Paper - The origin and development of the human extrinsic ocular muscles (1957)
| Embryology - 30 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Gilbert PW. The origin and development of the human extrinsic ocular muscles. (1957) Carnegie Instn. Wash. Publ. 611, Contrib. Embryol., Carnegie Inst. Wash. 36: 59-78.
| Online Editor |
|---|
|
In all the vertebrates:
In several groups of vertebrates:
|
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
The Origin and Development of the Human Extrinsic Ocular Muscles
Contributions To Embryology, No. 246
Perry W. Gilbert
Department of Embryology, Carnegie Institution of Washington, Baltimore, Maryland, and Department of Zoology, Cornell University, It/mm, New York
With fourteen plates and two text figures
Introduction
Few muscles in vertebrates have been more challenging to the comparative anatomist than those that move the eyeball, yet, interestingly enough, few muscles have, during their evolution, remained more conservative. In all the vertebrates, the superior rectus, the medial rectus, the inferior rectus, and the inferior oblique are innervated by the oculomotor nerve, the superior oblique is supplied by the trochlear nerve, and the lateral rectus by the abducens nerve. A seventh eyeball muscle, the retractor bulbi, found in several groups of vertebrates, is, like the lateral rectus, supplied by the abducens nerve.
The uniform appearance of these muscles throughout the different classes of vertebrates, and their consistent innervation, lead one to anticipate some uniformity in their pattern of development. Marked similarity has indeed been found in elasmobranchs, reptiles, and birds, in which groups the eyeball muscles consistently arise from the walls of three pairs of head cavities or from homologous solid condensations. In these vertebrates, the four eyeball muscles innervated by the oculomotor nerve arise from the walls of the premandibular head cavity; the superior oblique arises from the walls of the mandibular head cavity or from a homologous solid condensation; and the abducens musculature arises, at least in part, from the hyoid head cavity or condensation. A comprehensive review of eyeball—muscle development in these groups may be found in the papers of Adelmann (1927), Edgeworth (1935), and Wedin (1949); specific groups have been treated fairly exhaustively by the following: elasmobranchs, Balfour (1878), Dohrn (1904), Lamb (1902), Marshall (1881), Neal (1918), Platt (1891), Wedin (1949), van Wijhe (1883); reptiles, filatofl (1907), Iohnson (1913), Orr (1887): Wedin (1953a); birds, Adelmann (1926, 1927), Rex (1900, 1905), Wedin (195312) As for the Metatheria, the eyeball muscles have been intensively studied embryologically in Trio/tosurus and other marsupials by Fraser (1915); Gilbert (1954) has investigated their early development in the opossum, Didelphys virginiana. In both Tric/xorurus and Didelphys, the oculomotor muscles arise from the wall of a well defined premandibular cavity situated on each side of the head, whereas the superior oblique and the abducens muscles are derived from distinct primordia which may be homologized with the mandibular and hyoid condensations of lower vertebrates. The extrinsic ocular muscles in marsupials thus arise from three separate primordia, as they do in elasmobranchs, reptiles, and birds.
The innervation of the extrinsic ocular muscles in certain cyclostomes is somewhat unusual in that the abducens nerve appears to supply both the lateral rectus and inferior rectus muscles. P. fiirbringer (1875) first noted this condition in the lamprey, Petromyzan nmrhmx; his observations were subsequently confirmed by M. Firbringer (1897), Coming (1900), and Cords (1929). Addens (1933) cleared up this apparent inconsistency when he showed that “the III nucleus of petromyzonts, in contradistinction to that of all other vertebrates, which is always single, consists of two widely separated divisions, viz., a large rostral part, situated at about the same place as the single nucleus in gnathostomes, and a small caudal part lying far backward, viz., at the caudal level of the V root.“ The root arising from the caudal division of the Ill nucleus does not join that arising from the rostral division, but issues separately from the brain as a part of cranial nerve VI. Addens proposed, therefore, the name “oculomotor—abducens" for this composite nerve of petromyzonts, and showed that the oculomotor portion innervates the inferior rectus whereas the abducens portion supplies the lateral rectus.
In placental mammals, accounts of the ontogeny of the eyeball muscles have been conflicting and, until recently, incomplete. Basing their observations on relatively late stages of development, Reuter (1897) and Keil (1906) believed that all the extrinsic ocular muscles of the pig arise from a single condensation of mesoderm adjacent and caudal to the optic stalk. Leser (1925), after observing early stages of the ground squirrel, bat, rabbit, dog, mouse, and guinea pig, concluded that the eyeball muscles of these mammals likewise originate from a single condensation on each side of the head.
On the other hand, Corning (1899) found in rabbit embryos of 12 and 17 somites a solid pair of premandibular condensations arising from the prechordal plate, which he suggested are the source of the oculomotor muscles. He was unable to find in these early stages the primordia of the superior oblique and abducens muscles, but believed that they arise independently of those innervated by the oculomotor. Bonnet (1901) likewise found, in a 16-somite dog embryo, a pair of mesodermal condensations derived from a medial mass of cells at the anterior end of the notochord intimately associated with the roof of the preoral gut. Bonnet called these condensations rudimentary head cavities (Kopf/16/ilenrudimem ten), but did not follow their fate. His illustration leaves no doubt that these masses are premandibular condensations of prechordal-plate origin.
The investigations of Coming and Bonnet suggested that an examination of mammalian embryos younger than those that were available to Keil and Reuter, and more critically studied than those of Le‘s'er, might well reveal the true origin of the eyeball musculature in placental mammals and, at the same time, determine whether in this group the extrinsic ocular muscles arise from a single primordium or from three distinct but closely apposed primordia such as are found in elasmobranchs, reptiles, birds, and marsupials.
With this in mind, the author (Gilbert, 1947) examined 225 cat embryos, in stages ranging from 10 somites to 55 mm., and studied the origin and development of the extrinsic ocular muscles. It was found that the oculomotor muscles arise from a pair of premandibular condensations of prechordal-plate origin, and that the eyeball muscles innervated by the IVth and Vlth nerves arise from separate condensations in the maxillomandibular mesoderm. It was concluded that “the origin, relations, and fate of the 3 condensations giving rise to the eye muscles in the cat indicate clearly their homology with the so-called premandibular, mandibular, and hyoid eye-muscle anlagen of lower vertebrates." Is it possible that the eyeball muscles of other placental mammals, including man, follow a similar pattern in their development?
References to the origin and development of the extrinsic ocular muscles in man are to be found in the texts of Mann (1928) and Wolff (1940). Mann states (p. 254) that “the eye muscles in man seem, at their earliest stage, to be represented by a single undifferentiated mass. The earliest trace of these muscles in man can be seen at 7 mm. when they are represented by a massed condensation in the paraxial mesoderm surrounding the optic vesicle.” Wolff reaches a similar conclusion, reporting that the primordia of the human eyeball muscles at first “form one mass which Lewis (1910) found was supplied by the 3rd nerve only. Later (at 9 mm.) when the 4th and 6th nerves enter, this mass divides into separate muscles.”
To the author's knowledge, Lewis (1910) was the first investigator to study and describe the early development of the human extrinsic ocular muscles. Unfortunately, only a limited number of human embryos was available to him when he made his study; for this reason he was unable to investigate the very early stages, which would have thrown more light on this problem. Lewis‘ widely quoted statement concerning the origin of the eyeball musculature in man is as follows:
It has commonly been assumed that the muscles of the orbit are derived from anterior head somites; the first somite giving rise to the Mm. levator palpebrae superioris, rectus superior, rectus internus, rectus inferior, and obliqnus inferior, supplied by the N. oculomotorius; the second, to M. obliquus superior, supplied by the l\'. trochlearis; and the third, to the M. rectus lateralis, supplied by the N. abducens. Anterior head somites have, however, never been observed in man or mammals. Zimmermann (1898) [actually, 1899, P.W.G.] observed in a 3.5-mm. embryo several small head cavities in the region where later the eye muscles develop.
It is uncertain even that they represent rudimentary head somites, and no connection has been established between them and the eye muscles. On the contrary, the observations of Renter (1897) on the pig and my own observations on human embryos, which follow here, show that all the eye muscles arise from a common premuscle mass, which occupies, both in the pig and in man, the same general position in the early embryo. This common premuscle mass is first to be recognized in human embryos of about 7 mm. in length (fig. 367). It consists of a lens-shaped mass of condensed mesenchyme, outlined from the surrounding mesenchyme by a capillary network over its surface. This premuscle mass lies dorsal to the optic stalk, between it and the ganglion Gasseri, and medial to the optic cup. The N. ophthalmicus passes in front of and lateral to the mass, and the N. maxillaris behind and lateral to it. At this stage only the N. oculomotorius enters the anterior end of the muscle mass. The Nn. trochlearis and abducens do not appear until later.
In a 9-mm. embryo the eye premuscle mass occupies much the same position as in the earlier stage, lying on the dorsal side of the optic stalk and medial to the N. ophthalmicus and maxillaris (figs. 368, 369). It has enlarged somewhat and extends along the caudal side of the optic stalk. It also begins to show cleavage into separate muscle masses, each supplied by its respective nerve. The N. trochlearis now enters the anterior portion of the mass which later forms the rudiment of the M. obliquus superior. The N. abducens enters the caudal end of the mass, which has begun to extend out along the path of the nerve and also shows indications of separation from the rest of the mass which lies closer to the optic stalk and into which the N. oculomotorius runs. The muscle mass at this stage has no very distinct attachment either to the precartilage or to the sclera, but there exists a direct continuity with the mesenchyme from which these structures differentiate.
In an 11-mm. embryo the premuscle mass has still farther enlarged and partially split into the rudiments supplied by the three nerves. The anlagen lie on the dorsal and caudal sides of the optic nerve and for the most part medial to the eyeball (figs. 370, 379). They are continuous laterally with the primitive sclera, which is now beginning to form as a definite condensation of the mesenchyme about the eyeball, and medially with the precartilaginous tissue about the optic nerve. The M. rectus lateralis has extended farther out along the path of the N. abducens and has as yet no skeletal attachment. The N. ophthalmicus passes above these muscle masses and nearly at right angles to them.
As differentiation progresses the muscle mass of the N. oculomotorius gradually extends around the optic nerve and splits into the various muscles supplied by this nerve. In a I4-mm. embryo all the orbital muscles are to be distinguished and have nearly the adult relations to the bulbus oculi. The M. obliquus inferior, however, does not completcly separate 0H from the M. rectus inferior until a later stage.
Recently, in a doctoral thesis, Carnicer (1950) described the origin and development of the extrinsic ocular muscles of three placental mammals, Lepus, M us, and Homo. He believes that mesenchymal thickenings, the primordia of the eyeball muscles, arise in situ about the distal ends of cranial nerves III, IV, and V1, and is of the opinion that the nerves in some way are responsible for the appearance of the eye-muscle primordia. He ignores the fact that the eye-muscle primordia in both cat and man are evident before the cranial nerves III, IV, and VI make their appearance. Although he does not state clearly the number of mesenchymal thickenings, he gives the impression that there is but a single mass on each side of the head which thickens unevenly to form the eye-muscle primordia.
Carnicer’s conclusions are based on an examination of the following material: Lepu: (6 specimens: 10, 11, 11 to 13, 13, 15, 15 to 18 days), Mus (6 specimens: 10, I2, 12, 15, I6, 16 to 17 days), Homo (4 specimens: 2 to 4 weeks, 5 to 6 weeks, 2 months, 3 to 4 months). The youngest human embryo he examined appears to belong to late horizon xiv or early horizon xv, for the lens vesicle has nearly closed off from the surface ectoderm. Insufficient data are given for the other human embryos to permit assignment to a specific horizon, but, from their condition, it must be concluded that two and possibly all three are much too old to be of value in reaching any conclusions as to the origin and early development of the eyeball musculature. Even the youngest human embryo he examined, belonging to horizon xiv to xv, hardly provides an acceptable starting point for such a study. Moreover, the paucity of material with which Carnicer worked, not only in man, but also in the mouse and rabbit, would provide a serious handicap even for an experienced investigator. It is not surprising, therefore, that Carnicer's study does little to elucidate the early ontogeny of the mammalian eyeball musculature.
It should be remembered that the observations of Lewis (1910) and Carnicer (I950) on the eyeball muscles of man, like those of Reuter (1897) and Keil (1906) on the eye muscles in the pig, were made on embryos belonging to relatively late stages of development. The present study, based on a younger and much more extensive series of human embryos than was available to Lewis and Carnicer, reveals that: (I) transitory head cavities do exist in a premandibular condensation on each side of the head, and that this condensation subsequently gives rise to the extrinsic ocular muscles innervated by the oculomotor nerve; (2) the premandibular condensations are formed by cells proliferated from the prechordal plate; (3) the superior oblique and lateral rectus muscles arise from separate primordia adjacent to the premandibular condensations; (4) the eyeball muscles in man, therefore, do not arise from “a common premuscle mass," but rather from three separate primordia on each side of the head; (5) the development of the extrinsic ocular muscles in man follows a pattern which conforms in essential features with that found in elasmobranchs, reptiles, birds, marsupials, and the cat.
Acknowledgments
Much of this investigation was conducted at the Department of Embryology, Carnegie Institution of Washington, in Baltimore, Maryland, during a sabbatic leave from Cornell University. It was made possible in part through a generous research grant from the Carnegie Institution of Washington; to both that Institution and Cornell University the author is most grateful. The author wishes to express his appreciation to Dr. George W. Corner, Director of the Department of Embryology of the Carnegie Institution of Washington, for his many kindnesses and for the privileges of the laboratory. It is also a pleasure to thank Dr. Chester H. Heuser and Dr. Robert K. Burns, of the Carnegie laboratory, and Dr. Howard B. Adelmann, of the Cornell University Department of Zoology, for their helpful advice and interest in this problem. The author is grateful to the late Mr. James Didusch and to Mrs. Mary M. Cope for the skillfull preparation of the illustrations; their patience and care in the execution of those drawings based on the plastic and plaster reconstructions are particularly appreciated. To Mr. O. 0. Heard and Mrs. Cope the author is indebted for assistance in making the plaster and plastic models, and to Mr. Richard Grill for the preparation of the photomicrographs.
Material And Methods
The material for this study consisted of approximately 100 human embryos (table 1), ranging in size from presomite to 32+ mm. All the embryos belong to the collection of the Department of Embryology, Carnegie Institution of Washington. The fact that these embryos, largely through the efforts of the late Dr. George L. Streeter, are arranged in the collection according to their developmental age (horizon) contributes immeasurably to the ease and efliciency with which a study of this kind can be pursued.” Paper, plastic, and plaster reconstructions of important stages in the development of the eyeball muscles were made.
The Prechordal Plate And The Origin Of The Premandibular Condensations
In most, and possibly all, fishes, amphibians, reptiles, and birds, the eyeball muscles innervated by the oculomotor nerve arise from mesoderm derived from the prechordal plate. Parker (1917) found this to be true in marsupials also, and the author has pointed out the sig nificance of the prechordal plate in this connection in the cat (Gilbert, 1947) and opossum (Gilbert, 1954). An understanding of the prechordal plate is, therefore, of paramount importance in ascertaining the origin of the oculomotor eyeball musculature.
- For a description of horizons xi to xxiii, consult Streeter (1942. 1945. 1948, 1951)
Table 1
| Table 1 - Human Embryos of the Carnegie Collection Utilized in Studying the Development of the Extrinsic Ocular Musculature | |||||
| Horizon | Approximate age (days) | Embryo number | Somites, or crown-rump length (mm) | Plane of section | Thickness of sections (microns) |
|---|---|---|---|---|---|
| x | 22 ± 1 | 4216 | (7) | T | 15 |
| 5074 | (10) | T | 10 | ||
| xi | 24 ± 1 | 2053 | (20) | T | 10 |
| 6050 | (19) | FT | 10
6750 10.0 T 10 | ||
| 6344 | (13) | T | 6 Z115 9'? F 1° | ||
| 6784 | (17) | T | 6 Template:CE1629 11.5 F 10 | ||
| 7611 (16) T 8 7804 9.5 T 10 | |||||
| 8005 <19 T 8 Template:CE3312 133 E ‘S | |||||
| xii | 26 ± 1 | 486 E21) T 10 Template:CE3179 11.9 F 10 | |||
| xiii | 27 ± 1 | ||||
| xiv | 29 ± 1 | ||||
| xv | 31 ± 1 | ||||
| xvi | 33 ± 1 | 6509 | 8.1 | F | 10 |
| 6511 | 8.1 | S | 10 | ||
| 6513 | 7.2 | F | 10 | ||
| 5515 | 11 | 15 S 3 | |||
| xvii | 35 ± 1 | ||||
| xviii | 37 ± 1 | ||||
| xix | 39 ± 1 | ||||
| xx | 41 ± 1 | ||||
| xxi | 43 ± 1 | ||||
| xxii | 45 ± 1 | ||||
| xxiii | 47 ± 1 | 4289 32.2 T 15,20 | |||
| 4525 | |||||
| 4570 | |||||
| 5154 | |||||
| 5725 | |||||
| 7425 | |||||
| Unclassified.. ca. 9 weeks | Template:CE5017 | 32.2 | S | 40 | |
| Unclassified.. ca. 9 weeks | 9226 | 31.0 | T | 10 | |
6506 7.5 F 10
163 3.0 T 20
5154 310 T 20 617 /-0 T 15
23.0 F 25 2 fig
|} 4245.7 ca. 24) T 10 .. 4736 (26) F 10
6519 10.8 s 8
6097 (25) T 10
6631 13.0 F 10
6144 (27) T 10
8118 12.6 F 10
{{CE7724 en. (29) s 8
1909 14.6 F 20
7852 (25) T 10
4430 13.4 T 15
7999 ca. (28) T 10
6522 13.2 F 10
{{CE8505A (24) T 8
6524 11.7 T 10
850513 (23) s 8
6528 13.4 F 8 xiii ....... ,. 27:1
463 3.9 F 10 1; 6 8
588 4.0 F 15 “' ’
(B6 4 0 T 15
8097 15.5 T 10
Gjm 510 F 6
8235 14.0 s 10
7433 5.2 F 8
409 18.0 T 20
Template:CE7613 4,8 F 10
1390 18.0 S 20
8119 5.3 T 8
5609 18.0 F 25
8239 4.3 s 8
6824 18.5 s 12 35111 4-3 5 3-111 .6
431 19.0 s 20
1620 6.6 s 20
4059 21.6 F 15
6428 7.0 F 6, 10
6202 21.0 s 20
6500 4.9 s 10
6426 21.5 T 20
6503 6.3 F 10
8157 20.8 F 20
533° 55 F 3 xxi ....... .. 43:1
4960 22.0 T 15
€343 2-3 1’ 10
7254 22.5 T 20
E 3
7392 22.7 T 20
8553 22.0 T 12 1” ------- -- 31’—"1 3" xxii . . . . . . . .. 45:1
4304 25.0 T 20
4339 24.5 T 15
Template:CE1005 9-11 F 211
6701 24.0 F 20
8394 25.3 T 20
Template:CE4502 9:3 3 ]5 ma; ....... .. 47:1
‘ Number of somitcs given in parentheses.
T T, transverse; F, frontal; S, sagitml.
The prechordal plate is a median mass of cells, located at the anterior end of the notochord; it appears in early embryos as an integral part of the roof of the foregut.” From it, prechordal mesoderm is proliferated laterally. According to Adelmann (1922), it is to be regarded as “an essentially mesodermal structure” which participates in the marked bilateral growth of the embryonic head. "The extent to which this bilateral growth occurs and the stage of development at which it is initiated,” Adelmann believes, “probably differs to some extent in different forms, and this may account for the variations in the extent to which the prechordal plate is developed in various forms. Marked bilateral growth occurring early in development would cause a less noticeable medial accumulation of material anterior to the notochord. This may possibly account for the poorly developed prechordal plate which has been described in mammals.”
The prechordal or prochordal plate has been described by Heuser (1932) in a presomite human embryo (no. 5960) having an ovulation age of approximately 18 days. Traced forward from the anterior end of the notochord, the plate, according to Heuser, “is found to gradually increase in thickness until in its middle portion as many as eight rows of nuclei can be counted. However, the dorsal surface is very irregular, since cells are being given off to the surrounding tissue and some of them should no doubt be classified as mesodermal cells.” One would surmise, therefore, that the prechordal plate has, at this early stage, already begun to proliferate prechordal mesoderm. Heuser further noted that the plate “becomes thinner and broader in the anterior part; in the first six sections it is composed of two layers of cells. The front end of the prochordal (prechordal) plate can be definitely located in section 205, since in front of it the entoderm abruptly changes to a layer of one cell thick. Sections through the plate, especially in its middle portion, show irregular outlines with bulgings from the ventral surfaces into the cavity of the yolk-sac. Ridges and grooves thus appear on the ventral surface of the plate."
Until more material is available that is sectioned and I stained to show the prechordal plate clearly, it will not be possible to follow in detail its cytological development in the human embryo. Glimpses of what is going on in this area may be obtained, however, by examining the prechordal region of human embryos ranging from 7 to 20 somites.
3 Several investigators have confused the prechordal plate with an extensive area of endoderm (and mesoderm?), known as the protochordal plate, located at the anterior end of the notochord of early embryos which develop from yolk-rich eggs or eggs which behave as if they were yolk-rich. Some workers have employed the term prochordal plate for protochordal plate, whereas others have used it in a more restricted sense as a synonym for prechordal plate. A discussion of the difference between prechordal, prochordal, and protochordal plate lies beyond the scope of this paper; the subject has been treated by Parker (1917), Hill and Tribe (1924), and Adelmann (1922, 1926).
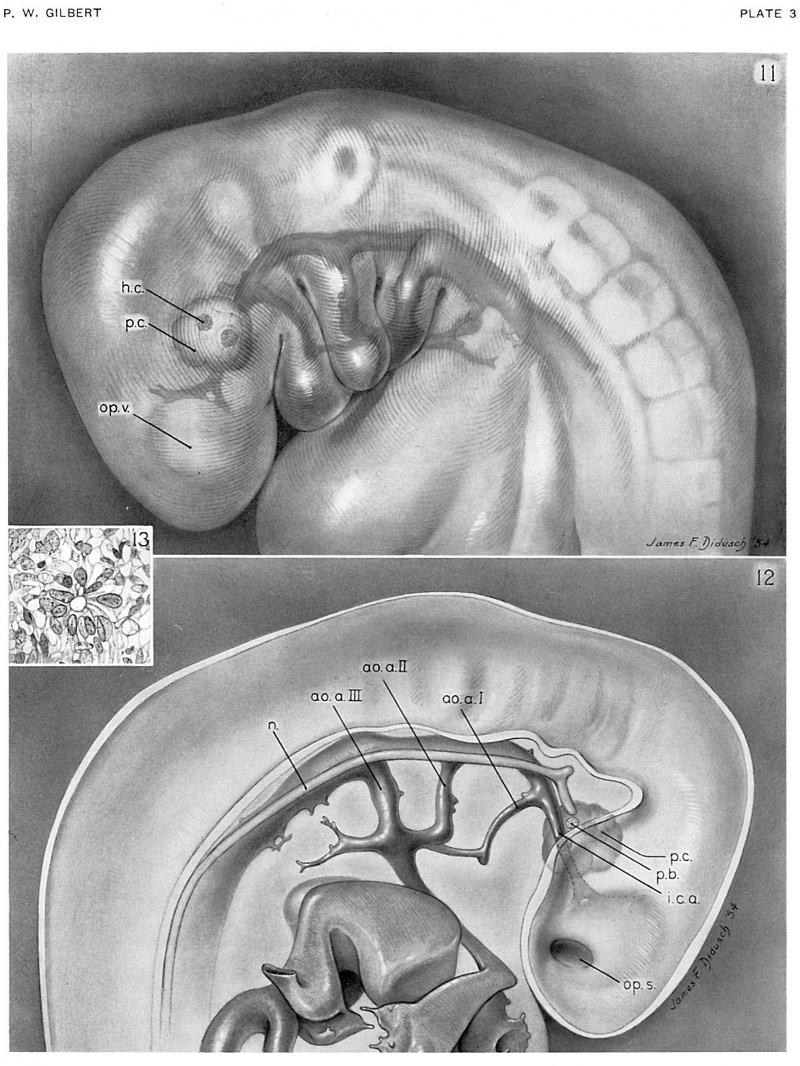
In a 7-somite human embryo (no. 4216), the youngest examined in this study, the neural folds have met in the region between somites 3 and 7; otherwise, the central nervous system is widely open dorsally, and the troughlike brain has differentiated into three primary divisions. The head is slightly bent in the region of the midbrain so that the forebrain is directed toward the yolk sac. The optic sulcus is just beginning to form, and traces of the otic plate can be seen. The heart, the dorsal aorta, and the first pair of aortic arches have appeared. Only in the region immediately ahead of the primitive streak does the notochord exist as an independent structure, lying between the floor of the neural tube and the endoderm of the gut roof. Anterior to this region the notochord appears as a thickened plate of cells on the dorsal wall of the gut.
The prechordal plate of this 7-somite embryo was first noted by Payne (1925), who described it as “a thickened mass of cells continuous dorsally and posteriorly with the notochord, and extending caudally along the ventral surface of the gut for a short distance. Dorsally, it is in Contact with the ectoderm of the neural groove but is not fused with it.” The author has verified the presence of this mass of cells, 15 to 20 in number, and is in full agreement with Paync’s interpretation. The structure Payne describes corresponds closely to the prechordal plate from which prechordal mesoderm is proliferated in slightly older embryos.
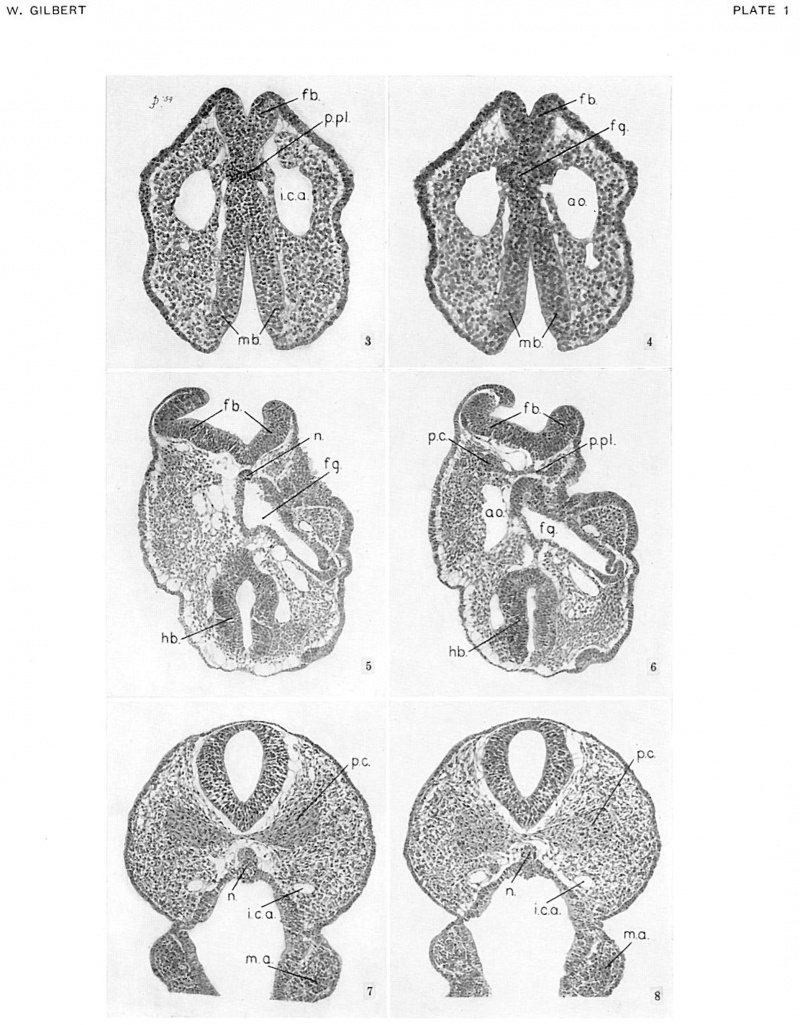
As development proceeds, the cephalic flexure in the region of the midbrain becomes more and more pronounced, so that, in embryos with 10 to 13 somites, the long axis of the forebrain forms a right angle with that of the hindbrain. Concomitantly with the bending of the brain, the prechordal portion of the roof of the foregut in contact with the brain is bent ventrally, the prechordal plate becoming vertical in the process. Transections through this region of the head are, therefore, most unfavorable for observing the prechordal plate at this stage, since it appears in only one or two sections between the ventral (now posteriorly directed) floor of the forebrain and the lumen (originally dorsal) of the foregut. This is probably one reason why the prechordal plate of young mammalian embryos has so often been overlooked. In the Io-somite embryo (no.- 5074), for example, the prechordal plate appears in two 10-micron sections only, crowded in between the floor of the forebrain and the anterior wall of the foregut (fig. 3, pl. 1). Posteriorly, the prechordal plate is continuous with the notochord, which is still embedded in the roof of the gut throughout most of its length. At this stage, the prechordal plate consists of about 35 to 40 cells. These cells can be clearly delimited from the forebrain anteriorly, but not from the cells that will ultimately line the roof of the foregut. Determination of the caudal limit of the prechordal plate is also difficult, for the cells blend imperceptibly with the tip of the notochord.
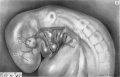
‘Nith its expansion restricted by the floor of the fore-brain, the prechordal plate, of necessity, can proliferate prechordal mesoderm only laterally over the “knee” of each aorta. For a short time the prechordal mesoderm appears as a thin lamina of cells anterior to the first aortic arch of each side. As more and more prechordal mesoderm is proliferated, it forms a globular mass of cells cranial and lateral to the ‘‘knee’' of each aorta. These masses of prechordal mesoderm are known as the premandibular condensations; they abut against the maxillomandibular mesoderm laterally, and from the beginning are connected with each other across the mid-line by a thin lamina or bridge of mesodermal cells of prechordal—plate origin. The development of the premandibular condensations in the human embryo is illustrated schematically in figure 1.
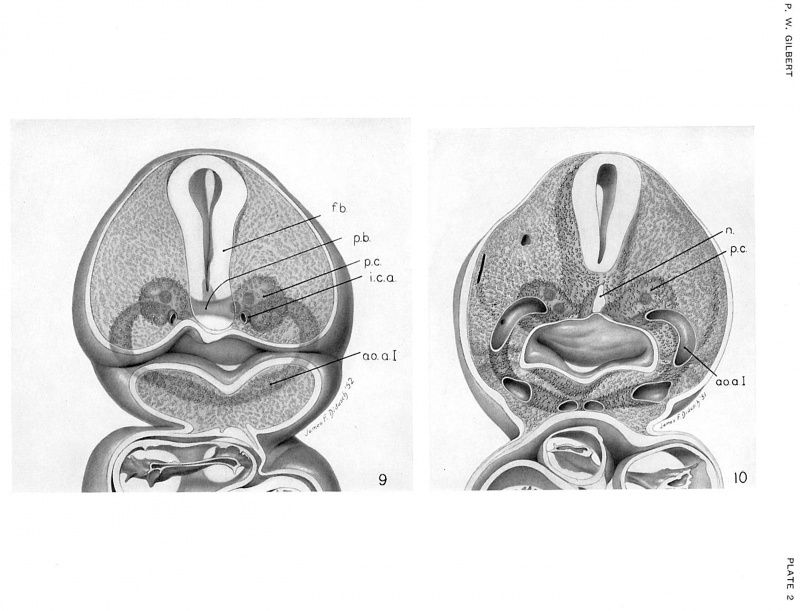
A pair of small premandibular condensations first appears in human embryos of 14 somites between the “knee" of the aorta and the floor of the forebrain (fig. 6, pl. 1). As development proceeds, and an internal carotid artery grows forward from the "knee" of each aorta, the premandibular condensations, connected by a narrow bridge of prechordal mesoderm, increase in size and occupy a position above and external to each internal carotid artery. By the 19—somite stage (figs. 7, 8, pl. 1), each premandibular condensation is well developed and occupies a position adjacent and lateral to the internal carotid artery, and somewhat above and medial to the optic vesicle. Laterally, the premandibular condensations abut against, and almost imperceptibly blend with, the maxillomandibular mesoderm; medially, they remain joined across the mid-line by a solid transverse bridge of cells.
In the stage that immediately follows (20 to 30 somites), lateral expansion of the head in this region continues, and the premandibular condensations enlarge. During this period of rapid growth of the head as a whole, small epitbelit1m—lined spaces, the head cavities, appear in the premandibular condensations. It is remarkable that, of all the placental mammals that have been thus far studied, man is the only one in which premandibular head cavities have been found. Their origin, structure, and fate in the human embryo are, therefore, of special interest.
In this study, head cavities were observed in a total of ten human embryos belonging to horizons xii, xiii, and xiv (table 2). Nine of the embryos belong to horizon xii or xiii and range in size from 24 somites to 5.2 mm.[1]
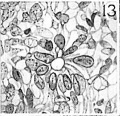
An embryo of 28 somites (no. 7999), belonging to horizon xii, about 26 days of age, may be considered as representative. In this embryo (figs. 32, 33, pl. 8), three small cavities appear in the premandibular condensation of the right side, and one somewhat larger head cavity is located in the left premandibular condensation. The lumen of this last cavity is about equal in diameter to the height of the single layer of columnar epithelial cells surrounding it, and occupies a position in the premandibular condensation about 40 microns above the internal carotid artery. The entire head cavity, including its wall of radially arranged columnar epithelial cells, measures about 40 microns by 45 microns, and is only slightly greater in diameter than that of the internal carotid artery at this level. The three head cavities located along the_ upper cranial border of the right premandibular condensation of embryo no. 7999 are arranged in an oblique row which parallels the floor of the forebrain. The most caudal and medial of the three head cavities on this side has the largest lumen, and, like its somewhat larger fellow on the left, is situated about 40 microns above the internal carotid artery.
In certain embryos, for example no. 7724, the ventral wall of a head cavity may actually abut against the dorsal wall of the internal carotid artery (fig. 27, pl. 7). In others, for example no. 8239, a small head cavity is found some distance (about 30 microns) medial to the internal carotid artery in the tapered medial end of the premandibular condensation near its junction with the bridge. For the most part, however, head cavities, when present, are situated in the premandibular condensation above or lateral to the level of the internal carotid artery. A glance at table 2 reveals no uniformity in their location or number, and they have been as frequently encountered on the right side of the head as on the left.
The head cavities in the human embryo are never as large as those in many reptiles and birds. They resemble very closely, however, early stages in the development of these structures in both the turtle and the chiclt; that is, they appear as small isolated lumina, lined with cuboidal or columnar epithelium, in the premandibular condensations. In reptiles and some birds, it will be recalled, the separate lumina eventually coalesce to form one or more larger cavities on each side, from the walls of which the oculomotor eyeball muscles presently arise. But, in man, no such fusion of separate lumina to form a large single cavity has been observed.
It cannot yet be stated whether head cavities develop in the premandibular condensations of all human embryos. Certainly many of the embryos belonging to horizons xii and xiii in the Carnegie Collection do not possess them. Although it is possible that these embryos never did have, or never would have developed, head cavities, it is more likely that cavities do form with some regularity in embryos of horizons xii and xiii, last but a few hours, and then become crowded out by the rapid multiplication of cells in the premandibular condensations. It is probable, therefore, that the head cavities vanish as suddenly as they appear, and by the time the human embryo reaches its goth day no trace of these transient structures can be found.
Table 2
Human Embryos or Tm: C.-\a.\'F.r.u: COLI.P.CTIO.\' nr..\io.\'sra.\Tmc HEAD CAVITIES
Head ca\'i- Total head
xii . . . . . . . . . . . . . . . . 4736 26 som. F 1 2 3 2-5-3, 4, 5, 6, 7 xii . . . . . . . . . . . . . . . . . . 6097 25 som. T 0 1 I l-4-2 xii . . , . . . . . . . .. 7724 ca.29so1n. S 1 4 5 2-1-6, 8, 12; 2-3-1], I2 xii . . . . . . . . . . . . . . . . . . 7999 ca. 28 som. T 3 l 4 2-1-1 1, 12 xii . . . . . . . . . . . .. 3505A 24 som. T 0 l 1 2-7-9; 3-1-1 xiii. . . . . . . . . . . . . . 463 3.9 mm. F 1 0 1 5-2-5, 6, 7 xiii . . . . . . . . . . . .. 7433 5.2 mm. F 3 l 4 2-3-I5; 3-1-1, 2 xiii. . 8239 4.3 mm. S 1 1 2 2-3-7; 3-1-8 xiii . . . . . . . . . . . . . .. 8581 4.8 mm. 5 1 1 2 1-6-4; 2-4-4 xiv. . . . . . 6830 5.5 mm. F I 0 l 4-3-1, 2, 3| Horizon | Embryo no. | Size of embryo section | Plane of Head cavities, right ties, left cavities Slide, row. section |
|---|
' F, frontal; S, sagittal; T, transverse.
The Maxillomandibular Mesoderm
In embryos belonging to horizon xi, a diffuse mass of mesenchymal cells, situated just beneath the surface ectoderm at the upper end of the mandibular arch and maxillary process, can be distinguished. For convenience, this mass is referred to as the maxillomandibular mesoderm. Part of its medial surface adjoins the developing premandibular condensation; anteriorly, it crowds against the optic vesicle, and ventrally it is continuous with the somewhat denser mesoderm of the mandibular arch. At this stage a prominent cephalic flexure exists in the region of the midbrain, so that the forebrain forms an angle of about 90° with that of the hindbrain. It is important to bear in mind that at this time the maxillomandibular mesoderm crowds against the caudal surface of the optic vesicle that is evaginating from the cephalic end of the forebrain. As development proceeds (horizon xii), the cephalic flexure becomes more prominent, and the angle formed by the long axis of the forebrain with that of the hindbrain becomes more acute (see Streeter, 1942, p. 241, fig. 7). During this period the maxillomandibular mesoderm becomes more compact; because of the ventral rotation of the forebrain, its cranial end now lies adjacent to the dorsocaudal surface of the optic vesicle (figs. 30, 31, pl. 7).
The cephalic flexure continues to increase in prominence in embryos of horizon xiii; from the dorsocranial border of the maxillomandibular mesoderm an arm of cells extends forward over the optic vesicle. Since the head is expanding rapidly in this region, the arm of maxillomandibular mesoderm above the optic vesicle keeps pace and moves laterally as well as cranially. The caudal portion of the maxillomandibular mesoderm, lying in a region expanding less rapidly, curves, therefore, into a plane lying medial to its cranially directed arm.
The position of the maxillomandibular mesoderm relative to the optic vesicle and premandibular condensation at this stage is illustrated schematically in figure I.
In embryos belonging to late horizon xiii, a condensation appears in the caudal and more medial portion of the maxillomandibular mesoderm, medial to the Gasserian ganglion and vena capitis medialis. A homologous condensation found in cat embryos at a comparable stage of development has been termed the Vlth condensation, and from it arise the retractor bulbi and lateral rectus muscles, both innervated by cranial nerve VI. Since man lacks a retractor bulbi muscle, this condensation in the human embryo will be referred to as the lateral rectus primordium. Anteriorly, this condensation abuts against the premandibular condensation, and at later stages is very difiicult to delimit from it.
Shortly after the primordium of the lateral rectus becomes established, a second condensation appears in the maxillomandibular mesoderm. This condensation, the primordium of the superior oblique, is first observed in embryos of horizon xiv in the cranial arm of maxillomandibular mesoderm that has extended forward above the eye. It is somewhat smaller and less dense than the lateral rectus primordium, and occupies a position between the vena cerebralis anterior and the dorsocranial surface of the optic cup. Situated between the Gasserian vanglion and optic cup, and lateral to the bifurcation in the vena capitis medialis, a more diffuse mass of mesodermal cells connects the condensations for the superior oblique and lateral rectus muscles. This mass of cells, which Fraser (1915) has termed the “intermediate mass” in Tric/zosurus and Adelmann (1927) the “intermediate region” in the chick, abuts medially against the premandibular condensation and is continuous ventrally with the denser mesoderm of the mandibular arch. These relations are illustrated schematically in figures I and 2.
While the superior oblique and lateral rectus primordia are increasing in density, the intermediate mass adjacent to the caudal surface of the optic cup spreads forward above and below the outer margin of the eye, beneath the surface ectoderm. In this mesoderm around the periphery of the optic cup there appear in older embryos, belonging to horizon xiv, four faint condensations, each approximately equal in diameter to that of the lens vesicle, which is in the process of invagination. These peripheral condensations, first described by the author (Gilbert, 1947) in the cat, and termed the superior, anterior, inferior, and posterior peripheral condensations, are believed to contribute to the formation of the scleral coat of the eye, and they mark the points of insertion on the sclera of the four rectus muscles which, at a later period, grow out into them. The peripheral condensations in the human embryo are less dense than in the cat, and are most readily observed in parasagittal sections through the eye in embryos belonging to horizons xv, xvi, and xvii. The position of the four condensations relative to the eye and the superior oblique primordium, in embryos of horizons xv and xvi, is illustrated in figures 2, 52 (pl. 11), and 53 (pl. 11). Because of the lack of material suitably sectioned and the lack of density of those condensations in the human embryo, their fate could not be followed as clearly as it was in the cat. However, since in the time of their appearance and in their location relative to the periphery of the optic cup they agree with the similar condensations found in the cat, we may assume that they probably have the same fate.
Before discussing the subsequent development of the lateral rectus and superior oblique muscles, the relation of these two eye-muscle primordia to the premandibular condensation (from which the oculomotor eyeball muscles develop) in the human embryo and in other vertebrates deserves comment. There is a striking similarity in these three separate and distinct eyeball-muscle primordia at this stage of development (lens vesicle invaginating and closing off or closed off from surface ectoderm) in mammals, birds, reptiles, and elasmobranchs. The premandibular condensations of the mammal are directly comparable to the premandibular head cavities of birds, reptiles, and elasmobranchs. In all groups, a solid transverse band or hollow cross canal at first connects the premandibular condensation or cavity of the right side with its fellow on the left. This transverse connection passes above the internal carotid arteries and across the confined area between the cranial tip of the notochord and the floor of the forebrain. The maxillomandibular mesoderm, exclusive of the lateral rectus primordium, is similar with respect to location and fate to the mandibular head cavity or homologous condensation of birds,“ reptiles, and elasmobranchs. finally, the Vlth condensation of the cat or lateral rectus primordium of man has its counterpart in the hyoid cavity or condensation in the embryonic head of lower vertebrates. In both cat and man, this eyeball—muscle primordium, in its relations to the eye, first pharyngeal pouch, Gasserian ganglion, superior oblique primordium, and premandibular condensation, bears a conspicuous resemblance to the hyoid condensation of birds and the hyoid head cavity of reptiles and elasmobranchs.
The Development of the Lateral Rectus
The earliest primordium of the lateral rectus, it will be recalled, is situated caudal and slightly dorsal to the premandibular condensation and medial to the maxillary division of the Gasserian ganglion in embryos belonging to horizon xiii. Although poorly defined in embryos of this horizon, the lateral rectus condensation quickly increases in density, and in several embryos of horizon xiv it is easily recognized. Its shape is roughly ovoid; its long axis extends cephalocaudall '; and its total volume is approximately one-third that of the premandibular condensation. As development proceeds (horizon xv), the lateral rectus condensation, while retaining its confluence with the intermediate mass of the maxillomandibular mesoderm, increases in density and size as it swings craniolaterally across the dorsocaudal surface of the premandibular condensation, through the fork in the vena capitis medialis formed by its bifurcation into the vena cerebralis anterior and the vena orbitalis inferior. The abducens nerve, which has emerged by several rootlets from the floor of the hindbrain, extends forward ventrally and laterally, and penetrates the dorsocaudal surface of the lateral rectus condensation. The ophthalmic nerve has, in the meantime, become conspicuous above, and cranial to. the distal end of the lateral rectus condensation; the maxillary nerve now extends ventrally behind the distal end of the condensation into the maxillary process. Medially, a portion of the lateral rectus condensation appears to be confluent with the premandibular condensation, and. were it not for the fact that the oculomotor nerve, which has grown ventrally from the midbrain, now passes between the premandibular and lateral
- Variation exists among vertebrates as to which end of the superior oblique primordium grows medially and eventually attaches to the orbital wall. In C/zclyzlm (Johnson, 1913)a the cranial end of the superior oblique primordium swings medially during development and eventually attaches to the orbital wall. On the other hand, in Torpn/o (Wedin, 1949), chick (Adelmann. 1027), xlrdcu (Wedin, 195312), Tric/mmru.r (Fraser, 1915), cat (Gilbert, 1947), and man, the caudal end of the superior oblique primordium grows medially to gain insertion on the wa|l of the orbit, whereas the cranial end eventually inserts on the sclera.
Fig. 1. Diagrams, based on reconstructions of embryos of horizons xiii and xiv, illustrating the relations of the maxillomandibular mesoderm (figs. A, D) and the premandibular condensation (figs. B, E) to the brain, eye vesicle, Gasserian ganglion, and mandibular arch. figures C and F represent transeetions through the brain of embryos of horizons xiii and xiv respectively at the levels indicated in figures A and D; the maxillomandibular mesoderm and premandibular condensation have been superimposed on the left side; the developing eye is illustrated on the right side of each transection.
In embryos of horizon xiii :1 prominent pear-shaped premandibular condensation (p. c.) is situated dorsal and caudal to the medial surface of each optic vesicle and is in contact laterally with the maxillomandibular mesoderm (m. m. m.). The rapid expansion of the head anterior to the end of the foregut, in embryos of horizons xi and xii, is reflected not only in the growth of the premandibular eondensations but also in the lateral movement of the maxillomandibular mesoclerm above the expanding optic vesicles, so that in embryos of horizon xiii the caudal portion of the maxillomandibular mesoderm, situated in a region which has expanded less rapidly, lies in a plane some distance medial to its cranial portion above the eye.
In embryos of horizon xiv, a small process, the primordium of the superior rectus (5. r.), extends forward and outward from the dorsolateral border of the premandibular condensation. The primordium of the superior oblique (5. 0.) and that of the lateral rectus (I. r.) have appeared as condensations in the cranial and caudal portions of the maxillomandibular mesoderm respectively, and are connected by a more diffuse area of maxillomandibular mesoderm known as the intermediate mass (int.). rectus condensations, it would be difficult, if not impossible, to distinguish one condensation from the other. It is not surprising, therefore, that earlier investigators, when examining embryos of this and older stages, failed to distinguish the two condensations and believed them to be one.
In embryo no. 3385, a typical representative of horizon xv, the lateral rectus condensation is well defined, and its relation to the premandibular condensation, principal nerves, and blood vessels of the head is readily ascertained. The caudal and more medial end of the condensation is situated midway between the internal carotid artery and that portion of the vena capitis medialis which lies adjacent and medial to the root of the maxillary nerve. A few sections cranial to its caudal end, the lateral rectus condensation receives the abducens nerve, and from this point proceeds cranially and laterally along the dorsocaudal face of the premandibular condensation, from which it is separated for a short distance by the oculomotor nerve, and thence laterally in front of the fork in the vena capitis medialis into the diffuse “intermediate mass” of maxillomandibular mesoderm. To what extent this mass, situated between the roots of the ophthalmic and maxillary nerves, participates in forming the distal end of the lateral rectus is difficult to ascertain. '
At horizon xv the “intermediate mass” is less dense than the medial portion of the lateral rectus condensation, and is so situated that the lateral rectus condensation would have to grow through it in the next few days, or receive a contribution from it, before reaching the eyeball. In the eat, it was concluded that the latter phenomenon takes place, and, from the limited material at hand for horizon xv, it appears probable that the “intermediate mass” makes some contribution to the distal end of the lateral rectus in man also.
By horizon xvi, the lateral rectus condensation is sufficiently dense to be traced from its caudomedial extremity forward and laterally in front of the fork in the vena capitis medialis to the posterior peripheral condensation on the outer margin of the optic cup (figs. 47-50, pl. 10). The relation of the lateral rectus condensation at subsequent stages (horizons xvii, xix, xxiii) to the other eye muscle primordia, the eyeball, and the ophthalmic and maxillary nerves is illustrated in plates 4, 5, and 6. The lateral rectus has a well defined insertion in embryos of horizon xix, and its proximal (origin) end becomes clearly separated from the other extrinsic ocular muscles in embryos of horizon xx. Muscle fibers appear in the lateral rectus condensation as early as horizon xx; Bartelmez (personal communication) has recently found, in teased preparations of this muscle, that Q, J, and Z lines are present in the myofibrillae of a 25 mm. embryo (no. Template:CE5635) belonging to horizon xxii.
The Development of the Superior Oblique
The superior oblique primordium, it will be recalled, appears first as a condensation in an arm of maxillo-mandibular mesoderm extending from the upper end of the mandibular arch forward over the dorsal surface of the optic vesicle, close to the surface ectoderm. The condensation is first visible slightly dorsal and medial to the superior peripheral condensation in older embryos of horizon xiv (fig. I). At this stage the superior oblique condensation is less dense and somewhat smaller than the lateral rectus primordium, and considerably smaller than the premandibular condensation. The ophthalmic nerve lies close to the superior oblique condensation, if not actually embedded in it.
During the next 6 days (horizons xv to xvii), the head continues to expand rapidly, the ophthalmic nerve divides into frontal and nasoeiliary branches, the optic stalk lengthens, and the optic vesicle increases markedly in diameter. Concomitantly with the enlargement of the optic vesicle, and possibly as'a result of it, the caudal end of the superior oblique primordium shifts medially, retaining an intimate association with the frontal branch of the ophthalmic. In an embryo of horizon xvii (no. 6258), the superior oblique primordium has become more elongated and is crescentic (fig. 16, pl. 4). Its center lies medial and cranial to the superior peripheral condensation, and its cranial (insertion) end bends downward and backward (in the region of the future trochlea), almost touching the periphery of the optic vesicle between the superior and anterior peripheral condensations. The caudal end of the superior oblique primordium is situated in a plane medial to that of its cranial end, and is penetrated by the trochlear nerve, the last of the eye—muscle nerves to appear, at a point immediately ventral and lateral to the vena cerebralis anterior.
The relation of the frontal branch of the ophthalmic to the superior oblique primordium is of interest at this stage. As the caudal end of the superior oblique condensation moves medially, it passes obliquely above the frontal nerve. More anteriorly, the frontal nerve still retains intimate contact with the ventral surface of the superior oblique, and emerges lateral to it in the region where the trochlea will presently form. The caudal half of the superior oblique condensation, therefore, “pivots" medially on the dorsal surface of the frontal nerve.
Four days later (horizon xix, no. 5609), the caudal end of the superior oblique primordium has grown much farther medially and caudally, practically reaching the point where it will ultimately become attached to the orbital wall (Fig. 20, pl. 5). The progressive growth medially of the caudal portion of the superior oblique primordium, especially pronounced in embryos of horizons xix and xx, is shown in table 3 for a series of representative sagittally sectioned embryos belonging to horizons xvi to xx. In each case, the number of sections in which a portion of the superior oblique primordium appears has been counted and multiplied by the thickness of the section to give the lateromedial (transverse) extent of the condensation. The frontal division of the ophthalmic nerve now lies dorsal to that portion of the superior oblique about which the trochlea will soon form; the nasociliary division lies ventral to it.
Table 3
Transverse Extensor or superior onuous Primordium Transverse extent of superior
| Horizon | Embryo | embryo (mm) | Length of oblique (microns) |
|---|---|---|---|
| xvi | 1197 | 10.0 | 160 |
| xvii | 6519 | 10.8 | 224 |
| xviii | 6533 | 12.5 | 300 |
| xix | 1390 | 18.0 | 640 |
| xx | 431 | 19.0 | 880 |
By the time the human embryo is 42 days old (horizon xx), striated fibers are recognizable in the superior oblique muscle, which extends from its narrow tendon of origin on the anterior margin of the optic foramen forward along the dorsomedial wall of the orbit, terminating anteriorly in a round tendon which, after passing through the developing trochlea (figs. 73, 74, pl. 14), is deflected ventrally, laterally, and caudally beneath the superior rectus to its insertion on the sclera on the equator of the eyeball. The frontal nerve at this stage extends forward midway between the bellies of the superior oblique and levator palpebrae superioris muscles. As the two eyes move forward, the frontal nerve comes to lie directly above the levator palpebrae superioris. During this period, the angle formed by the proximal and distal portions of the superior oblique tendon at the trochlea (trochlear angle) becomes more acute.
It is interesting that the trochlear angle appears in the superior oblique condensation as early as horizon xvii, and in a sense anticipates the formation of a trochlea which does not become evident until 5 or 6 days later (horizon xx). From the beginning, the trochlear angle is obtuse; even in an embryo of horizon xix, just before the trochlea appears, it is about 120°. After the U-shaped trochlea forms about the superior oblique tendon and becomes attached to the developing frontal bone near the anterior margin of the orbit, the trochlear angle becomes progressively more acute. This increase in the acuity is probably the result of the forward growth of the portion of the frontal bone to which the trochlea is attached. Thus the trochlear angle, which was about 120° in horizon xix, becomes about 95° in horizon xxii (fig. 75, pl. 14), about 80° in horizon xxiii (fig. 24, pl. 6), and about 55° in adult man.
The Fate Of The Premandibular Condensations
The early history of the premandibular condensations and the development of small, transitory, epitheliumlined head cavities within them, in embryos belonging to horizons xii to xiv, has already been sketched. Each condensation is formed from mesodermal cells proliferated laterally from the prechordal plate; as the head expands, each condensation, which from the beginning is connected with its fellow by a solid transverse bridge of mesoderm, rapidly enlarges. In early embryos belonging to horizon xiv, each pear-shaped premandibular condensation is well developed and anteriorly almost reaches the caudomedial surface of the optic vesicle, its tapered medial end passing above the internal carotid artery; caudally, it is in contact with, and difficult to delimit from, the primordium of the lateral rectus. At this time the premandibular condensations are about twice the size of the lateral rectus primordium (described above) and are still connected across the mid-line by a rather tenuous transverse bridge of mesodermal cells which, in the mid-plane, is in contact with the cranial tip of the notochord.
In older embryos of horizon xiv, the middle of the transverse bridge ruptures, and the two premandibular condensations are no longer connected. During this period, when the lens vesicle is invaginating, a small bud of mesodermal cells (the primordium of the superior rectus) appears on the dorsolateral portion of the premandibular condensation, and extends forward and laterally toward the upper surface of the optic vesicle to the level of the fork in the vena capitis medialis (fig. 35, pl. 8).
During the next 2 days (horizon xv), the lens vesicle closes of? from the surface ectoderm, and cranial nerve III and the frontal branch of the ophthalmic nerve appear. The superior rectus primordium is closely apposed to the caudomedial surface of the superior oblique condensation, from which it may with some difliculty be distinguished, since the frontal nerve usually passes between these two condensations. In slightly older embryos (horizon xvi), the superior rectus primordium extends cranially and laterally as a rather dense arm of cells and establishes a diffuse connection with the superior peripheral condensation. Proximally, the frontal branch of the ophthalmic nerve passes above the superior rectus primordium, and the nasociliary branch passes below it near its junction with the parent premandibular condensation. The oculomotor nerve has grown down between the caudal face of the premandibular condensation and the cranial border of the lateral rectus primordium (now supplied by the abducens nerve), and is of considerable assistance in delimiting these two intimately associated condensations. In fact, were it not for our knowledge of the early independent origins of these two condensations, it would be extremely difficult to separate them at this time, and one might easily, as did Lewis (1910), erroneously consider them to be one.
Fig. 2. Diagrams, based on reconstructions of embryos of horizons xv, xvi, and xvii, illustrating the relations of the derivatives of the maxillomandibular mesoderm (figs. A, D) and the premantlibular condensation (figs. B, E) to the brain, eye vesicle, Gasserian ganglion, and mandibular arch. figure C represents a transection through the brain of an embryo of horizon xv at the level indicated in figure A. The derivatives of the maxillomandibular mesoderm and the premandibular condensation have been superimposed on the left side; the developing eye is illustrated on the right side of the transection.
In addition to the superior oblique and the lateral rectus primordia, four other condensations appear in embryos of horizon xv in the maxillomandibular mesoderm. These condensations, situated around the outer rim of the optic vesicle (figs. A, B, C), are known as the superior (I. p. 6.), inferior (i. p. an), anterior (a. p. 6.), and posterior (p. p. c.) peripheral condensations, and they anticipate in a sense the insertion points of the four rectus muscles on the eyeball and probably contribute to the formation of the sclera. At this stage, cranial nerves III and VI have grown from the brain into their respective eye-muscle primordia; the lens vesicle has pinched off from the surface ectoderm.
In embryos of horizon xvi (figs. D-G), the superior oblique and lateral rectus condensations have elongated, and, in addition to the superior rectus, two other processes, the primordium of the medial rectus (m. r.) and the common primordium of the inferior rectus and inferior oblique (i. r.+i. 0.), project laterally from the premandibular condensation. In figures F and G, the premandibular condensation and associated eye-muscle primordia, illustrated in figure E, are viewed from lateral and caudal aspects, respectively. A distinct inferior oblique primordium first appears in embryos of horizon xvii (fig. H), and remains intimately attached for some time to the distal end of the inferior rectus.
The superior rectus continues to increase in density, and in embryos of horizon xvii (fig. 16, pl. 4) establishes a firm connection with the superior peripheral condensation. Although the superior rectus is not yet clearly delimited from the premandibular condensation proximally, it is supplied by a branch of the oculomotor nerve. By the time the embryo attains a crown-rump length of 19 to 20 mm (horizon xx), muscle fibers appear in the superior rectus; its insertion on that portion of the sclera formerly occupied by the superior peripheral condensation is clearly defined; and its attachment medially to the developing sphenoid bone immediately above the optic foramen now closely adjoins that of the lateral rectus. It will be recalled that in the adult all four rectus muscles attach to a funnel-shaped tendinous ring, known as the annulus of Zinn, which encloses the optic foramen.
In embryos of horizons xxii and xxiii, a thin muscular sheet, the levator palpebrae superioris, splits off from the dorsomedial surface of the superior rectus and grows forward into the upper eyelid. The levator palpebrae superioris in an embryo of horizon xxiii (figs. 23, 24, pl. 6; fig. 72, pl. 13) is situated adjacent and lateral to the frontal nerve and immediately above and slightly medial to the belly of the superior rectus. As the eyes move forward, the frontal nerve ultimately comes to lie above the levator palpebrae superioris, which in turn comes to occupy a position immediately above the superior rectus.
In older embryos of horizon xv, while the superior rectus is growing forward toward the superior peripheral condensation, a broad bulge develops on the ventrolateral surface of the premandibular condensation and extends laterally as far as the fork in the vena capitis medialis. Presently, in embryos of horizon xvi, two buds appear on the outer surface of the bulge. The more cranial bud, the primordium of the medial rectus, at first consists of a small, diffuse cluster of cells projecting craniolaterally for two or three 10-micron sections. It soon increases in density, and stretches forward beneath the optic stalk toward the anterior peripheral condensation on the outer margin of the optic vesicle. In embryo no. 6258 of horizon xvii, the medial rectus primordium has extended cranially and ventrolaterally until its distal end is continuous with the anterior peripheral condensation (figs. 16, 17, pl. .j). The nasoeiliary branch of the ophthalmic nerve passes immediately above and cranial to the point where the medial rectus joins its peripheral condensation. For some time the proximal end of the medial rectus is indistinguishable from the proximal ends of the other rectus muscles; it is not until horizon xix that a clearly defined origin for this muscle may be detected on the orbital wall. The medial rectus first receives a branch of the oculomotor nerve in embryos of horizon xix; muscle fibers are observable in this muscle soon thereafter (horizon xx).
The second and more caudal bud that develops on the bulging ventrolateral surface of the premandibular condensation in embryos of horizon xvi is destined to form the inferior rectus and inferior oblique muscles. From the beginning, the primordium of the inferior rectus is more dense and better defined than that of the medial rectus. It grows laterally, and in younger embryos of horizon xvii its distal end, which now nearly reaches the inferior peripheral condensation, becomes conspicuously expanded to form the primordium of the inferior oblique. At this stage, a branch of the oculomotor nerve extends along the primordium of the inferior rectus almost up to the inferior oblique condensation, if it does not actually enter it. Medially, ventrally, and caudally, the compact inferior oblique primordium is bounded by branches of the vena orbitalis inferior. Proximally, the inferior oblique condensation is continuous with that of the inferior rectus; in sagittally sectioned embryos, it is impossible to delimit the two primordia. In embryos sectioned frontally, however, the fate of the inferior oblique and inferior rectus condensations may be easily followed.
In older embryos of horizon xvii, the primordium of the inferior oblique has become ovoid, and its long axis is situated at approximately right angles to that of the inferior rectus (figs. I4, 17, pl. 4). The inferior rectus primordium has grown laterally between the inferior oblique and the optic cup to reach the inferior peripheral condensation. In embryos of horizons xviii and xix, the inferior oblique retains intimate contact with the inferior rectus primordium even after the latter has reached its peripheral condensation (fig. 65, pl. 12; figs. 68, 69, pl. 13); it is not until horizon xx, at which time the inferior oblique becomes attached to the eyeball, that all connection with the inferior rectus is lost. At this time a well defined branch of the oculomotor nerve can be traced to the inferior oblique; shortly thereafter (late horizon xx), muscle fibers appear in both the inferior oblique and inferior rectus, and separate attachments (origins) on the orbital wall are distinguishable. The inferior oblique becomes attached to the floor of the orbit near its outer medial rim, whereas the proximal end of the inferior rectus takes origin on the wall of the orbit immediately below the optic foramen, adjacent and caudal to the origin of the medial rectus.
Footnotes
References
Streeter GL. Developmental horizons in human embryos. Description of age group XI, 13 to 20 somites, and age group XII, 21 to 29 somites. (1942) Contrib. Embryol., Carnegie Inst. Wash. Publ. 541, 30: 211-245.
REUTER, K. 1897. Ueber die Entwickelung der Augenmuskulatur beim Schwein. Anat. Hefte, vol. 9, pp. 365-387. Rex, H. 1900. Zur Entwicklung der Augemnuskeln der Ente. Arch. f. mikrosk. Anat., vol. 57, pp. 22 271.
1905. Ueber das Mesoderm des Vorderkopfes der Lach— mowe (Lam: ridibumlm). Morphol. ]ahrb., vol. 33, pp. 1°7'347 ScA.\1.\ro.\', R. E. 1911. Normal plates of the development of Sqrralm acmzt/iiczs. In Keibel, Normentafeln zur Entwicl<lungsgeschichte der \Virheltiere, vol. 12. 140 pp. Iena.
Streeter GL. Developmental horizons in human embryos. Description of age group XI, 13 to 20 somites, and age group XII, 21 to 29 somites. (1942) Contrib. Embryol., Carnegie Inst. Wash. Publ. 541, 30: 211-245.
Streeter GL. Developmental horizons in human embryos. Description of age group XIII, embryos about 4 or 5 millimeters long, and age group XIV, period of indentation of the lens vesicle. (1945) Carnegie Instn. Wash. Publ. 557, Contrib. Embryol., Carnegie Inst. Wash., 31: 27-63.
1948. Developmental horizons in human embryos. Dc scription of age groups xv, xvi, xvii, and xviii, being the third issue of a survey of the Carnegie Collection. Carnegie Inst. Wash. Pub. 575, Contrib. to Embryol., vol. 32, pp. I 33 203.
1951. Developmental horizons in human embryos. Description of age groups xix, xx, xxi, xxii, and xxiii, being the Fifth issue of a survey of the Carnegie Collection. Carnegie Inst. Wash. Pub. 592, Contrib. to Embryol., vol. 34, pp. 165I96.
van Wrjria, I. W. 1883. Ueber die Mesodermsegmente und die Entwickclung der Nerven des Selachierkopfes. 50 pp. Amsterdam. WEm.\', B. 1949. The anterior mesoblast in some lo\ver vertebrates. 225 pp. Lund.
195311. The origin and development of the extrinsic ocular muscles in the alligator. Iour. t\I0rphol., vol. 92, pp. 3°3‘335 1953b. The development of the eye muscles in Ardca cincrca L., Acta Anat., vol. 18, pp. 30-48.
1955. Embryonic segmentations in the head. Malmo. \VoL1=t=, 1940. The anatomy of the eye and orbit including the central connections, development, and comparative anatomy of the visual apparatus. 2d ed. 374 pp. London.
ZIMMERMAN, K. W. 1899. Ueber Kopfliiihlenrudimente beim Menschen. Arch. f. mikrosk. Anat., vol. 53, pp. 481-484.
Abbreviations Used in Figures
| Abbreviations Used in Figures | |
| ao., aorta
ao. :1. I, aortic arch I ao. (I. II, aortic arch II (to. (I. III, aortic arch III a. p. c., anterior peripheral condensation c. g., ciliary ganglion fix, forebrain fg., foregut fr. 9., frontal nerve g. V, Gasserian ganglion hb., hindbrain h. c., head cavity i. c. a., internal carotid artery inn, intermediate mass i. o., inferior oblique i. p. c., inferior peripheral condensation I. r., inferior rectus I. p., lens placode I. p. s., levator palpebrae superioris I. r., lateral rectus I. an, lens vesicle |
m. a., mandibular arch
nmmI. V, mandibular branch of cranial nerve V max. V, maxillary branch of cranial nerve V mb., midbrain m. m. m., maxillomandibular mesoderm m. r., medial rectus N., notochord o. III, oculomotor nerve 11. IV, trochlear nerve 11. VI, abducens nerve m. N., nasociliary nerve op. S., optic stalk op. V., optic vesicle op. V, ophthalmic branch of cranial nerve V p. b., prechorclal bridge p. 6., premandibular condensation p. pl., prechordal plate p. p. c., posterior peripheral condensation s. o., superior oblique s. p. c, superior peripheral condensation s. r., superior rectus t., trochlea v. c. a., vena cerebralis anterior v. c. m., vena capitis medialis v. o. i., vena orhitalis inferior |
ao., aorta
a0. :1. I, aortic arch I
ao. (I. II, aortic arch II
(to. (I. III, aortic arch III
- 1. p. 5., anterior peripheral condensation
c. g., ciliary ganglion
fix, forebrain
/g., foregut
fr. 9., frontal nerve
g. V, Gasserian ganglion
I:b., hindbrain
Ii. 6., head cavity
i. c. a., internal carotid artery
inn, intermediate mass
i. o., inferior oblique
i. (2. c., inferior peripheral condensation
I. r., inferior rectus
I. (1., lens placode
I. p. 5., levator palpebrae superioris
I. r., lateral rectus
I. an, lens vesicle
m. 11., mandibular arch
nmmI. 1-’, mandibular branch of cranial nerve V
max. I", maxillary branch of cranial nerve V
mb., midhrain
m. m. 111., maxillomandibular mesoderm
m. r., medial rectus
13., notochord
- 1. III, oculomotor nerve
11. IV, trochlear nerve
11. VI, abducens nerve
m‘. 11., nasociliary nerve
ofr. 5., optic stalk
op. L'., optic vesicle
op. V, ophthalmic branch of cranial nerve V
(I. 12., prechorclal bridge
p. 6., premandibular condensation
p. pl., prechordal plate
p. p. c., posterior peripheral condensation
5. 0., superior oblique
.r. [L t., superior peripheral condensation
3. r., superior rectus
t., troehlea
v. c. (1., vena cerebralis anterior
z». c. m., vena capitis medialis
1'. 0. i., vena orhitalis inferior
Figures
Plate 1
Plate 2
Plate 3
Plate 4
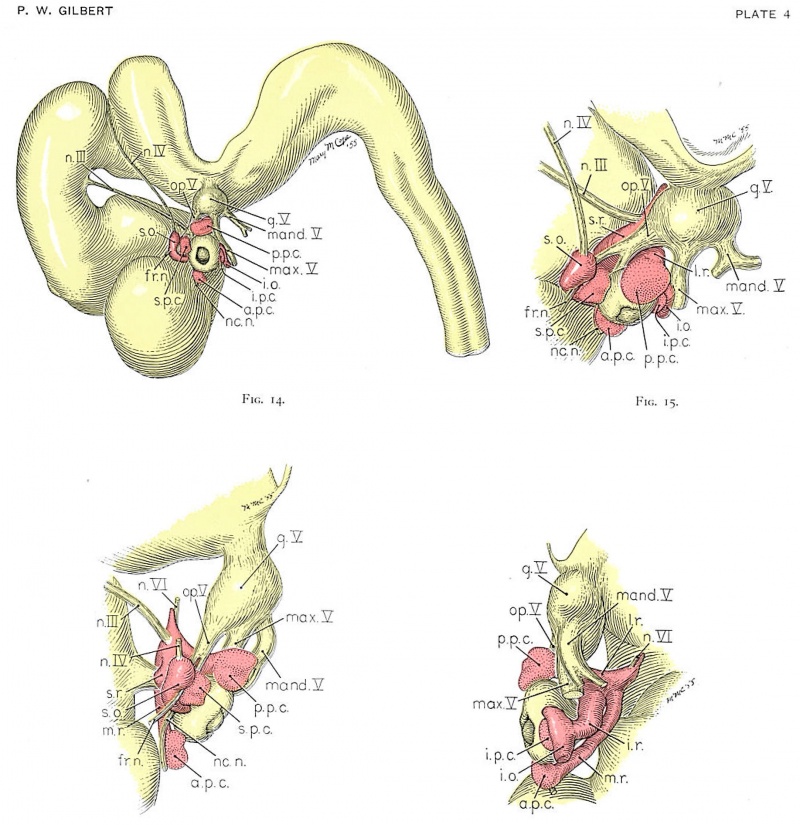
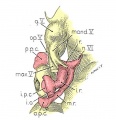
Four drawings of a model of the eye, eye-muscle primordia, and associated nerves. Embryo No. 6258, horizon xvii.
Fig. 14. The eye-muscle primordia of embryo no. 6258 are superimposed on the brain of another embryo, no. 6520, of approximately the same age and size. The relation of the principal branches of cranial nerve V to the four peripheral condensations into which the four rectus muscles grow is illustrated. Lateral aspect. X 15.
Fig. 15. A portion of the brain, the eye, principal nerves, peripheral condensations, and the eye—muscle primordia. Dorso-lateral aspect. No. 6258, X 30.
Fig. 16. Dorsocranial aspect of the same model illustrated in figure 15. X30.
Fig. 17. Ventrocaudal aspect of the same model illustrated in figure 15. X30.
Attention is called to the four peripheral condensations, about the outer margin of the optic vesicle, into which the primordia of the four rectus muscles have grown. Cranial nerves III, IV, and VI have reached their respective eye-muscle primordia: the primordium of the inferior oblique has appeared as a conspicuous condensation at the distal end of the inferior rectus; a prominent bend (at the point where the trochlea will subsequently develop) has appeared near the distal end of the superior oblique primordium, and the proximal end of the superior oblique has begun to shift medially.
Cite this page: Hill, M.A. (2024, April 30) Embryology Paper - The origin and development of the human extrinsic ocular muscles (1957). Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_The_origin_and_development_of_the_human_extrinsic_ocular_muscles_(1957)
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G