2017 Group Project 3: Difference between revisions
| Line 276: | Line 276: | ||
===Ventricular Septal Defect=== | ===Ventricular Septal Defect=== | ||
[[File:Ventricular Septal Defect (VSD).png|400px|thumb|right|'''Figure :''' Ventricular Septal Defect (as viewed from the left ventricle)]] | |||
Ventricular septal defects are the most prevalent of all congenital heart defects (excluding patent foramen ovales), accounting for approximately 40% of all cardiac abnormalities among newborns.<ref name="PMID21349577><pubmed>21349577</pubmed></ref> The exact definition of a VSD is still a point of contention within the scientific community, but is generally referred to as a communication between the left and right ventricles; this anomaly can manifest as a lone defect, but is often present with other abnormalities such as Tetralogy of Fallot or a complete AVSD.<ref name="PMID4316658><pubmed>PMC4316658</pubmed></ref> occur as a result of abnormal development (or absence) of any of the components forming the interventricular septum.<ref name="PMID7437181><pubmed>7437181</pubmed></ref> Hence, a VSD can occur in either the muscular or the membranous portion of the septum, with the latter being more common.<ref name="PMID7437181"/> | Ventricular septal defects are the most prevalent of all congenital heart defects (excluding patent foramen ovales), accounting for approximately 40% of all cardiac abnormalities among newborns.<ref name="PMID21349577><pubmed>21349577</pubmed></ref> The exact definition of a VSD is still a point of contention within the scientific community, but is generally referred to as a communication between the left and right ventricles; this anomaly can manifest as a lone defect, but is often present with other abnormalities such as Tetralogy of Fallot or a complete AVSD.<ref name="PMID4316658><pubmed>PMC4316658</pubmed></ref> occur as a result of abnormal development (or absence) of any of the components forming the interventricular septum.<ref name="PMID7437181><pubmed>7437181</pubmed></ref> Hence, a VSD can occur in either the muscular or the membranous portion of the septum, with the latter being more common.<ref name="PMID7437181"/> | ||
Revision as of 13:48, 21 October 2017
| 2017 Student Projects | |||
|---|---|---|---|
|
Heart
Introduction
The cardiovascular system is the first system to develop and function in the human embryo. Rapid cardiac development is essential as the growing embryo can no longer receive oxygen and essential nutrients via diffusion alone, hence a circulatory system and a contractile heart mechanism is required to supply the embryo. We recognise the hearts normal development is vital for foetal life, and hence we have chosen to document the development of the heart from gastrulation to birth. Any defects occurring during the developmental processes can lead to congenital heart abnormalities. However, early cardiac development is a multifaceted procedure and is associated with other developmental processes such as: embryonic folding, coelom formation, and vascular development [1].
Through researching the advances in technology, coupled with the biological use of suitable animal models [2] our understanding of embryological cardiac development has evolved, and we are piecing together the mechanism underlying this development. This page will outline the importance of how heart abnormalities arise, the treatments available and the possible treatments to be developed in the future. Due to the major knowledge gaps in current embryological heart research, we acknowledge that this will impact our assignment, and aim to address further research concepts that will improve our understanding.
Anatomy of the Heart
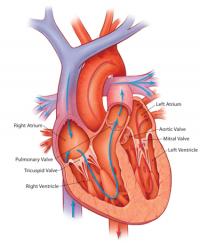
The heart is the muscular organs that pumps blood around the body via the circulatory system. It is located within the thoracic cavity, in a compartment called the mediastinum. The heart is divided into four chambers including: left and right atria and ventricles which are compartmentalised by semilunar and atrioventricular valves. Blood moves via the systemic circuit to the organs of the body and back to the heart. The pulmonary circuit is responsible for the flow of blood between the lungs and the heart. Deoxygenated blood enters the right side of the heart, while Oxygenated blood returning from the lungs exits the left side. The heart’s electrical system uses electrical signals to cause the muscular walls to contract. The mechanical pumping of the heart is essential for movement of blood which exchanges gases and essential nutrients between organs of the body[3].
Developmental Origin
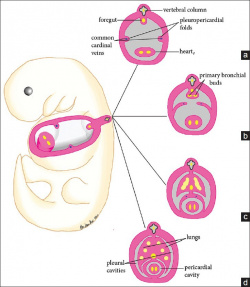
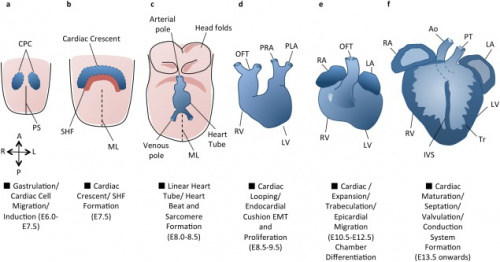
In the developing embryo the lateral plate mesoderm splits into somatic and splanchnic layers, the latter is comprised of cardiac progenitor cells [4] . The somatic mesoderm lines the ectoderm, the splanchnic mesoderm lines the endoderm, and in between lies an embryonic coelom [5] . At the end of week three, the heart develops from splanchnic mesoderm of the cardiogenic region of the embryonic plate. At the cranial end of the embryo, anterior to the developing neural tube is the initial origin of heart formation.
Originally, the cardiogenic region forms laterally of the paraxial mesoderm and primitive streak of the embryonic disc. Due to the natural mesenchymal cell organisation of the mesoderm, cells ‘migrate’ and fuse at the midline of the cranial end of the embryo forming the cardiac crescent just prior cardiac tube formation and folding. Endoderm surrounding the primitive gut contracts bringing the cardiogenic precursor regions of the splanchnic region towards the midline. Early heart formation begins with angioblastic cords of the splanchnic mesoderm. The angioblastic cords develop into separate endochondral heart tubes moving closer together as the foregut pinches together and the yolk sac contracts into the embryo. Fusion of the two heart tubes is facilitated by apoptosis [6]. The origin of the primitive heart is in the early pericardial coelom, which is later developed into the pericardial cavity, through fusion of the pleuropericardial folds to separate the pleural cavities at the later development of the lungs [7].
Developmental Timeline
| WEEK | DEVELOPMENT |
|---|---|
| 2 | Bilateral cardiogenic areas form |
| 3 | Mesoderm splits, Heart tubes are brought to the midline, Heart tube fusion, Heart beat |
| 4 | Heart looping, Neural crest migration commences, Dorsal and ventral endocardial cushions fuse |
| 5 | Foramen primum closed, Septum secundum srtats developing, Muscular interventricular septum develops, Bulbar ridges and trabeculation become evident |
| 6 | Aortic and pulmonary trunks cleave |
| 7 | Valves develop |
Primary Heart Field
Gestational Week 4-5
The first cells start to migrate through the primitive streak to the anterior and lateral sections of the cranial end of the embryonic disc, forming bilateral primary heart fields. These primary heart fields resemble a crescent shape.
The lateral plate mesoderm is split into two layers, namely the splanchnic mesoderm, facing the endoderm and the somatic mesoderm, facing the ectoderm. The former portion of the mesoderm gives rise to the heart. The portion between the splanchnic and somatic mesoderm is the presumptive pericardial space. Cells from the splanchnic mesoderm will merge to form 2 lateral endocardial tubes (also known as angioblastic cords) and as they form a lumen, are enveloped by myocardium. These endocardial tubes are as of now located inferior to the presumptive pericardial space.
Heart Tube Formation
Gestational Week 5
The embryonic disc starts to fold. This folding begins cranially and extends in a caudal direction. The endocardial tubes fuse and is now located between the pericardial space and newly formed foregut that becomes surrounded by pericardial space (also known as the pericardial coelom). At this stage, the myocardium does not completely engulf the endocardial tubes. Instead, it remains in a continuous attachment with the non-cardiac splanchnic mesoderm through a structure called the dorsal mesocardium.
At this point, the primitive heart tube is bilaterally symmetrical and resembles an inverted Y shape. Starting from the inflow tract, there is the right and left sinus venouses that receives blood from the embryo, followed by the primitive atrium, primitive ventricle, bulbus cordis and then the truncus asteriosus which gives rise to the aortic and pulmonary trunk [8].
<html5media height="200" width="240">File:Heart_folding_001.mp4</html5media>
Secondary Heart Field
Gestational Week 5-6
The secondary heart field (SHF) is a region of subpharyngeal mesodermal progenitor cells located medially and ventrally to the adjacent primary heart field (PHF) (that forms the initial heart tube). The PHF and SHF cells make up a region known as the cardiogenic field. Following heart tube fusion around 19-21 days, cells from the SHF migrate to the cranial and caudal ends of the tube and continue elongation [9].
It is understood that within the cardiac regions the progenitor cell population of the SHF is “pre-patterned”[10], hence being termed “specified but undifferentiated”. It is the patterning of cells within the soon to become myocardium, that is responsible differentiation into chamber-specific myocytes (atrial and ventricular) and the conduction of cells [11]. It is essential that the SHF cells remain undifferentiated and do not add prematurely to the heart tube [12].
The SHF cells gives rise to endocardial, myocardial and smooth muscle cells through expression of transcription factor Islet -1 [13]. It also contributes to the right ventricle, inflow and outflow tract (OFT), and the arteries and semilunar valves that meet to form the arterial pole of the heart [14] [15]. The OFT is the point of exit from the heart via structures of early cardiac development that give rise the essential structures of the aorta and pulmonary artery [16]. The SHF of the developing heart and its contribution the arterial pole involves complex and interconnected signalling pathways that will be covered later in further detail.
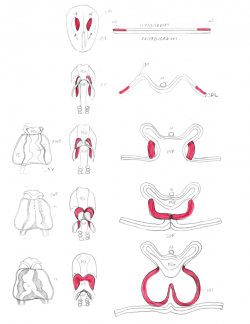
Cardiac Looping and Steps
Gestational Week 6
In the early embryo, the same progenitor cells that constitute the SHF are responsible for the rapid growth occurring during cardiac looping morphogenesis [17] . Looping is essential for cardiac development as it assist further growth of the originally straight heart tube, and ensures it fits within the pericardial celom. Additionally, looping of the heart tube is known to be the first visual evidence of embryonic asymmetry.
- Straight heart tube continues to elongate
- Rapid growth of the bulbus cordis and the primitive ventricle causes ventral bending and right rotation = C shaped loop (convex side on the right)
- The ventricular bend continues to move caudally, the inflow and outflow tracts are brought together at the atrial pole of the heart = S shape
- The truncus arteriosus if formed by adding myocardial cells at the top end of the heart. This portion will form roots and the proximal portion of the aorta and the pulmonary artery [18].
<html5media height="240" width="280">File:Heart looping 002.mp4</html5media>
[1]- Internal hyperlink to page?
Cardiac Septation
This stage of heart morphogenesis refers to the development of the four main cardiac chambers from the primitive atrium and ventricle. Cardiac septation is comprised of three main events:
- Division of the atrioventricular canal
- Atrial septation
- Ventricular septation
Division of the atrioventricular canal
Division of the atrioventricular canal (AVC) begins with the formation of the superior and inferior endocardial cushions, which are located on the dorsal and ventral aspects of the AVC respectively.[19] These cushions develop as mesenchymal cells invade and proliferate within swollen regions of cardiac jelly of the AVC; this mesenchyme is derived from endothelial cells that have transdifferentiated in the process of epithelial-mesenchymal transformation (EMT).[20] Throughout the fifth week of development, the endocardial cushions project inwards and eventually fuse to partition the AVC into the left and right atrioventricular canals; these canals will serve as the orifices in which the tricuspid and mitral valves are situated.[20]
Atrial septation
As the AVC is undergoing division, a muscular outgrowth, referred to as the septum primum, extends inferiorly from the roof of the primordial atrium.[21] This septum partially divides the atrial chamber into left and right halves, leaving a temporary communication located between the inferior border of septum primum and the endocardial cushions known as the foramen primum.[19] As the size of the foramen primum diminishes, perforations in the superior portion of the septum primum develop as a result of apoptosis, forming a second communication between the atrial chambers called the foramen secundum.[20] Concurrently, an additional muscular septum, known as the septum secundum, projects inferiorly to the right of the septum primum.[19] Eventually, the septum secundum will extend beyond the length of the foramen secundum, generating a partial division of the atria that forms the upper boundary of the foramen ovale.[21] The development of the foramen ovale is critical as it allows oxygen-rich blood from the placenta to bypass pulmonary circulation of the embryo and directly enter systemic circulation.[22] Following birth, the foramen ovale is obliterated as the septum primum and septum secundum fuse, resulting in complete formation of the interatrial septum.[19]
Ventricular septation
As with atrial septation, differentiation of the ventricles is initiated within week 4 of development.[19] A muscular ridge, referred to as the interventricular septum primordium, develops and extends superiorly from the caudal aspect of the primitive ventricular chamber.[20] Growth of the septum is attributed to the expansion of the ventricles, which involves the development of muscular trabeculae as cardiomyocytes proliferate within the chamber walls. [21] The end result is a partial division between the left and right ventricles, which forms the muscular portion of the interventricular septum.[20] A communication between the ventricles, known as the interventricular foramen, remains until approximately week 7 of development.[23] The foramen is obliterated by the fusion of the septum intermedium and the bulbar ridges of the bulbus cordis; this constitutes the membranous portion of the interventricular septum.[19]
z5059996
Cardiac Neural Crest and Outflow tract
Gestational Week 8
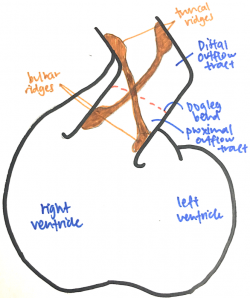
The outflow tract is a tube that runs from the right ventricle to the aortic sac and presents with a distinctive dog-leg bend that separates the proximal (bulbus cordis) and distal (truncus arteriosus) ends of the tract. The endocardial jelly that lines the lumen of the outflow tract concentrates to form the endocardial cushion facing each other that spirals in a 180-degree twist through the length of the outflow tract. Like the outflow tract, these endocardial cushions can be divided into distal and proximal moieties. The distal endocardial cushions are also known as the truncal ridges and the proximal ones are also known as the bulbar ridges. Cells from the cardiac neural crest migrates out of the neural tube, through the pharyngeal arches and aortic sac and into the outflow tract, where it condenses in the ridges to support the septation of the outflow tract. [24] [25]
The fusion of the endocardial cushions starts from the distal end of the outflow tract and proceeds proximally. Fusion of the truncal endocardial cushions forms the aorticopulmonary septum that separates the truncus into an aortic and pulmonary trunk. The bulbar endocardial cushions fuse as they extend towards the interventricular septum, separating the proximal outflow tract into the prospective aortic and pulmonary trunks. As the outflow tract separates, the aortic trunk leads to the 3rd and 4th pharyngeal arch arteries and the pulmonary trunk leads to the 6th pharyngeal arch artery. [26][27]
Proepicardium and Coronary Heart Development
Coronary vasculature arises when the fourth layer of the primary heart tube is formed. This fourth layer is known as epicardium that surrounds the myocardium which allows the development of vascular structures over the heart surface.
The monolayered embryonic epicardium derives from mesothelial cells of the septum transversium and mainly from clustered of proepicardium cells. The process involves either of these two mechanisms, one is by detaching of these cells and attach to the pericardial cavity and spread over the heart surface or two is by directly attaching the proepicardium to the myocardium forming a permanent tissue bridge. The spreading of the monolayered embryonic epicardium over the bare myocardium initiates the vascularisation of coronary system. A new extracellular matrix (ECM) layer, the subepicardium forms between the epicardium and the myocardium actively promotes the development of coronary blood vessels. The coalescence of three lineages of the coronary vessel cells: smooth muscle, endothelial and connective tissue and fusion of vascular cell progenitors (angioblasts) form a vascular structure de novo via the process of vasculogenesis. They then migrate onto the primary heart tube between day 22 and day 28 of human development. [28],[29]
z5018962
Developmental Signalling Processes
Heart development is a very complicated and dynamic process that requires a high degree of control and regulation. This control is achieved by several temporally regulated signalling cascade (Fig. 1) expressed at different stages of heart development.
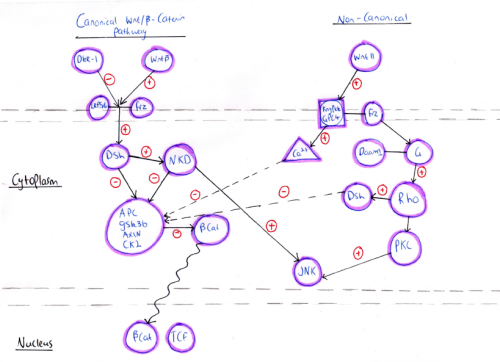
Wnt signalling
According to the primary mode of action, Wnt signalling pathways have been divided into two major classes, canonical and non-canonical Wnt signalling pathways. Both the pathways have a role in different stages of cardiac development which could be overlapping or independent of each other. The canonical Wnt signalling pathway involves β-catenin and is activated by a number of ligands such as Wnt-1, Wnt-2, Wnt-3A, Wnt-8A, Wnt-8B, Wnt-8C, Wnt-10A, and Wnt-10B. However, the non-canonical signalling pathway is associated with planar cell polarity and Wnt/Ca2+ pathways that are activated by different ligands such as Wnt4, Wnt5A, Wnt5B, Wnt6, Wnt7A, Wnt7B, and Wnt11 [31].
Canonical/β -catenin signaling is essential for mesoderm formation. Upon binding of the Wnt ligands to the Frizzled receptor, a seven-transmembrane receptor, or the co-receptor LRP-5/6, the Canonical signaling pathway is activated. This leads to cytoplasmic accumulation of β -catenin in one side of the embryo and its translocation to the nucleus where it drives the activation of transcription factors required to determine the site at which mesoderm and endoderm formation will occur in the embryo. Animal studies have shown that the lack of nuclear accumulation of β -catenin results in inability of axis formation and mesoderm development whereas overexpression of β -catenin leads to the formation of secondary axis and ectopic expression of mesoderm signals. Also, homozygous deletion of β -catenin in mouse models results in absence of primitive streak and mesoderm formation [32].
Once mesoderm formation proceeds, the β -catenin signal must be shut down by inhibiting factors otherwise cardiac mesoderm formation will be stopped. This indicates that canonical signaling act as a switch that either induces or suppresses cardiac development. One of the inhibiting factors include dickkopf-1(DKK1) which is an extracellular Wnt inhibitor promotes the expression of cardiac-inducing factor in the endoderm such as Hex. The latter activates paracrine factors to direct adjoining cells towards cardiac fate [33].
Transforming growth factor-β
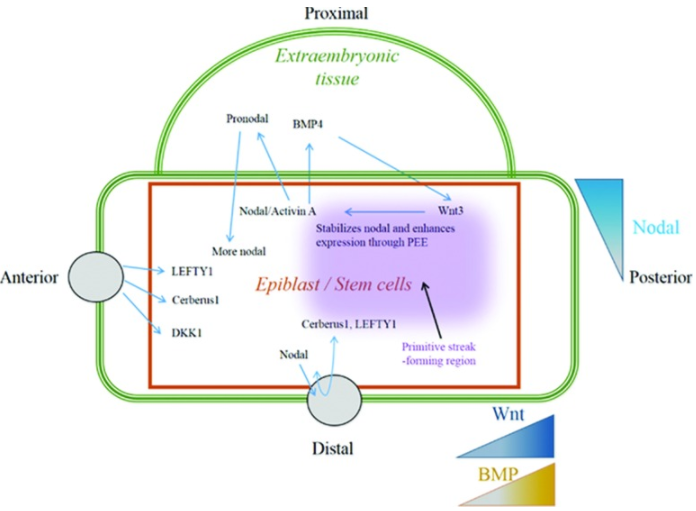
Transforming growth factor-β superfamily involves a large number of growth factors that are structurally related. These growth factors, including Nodal or its mimic Activin, bone morphogenic protein (BMP) and growth and differentiation factors, signal through SMAD-dependent and SMAD-independent pathways [34]. Nodal and its mimic Activin act through SMAD-2 and -3 to activate transcription. During embryo development, epiblast cells produce high levels of Nodal growth factors leading to a gradient of nodal in these cells. This gradient is important for mesoderm patterning (left-right asymmetric heart development) and lineage specification. For this reason, this gradient is maintained by Nodal antagonist secreted from the anterior visceral endoderm or by BMP-4 and Wnt-3 feedback loop in the extraembryonic tissues (Fig.3) [35]. Nodal/Activin’s important role in heart formation is demonstrated by animal models. Presence of Activin in amphibian embryo lead to activation of heart formation whereas the absence of Nodal co-receptor in, called Cripto, in mouse embryo resulted in failure of ES cells differentiation into cardiomyocytes [36]. Also, knocking out copies of SMAD-2 alone or both SMAD-2 ND -3 result in inappropriate specification of axial mesoderm [37].
 [38].
[38].
FGF signalling
Fibroblast growth factors (FGF) serve a variety of functions in development, the maintainence of health and disease. FGFs are signaling proteins mostly as paracrine growth factors or endocrine hormones. There are 22 types of human FGFs, whereby paracrine FGF8, FGF9, FGF10 and FGF16 serve a major role in embryonic heart development. Additionally, FGF2, FGF9, FGF10 and FGF16 are involved in postnatal heart pathophysiology[39].
Communication between cardiac progenitor cells is required during embryonic heart development. During heart development, neural cress cells which migrate from the neuroectoderm of the dorsal neural tube will contribute to cushion formation and dictate the correct septation and alignment of the heart[40]. In conjunction to this process FGF3, FGF8, FGF9, FGF10, FGF15, FGF19 and FGF16 function was paracrine signals in embryonic heart development.
A study conducted demonstrated how a combination of both FGF2 and Bone morphogenic protein 2 (BMP2) efficiently enhances the cariomyogenic differention of embryonic stem (ES) cells at an optimal concentration. When FGF2 was inhibited, this ultimately suppressed cardiomyogenic differentiation, thus indicating that FGF signaling play a crucial role in early cardiomyogenesis. The ability for FGF2 to induce cardiac differentiation may serve a key role in treatment for heart diseases. [41] In addition, FGF10 also promotes cardiomyocytes differentiation from ES and induced pluripotent stem cells (iPS). In experiments, when FGF10 and ES cells were administered, this lead to the promotion of cardiomyocyte differentiation in the myocardium of the heart. This may thus serve as a treatment for those who have suffered from myocardial infarctions, where the myocardium is replaced by scar tissue. Providing new and fully functional cardiac muscle may help reduce complications. [42]
The table below outlines the different types of FGFs invovled in heart development and their functions. [43]
| Fibroblast Growth Factor | Function |
| FGF8 | FGF8 is expressed in the early embryonic stages. Required for cardiac looping and migratory cardiac neural crest cell survival. FGF8 is also required for anterior heart field development. |
| FGF9 | Activates FGFR1c with heparin sulfate as a co-factor in a paracrine manner. In doing so, this FGF plays a major role in stimulating the proliferation of cardiomyocytes. |
| FGF10 | Preferentially activates FGFR2b with heparin sulfate as a co-factor. Serves to regulate regional-specific cardiomyocyte proliferation in the embryonic heart in an autocrine/paracrine manner. This FGF type is also essential for the movement of cardiac fibroblasts in the compact myocardium. |
| FGF15/19 | These FGF types are required for proper morphogenesis of the cardiac outflow tract. In addition these FGFs play a major role in paracrine signaling in heart development. |
| FGF16 | FGF16 serves as a major growth factor involved in stimulating the growth of embryonic cardiomyocytes. |
The Notch pathway
Sonic Hedgehog
Retinoic Acid
Z5076466
Current Research And Findings
CTCF counter-regulates cardiomyocyte development and maturation programs in the embryonic heart
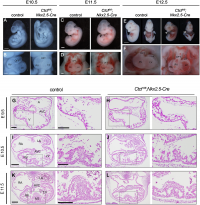
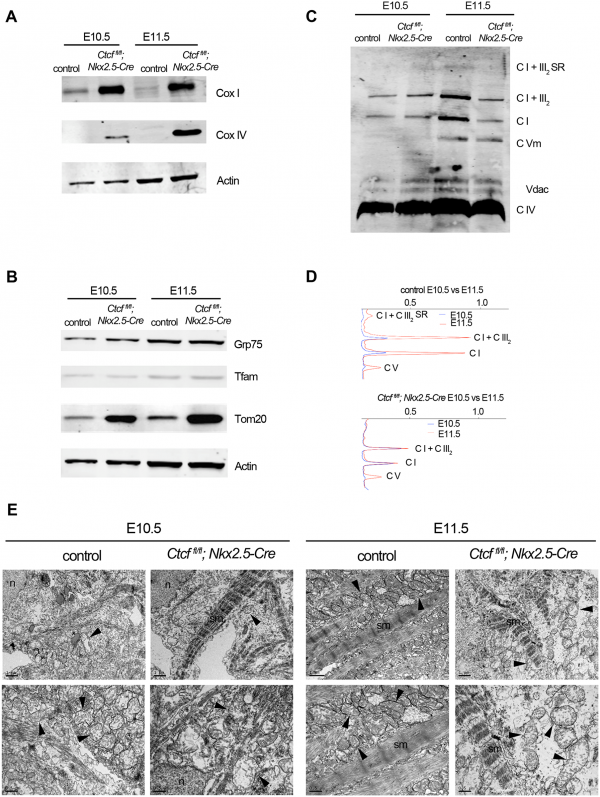
CTCF is a DNA binding factor that is essential in the process of genome binding and cardiogenesis. It mediates genomic interaction and maturation in the developing heart, coordinates cardiomyocyte differentiation by facilitating enhancer-promoter interactions. Although the role of CTCF in genome organisation is not fully understood. In this study by Gomez-Velazquez et.al (2017), they studied the effect of genetically deleting CTCF in differentiating cardiomyocytes at early stages of mouse development. They deleted CTCF in a population of cardiac progenitor cells which results in malformation of the heart and death of embryo (Figure 1)
From Figure 1, they presented control and CTCF-absent mice in stage E9.5, E10.5, E11.5 and E12.5. E10.5 and E11.5 mutant embryos appeared normal (Fig 1A - 1D). Pericardial edema (Fig 1E) and non expanding cardiac chamber (Fig 1F) presented in E12.5 mutant embryo. Histological examination showed slightly disorganised interventricular septum and normal 4 chambers and atrioventricular canal in E10.5 mutant embryo (Fig 1I and 1J). Fig 1G and 1H showed no defects in E9.5 mutants hearts as compared to controls. F 1K and 1L showed the thinning of myocardial wall in E11.5.
They also carried out Western blot to analyse the component of oxidative phosphorylation pathway in mutant and control embryonic cardiomyocytes. They revealed that there was an increase in Complex IV subunit 1 (Cox 1, encoded in mitochondrial DNA) and Complex IV subunit IV (Cox IV) in CTCF mutants hearts at E10.5 and E11.5 (Figure 2A). Fig 2B showed an increased in Tom20 which is the major receptor of mitochondrial outer membrane translocase. Complex I and Complex V increased from E10.5 to E11.5 in control hearts, consistent with maturation of mitochondrial oxidative phosphorylation. However, they did not increase in CTCF mutant hearts and Complex V was similarly increased between E10.5 and E11.5 in both control and mutant hearts (Fig 2C and 2D). Thus, this suggest that despite increased transcription of subunit in Complexes and supercomplexes at the mitochondrial inner membrane, maturation of the respiratory chain is blunted in the CTCF mutant heart.
Transmission electron microscopy (TEM) analysis (Fig 2E) presented immature but normal mitochondria in CTCF mutant cardiomyocytes. At E11.5, mutant cardiomyocyte's mitochondria are swollen, and larger than controls and are disorganised at E10.5.
They concluded that when CTCF is removed, genes are misregulated which leads to faulty mitochondria and incorrect expression of cardiac patterning gene. These misregulated genes control opposing genetic program incharge of development and cardiomyocyte maturation. Subsequently, embryo lethality. [44]
z5018962
Animal Models
β-catenin requirement in anterior-posterior axis formation in mice
Over the past few decades, intensive studies have been carried out in mice, Xenopu, zebrafish and chicken embryos to establish the role of Wnt signaling in cardiac development. Such studies, in particular, loss of function analysis studies, have allowed for the determination of specific roles of this signaling pathway in specific stages of cardiac development. For example, one of the studies conducted in 2000 on mice embryo established the role of β-catenin Wnt signaling in anterior-posterior axis formation. In this study, Huelsken et al. [45] produced β-catenin deficient mice and observed its impact on the anterior-posterior axis formation. At E6.0, the mice embryo showed block and inappropriate formation of anterior-poterior axis, mislocation of anterior visceral endoderm indicated by the Cerberus-like and Lim1 markers. Consequently, mesoderm and head structures were not developed and markers of posterior and anterior differentiation like Brachyury and Hex respectively were not expressed. However, adhesion was not affected by the lack of β-catenin due to the presence of substituents such as plakoglobin. In parallel to Xenopus and zebrafish [46] [47] , the observed block in axis formation of β-catenin null mice demonstrates the signaling functions of β-catenin.
Lineage of the cardiac valves
The developmental processes of cardiac valves formation have been well understood through animal models. Since the origin of cells contributing to different parts of the valves remains controversial, this study on mice models [48] provides further insight into the origin of these cells. In this study, myocardial, endocardial and neural crest cells were labeled irreversibly using a genetic system during embryonic development to determine their eventual contribution to different valvar components. At E17.5 majority of cells present in the cardiac valves were positive for β-galactosidase expression which is indicative of endocardial origin (Fig.?) Cells of the endocardial lineage were detected in leaflets and tendinous cords of the mitral and tricuspid valves (Fig. ?A - ?C), the atrioventricular fibrous continuity (Fig. ?G) and the leaflets of the aortic and pulmonary valves (Fig. ?I and ?J). However, these components showed minimal contribution from cells of the myocardial and neural crest lineages.
z5076466
Abnormal Development
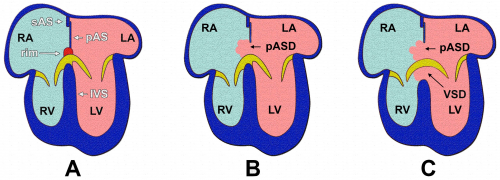
Atrial Septal Defect
Atrial septal defects (ASD) are one of the most prevalent congenital heart malformations, with an estimated 56 cases per 100,000 births.[49] This heart defect involves a communication between the left and right atria, and can be subdivided into five main types based on the nature of the defect.
The most common ASD is a patent foramen ovale. Throughout fetal development, the foramen ovale functions to shunt oxygenated blood from the right atrium into the left atrium.[50] After birth, the now functioning respiratory system of the newborn results in elevated pressure in the left atrium relative to that of the right.[51] This induces fusion of the septum primum and septum secundum which closes the foramen ovale, leaving a remnant known as the fossa ovalis.[50] Incomplete closure results in a patent foramen ovale. This condition is present in nearly all newborns and typically closes within the first month, however complete closure is achieved in only 70-75% of adults.[49] A patent foramen ovale is often asymptomatic and thus is not usually considered a clinically significant condition, however it can be problematic when other cardiac abnormalities are present.[52]
A secundum ASD is the most common of the true (clinically significant) atrial septal defects. This malformation occurs within the region of the fossa ovalis and typically results from abnormal resorption of the septum primum during cardiac development.[52] Secundum ASDs enable left-to-right atrial shunting of blood, which increases the load on the right side of the heart and, consequently, the pulmonary circulation; this can result in the development of atrial arrhthymias and right ventricular failure.[50]
Sinous venosus ASD refers to a communication between one of the right pulmonary veins (typically the upper vein) and the wall of the right atrium adjacent to the superior vena cava.[52] This defect can occur as a result of the resorption or partial absence of the septum secundum,[52] and accounts for 5-10% of all atrial septal defects.[53]
A primum ASD is the result of an abnormality in the development of either the septum primum or septum secundum.[52] This condition is a type of atrioventricular septal defect, and will be discussed in more detail in the next section.
The most extreme ASD is a common atrium. This occurs when the septum primum and septum secundum fail to develop.[49] This lack of an interatrial septum is often accompanied by other cardiac abnormalities, including clefts in the leaflets of the atrioventricular valves.[52]
z5059996
Atrioventricular Septal Defect
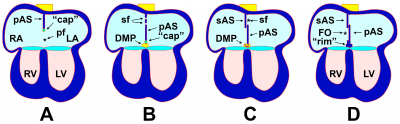
An atrioventricular septal defect (AVSD) refers to a malformation involving the interatrial and/or interventricular septa, and has an estimated incidence of 1 in 2000 births.[54] This defect typically occurs as a result of incomplete fusion of the endocardial cushions,[53] although recent research has concluded that malformation of the dorsal mesenchymal protrusion (DMP) also contributes to the development of AVSDs.[54] This can result in abnormal development of the interatrial septum, interventricular septum and/or the mitral and tricuspid valves.[52]
There are two main subdivisions of AVSDs, which differ according to whether one or both of the cardiac septa are affected. In a partial AVSD, there is a defect present in the inferoanterior region of the fossa ovalis, which allows the right-to-left shunting of blood across the atria.[54] In this scenario, the lack of fusion of the endocardial cushions can also result in the presence of clefts in the septal leaflets of the atrioventricular valves.[52] However, in a complete AVSD, a defect in the membranous portion of the interventricular septum is also present.[53] This condition results in direct communications between the atria and the ventricles, and pulmonary hypertension or congestive heart failure can occur if the anomaly is severe.[54]
z5059996
Ventricular Septal Defect
Ventricular septal defects are the most prevalent of all congenital heart defects (excluding patent foramen ovales), accounting for approximately 40% of all cardiac abnormalities among newborns.[55] The exact definition of a VSD is still a point of contention within the scientific community, but is generally referred to as a communication between the left and right ventricles; this anomaly can manifest as a lone defect, but is often present with other abnormalities such as Tetralogy of Fallot or a complete AVSD.[56] occur as a result of abnormal development (or absence) of any of the components forming the interventricular septum.[57] Hence, a VSD can occur in either the muscular or the membranous portion of the septum, with the latter being more common.[57]
A membranous VSD occurs when there is an incomplete occlusion of the interventricular foramen, which is due to the membranous portion of the septum failing to develop.[52] This is the result of a failure of fusion between the muscular portion of the interventricular septum and the endocardial cushions.[52] This defect is most commonly located in the outflow tract of the left ventricle.[52]
A muscular VSD results from abnormal development of the interventricular septum primordium during cardiac septation.[56] This can refer to incomplete formation of the primitive septum, or to errors in the proliferation of trabeculae within the septum.[55] VSDs can form anywhere within the muscular wall of the septum, and can extend into various regions of the right ventricle including the apex, inlet or outflow tract.[55]
The most extreme form of VSD is a common ventricle. This very rare defect occurs when the interventricular septum primordium fails to develop.[52] Infants with common ventricle often die from congestive heart failure.[52]
z5059996
Persistent Truncus Arteriosus
Cardiac Stem Cells
Following a myocardial infarction (MI), a significant number of cardiomyocytes becomes damaged and the heart has very little regenerative functions. The loss of cardiomyocytes pose as a problem when an individual has had repeated MI or those suffering from end stage heart failure. For these individuals, heart transplantation remains as the main solution. However, there are not enough donors available. Therefore, stem cell therapy provides as an upcoming possibility in replacement for donor transplant. There are two types of cells that could be use which includes human embryonic Stem cells (hESCs) and Human Induced Pluripotent Stem Cells (hiPSCs). However, there are differences between these two cells and further testing and applications are still required to produce highly pure and mature cardiomyocytes.
| FACTOR | Human Embryonic Stem Cells | Human Induced Pluripotent Cells |
|---|---|---|
| Ethical Issues |
|
|
| Immune Reactions |
|
|
| Availability of Cells |
|
|
| FACTOR | Human Embryonic Stem Cells and Human Induced Pluripotent Stem Cells |
|---|---|
| Teratoma formations | Both have tendency to form teratomas. Cardiomyocytes thus have to be highly purified. A few methods have been explored to obtain a highly purified culture of stem cells to prevent teratoma formation. The following methods include:
Mitochondrial Based Separation[60]
Biochemical Differences between Differentiated and Undifferentiated Cardiomyocytes[61]
However, because both methods do not have a 100% purity, teratomas could still be formed. |
| Cardiac Maturation |
Ultrastructural Analysis
Electrophysiological Properties
Contraction Properties
Since hESCs and hiPSCs are required to replace damaged cardiomyocytes, their obvious immaturity problem would have to be resolved before it can be utilized for transplantation or therapy. |
(z5178463)
Future Questions
What is the secreted protein that flows across the node, and what is the mechanism that ensures that the loop turns in rightward direction?
What are the developments of stem cell usage for developmental abnormalities?
http://circres.ahajournals.org/content/91/3/189.short
Glossary of Terms
| Term | Definition |
| Axial mesoderm | Also referred to as notochord. It is an embryonic structure lying within the midline of the trilaminar embryo. |
| Blastocoel | A fluid filled cavity formed during early embryonic development (Week 1-2) within the blastocyst. Once Morula is formed as a result of the initial cell division, further cell division followed by compaction leads to the formation of this cavity. |
| Cardiocyte | Mature muscle cell of the heart. These cells are characterized by striation and separated by intercalated discs. |
| Angioblastic cords | Groups or ‘columns’ of embryonic precursor cells which will form the walls of both arteries and veins. |
| Bulbus cordis | A region of the early developing heart tube forming the common outflow tract, will differentiate to form three regions of the heart. |
| Sinus venosus | An early developmental cardiovascular structure, thin walled cavity, forming the input to developing heart which has 3 venous inputs (vitelline vein, umbilical vein, common cardinal vein). Later in heart development this structure gets incorporated into the wall of the future right atrium. |
| Outflow tract | Exit of blood from the heart tube formed by the truncus arteriosus. |
| Inflow tract | Entrance of blood into the heart tube; the sinus venosus portion of the tube. |
| Mesothelial cells | Epithelial cells of mesodermal origin |
| Pericardium | The membranous sac filled with serous fluid that encloses the heart, aorta and large blood vessels |
| Epicardium | The inner layer of the pericardium that is in contact with the surface of the heart |
| Proepicardium | Group of progenitor cells that forms near the venous pole of the heart gives rise to the epicardium |
| Mitochondrial oxidative phosphorylation | A synthesis of ATP powered by the free energy of reduced compounds that are produced by other metabolic pathways, including glycolysis and the TCA cycle, occurs in the mitochondria |
Below are links to a more extensive glossary if additional definitions are needed
A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z
References
- ↑ http://journals.plos.org/plosone/article?id=10.1371/journal.pone.0022055
- ↑ <pubmed> PMC1767747 </pubmed>
- ↑ https://www.nhlbi.nih.gov/health/health-topics/topics/hhw
- ↑ http://www.mdpi.com/2308-3425/3/2/12/htm
- ↑ <pubmed> PMC2691808 </pubmed>
- ↑ https://embryology.med.unsw.edu.au/embryology/index.php/Cardiac_Embryology
- ↑ <pubmed> PMC4374196 </pubmed>
- ↑ <pubmed> PMC1767747 </pubmed>
- ↑ <pubmed> PMC1767747 </pubmed>
- ↑ http://perspectivesinmedicine.cshlp.org/content/4/10/a015750.full
- ↑ http://www.sciencedirect.com/science/article/pii/S1875213613002817
- ↑ <pubmed> PMC2794420 </pubmed>
- ↑ http://www.sciencedirect.com/science/article/pii/S1875213613002817
- ↑ http://circres.ahajournals.org/content/104/8/933
- ↑ <pubmed> PMC2794420 </pubmed>
- ↑ https://embryology.med.unsw.edu.au/embryology/index.php/Intermediate_-_Outflow_Tract
- ↑ http://circres.ahajournals.org/content/104/8/933
- ↑ <pubmed> PMID16479500 </pubmed>
- ↑ 19.0 19.1 19.2 19.3 19.4 19.5 <pubmed>PMC3424040</pubmed>
- ↑ 20.0 20.1 20.2 20.3 20.4 <pubmed>PMC1767797</pubmed>
- ↑ 21.0 21.1 21.2 <pubmed>24138816</pubmed>
- ↑ <pubmed>27188965</pubmed>
- ↑ <pubmed>12739611</pubmed>
- ↑ <pubmed> PMC1767864 </pubmed>
- ↑ <pubmed> 23633400 </pubmed>
- ↑ <pubmed> PMC1767864 </pubmed>
- ↑ <pubmed> 23633400 </pubmed>
- ↑ Pérez-Pomares J, Pires-Gomes A, (2013) The Epicardium and Coronary Artery Formation, Journal of Developmental Biology, Vol.1(3), pp.186-202, ISSN: 2221-3759, E-ISSN: 2221-3759, https://doaj.org/article/b3f5aa0ae8df409ab1664975469b95d1
- ↑ Martinsen B, Lohr J, (2005) Cardiac Development, Handbook of Cardiac Anatomy, Physiology and Devices Iaizzo, P.A. (Ed.), http://www.springer.com/cda/content/document/cda_downloaddocument/9781588294432-c2.pdf?SGWID=0-0-45-387660-p173728389
- ↑ <pubmed>12781678</pubmed>
- ↑ <pubmed>25813860</pubmed>
- ↑ <pubmed>16860783</pubmed>
- ↑ <pubmed>4533091</pubmed>
- ↑ <pubmed>18290874</pubmed>
- ↑ <pubmed>25813860</pubmed>
- ↑ <pubmed>20830688</pubmed>
- ↑ <pubmed>12842913</pubmed>
- ↑ <pubmed>25813860</pubmed>
- ↑ <pubmed>11493531</pubmed>
- ↑ <pubmed> 23799628</pubmed>
- ↑ <pubmed>26793421</pubmed>
- ↑ <pubmed> 24307297 </pubmed>
- ↑ <pubmed>26793421</pubmed>
- ↑ <pubmed>28846746</pubmed>
- ↑ <pubmed> 10662781 </pubmed>
- ↑ <pubmed>9914418 </pubmed>
- ↑ <pubmed> 7876319 </pubmed>
- ↑ <pubmed>15297379</pubmed>
- ↑ 49.0 49.1 49.2 <pubmed>24725467</pubmed>
- ↑ 50.0 50.1 50.2 <pubmed>PMC4711537</pubmed>
- ↑ <pubmed>PMC5505397</pubmed>
- ↑ 52.00 52.01 52.02 52.03 52.04 52.05 52.06 52.07 52.08 52.09 52.10 52.11 52.12 <pubmed>PMC325204</pubmed>
- ↑ 53.0 53.1 53.2 <pubmed>PMC4373719</pubmed>
- ↑ 54.0 54.1 54.2 54.3 <pubmed>PMC5267359</pubmed>
- ↑ 55.0 55.1 55.2 <pubmed>21349577</pubmed>
- ↑ 56.0 56.1 <pubmed>PMC4316658</pubmed>
- ↑ 57.0 57.1 <pubmed>7437181</pubmed>
- ↑ <pubmed>9804556</pubmed>
- ↑ <pubmed>20964482</pubmed>
- ↑ <pubmed>19946277</pubmed>
- ↑ <pubmed>23168164 </pubmed>
- ↑ <pubmed>21883888</pubmed>
- ↑ <pubmed>12791707</pubmed>
- ↑ <pubmed>21614516</pubmed>
- ↑ <pubmed>16322641 </pubmed>