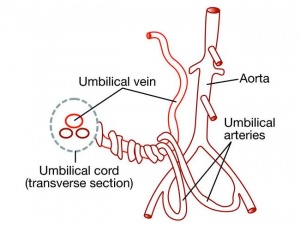
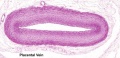
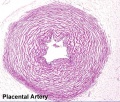
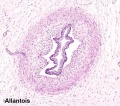
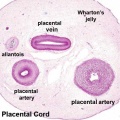
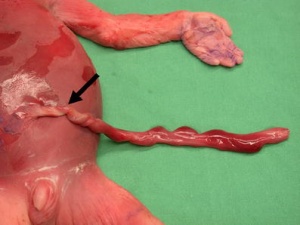
Placental Cord
Histology
Cord Length
The following are lengths and classifications at term.
- Normal range - 50 to 60 cm.
- Short cord - less than 35 cm.
- Long cords - over 70 cm can be associated with wrapping around the fetus and other abnormalities.
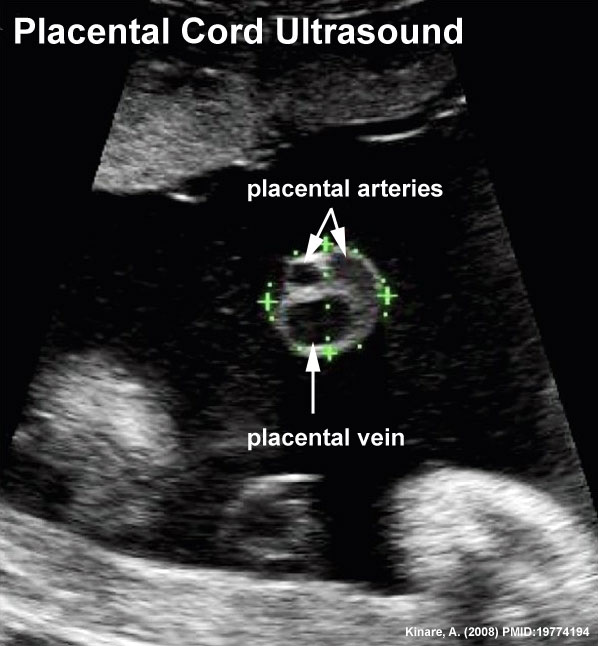
Ultrasound
Ultrasound image of transverse scan through the cord show the method of estimation of the cross-sectional area.
| Vessel Anomalies |
|---|
Persistent Right Umbilical VeinA study of 15,237 obstetric ultrasound examinations performed after 15 weeks' gestation identified only 33 cases of persistent right umbilical vein. <pubmed>7970470</pubmed> One artery and one veinIntra-abdominal umbilical vein varixA focal dilatation of the fetal intra-abdominal portion of the placental vein. |
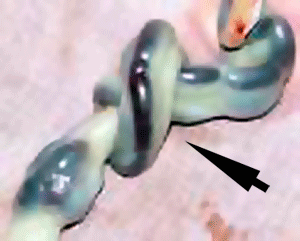
| Cord Coiling and Knotting |
|---|
HypocoilingAssociated with increased incidence of fetal demise, intrapartum fetal heart rate decelerations, operative delivery for fetal distress, anatomic-karyotypic abnormalities and chorio-amnionitis. HypercoilingAssociated with increased incidence of fetal growth restriction, intrapartum fetal heart rate decelerations, vascular thrombosis and cord stenosis. Cord KnottingCord knotting can occur (1%) in most cases these knots have no effect, in some cases of severe knotting this can prevents the passage of placental blood. Cord TorsionRare umbilical cord torsion, even without knot formation can also affect placental blood flow, even leading to fetal demise. |
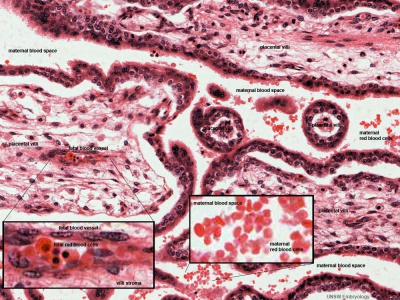
Exchange

|
|
| Crossing the placenta |
|---|
|
Term Placenta
Maternal and Fetal Side
Placenta Vasculature - MRI and CT
Term placenta viewed from the fetal side.[1]
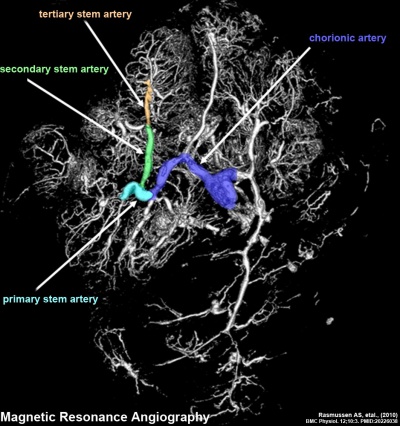
|
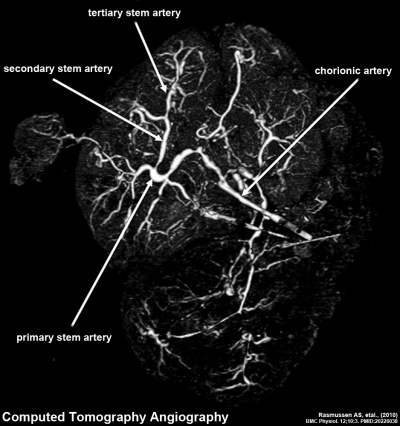
|
| Magnetic Resonance Angiography (MRA) | Computed Tomography Angiography (CTA) |
Legend
|
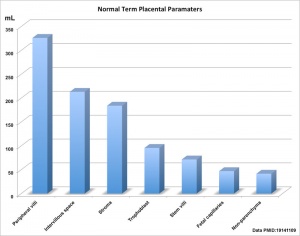
Placenta Measurements
Morphometric indices at term of placental composition, villous capillarization and the mean cross-sectional areas of peripheral villi and capillaries.
| Variable | Unit | Placenta |
| Intervillous space | mL | 213 |
| Stem villi | mL | 71.4 |
| Peripheral villi | mL | 326 |
| Trophoblast | mL | 95.5 |
| Stroma | mL | 184 |
| Fetal capillaries | mL | 46.9 |
| Non-parenchyma | mL | 41.5 |
| Peripheral villi | km | 89.2 |
| Fetal capillaries | km | 310 |
| TS area villi | µm2 | 3700 |
| TS area capillary | µm2 | 150 |
| Capillaries | mL mL-1 | 0.147 |
| Length ratio | km km-1 | 3.6 |
Data from a study sample of 15 normal placenta, mean placental volume, 652 ml.[2][3]
| Measurement methodology |
|---|
|
Abnormalities
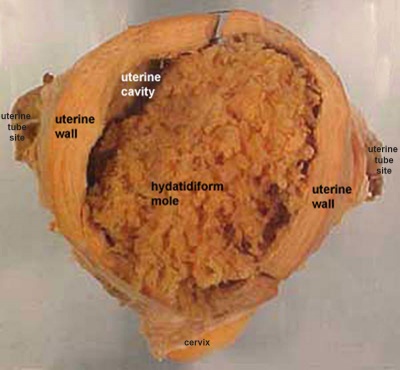
Hydatidiform Mole
Hydatidiform Mole
When only the conceptus trophoblast layers proliferates and not the embryoblast, no embryo develops, this is called a "hydatidiform mole" (HM), which is due to the continuing presence of the trophoblastic layer, this abnormal conceptus can also implant in the uterus.
| More information |
|---|
|
The trophoblast cells will secrete human chorionic gonadotropin (hCG), as in a normal pregnancy, and may appear maternally and by pregnancy test to be "normal". Prenatal diagnosis by ultrasound analysis demonstrates the absence of a embryo. There are several forms of hydatidiform mole: partial mole, complete mole and persistent gestational trophoblastic tumor. Many of these tumours arise from a haploid sperm fertilizing an egg without a female pronucleus (the alternative form, an embryo without sperm contribution, is called parthenogenesis). The tumour has a "grape-like" placental appearance without enclosed embryo formation. Following a first molar pregnancy, there is approximately a 1% risk of a second molar pregnancy.
|
| Velamentous Cord Insertion of the Placenta | |
|---|---|
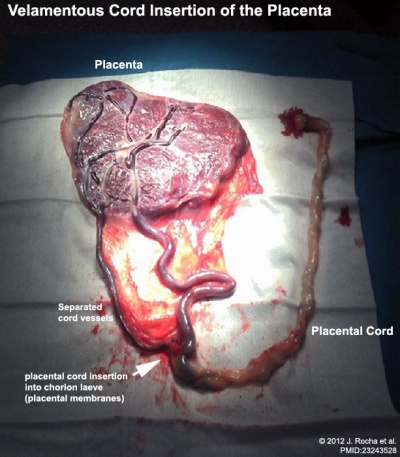
|
The placental cord inserts into the chorion laeve (placental membranes) away from the edge of the placenta.[4]
The placental vessels are therefore unprotected by Wharton's jelly where they traverse the membranes before they come together into the umbilical cord. This can cause hemorrhage if the vessels are damaged when the membranes are ruptured prior to birth. The condition is more common in monozygotic twins (15%) and triplets. |
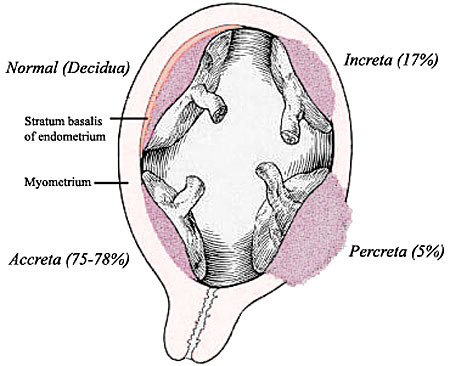
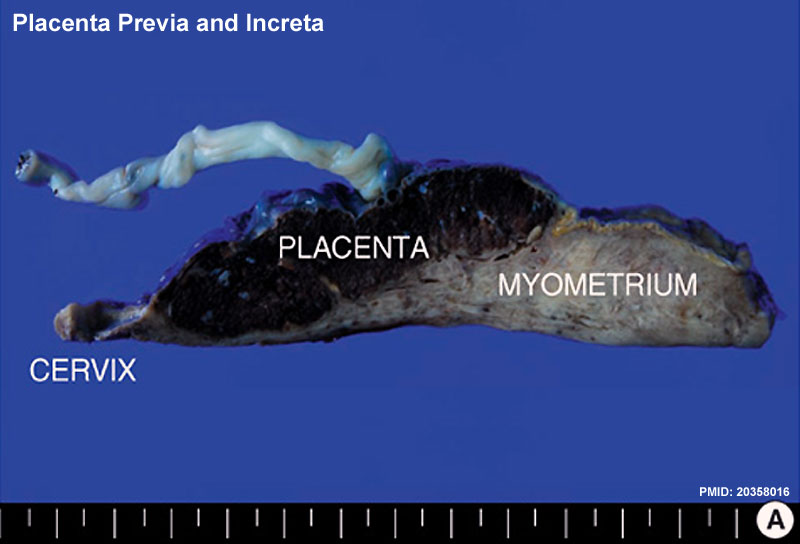
Implantation Abnormalities
Placenta previa and increta
| Implantation abnormalities |
|---|
|
Online Placenta Links
| Historic Embryology - Placenta |
|---|
| 1883 Embryonic Membranes | 1907 Development Atlas | 1909 | 1910 Textbook | 1917 Textbook | 1921 Textbook | 1921 Foetal Membranes |1921 human | 1921 Pig implantation | 1922 Single placental artery | 1923 Placenta Review | 1939 umbilical cord | 1943 human and monkey | 1944 chorionic villus and decidua parietalis | 1946 placenta ageing | 1960 monkey | 1972 Placental circulation | Historic Disclaimer |
References
- ↑ Rasmussen AS, Lauridsen H, Laustsen C, Jensen BG, Pedersen SF, Uhrenholt L, Boel LW, Uldbjerg N, Wang T & Pedersen M. (2010). High-resolution ex vivo magnetic resonance angiography: a feasibility study on biological and medical tissues. BMC Physiol. , 10, 3. PMID: 20226038 DOI.
- ↑ Mayhew TM, Jenkins H, Todd B & Clifton VL. (2008). Maternal asthma and placental morphometry: effects of severity, treatment and fetal sex. Placenta , 29, 366-73. PMID: 18328557 DOI.
- ↑ Mayhew TM. (2009). A stereological perspective on placental morphology in normal and complicated pregnancies. J. Anat. , 215, 77-90. PMID: 19141109 DOI.
- ↑ Rocha J, Carvalho J, Costa F, Meireles I & do Carmo O. (2012). Velamentous cord insertion in a singleton pregnancy: an obscure cause of emergency cesarean-a case report. Case Rep Obstet Gynecol , 2012, 308206. PMID: 23243528 DOI.
Books
- Wang Y, Zhao S. Vascular Biology of the Placenta. San Rafael (CA): Morgan & Claypool Life Sciences; 2010. Available from: http://www.ncbi.nlm.nih.gov/books/NBK53247
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, April 21) Embryology ASA Meeting 2013 - Placenta. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/ASA_Meeting_2013_-_Placenta
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G