Paper - The development of the lungs
| Embryology - 5 Apr 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Flint JM. The development of the lungs. (1906) Amer. J Anat. 6: 1-137.
| Online Editor |
|---|
| This historic 1906 paper describes the development of the human lung and pig from the embryonic to the early fetal period based upon 6 specimens.
See also 1936 papers by Palmer: Palmer DW. Early Developmental Stages of the Human Lung. (1936) Ohio J. Science. 36(2): 69-79. Palmer DW. The lung of a human foetus of 170 mm C.R. length. (1936) Amer. J Anat. : 59-72.
Harvey SC. (1945). Joseph Marshall Flint. Yale J Biol Med , 17, 503.b1-515. PMID: 21434224 Schittny JC. (2018). How high resolution 3-dimensional imaging changes our understanding of postnatal lung development. Histochem. Cell Biol. , 150, 677-691. PMID: 30390117 DOI. Schittny JC. (2017). Development of the lung. Cell Tissue Res. , 367, 427-444. PMID: 28144783 DOI. O'Rahilly R & Tucker JA. (1973). The early development of the larynx in staged human embryos. I. Embryos of the first five weeks (to stage 15). Ann. Otol. Rhinol. Laryngol. , 82, 1-27. PMID: 4746614 DOI.
|
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
The Development of the Lungs
By Joseph Marshall Flint, M. D.,
Professor of Anatomy in the University of California.
(From the Hearst Anatomical Laboratory of the University of California.)
With 4 Plates and 29 Text Figures.
Introduction
It requires only a cursory inspection of the literature on the lungs to show the unsatisfactory state of our knowledge concerning the development of these organs. In the first place, the ontogeny and phylogeny of the mammalian lungs have stood in apparent conflict. There are, moreover, few features of their anatomy upon which there is any agreement among the various investigators who have contributed to this field.
As a reworking of the entire subject has seemed desirable, the author was guided in choosing the pig, first of all, by the practically unlimited supply of the different embryonic stages and, secondly, by the fact that the artiodactyls possess in well developed form, all of the most discussed types of bronchi.
For the study of the early stages of the development of the respiratory system, the Born reconstruction method was employed. Fruitful suggestions for its use have been obtained from the contributions of Bardeen and Huber, Whose applications of the Born method have been followed in this study. Sections of a series of pigs were cut at 20 micra and stained in haematoxylin and congo red. The reconstructions were made at a magnification of 100 diameters. In order to obtain an accurate orientation of the subdivisions of the bronchi, the piling of the plates according to the external form of the lung was controlled by dissections of the lungs of a series of embryos of a corresponding age as those used for reconstruction after the method suggested by Minot.
Liberal use has been made of the various corrosion methods to follow the evolution of the bronchial tree in pigs from 4 cm. to those of adult life. The use of Wood’s metal and of eelloidin corrosions gave fruitful results, although the majority of the stages were obtained by the use of celluloid corrosions. For this purpose celluloid is dissolved in acetone and injected from aspiration bottles into the lungs through the trachea. Like the cellodin corrosions these were digested or macerated in concentrated hydrochloric acid. The advantage of celluloid over celloidin casts lies in the fact that the former, like Wood’s metal, may be left in the air and handled freely without the disadvantages of the glycerine bath, which often makes it either difficult or impossible to study certain parts of the celloidin preparations. For the study of the development of the respiratory lobules a combination of celluloid and Wood’s metal preparations proved most advantageous. Preparations of the entire embryonic lung cleared in oil of cloves were also found serviceable as control preparations for the reconstructions. They are, however, of doubtful value save for this purpose as the young dorsal and ventral buds on the stem bronchus are almost invisible until they have reached a considerable size.
The organogenesis was followed in a series of stained sections from embryos and lungs hardened in Zenker’s fluid and stained by Mallory’s method. At the period of birth the alveoli were distended by injecting them, under low pressure, with Zenker’s fluid, thus obviating the obscure and uncertain ‘pictures which are obtained when the lung is collapsed and contracted. In following the development of the epithelium, the well-known silver nitrate method has been used.
Review of the Literature
To von Baer, 28, we are indebted for the first description of the development of the pulmonary apparatus. In the chick it consists of two small hollow swellings about the middle of the head gut, which appear on the third day. These projections give rise to the lungs, while the hollow cavities represent the rudiments of the bronchi although the trachea up to this time is unformed. On the fourth day the lungs, still in connection with the (esophagus, lie more ventralwards, but the bronchi in growing backwards have dilated into small sacs. Anteriorly, however, the bronchi join each other at an acute angle and terminate in a short canal, the anlage of the trachea which communicates with the oesophagus behind the pharynx. These observations were amplified by the work of Remak, 55, Selenka, 66, Giitte, 67, and especially His, 68, who believes the larynx and trachea arise from a ventral groove in the head gut.
Caudalwards, this structure has two lateral projections representing the rudiments of the bronchi which are bilateral and paired in contradistinction to the unpaired anlage of the larynx and trachea. Less in accordance with our modern ideas on the development of the lungs are the papers of Rathke, 28, and Seessel, 77, while more recent contributions are those of fischelis, 85, and Kastschenko, '87. The work of the latter has been especially emphasized by Weber and Buvignier, 03, who support his views on the serial homology of the lungs with the brancliial pouches. They believe, from their work on the duck, that in birds as well as mammals the anlage of the lungs are paired derivatives of the respiratory tube. The lungs, therefore, while not representing actually existing branchial pouches, indicate the reappearance of endonlermic evaginations of the head gut which has carried gills among the ancestors of vertebrates.
The study of the development of the amphibian and reptilian lung was taken up somewhat later when Rathke, 39, in Coluber natrix described its appearance from paired projections from the head gut. He states that the right lung increases in size until it is larger than the stomach while the left remains, in consequence of regressive changes, as a slight appendix of the trachea. Baumann, 02, in Tropidonotus natrix confirms these observations of Rathke by finding the right lung is threetimes larger than the left in an embryo 3 mm. long, while at 5‘mm. it is some forty times larger. But he is inclined to believe, however, that the discrepancy in size is due to arrested development of the left lung sac rather than a true regressive process. Betrachians were studied by Remak, 55, who found the first rudiments as paired buds from the head gut passing laterally and caudally, while Gotte, 75, describes the origin of the lungs in Anura from endodermal projections immediately behind the last branchial pouch. Gotte, in Anura, suggested the possibility of transformed branchial pouches taking part in the formation of the lungs, before Kastsclienko described the origin of the avian lung from the respiratory tube. Naturally, the observations of Gotte, like those of Kastschenko, are supported by Weber and Buvignier, 03, while Gotte, 04, himself, more recently reaffirms that theory.
Greil, 05, however, who also Worked on Anurans comes to the opposite conclusion from these investigators. Primitively the lungs appear, according to Greil, in the form of two bilaterally symmetrical grooves in the ventral wall of the heat gut about the time the first four gill pouches are formed. The fifth and sixth pouches appear later and are separated from the lung anlage by an appreciable space which is greater than the interval between the individual pouches. He concludes, therefore, that the gill pouches have nothing whatever to do with the formation of the lungs. In subsequent stages the pulmonary grooves deepen and are covered with a thickened splanchnopleure t.o form the primitive lung sac.
Between these structures a transverse gutter appears, while the portion of the head gut anterior to this, produced by the narrowing of its lateral Walls, forms a longitudinally placed laryngo-tracheal groove, which gives rise to the trachea and larynx. The separation from the oesophagus then begins at the caudal extremity and proceeds forwards.
Among the earlier investigators there was an apparent unanimity of opinion that the subsequent differentiation of the amphibian and reptilian lung was due to a centripetal ingrowth of septa from the lung wall dividing and subdividing the primitive lung cavity into a series of smaller peripheral spaces. Furthermore, as early as the middle of the last century Leydig, 57, taught that the complicated lungs of the higher vertebrates represented a complex of a series of simpler lungs, or, in other words, that the infundibulum of the mammalian lung might be compared with an entire frog’s lung with its parietal alveoli. Miller, 93, in a comparative study of the reptilian, avian, and mammalian lung, states that the complexity of the reptilian lung is due to a system of septum formation while the process of budding plays a secondary role. In the avian lung, however, budding becomes more important and septum formation is secondary. Thus Miller looked upon the avian lung as a transition stage between the reptilian lung with its septum formation and the mammalian lung produced by the budding process.
In an extensive study of the dried lungs of adult reptiles Milani, 94, 97, emphasizes the importance of septum formation for the differentiation of the pulmonary apparatus as one ascends the animal scale. The formation and enlargement of primary septa upon the dorsal and ventral walls of the lung cavity which extend horizontally from the median to the lateral wall of the lung as well as the further subdivision of these spaces by secondary septa is responsible for the gradual evolution of the complex from the simple lung.
Ever since the work of Kolliker, 79, the architecture of the mammalian lung has unanimously been conceeded by all who have worked upon the embryonic stages to rest upon a process of centrifugal budding. The centripetal formation of septa, apparently, plays no part in its evolution.
There has been, therefore, a great gap between the developmental processes in the reptilian, amphibian, and avian lung, on the one hand, and the mammalian lung on the other, for, as Gegenbaur has pointed out, ontogeny and phylogeny have apparently stood in conflict, as the pulmonary apparatus in the ancestors of the mammals was produced by a process exactly opposite to that which ontogeny shows is responsible for the growth of the mammalian lung.
The first work which has offered us a suitable explanation of this apparent discrepancy between the ontogeny and phylogeny of the mammalian lung is that of Moser, 00, who, in studying the comparative embryology of the respiratory apparatus in vertebrates, comes to the important conclusion that all Vertebrate lungs are formed by a common growth process. In birds the respiratory apparatus is developed from a projection of the head gut, and its bronchial system results solely from a. process of budding. In reptiles the growth process is exactly like that in birds, namely, by a bronchifugal system of sprouts while the septa are produced by relatively resistant points in the lung wall remaining between two of its outgrowing portions. This same method of growth, furthermore, is again repeated in a less localized and more diffuse form in amphibians where it gives rise, in the first place, to the dilated lung cavity, and, later, to the semispherical projections on the peripheral wall of the lung. In amphibia, as in reptilia, septa are formed by more resistant points in the lung wall remaining between two projecting portions.
Any doubts of Moser’s method or results seem to be effectually silenced by the appearance of Hesser’s, 05, careful and convincing paper on the development of the reptilian lung. Hesser finds the endodermal anlage of the reptilian lung appearing as a fold projecting from the head gut immediately behind the last gill pouch. This separates from the oesophagus in a caudocranial direction. From the cranial portion the trachea is formed, while the caudal part gives rise to the bronchi. The latter grow out as long, narrow tubes, at first in a dorsolateral direction, and, later, parallel to the median plane of the embryo. In the lizards, the bronchi begin to widen at the lateral side, making a sharp distinction between the extra-pulmonary bronchus and the future lungs. In species, however, where there is no extra-pulmonary bronchus, the dilatation affects the whole tube. We have then, at this stage, a respiratory anlage consisting of a long narrow trachea with two narrow bronchi arising from it. These terminate in two enlarged primitive lung sacs. At this point the inner surface of the lung becomes complicated by the more rapid growth of certain portions of the wall of the lungs by a hernia-like produetion of buds. This process begins in a cranial portion of the lung and proceeds gradually to its caudal extremity until finally a large number of buds surround the sac. In Tarentola, the most prominent group appears along the dorsal side of the stem bronchus, while the remaining sprouts occupy transverse rows alternating with the dorsal series.
While, in lizards, the stem bronchus is dilated, in turtles (with the exception of the caudal end which contains a large lumen) it remains a relatively small tube. The bronchi grow to considerable length before branches appear. These are produced by buds or hernial projections from the wall of the bronchus. Upon the stem bronchus are produced, according to Hesser, a lateral and medial row of buds, a result in which he is not in accord with Moser, who believes that there are three series, a lateral, ventral, and dorsal. Especially noteworthy is the fact that in land turtles the lateral bronchi form dilated sacs which later grow into wide ducts, while in the sea turtles the buds grow out as small tubes somewhat dilated at the ends.
The question of the unequal development of the snake’s lung has recently been taken up again by Schmalhausen, 05, who finds in Tropidonotus natrix an unpaired pulmonary anlage. From its caudal end, appears later the two projections for the lungs, which grow unequally but continuously throughout embryonic life. The enormous overgrowth of the right lung leaves the left as a slight appendix upon it. There is, apparently, no regressive change such as Rathke supposes takes place in Coluber natrix. Schmalhausen’s observations support Baumann’s supposition on this point. More important, however, is a still further confirmation of the work of Moser and Hesser as the lung of Tripidonotus natrix grows not through the development of axipetal septa production but from an outward budding of the lung wall.
In view of these researches of Moser, Hesser, and Schmalhausen, then we may look upon respiratory apparatus of vertebrates as the resultant of a common principle of growth, and, in turning to ‘the consideration of the ontogeny of the mammalian lung, there is good ground for believing that its developmental processes no longer conflict with its phylogeny. The evolution of the pulmonary system of mammals was first studied by Kolliker, 79, who traced the development of the organ in rabbits. It appears from an unpaired anlage which arises behind the gill pouches. This is produced by longitudinal furrows which separate the head gut into a dorsal and ventral portion from the latter of which the lungs arise, while the former forms the oesophagus. On the tenth day, the lower part widens so that the lung anlage forms a half canal which ends caudalwarcls in two round depressions. Through a longitudinal fissure, the anlage is still in communication with the oesophagus, while both structures are surrounded by a mass of mesoderm. The projections forming the rudiments of the lungs grow rapidly and bend dorsalwards, and, at the same time, the trachea and oesophagus begin to separate. This process starts at the posterior end of the juncture and progresses towards the head.
A few years later Uskow, 83, confirmed Kolliker’s observations on the rabbit by finding on the tenth day evidences of separation of the head gut into dorsal and ventral portions. From the ventral segment arises the respiratory system, while the dorsal is transformed into the oesophagus. About the level of the sinus venosus, the lungs appear as an unpaired dilatation of the ventral section and, synchronously, the trachea, also unpaired, is developed from the head gut just above it. Although the two structures appear simultaneously, the anlages, according to Uskow, are quite independent. Fol, 84, finds the origin of the lungs in a human embryo 5.6 mm. long as lateral divcrticulae on the head gut just behind the series of gill pouches. He is inclined to believe with Gotte in the transformation of the last pair of gill pouches which have disappeared in the phylogeny of vertebrates into the respiratory apparatus.
His, 87, recognized the anlage of the human lung before the flexion of the embryo, that is to say, about the third week. It appears as a groove in the ventral part of the anterior segment of the intestine which becomes flattened just below the Fundus branehiales into a sagittal fissure and divides into an anterior and posterior half. From the former the trachea is formed, while the latter develops into the oesophagus.
The respiratory portion begins above as a groove and ends below at the level of the auricles in a widened projection. From the latter, the lungs are evolved, while the former yields the trachea. At first, there is no medial division of the unpaired anlage which, save through the thickness of its epithelial lining, it is difficult to differentiate in the early stages.
At the end of the first month the separation of the trachea from the oesophagus, beginning at the caudal extremity and proceeding upwards, is complete. And, as this separation takes place, there is a bilateral division of the anlage, which yields the primitive bronchi. These bend sharply dorsalwards, like a horseshoe, to embrace the oesophagus. The dilated primary lung sacs formed on these divisions are asymmetrical, the cause of which is probably to be sought in the first anlage of the lungs, which, according to His, does not show bilateral symmetry.
Up to this time Kolliker, Uskow, and His have agreed in their observations that the respiratory apparatus of mammals is derived from an unpaired anla.ge, but Willach, 88, in following the pulmonary system of the mole believes the trachea arises from an unpaired anlage, while the lungs originate as paired structures. The asymmetry of the anlage according to Willach is probably responsible for the greater development of the right over the left lung. In rats and mice, the process of development as described by Robinson, 89, agrees, in general, with the results obtained by His, Kolliker, and Uskow. Stoss, 92, and Bonnet, 92, in the study of sheep give results which accord with the findings of Uskow and Kolliker in rabbits, while Minot, 92, in his account of the evolution of the pulmonary system in man, differs from His in looking upon the first anlage as symmetrical. Its subsequent asymmetry Minot believes is due to the unequal development of the heart.
In sheep, Nicholas and Dimitrova, 97, find by the reconstruction method in an embryo of 5 mm. the main bronchi resulting not from a bifurcation of the primitive pulmonary projection, but as asymmetrical buds on its lateral face. Later stages, 7-9 mm., show an exaggeration of the precocious asymmetry as the right side is considerably more developed than the left, and the two primitive bronchi with the trachea form an inverted T.
Narath, 01, followed. the development of the lungs in rabbits and guinea pigs. In the latter, the development begins as a lateral flattening of the head gut just under the Fundus branchiales. This process continues until the lumen of the head gut forms a sagittal fissure just above the lower anlage, which, as it passes upwards, soon resumes its rhomboidal form. The ventral groove deepens and thickens, while, at the same time, the dorsal groove becomes narrower. Lungs and trachea arise from the ventral, while the dorsal part yields the oesophagus.
Somewhat later a longitudinal furrow separates the two and the projection at the most caudal portion of the ventral groove, forming the first unpaired anlage of the lungs, shows a slight asymmetry as the right side is somewhat larger than the left. The lung anlage increases in size, ventrally, but even more markedly to the right and left. These two outgrowths, the anlage of the bronchi, show diiferent relationships, as the right bends dorsally and caudally, while the left remains practically transverse. About this time begins the separation of the trachea from the oesophagus, which proceeds in a caudocephalic direction until the mesoderm surrounding the lung sacs not only projects into the cavity of the coelom, but also passes in and separates the respiratory from the digestive portion of the head gut. The end of the lung sacs dilate, while still maintaining a marked asymmetry and, as this takes place, they extend dorsalward and embrace the oesophagus. In the development of the cat’s and rabbit’s lung, the transformation in general agrees with the conditions in the guinea pig so that Narath finds himself in accord with the earlier researches of Kolliker and Uskow, who also worked on the latter animal. Somewhat later Weber and Buvignier, 03, in a corn- parative study of the origin of the lungs, especially in Minopterus Schreibersii, followed, by the reconstructive method, the lateral flattening of the post branchial region of the head gut. They describe a branchial crest, which descends from the last pair of gill pouches and terminates just before reaching the region in which the pulmonary apparatus appears. The latter is formed from two asymmetrical thickenings of the lateral wall of the head gut, the left of which appears first in an embryo with 18 primitive vertebrae a little below and ventralwards to the last trace of the branchial crest. A constructive process, which these authors hypothecate, isolates the entire ventral segment of the head gut carrying with it the rudimentary lungs and extending as far cephalad as the last gill pouches. Weber and Buvignier obviously abandon the idea of the primitive unpaired anlage described by Kolliker, Uskow, and His, and with it the conception of a pulmonary groove formed syncronously with or before the lungs. Thus, the trachea is post-pulmonary in origin and is formed by this constructive process involving the ventral part of the head gut in the region behind the gill pouches. Like Gotte in Anura, Kastschenko in the chick, and Fol in man, Weber and Buvignier look upon the pulmonary apparatus as diverticulae of the head gut serially homologous with gill puoches.
Very briefly Blisnianskaja, 04, describes the anlage of the human lung as a projection in the ventral portion of the foregut, which, in an embryo of 4.5 mm., shows by two lateral grooves the beginning separation of the respiratory from the digestive system. At this stage, however, the two systems are still in open communication.
lt is apparent that here is a practical unanimity of opinion among those who have contributed to our knowledge of the development of the mammalian lung as to the nature of the anlage and the process by which the primitive lung sacs are produced. Slight differences of opinion may be explained by the nature of the material and the methods by which the different observers have worked. Fol, who believes in a. paired anlage for the human lung, studied an embryo somewhat older than the specimens of His and Blisnianskaja, while Weber and Buvignier and Willaeh, with this single exception, stand alone in regarding mammalian respiratory apparatus as arising from primitively paired structures. In turning, on the other hand, to the consideration of the organogenetic processes by which the bronchial tree is produced not only are few authors in accord, but, also, there is scarcely a chapter in the whole of embryology in which we find so many different opinions based apparently upon objective work. It will be wise, therefore, to consider briefly first the results which have been obtained by the different contributors to this chapter on the development of the lungs, and then attempt to make therefrom a fair statement of our knowledge of the architecture and origin of the bronchial tree at the present time.
Before the appearance of Aeby’s paper we had no general conceptions concerning the architecture of the bronchial tree. According to the current belief, as he himself points out, the division of the bronchi was dichotomous. Little of the origin, the relations, and mode of division of the bronchi ‘was known and even less of the significance of the lobes either to each other or to the species in which they were found. Aeby, 8o, graphically dmeribes the darkness which surrounded our knowledge ofthe lung and blames the widely-accepted dogma of dichotomy for the condition. It is noteworthy, however, how the few objective investigators whose publications immediately preceded Aeby’s also held his conception of the growth process of the tree. Among the first of these was Kiittner, 76, who followed certain stages of the growth of the bronchi in the older stages of cow embryos and described the method of their proliferation as undivided from the end, that is to say, monopodial. From the stems of the bronchi, he says, lateral buds appear having their axes directed at right angles to the mother bronchus. By the subsequent rapid growth of these branches the monopodial character of the division is lost and an apparent dichotomy ensues. A year later Cadiat, 77, in sheep embryos measuring 12-15 mm. and upwards finds the trachea and main bronchi already well formed and describes the growth process as occurring not from the dilated ampullae at the end of the bronchi but rather from lateral outgrowths from their walls. In a slightly different way Stieda, 78, who also used sheep embryos supplemented by rabbits, came to practically the same conclusion.
In the year preceding Aeby’s publication, Kolliker, 79, describes the appearance of secondary branches upon the primitive lung sacs in rabbits on the 12th day, when the stem bronchus of each lung has three projections. From this period the subdivisions become so numerous that it is diflicult to follow them step by step, but, in general, the first branches pass dorsalwards and lateralwards. This branching, according to Kolliker, occurs from hollow buds or projections from the epithelial tube which multiply rapidly until each lung consists of a small tree of hollow canals with swollen terminal buds.
From these citations it is of course obvious that the idea of monopody was not new at the time Aeby wrote, and so the ignorance of the times concerning the architecture of the pulmonary tree was not, as Aeby supposed, so much due to the dogma of dichotomy as to the lack of a thorough piece of objective research such as he himself attempted to supply.
And while many of his conclusions may find no place in our final conceptions concerning the structure of the lung, still they must always receive the credit of having furnished us with a working hypothesis by the aid of which the problem might be attacked by objective methods.
His suggestive appeal to embryologists, of which His, 87, speaks later, indicates his belief in the final solution of the question through embryological investigations. An interesting parallel, in a more limited way, might be drawn between the effects of Aeby’s stimulating paper and the energetic investigations in the field of experimental biology which followed the annunciation of Weismann’s views on heredity.
Aeby abandoned entirely any idea of dichotomy and substituted in its place a strict rnonopodial explanation of the arrangement of the branches of the bronchial tree. Each lung, according to this author, possesses a stem bronchus which forms its axis and leaves the lung at the hilum to fuse with its mate on the opposite side as they join the trachea. Of great importance is the relationship which the pulmonary vessels, especially the arteries, bear to the bronchial tree. The veins run in front of the bronchi, the arteries behind, as the latter are forced in leaving the heart to cross over the large air passages to reach their place. This crossing occurs near the upper end of the stem bronchus and divides the tree into two distinct segments of different importance. These are termed eparterial and hyparterial, according to their position with reference to the point where the pulmonary arteries cross the bronchi.
The arrangement of lateral bronchi is throughout typical and regular.
Few occur in the eparterial while most are in the hyparterial zone. Tl1e former may be absent, but the latter are always present. The hyparterial systems of both lungs are symmetrical, but the eparterial systems, on the contrary, are ordinarily asymmetrical. The hyparterial bronchi always appear in two series, a dorsal and a ventral, which usually alternate and have their origin from the stem bronchus relatively close to each other, leaving the greater portion of the large bronchus free from branches.
This forms then the angle of a three-sided prism from which the two series of lateral bronchi extend into the adjacent space bounded by the chest wall. The dorsal bronchi are shorter. The lateral bronchi give up some of their branches to the stem bronchus, a process which may be followed, according to Aeby, step by step, with the greatest clearness. These wander medialwards and finally cover the previously naked portion of the stem bronchus with dorsal and ventral accessory bronchi. ‘These either remain close to the parent stem or else wander downwards. Their development begins usually quite fa.r down the left lung, while in the right, they appear higher up and often produce a special bronchus supplying the Lobus infracardiacus known as the Bronchus cardiacus.
Eparterial bronchi are always single and never give off accessory branches. They arise from the stem bronchus at a point midway between the sites of origin of the lateral bronchi and divide generally into dorsal and ventral branches. One, especially the left, or both may be absent, thus giving to us three principal forms to the bronchial tree, namely, (1) Lungs with an eparterial system on both sides; (2) Lungs with an eparterial system on the right side; (3) Lungs without an eparterial system.
In some instances the eparterial bronchus is shifted back on to the trachea while in certain lower animals, especially the birds and reptiles, the eparterial system is more highly developed than in mammals. In the phylogeny of the lung, however, it becomes smaller until it may disappear entirely in some of the higher series.
In the further development of the lung sacs in the human embryo as described by His, 87, all secondary bronchi arise from the first five primary divisions. Three of these occur on the right lung sac and two on the left. On the right side they are termed upper, middle, and end buds while those on the left are respectively lateral and end buds. With Aeby, His finds the primitive lungs prismatic in transection with one attached and two free angles between which lies its dorsal or costal surface. The stems give rise to the so-called ventral bronchi, which, His believes, should have been termed lateral bronchi. Owing, -however, to the general acceptance of Aeby’s nomenclature, he has followed it. From the stem bronchus dorsal branches appear which like the ventral group subdivide regularly. These secondary branches are accordingly designated as follows:
- Bronchus dorsalis posterior.
- Bronchus dorsalis lateralis.
- Bronchus ventralis lateralis.
- Bronchus ventralis anterior.
His agrees with Aeby with reference to the interpretation of the eparterial bronchus and looks upon it as an unpaired branch which, if it were in the hyparterial region, would divide into dorsal and ventral elements.
As a matter of fact, after its appearance in the human embryo, it gives off branches which have these two general directions. On the other hand, he looks upon the Bronchus cardiacus as a true side bronchus, which, in opposition to the dorsal series, passes in a ventral direction.
Its independence is shown in its early appearance as well as by the distance which separates it from the first and second ventral bronchi. It is regarded by His as an element which appears out of the schematic order and follows its own development. In the left lung, cardiac and eparterial bronchi are lacking, but the first ventral bronchus sends up a strong dorsal branch, which mounts up into the apical region of the left lung and is designated the Bronchus ascendens. In this way a substitution is made for the eparterial bronchus of the right side which, with the absence of the Bronchus cardiacus, destroys the absolute symmetry of the hyparterial region. His followed the successive appearance of the chief bronchi and their main branches by the reconstructive method as far as embryos of the second month.
The growth of the tree occurs according to His by an extension of the root branches and a division of the end buds. In no place did he find evidences of lateral budding. The end buds during the process of division lose their conical form and flatten to some extent, while an elevation appears on one side which through the formation of a furrow leads to the outgrowth of two separate enlargements from the original bud. By the acquisition of cylindrical status on the part of these secondary buds the process can repeat itself. Below the region of the 3d hyparterial bronchus a point is reached where one cannot hold strictly to the principle of monopodial division, for it is impossible, His believes, to make as Aeby does the principles of monopodial and dichotomous division mutually exclusive. This, His remarks, is a conception of a somewhat transcendental nature, which leads the zealous investigator to personify his own ideas in the organ. The causes which control the form development of a growing tissue need not always remain the same, but may change its character once or several times. Accordingly, His summarizes the growth process from the unpaired anlage of the lung, which extends to either side in paired dilatations. From these primary sacs, lateral sprouts appear by monopodial growth. Further division is by dichotomy and finally a point is reached where the division occurs by more or less abundant lateral budding.
Willach, 88, studied several stages of the development of the lungs in the mole and pig, but his material, however, was not sufficient to give him a very complete picture of the gradual evolution of the pulmonary apparatus so he used the findings of other investigators to fill the gaps.
Although Willach’s own specimens did not include the stages of the first division of the primitive bronchi he believes the growth from first to last is monopodial, the end bud developing a lateral bud before its lumen narrows. These lateral buds become cylindrical as the parent bronchus continues to grow. Willach concludes from a study of the illustrations in His’ paper that the eparterial bronchus is a derivation of the first ventral bronchus and looks upon it as an accessory branch in the sense of Aeby. He likewise believes that the apical branch of the 1st left ventral bronchus is analogous to the cparterial branch because, on its side, it bears the same relationship to the first lateral branch of the pulmonary artery that the eparterial does on the left. Willach follows the ideas of His in believing the Bronchus cardiacus is an independent lateral bronchus and not an accessory bronchus in the sense of Aeby. In the case of the other so-called accessory bronchi, however, this author is in accordance with the views of the latter. Robinson, 89, studied the development of the lungs in rats and mice, and finds about the eighth day the primitive lung sacs growing lateralwards and dorsalwards, forming the bud-like projections into the coelom from which the primitive and stem bronchi arise. The eparterial bronchus, according to Robinson, arises as the first division of the right lung bud. As a distinct branch, it is absent on the left side, although it is compensated for by a branch of the first lateral hyparterial bronchus, which is totally unrepresented on the right side and passes up to the apex of the lung. Robinson, in this view, is in accord with the findings of His. He believes the growth of the tree occurs by a flattening of the terminal bud opposite the axis of the bronchus and a subsequent division into two unequal segments of which the smaller becomes the lateral branch giving rise to what he terms an unequal or sympodial dichotomy. Robinson also describes branches arising as hollow buds from the main bronchus after it has resumed its cylindrical form, allowing the interpolation of secondary bronchi between those already existing, while the dorsal accessory bronchi of Aeby arise, according to Robinson, by a division of the primary dorsal bronchi, not by budding but by having the dorsal stalk split from the point of origin of the first median bud as far back as the stem bronchus, allowing this medial bronchus to obtain a secondary origin from the stem bronchus itself instead of from the primitive dorsal branch. The bronchus infracardiacus is ontogenetically a derivative of the main stem bronchus, but phylogenetically it is, as Aeby suggests, an original branch of the 1st hyparterial bronchus.
With the exception of the Bronchus cardiacus, Robinson has nothing to say concerning the ventro-accessory bronchi of Aeby. He calls them ventral bronchi, but it is not clear whether either ontogenetically or phylogenetically, as in the case of the most prominent one of the group, he considers them accessory branches of his lateral bronchi.
Ewart, 89, published a large monograph containing a criticism of Aeby’s ideas on the architecture of the lungs. Ewart, like Aeby, used material consisting of dissections and corrosions of the adult lung, but only of one species, namely, man. Apparently this author did not perceive as clearly as Aeby that the hyparterial and eparterial theory was in reality a working hypothesis, which could only receive from embryological investigations the evidence necessary for its final substantiation or disproof. From his investigations Ewart believes that dichotomy, more or less equal, is the principle governing the division of the bronchi from beginning to last. He abandons the distinction between the hyparterial and eparterial regions as well as Aeby’s simple nomenclature and substitutes in its place a method of topographical designation which, besides going into endless detail, is constructed entirely independent of embryological considerations and has received, thus far, no support from subsequent investigators.
In a series of papers the first of which appeared the same year, Zunistein, 89, 91, 92, 00, by the study of corrosion specimens of the lungs of a series of mammals and birds in which the pulmonary artery as well as the bronchial tree was injected is unable to support Aeby’s conclusions with reference to the influence of the pulmonary artery on the architecture of the bronchial system. The division of the tree into eparterial and hyparterial bronchi according to Zumstein is not based on sound conclusions as he finds a series of variations in both arteries and bronchi, indicating that a formative influence in the sense of Aeby cannot exist.
At the same time Zumstein studied the development of the lungs in the mole and the duck by the Born reconstruction method. With other investigators, he agrees in the precocious development of the right lung He does not describe in detail, however, the gradual evolution of the mammalian lung but simply states that the dorsal and medial bronchi arise later than the lateral branches but do not attain the extensive development of the latter. Whether or not he considers them accessory bronchi in the sense of Aeby is not clear from his description. The Bronchus infracardiacus may originate, according to Zumstein, either from the stem bronchus beneath the second lateral bronchus or from this bronchus itself. The eparterial branch of Aeby he designates as the first lateral bronchus. In the early stages the Arteriae pulmonales originate far eranialwards and accompany the trachea ventro-lateralwards on both sides. The left is more dorsal even before the trachea is reached while the right artery passes Ventralwards of the first lateral branch of the right bronchus (Aeby’s eparterial). It is scarcely possible, Zumstein concludes, for the arteries to have an influence upon the structure of the tree as the first bronchi have appeared on the stem bronchus before the arteria pulmonalis can be traced into the lung.
In a preliminary note Narath, 92, published a résumé of a large monograph upon the embryology and comparative anatomy of the bronchial tree of the mammalian lung, which appeared some nine years later, 01. Before this work was published, however, Narath, 97, described the development of the lung in Echidna aculeata. In all of the papers, he takes exception to Aeby’s fundamental conception of the architecture of the bronchial tree. From a rich embryological material, echidna. rabbit, and guinea-pig, he describes the growth of the tree after the formation of the primitive lung sacs as taking place by monopodial growth with acropetal development of lateral twigs. In this process the stem bud is the principal structure, which grows on undivided with the ventral bronchi originating as lateral outgrovcons upon it. The primitive lung sacs are to be looked upon, according to Narath, as the first stem buds.
By this process arise from the stem bronchus two series of lateral branches, the ventral and dorsal bronchi. While the former are true derivatives of the stem bronchus, the latter, Narath is inclined to regard, as branches of the ventral bronchi which in course of ontogenetic and phylogenetic development are given up to the stem bronchus. From his embryological investigations, Narath supports Aeby’s conclusions with reference to the dorsal and ventral accessory bronchi. They are formed first on the ventral and dorsal branches and then wander to their positions on the inner and ventral side of the stem bronchus. In this group and in complete accord with Aeby, he would also classify the Bronchus cardiacus except that, unlike Aeby, he believes it can arise in some instances from the second or third ventral bronchus. The pulmonary artery according to Narath’s view has no great influence on the growth of the bronchial tree as he, like Zumstein, has found a whole series of variations in the artery without any important changes in the bronchi.
Furthermore, he reiterates Zumstein’s view that, both at the time the primary bronchi are formed, as well as later, the pulmonary arteries are thin, weak vessels of insuflicient strength to influence these relatively thick and well-developed epithelial structures. Of equal importancein this connection is the observation that the arteries cross over the bronchi to pass down on its lateral, instead of its dorsal, side. ‘Only at the end of the stem bronchus is its position distinctly dorsal. In consequence of this course, it forms a half spiral round the stem bronchus. Of a crossing in the sense of Aeby no true case exists. Narath accordingly proposes to abandon the distinction between the so-called eparterial and hyparterial regions of the bronchial tree.
The eparterial bronchus.of Aeby has, according to Narath, the same area of distribution as a dorsal bronchus. He not only regards it such, but believes it is in reality, the first dorsal bronchus. To emphasize its special meaning for the topography of the lung, he terms it the apical bronchus. It is never suppressed nor does it degenerate in certain animals as Aeby suggests. It is, furthermore, always present normally as a lateral branch of the first ventral bronchus and possesses, moreover, the power of wandering up either onto the stern bronchus or the trachea.
In speaking of his conviction that it is a real dorsal bronchus he continues: “ Mit diescr eiuen '1‘liatsa<-lie fiillt die gauze .-\eby’sel1e Theorie Von den ep- und hvxpartcr-iellen Broncliien ein— fiir allemal.” This view for which Narath has appareritly received the entire credit in the literature was, as we have already seen, first announced by Willaeh. Narath’s single addition to Willaclfs statement is in the designation of the eparterial branch as a dorsal element in conf0rmit_\_' with his idea as to the possible derivation of the whole series of dorsal bronchi. In his belief, that the eparterial bronchus has the area of’ distribution of a. dorsal bronchus, his observations are not in accord with those of Aeby, His, and Robinson.
llinot, 92, thinks the ideas of Acby and His are erroneous with reference to the monopodial growth of the tree. He, on the other hand, looks upon the branching as charaetcristieall_v dichotomous, describing the branches as having rounded ends. After division they develop unequally with the ventral fork, as a rule, serving as the stem. The first branches correspond to the lobes, but he does not agree with the findings of His and Achy with reference to the presence of a bronchus in the right lung which is not represented in the left. With \Villach and Narath he regards the cparterial bronchus of the right side and the apical branch of the first ventral on the left as homologous. The differcnce between the two. .\l’inot holds, is due to the more precocious development of the right side and the secondary modifications in the arteries.
The relationship of the veins confirms this view. 'l‘hc peculiar course of the right pulmonary artery is due to the abortion of the 5th arch on the rigrht side and the subsequent transfer of the origin of the artery to the left.
In a series of papers d’Hardiviller, 96, 1, 97, 1, 2, 3, describes in the rabbit and sheep, the evolution of the tree after the trachea and main bronchi are laid down. There is. according to this author, a stem bronchus which transverses the whole lung and from which all of the primary bronchi are derived by means of collateral ramifications, that is to say, through epithelial hernize from the walls of the stem bronchus, a process in which the terminal bud of the bronchus takes no part. In this way appear, in the rabbit, two buds on the right side and one on the left which, with the stem bronchi, enter into the formation of the five lobes of the lungs and produce all t'urthcr ramifications. In the sheep, on the other hand, there are, including the stem bronchi, four buds on the right and two on the left giving rise to the six lobes of the sheep’s lungs. The primary branches of the stem bronchus occur in four series, external, internal, anterior, and posterior, according to their position on the stem bronchus. Of the four series, the primary, external, and posterior are the most important and are extensively developed, forming the principal bronchi of the adult lung. On the other hand, the anterior and internal proliferate to some extent but do not form extensively developed branches of the adult tree and are, therefore, termed by d’Hardiviller accessory bronchi using a similar nomenclature with a dissimilar meaning from Aeby and Narath. The further growth of the tree after the origin of the principal bronchi by collateral ramification, is by unequal dichotomy at first, and later, equal dichotomy. The processes differ with the different primary bronchi and appear earlier in the sheep than in the rabbit. The cardiac bronchus, according to d’Hardiviller, arises from the stem bronchus and, in this animal, remains independent. In the sheep, it emigrates on to the 1st lateral bronchus.
The bronchus on the left side, he believes, always originates on the stem bronchus and wanders onto the 1st lateral thus forming the Bronchus cardiacus of Hasse. In the rabbit, d’Hardiviller finds the eparterial bronchus originating on the right side by collateral ramification, but unlike other investigators, he believes there is also an eparterial bronchus on the left. It appears on the 13th day and in 24 hours begins to degenerate and remains as a solid epithelial mass in connection with the mother bronchus. In consequence of his belief of the presence of this left eparterial element, d’Hardiviller thinks Aeby’s classification of the lungs of mammals is only of secondary value. It also emphasizes its independent character and forces him to conclude that it is independent of Narath’s apical bronchus as it is not a lateral branch of the first ventral bronchus.
d’Hardiviller’s series of papers was interrupted by. the appearance of a study by Nicholas and Dimatrova, 97, upon the development of the lungs in sheep by the Born reconstruction method in which they supported, in most respects, his observations. In an embryo of 5 mm. they find the main bronchi appearing as asymmetrical buds on the lateral faces of the anlage. In their later growth, this asymmetry is exaggerated.
After the origin of the primitive pulmonary sacs two buds appear on their lateral walls (embryo 9 mm.) representing the first two lateral bronchi while simultaneously the tracheal bronchus is seen as an elongated projection on the right side of the trachea. No trace of a symmetrical bronchus, however, is found on the other side. They regard this element as being entirely independent of the bronchial system which must be regarded as a supernumerary bronchus originating from the future trachea just as the collateral bronchi are formed from the stems.
The collateral bronchi, of which there are three sets, a lateral, a dorsal, and a ventral, originate in the form of buds upon the bronchial stems.
Each is an independent structure and does not show any ontogenetic relationship with the other bronchi, indicating a wandering of the accessory bronchial groups as described by Aeby, Willach, and Narath or d’Hardiviller, in the case of the cardiac bronchus of Hasse. From the division of the first lateral bronchi, a branch passes up towards the head on the left side which is unpaired, for on the opposite side this region is supplied by the tracheal bronchus. The infracardiac bronchus, Nicholas and Dimitrova regard as an unpaired precocious ventral branch for which there is no symmetrical structure in the left lung. The remaining ventral bronchi appear later as in an embryo of 18 min. they find one between the second and third, and another between the third and fourth lateral element. , Huntington, 98, in studying the eparterial system of a series of adult mammals, comes to the conclusion that the right and left lungs agree morphologically in the type of their bronchial distribution and that the asymmetry is apparent and not real. These apparent differences are due to the shifting of a branch of the upper bronchus (cephalic trunk) which wanders up and becomes topographically eparterial. At times, the asymmetry may be more exaggerated by the migration of the entire branch. As the factor involved in this change is the bronchus itself and not the pulmonary artery, Huntington proposes to abandon Aeby’s distinction between the hyparterial and eparterial regions of the bronchial tree except in a topographical sense. In the left lung there is a morphological equivalent for every eparterial element that may occur in the right lung and, accordingly, this author believes in the equivalent morphological value of the upper and middle lobes of the right side with the upper lobe on the left. This, it will be remembered is the conclusion of Willach and Narath except that Huntington, like Willach, does not believe that the eparterial element is primarily a dorsal bronchus.
As the pulmonary artery does not run dorsal to the stem bronchus, but lateral, or dorsolateral, as Narath has shown, Huntington proposes to abandon also the distinction made by Aeby between the dorsal and ventral bronchi. From the study of his corrosions this author believes that the primitive type of division is practically dichotomous and later is changed into the monopodie system. Phylogenetically, the primitive type is the so-called bilateral hyparterial form, while the symmetrical eparterial type represents the end stage in the process of evolution and not the beginning as Aeby and Wiedersheim believe.
An ingenious effort is made by Gruyesse, 98, to support the monopodial theory of growth. This author has studied the transformation of the tracheal musculature into the muscle of Reisseissen in the successive branches of the bronchial tree. He finds the entire stem bronchus until it is past the divisions of the upper and middle lobe and projects Well into the lower lobe has a musculature like the trachea. On the other hand, the bronchus of the upper, middle, and then lower part of the stem has the muscle of Reisseissen. These findings, Guyesse believes, give evidence that the production of the main bronchi is by monopodial growth.
Miller, 00, while working chiefly on the anatomy of the lobule, agrees, apparently, with Aeby’s division of the eparterial and hyparterial region of the human lung, and, furthermore, he also speaks of monopodial division of the tree.
According to Justesen, 00, who studied the branching of the bronchial tree chiefly in cow embryos of well-advanced stages and in post-natal life, the division of the bronchi from first to last takes place by undoubted dichotomy after which the asymmetry is produced by unequal growth of the stem. This author approves of His’ attitude towards Aeby’s theory of monopodial development in general, but criticises his belief in the production of the first branches of the tree by monopody without having the material to follow their successive development. It seems rather strange, therefore, that Justesen, who was himself without these stages, should attempt to prove from His’ illustrations in which these branches were already formed, that they originated by sympodial dichotomy especially after remarking so wisely, “ Es ist kein Versuch, die Frage durch unberichtige Analogie folgerungen zu liisen. Ich will nur behaupten, dass die Frage nicht geliist ist, weitere Untersuchungen dagagen notig sind.” Justesen does not believe in the production of bronchi by lateral outgrowths of the mother stem. He believes, therefore, Stieda’s observation was faulty and states that no other investigator has since repeated this observation. He is ignorant, apparently, of the work of Robinson, d’Hardiviller, and Nicholas, and Dimitrova.
Justesen does not accept Aeby’s distinction between the eparterial and hyparterial regions of the bronchial tree and looks upon the accessory bronchi of Aeby as independent structures. Their irregularity he ascribes to the presence of the heart and vertebral column.
Merkel, 02, agrees with His, that the first divisions of the stem bronchi are produced by monopodial growth and that the later divisions arise by dichotomy. With Narath, he abandons Aeby’s distinction between the eparterial and hyparterial region as resulting from the influence of the pulmonary artery on the architecture of the tree, and looks upon the right apical bronchus, the so-called eparterial, as a derivation of the first ventral and homologous with the apical branch of the 1st ventral or lateral bronchus on the left side. Concerning the so-called accessory bronchi, Merkel seems to be in accord with the older observers in looking upon them as derivations of the dorsal and ventral lateral bronchi, and apparently follows Narath, instead of His, regarding the Bronchus infracardiacus as a possible derivative either of the first, second, or even third ventral bronchus instead of an independent branch of the stem.
The comparative embryology of the lungs in vertebrates has been studied by Moser, oo, whose material consisted chiefly of the lower vertebrates amplified to some extent by sections of rat, mouse, and rabbit embryos. All vertebral lungs, according to Moser, are developed through a common principle consisting in a general increase in size due to an increase of their constituent tissues. The epithelium is the principal factor which originates from the endoderm and passes as a single tube into a solid mass of connective tissue forming the framework of the lung. If this connective tissue is thin, the growth of the epithelium produces a widening of the intrapulmonary bronchus with simple projections on its walls as in amphibia. On the other hand, if the con— nective tissue is dense and resistant, the epithelial increase is localized in certain places, the cells are packed together until they force their way into the connective tissue forming buds such as we find in the lungs of all vertebrates from reptiles up. Certain points on the walls of the lung are more resistant and remain in the lung cavity as septa. At the same time, as we ascend the scale, the number of buds of the second order constantly increase. According to Moser, we may also observe at this time a gradual increase in the mass of connective tissue in passing from lower to higher vertebrates, and we obtain, in consequence, a system of long canals or bronchi passing through a connective tissue sac.
The division of the bronchi is always and exclusively by monopodial growth, and is a main bronchus, the intrapulmonary bronchus, which is a direct continuation of the extrapulmonary bronchus passes through the lung from the root to its distal end.
By means of the reconstruction method, Bremer, 04, studied the lung of the young opossum (Didelphys virginiana) and compared it with older stages. His youngest specimen measured from 10.5 to 12.5 mm. and were taken from the same pouch. Older specimens, 14 cm. long, and three adults were also used for comparison. In five out of six of the new-born animals Bremer found an eparterial bronchus on both sides, except that the one on the left bronchu is always smaller and placed slightly lower than the eparterial branch on the right. The air chambers supplied by it, however, do not form the apex of the lung.
In spite of its small size and low position, it is above the first ventral bronchus and behind the artery and thus, according to Bremer, makes the right and left side of the lung symetrical and reptilian in type as no placentalian lungs are. The complete symmetry of the young lung is marred by the presence of a cardiac lobe on the right side which is unrepresented in the left. Bremer states that the reptilian lung has the double eparterial bronchus and thus the lung of the opossum is reptilian in type. In its later phases, the lung is changed from the reptilian to the mammalian form by the loss of the left eparterial bronchus, the multiplication of its bronchi and the acquisition of a new type of air chamber. In a 14 cm. opossum no trace of the left eparterial bronchus remains but Bremer states he is unable to follow the degeneration of this element from lack of necessary stages. He believes, however, with Selenka, that in the opossum we have an epitome of the evolution of the reptilian lung to the mammalian lung by means of the changes noted above.
The observations of Bremer at once recall the views of d’Hardivi1ler, who believes the left eparterial bronchus is always present in rabbits but subsequently degenerates. If this observation is confirmed it would seem to support d’Hardiviller’s contention, although Narath, it will be remembered, believes that d’Hardiviller was dealing with a variation.
From Bremer’s statement that no other lungs of placentalia have the double eparterial system, it is apparent that he has overlooked Aeby’s description of the lungs of Phoca vitulina, Bradypus tridactylus, Didelphinus delphis, Auchenia lama, Equus caballus, and Elephas Africanus, and some other nine species described by Narath and two species of Cebus by Huntington, making in all seventeen species where the condition described by Bremer as exceptional in mammalia is permanent.
We must also consider the possibility that Bremer is dealing with a dorsal bronchus placed abnormally high on the stem bronchus, especially as he states this bronchus did not supply the apex of the lung. The observations of Narath, 96, on Echidna aculeata are also suggestive in this connection as he states the relationships of the vessels, while young marsupialia are in the pouch suffer no further change either in the case of the arteries or the veins. Furthermore, Narath does not support Selenka, 87, with whom Bremer is, more or less, in accord in his observations on the opossum lung as he finds the lung of Echidna develops like other mammalian lungs and is not differentiated from the developmental processes which are active in the production of the placentalian lung.
He, therefore, does not approve of a comparison of the lung of marsupials with that of reptiles. Moreover, Hesser was unable to find an eparterial bronchus or a bronchus which corresponded to it in his extensive work on the reptilian lung. (Personal communication.) Blisnianskaja, 04, from the study of a series of models of the lungs of human embryos concludes that His’ criticism of Aeby’s nomenclature is correct, and accordingly divides the branches of the main bronchus into two groups, namely, a dorsolateral representing Aeby’s dorsal series, and a ventrolateral including Aeby’s ventral group. She states that this revision is justifiable even from a study of Aeby’s own illustrations. These two series originate so that a line connecting their roots, from two more or less spiral lines on the stem bronchus. The eparterial bronchus, according to Bljsnianskaja, is a dorsal branch of the first ventrolateral bronchus, which emancipates itself and wanders up on the stem bronchus according to the ideas of Willach, Minot, Narath, and Huntington. The entire dorsolateral group are similarly placed originally upon the ventrolateral group, they separate and wander up on the stem bronchus to receive a separate origin. As the eparterial on the right side is the first dorsolateral bronchus, Aeby’s first dorsal bronchus becomes Blisnianskaja’s second dorsal element. The apical bronchus on the left side is homologous then to the eparterial on the right side. The Bronchus cardiacus is also a division of the 1st ventrolateral bronchus on the right side, which separates from the mother branch, passes downwards, and receives a final origin upon the stem bronchus. Since the eparterial bronchus arises from the 1st ventrolateral, Blisnianskaja believes that the upper and middle lobe with the cardiac bronchus on the right side are equivalent to the upper lobe on the left side, and that the lower right lobe is equivalent to the left lower lobe.
The form of the embryonic lung is influenced by the large foetal heart and by the long development through which the human trunk, especially the thorax, passes. Blisnianskaja believes the method of division is sympodial or unequal dichotomy. She has never observed a. bronchus originating from the complete bronchial tube by the monopodial growth.
A glance at this review of the literature shows a unanimous agreement among the various investigators only upon the independence of the lateral group of bronchi (ventral of Aeby, His, and Narath). There is, however, with the exception of Willach and F01 a general recognition of the fact that the mammalian lung arises from an unpaired anlage.
Although supported by objective investigations, the interpretation of the origin of the other groups of bronchi, the method of their growth, and their significance for the architecture of the bronchial tree have varied within Wide latitudes. We may be said at the present time to have no settled views upon the development of the bronchial system. In view of the Work of Moser and Hesser, the student of the mammalian lung, however, may look upon its phylogeny as being no longer in conflict with its ontogeny, and may also state his problem in the following series of questions: 1. Is the anlage of the lung unpaired or paired? 2. Is it symmetrical or asymmetrical? 3. Does the pulmonary artery exert any fundamental influence upon the growth of the bronchial tree, separating it into two regions of unequal significance as expressed in Aeby’s Ep- and Hyparterial theory? 4. Is the “eparterial bronchus” an independent structure or a derivation of the 2d lateral bronchus? Is it an unpaired or paired element? Does an “eparterial bronchus” always form on the left side and then degenerate or undergo atrophic changes? 5. Is the Bronchus ascendens of His, or the left apical bronchus of Narath, the equivalent of the “ eparterial bronchus ”? 6. Are the lateral bronchi independent structures? '7. Are the dorsal bronchi independent structures or derived from the lateral group? 8. Are the ventral bronchi independent structures or derived from the lateral group? 9. Are the medial bronchi independent or derived from the dorsal group ? 10. Is the Bronchus cardiacus an independent or accessory bronchus? 11. In what way do the bronchi grow? Does one system of growth predominate throughout the whole development of the bronchial tree? 12. What is equivalent value of the lobes of one lung in terms of the other
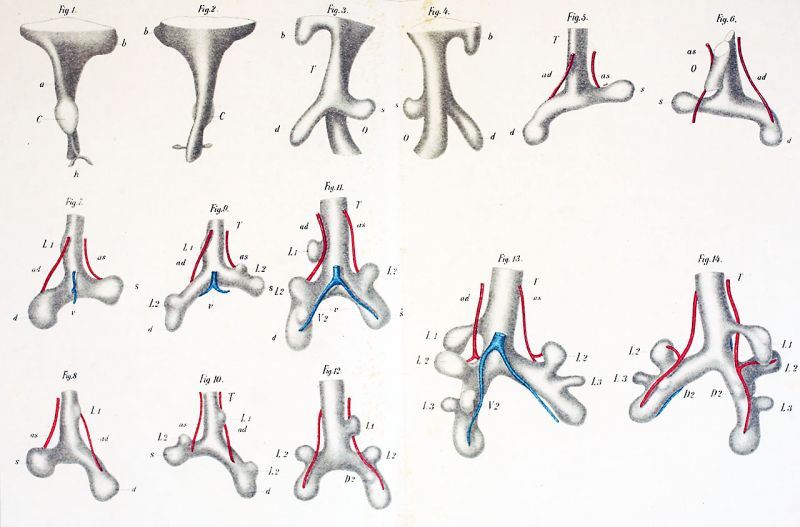
The development of the respiratory apparatus begins in a pig by a lateral flattening of the head gut just below the Fundus branchiales. At the age represented by an embryo, 3.5 mm. nape breech measurement, the last gill pouch has in transsection (fig. 1)* a flattened rhomboidal form with dorsal, ventral, and lateral angles. Below this gill pouch, lying behind the Sinus venosus, which already shows evidences of the increasing asymmetry of the heart, the ventral angle as it deepens to form the pulmonary groove (Pl. I, fig. 1) is pushed somewhat to the right of the median plane (fig. 1). The head gut in passing caudalwards,
- References to Text-figures may read simply fig. 1, or fig. 2, or fig. 3, etc., but every reference to figures on plates is accompanied by the proper plate number.
narrows gradually until its lumen in cross-section forms an asymmetrical sagitally placed fissure. A short distance above the Ductus hepaticus (fig. 3 DH) the pulmonary groove terminates caudalwards in an irregular enlargement (fig. 2P/1), the asymmetrical pulmonary projection forming the first unpaired anlage of the lungs. As yet, there is no trace of the main bronchi nor any evidence of a division. Ventralwards, it projects somewhat from the level of the ventral margin of the intestine below it (Pl. I, fig. 1), while laterally it is more marked on the right than on the left side, an asymmetry more apparent from a. transverse section (fig. 2) or a dorsal view of the reconstructed intestine (Pl. I, fig. 2). Whether the cause of this asymmetry lies primarily in the anlage itself or is due to the influence of the heart as Minot suggests, it is impossible to determine from these specimens. Below the pulmonary projection, the head gut while still asymmetrical lies more in coincidence with the median longitudinal plane.
|
|
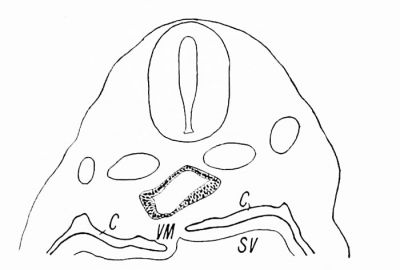
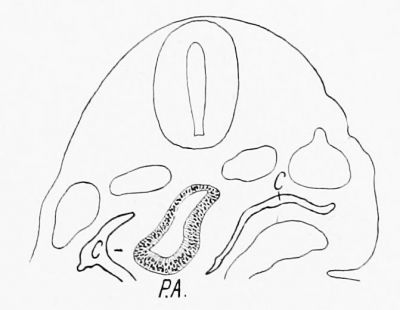
| Text Fig. 1. Section of embryo pig 3.5 mm. long, showing head gut in the region of the upper part of the Mesocardium posterior. C = Coelom. SV = Sinus venosus. VM = Mesocardium posterior. | Text Fig. 2. Section of embryo pig 3.5 mm. long, through the pulmonary anlage. C = Coelom. PA = Pulmonary anlage. |
At this stage, the epithelial lining of the head gut is quite variable in thickness. In the pulmonary enlargement (fig. 2PA) it is clothed by a columnar epithelium of several layers with mitoses taking place chiefly in the innermost row. In the dorsal segment of the head gut at this level, it is considerably lower especially at the dorsal angle where it consists of a single layer. Above the projection it is thinner in the bottom of the groove and thicker at its sides. The Mesocardium posterior (fig. 1 VM) begins just below the last gill pouch and extends down to a short distance below the pulmonary anlage. Between these points, the entire head gut is surrounded by a mesoderm composed of anastomosing cells in which the exoplasmic or fibrillar portion of the mesoderm is not well differentiated (compare Mall, 02, and chapter on organogenesis). In the upper part of the gut just below the gill pouches, the mesoderm, covered by coelomic epithelium forms slight asymmetrical projections into the coelom (fig. 1 0), while at the level of pulmonary swelling, the anlage of the mesodermic portion of the lung wings (fig. 2) takes the form of two irregular lateral projections into the coelornic cavity. The one on the right is much larger than that on the left (fig. 2), so much so that at this stage the latter is only faintly shown. This results in a marked asymmetry of the primitive lung wings themselves. The mesoderm in the two wings is characterized by the richness of its cellular content, as the portion behind the intestine already shows a differentiation preceding the stages of chondrification of the primitive vertebrae. The mesoblastic anlage of the lungs arises from the general mesoderm of the head gut. Just below the pulmonary anlage on the left side are evidences of the Recessus pleuroperitonalis which, as described by Stoss, 92, may at this stage be followed through a few sections.
| File:Flint1906 textfig03.jpg | File:Flint1906 textfig04.jpg |
| Text Fig. 3. Section of embryo pig 3.5 mm. long, through Ductus hepaticus. C = Coelom. DH = Ductus hepaticus. | Text Fig. 4. Section of embryo pig 4 mm. long at the beginning of the Mesocardium posterior. 0 : Coalom. VM = Mesocardium posterior. SV = Sinus venosus. O = OEsophageal portion of head gut. PG = Respiratory portion of the head gut. |
In a slightly later stage, 4 mm., for example, the embryo shows the next step in the development of the respiratory apparatus. The head gut is more symmetrical with reference to the median longitudinal plane (figs. 4, 5, 6). In the upper portion below the gill pouches, a longitudinal fissure appears on either side dividing it new info well-marked dorsal and ventral segments giving the gut in the respiratory level, more or less of an hour—glass appearance in transsections. These fissures mark the line of separation between the respiratory (figs. 4Pg, 5'PA) and digestive systems (figs. 4, 5 0) and extend from the region just below the gill pouches to the pulmonary anlage. In the upper portion, near the gill pouches, the lumen of the (esophageal part is somewhat larger, while, at the level of the pulmonary anlage, the respiratory segment is markedly dilated (fig. 5PA). Between these levels, the relationship between the two is practically equal (fig. 4). Above, the epithelium is lower in the dorsal and ventral angles, slightly so in the lateral fissures but somewhat thickened at the sides of both dorsal and ventral segments.
In these thickened portions there is a double layer, in the angles a single layer of epithelium. In passing caudalwards, the epithelium of the respiratory anlage thickens as its lumen increases in size until a double row of columnar cells line the floor of the pulmonary groove (fig. 5 PA), while at the sides, they are three cells deep. At the level of the pulmonary anlage, the asymmetry is again evident. The projection has now begun to extend lateralwards on each side to produce the main bronchi.
| File:Flint1906 textfig05.jpg | File:Flint1906 textfig06.jpg |
| Text Fig. 5. Section of an embryo pig 4 mm. long, through the upper part of the pulmonary anlage. C= Coe1om. PA = Pu1monary anlage. O: Digestive portion of the head gut. | Text Fig. 6. Section of an embryo pig 4 mm. long through the lower portion of the pulmonary anlage. G = Coelom. O = D1gestive portion of the head gut. BD = Right stem bronchus. |
To the left, the evagination is considerably higher than on the right and also less prominent. At the same time the asymmetry is exaggerated by the anlage of the right bronchus (fig. 6 BD) which points somewhat caudally. The epithelium lining the two primitive bronchi is columnar and consists of several layers. Rapid mitosis is taking place chiefly in the inner row of cells.
With the more marked symmetry of the head gut itself, there is also a greater symmetry of the mesodermal anlage (figs 5, 6) of the lungs.
While the two wings still show the influence of the asymmetry of the bronchial projections, they are somewhat more regular than in the preceding stage. The anlage of the right wing is larger than the left and the Mesocardium posterior is also pushed slightly to the right. The character of the mesoderm remains about the same as in the last stage, that is to say, rich in cells with scarcely any differentiation of the exoplasm into primitive connective-tissue fibrils. Below the lung anlage, the Recessus pleuro-peritonealis is patent on the right side.
| File:Flint1906 textfig07.jpg | File:Flint1906 textfig08.jpg |
| Text Fig. 7. Section of an embryo pig 4.5 mm. long at the beginning of the Mesocardium posterior. C= Coe1om. PG = Respiratory portion of the head gut. O = Digestive portion of the head gut. SV = Sinus venosus. | Text Fig. 8. Section of an embryo 4.5 mm. long, through the anlage of the stem bronchi. C = Cce1om. PA = Pu1monary anlage. BS = Left stem bronchus. O = Gflsophagus. |
In a still later stage, 4.5 mm., the conditions remain practically as in an embryo of 4 mm. The most apparent differences lie in the further development of the two main bronchi. That on the left (fig. 8 BS) grows practically at right angles to the axis of the pulmonary groove, while the right bronchus is directed laterally and caudally (fig. 9 BD) and extends through a number of sections after the other has disappeared. From the anlage at the point of origin of the bronchi, there is a crest-like projection of the epithelial tube in the midline which is exaggerated by the slight dorsal flexure of the two main bronchi. This is scarcely seen in cross—sections, but can be made out easily in embryos cut longitudinally. At this stage, we also note the beginning of the process of separation of the respiratory from the digestive tract in a sulcus (fig. 8) formed below the pulmonary anlage just behind it and in front of the ventral part of the oesophagus which is continuous above with the lateral fissures. In this particular embryo, the process seems a little precocious as I possess later stages where the two systems are in open communication at a lower level than is shown in this specimen. At the level where the Mesocardium posterior begins (fig. 7 VM), the epithelium lining the fore gut is columnar and consists, except in the ventral and dorsal angles, usually of a double layer of cells. In the anlage of the lungs (fig. 8), it is slightly higher and shows a more active karyokinetic process. A similar layer of endoblast extends out into the primitive bronchi. At the tips, cell division is proceeding rapidly. The mesoderm of the lungs remains, so far as its differentiation is concerned, practically unchanged, but the lateral extension of the left bronchus now makes the projection into the coelom at this level more marked than on the right side as the right bronchus, lying in a caudo-lateral direction nearer the median plane, does not carry the mesoderm quite so far into the right coelomic cavity. On both sides, the Recessus pleuroperitonealis may be seen. It is larger and extends higher on the right than on the left (fig. 9 RD, RS’). In fig. 9, the beginning of the formation of the dorsal mesentery at the lower level of the lungs is apparent.
Text Fig. 9. Section of an embryo 4.5 mm. long, through the lower part of pulmonary anlage. O = OEsophagus. C = Coelom. BD = Right stem bronchus. RD = Rlght Recessus pleuroperltonealis. R/S:Left Recessus pleuroperitonealis. VM =Mesocard1um posterior.
By the reconstruction process, the changes which have been occurring in the two preceding stages are demonstrated beautifully in a pig 5 mm. long where they are also considerably accentuated. Above (Pl. I; figs. 3, 4) is seen a segment of the last gill pouch, while below it, the head gut narrows rapidly to a sagittal fissure forming the ventral respiratory and the dorsal digestive portion. The pulmonary groove, still in open communication with the oesophagus, terminates below in the asymmetrical right and left bronchi. Of the two, the left (Pl. I, fig. 33) passes lateralwards almost at right angles to the axis of the groove, while the right (Pl. 1, fig. 3 d) extends caudalwards and lateralwards, giving a sharp asymmetry to the fork which they form with the trachea (Pl. I, fig. 3 T). From the slight crest in the midline which is not seen in the ventral view, both bronchi bend slightly dorsalwards. At the ends, there is a slight increase in the caliber of the bronchi, but end buds are not yet formed upon them. Underneath the point where the two unite, the sulcus from which the separation begins is already present, but it does not extend quite as far cranialwards as in the preceding stage.
Viewed in profile, the whole anlage now extends somewhat ventralwards from the head gut, an extremely important relationship as we shall see in the chapter on the relation of the blood-vessels to the bronchial tree (cf. Schema A). The head gut below the origin of the two bronchi bends slightly ventralwards and to the left. In this region, which may be considered the anlage of the stomach, a noticeable dilatation of the gut is taking place (Pl. I, figs. 3, 4).
In this stage the character of the mesoderm has not changed, the Mesocardium posterior begins at a lower level owing to the descent of the heart, while the dorsal mesentery is now well marked above the level of the lower extremity of the right bronchus. The two lung wings are more symmetrical and project further into the coelom than in the preceding stage. Nevertheless, they are still asymmetrical in so far as the projection forming the left lung is higher than that of the right.
Both on the right and left sides, the Recessus pleuroperitonealis is well marked. In another embryo of the same measurement, but evidently somewhat better developed, the process of separation of the bronchi from the oesophagus is well started. The sulcus between the trachea and the oesophagus extends just above the level of the origin of the two bronchi.
This is filled with mesoderm of a nature similar to-that about the head gut. The mechanical factors involved in the process are difficult to make out, but it begins by an approximation of the epithelium along the line of the two lateral fissures and then proceeds upwards from the sulcus formed behind the primitive bronchi which is filled with mesoderm.
At this stage the following formula of the derivatives of the pulmonary anlage may be made:
TRACHEA.
Right bronchus. Left bronchus.
At 6 mm. (Pl. I, figs. 5, 6) the process of separation is practically complete, the trachea and oesophagus remaining in communication only at the upper end. At the point of origin of the two bronchi, the oesaphagus and trachea are separated by a mass of mesoderm filling the intervening spaces. The simple bronchial system has increased in length and caliber, but the relationships are practically the same, save for the appearance of the rounded terminal buds on the end of the stem bronchi (Pl. I, figs 5, 6 d 3). While, in this embryo, the two bronchi still lie ventralwards to the head gut, they now begin at the ends to bend more dorsalwards than in the preceding stage, the right a trifle more than the left. The Mesocardium posterior is still lower than in the preceding stage, its upper level now beginning only a short distance above the origin of the left bronchus. The mesodermic syncytium is unchanged.
The lung wings are fairly symmetrical as they project on either side into the coelomic cavity. The diflference, however, between the right and left lung bronchi still suffice to give the two lungs a slight asymmetry.
The Recessus pleuroperitonealis is marked on the right side and ex TEXT fiG. 10.
TEXT fiG. 10. Section through the primitive lung sacs of an embryo 7.5 mm. long. C’=Coelom. BD=Right stem bronchus. BS=Left stem bronchus. DM = Dorsa1 mesentery. VM =Mesoca.rdium posterior.
tends some distance above the lower end of the right bronchus, while the left recessus is almost obliterated.
The next stage in the development shown in an embryo 7.5 mm. long consists in the complete production of the primitive lung sacs through the dilatation of the buds on the end of the right and left bronchi (Pl. I, figs. 7, Ssd). The size of the branches of the primitive tree have increased markedly, the two dilated lung sacs while still lying ventralwards of the oesophagus now bend sharply backwards forming a horseshoe-like curve around it (fig. 10). The left still preserves its position at right angles to the trachea with a slight growth caudalwards at the bottom of the sac. On the right side, the general direction of the bronchus is lateralwards, dorsalwards, and caudalwards. The form of the dilated sacs is different on the two sides, that on the right is larger and more nearly triangular in transsection (fig. 10 BD). It projects further dorsalwards than the left (fig. 10 BS). As yet there are no marked evidences of the production of lateral branches except a slightly more prominent angle at the upper lateral wall of the right sac and a similar irregularity of contour on the upper wall of the left. From these points, as we shall see in the next stage, the paired second lateral bronchi arise. Just above the origin of the stem bronchi, however, on the right side of the primitive trachea, one observes a slight bulging or outgrowth of its wall. At this level, the epithelium is a trifle thicker and numerous mitotic figures occur. The projection extends over an area of about 80 mikra and represents the anlage of the first lateral bronchus (Pl. I, figs. '7, 8, L. 1). The process by which this structure is produced is apparently a simple evagination to be compared, perhaps, with the evagination of the pulmonary swelling from the primitive head gut, on the one hand, and the primitive bronchi from the pulmonary anlage on the other. Thus we may consider the same process as repeating itself in the development of the first stages of the pulmonary apparatus. No similar evagination, however, can be observed on the left side.
In the mesoderm of the lungs, the dorsal mesentery (fig. 10 DM) now reaches as high as the forking of the trachea, while the Mesocardium posterior (fig. 10 VM) extends as high as the anlage of the tracheal bronchus. The mesodermic syncytium itself shows some differentiation, particularly under the pleura and in the region of the mesocardium and dorsal mesentery. Here the cells branch and anastomose and the differentiation of the exoplasmic portion into fibrils is in progress. About the oesophagus and pulmonary epithelium, however, there are dense masses of mesodermal cells without much differentiation. This group of cells is engaged in the production of the young basement membranes as the stems continue in their growth. In consequence of the more equal dilatation of the sacs, the simple lung wings are more symmetrical than at any other period of early embryonic life. Differences, however, between the two sides on inspection of the reconstructions are readily made out.
The right Recessus pleuroperitonealis extends slightly above the level of the lower end of the stem bronchus, while the left has disappeared.
At this age we may express the derivations of the pulmonary anlage in the following tabulation:
TBACHEA.
Lateral 1.
Right bronchus. Left bronchus.
Right lung sac. Left lung sac.
At 8.5 mm. the irregular contour of the lung sacs is lost and the two bronchi continue their growth after the production of the first two paired lateral bronchi. These appear as lateral evaginations from the walls of the primitive sacs. On the left side, however, the bud is directed more cranialwards owing to the horizontal position of the left stem. The trachea increases in diameter and length; the bronchi, however, still maintaining the same general relationships, have grown in both caliber and thickness. Now, the very slight evagination of the tracheal bronchus has increased considerably in size and projects from the right wall so as to be noticeable particularly in longitudinal sections from which the model shown in Pl. I, figs. 9 and 10, was reconstructed. It is quite as apparent as the paired Lateral 2 and is separated from the one on the right side by a distance of approximately 380 mikra. These three bronchi may be considered as practically contemporaneous branches of the primitive tree with the tracheal bronchus appearing as a very faint evagination before the lateral bronchi as such can be definitely seen in the primitive lung sacs.
The two stem bronchi now extend more caudalwards than in preceding stages; of the left particularly is this true. They also preserve, although not to such a marked extent, the horseshoe-like dorsal curvature observed in a pig 7.5 mm. long. On their lateral surfaces are two slight evaginations, the anlage of the second lateral bronchi (Pl. I, figs. 9, 10, L. 2). Of these the right project lateralwards, while the left points upwards. These two projections do not appear from a terminal portion of the end bud, but from its lateral surface. They are, therefore, the productions of a monopodial growth The epithelial lining in these primitive buds is a trifle deeper than in the other parts of the tubes and in the inner row karyokinetic figures are more numerous than in the other parts of the respiratory endoderm. The mesoderm about the buds does not appear either thicker or thinner than that on other parts of the respiratory tube. It is impossible, therefore, that this tissue can exert any marked growth influence in the production of these lateral branches.
Much more probable are the space relationships to which the tube adapts itself as, lateralwards in the coelom, we have one point of least resistance, while caudalwards between the thorax wall and the liver, is another.
The bending of the stomach anlage to the left (Pl. I, figs. 9, 10) for a time may have some influence on the growth of the left bronchus holding it in its more horizontal position. From this point the consideration of the development of the mesodermic portion of the lungs will be discussed in a separate chapter.
3 The branches of the primitive bronchial tree in a pig 8.5 mm. long, then, may be tabulated as follows:
TRACHEA.
Lateral 1.
Right stem bronchus. Left stem bronchus.
Lateral 2. Lateral 2.
At 10 mm., the trachea (Pl. I, figs. 11, 12 T) has increased considerably in size and Lateral 1, which appeared as a simple swelling in the earlier stages, has now grown to a button-like enlargement (P1. 1, figs. 11, 12, L. 1) sharply constricted from the wall of the trachea. It points lateralwards and also slightly ventralwards. The division of the trachea into right and left bronchi shows still the asymmetry of the preceding stages as the plane of the left stem is still more transverse than the right.
At the same time the general direction of the right bronchus does not extend so far dorsalwards, as the growth of the right bronchus has apparently been directed more towards the tail end of the embryo. Just at the point of bifurcation, the second lateral bronchi on either side are seen; the one on the right is somewhat larger than the corresponding branch on the left. Both, however, are now fairly symmetrically placed, although right Lateral 2 is slightly more ventral and the left more apical in its direction. Beneath right L. 2, a slight bulging is visible on the axial bronchus directed ventralwards. This is the anlage of Ventral 2, the infracardiac bronchus (Pl. I, fig. 11, V. 2) which arises directly from the stem bronchus and not from L. 2. At the same time, directly opposite the anlage of Ventral 2, there is also a slight dorsal evagination of the stem, indicating the first traces of Dorsal 2 (Pl. I, fig. 12, D. 2) on the right side. The appearance of Ventral 2 (Bronchus infracardiacus) and Dorsal 2 is accompanied by an apparent lateral flattening of the stem bronchus due to the extension of the buds dorsalwards and ventralwards from the axis of the mother branch giving it, in crosssection, a marked oval shape, while above and below, it resumes its cylindrical form. This may be nicely seen in figs. 11, 12, and 13, where 11 shows a transsection of the stem bronchus above, 13 below, and 12 at the level of the primitive dorsal and ventral branches (fig. 12, V. 2, D. 2). In the inner row of epithelium in these projections, karyokinetic figures are much more numerous than in other parts of the stem bronchus save in the neighborhood of the terminal bud. At the same time there is a packing of the nuclei at the base nearer the basement membrane which is now less distinct and gives the epithelium the appearance of having an extra row of cells at this point.
The left bronchus is considerably shorter than the right and projects more lateralwards. Its stem is cylindrical in form and it terminates in a rounded bud-like swelling in which mitoses are numerous. No evidences of Ventral 2 or Dorsal 2 are seen. If we turn for a moment to the consideration of the origin of L. 1, we find the bronchus is a trifle more precocious, but practically simultaneous with the second lateral branch in its origin. It is separated from Lateral 2 by a considerable distance. If the views of Willach and Narath were correct, this branch should not appear until later, and should be traceable, step by step, from the bud forming right Lateral 2 to its final position on the trachea. Its direction is practically lateralwards with a scarcely visible tendency to point ventralwards. It would not then, from the topography of its origin, bear any analogy to a dorsal bronchus. From this distinctly lateral position of its origin, I have classed it among the lateral group of bronchi, although, in its subsequent growth, one of its branches extends down into TEXT fiGS. 11, 12, and 13.
TEXT fiGS. 11, 12, and 13. Sections through the right stem bronchus of an embryo 10 mm. long. fig. 11 above, fig. 12 through, fig. 13 below the origin of Ventral 2 and Dorsal 2. V=Ventra1. D=Dorsa.i. C'=P1eura1 cavity. S’: Stem bronchus. V. 2=Ventral 2, the Bronchus cardiacus. D. 2 =Dorsa1 .2.
the dorsal region giving the bronchus a certain superficial resemblance to that series. On the other hand, the lower lateral elements grow ventralwards in the later embryonic stages and thus also lose their early strictly lateral character. This much is certain; if L. 1 arises phylogenetically from the dorsal group, a view for which there is no convincing proof, absolutely all trace of the migration is lost in the pig. It originates like one of the lateral bronchi and subsequently develops its superficial resemblance to the dorsal elements. Whatever support for the relationship of the bronchus to the dorsal series, can be drawn from this fact, is multiplied by the behavior of a lateral branch of left L. 2, which does exactly the same thing in an adaptative process on the part of the bronchus to a relatively unobstructed environment.
Similarly, Ventral 2 is produced after the formation of Lateral 2 simply as an evagination of the walls of the stem bronchus. It occurs at a level below the point Where the stem bronchus has already regained its cylindrical form after the production of the second lateral bronchus on the right side. Of the possibility of its being a branch of Lateral 2, in these specimens, there is not the slightest evidence. In this particular lung, D. 2 and V. 2 are given off at practically the same level. This is, however, not always the case as one, usually the ventral, may arise higher up.
It is this variability in the origin of these branches which gives rise in the adult tree to the series of stages, which simulate a transplantation of the Ventral 2 from Lateral 2 to the stem bronchus. They represent, however, simply a normal range of variation in the origin of the bronchus.
Narath states the wax-plate method is not adapted to the study of these branches and has, for the most part, used specimens cleared in oil of cloves.
In my experience, the latter method is valuable only for the lateral bronchi where the buds are seen in profile and, therefore, are sharply outlined.
In such specimens, either the dorsal or the ventral series must be studied not only through the mesoderm, but also through the entire thickness of the stem bronchus. In looking upon the surfaces of such buds as D. 2 and V. 2 in an embryo like that represented in P1. I, figs. 11 and 12, the slight projections forming the anlagen of these branches are invisible because they cannot be studied in contour. After they have developed into well-formed buds, they are quite apparent in cleared preparations, particularly when the stereoscopic microscope is used. By that time, however, the important stages of their origin are lost. So far as is known to me, reconstructions, controlled and supplemented by cleared and dissected specimens aiford us the only opportunity to see the first traces of these branches. For such schematic pictures as shown by Narath, 96 (Text figs. 1, 2, 3), which represent schemata of the origin of his apical bronchus and V. 2 from the bud of L. 2, I can find, in the pig, no parallel. Furthermore, the bud of V. 2 is shown in the schemata before the apical bronchus appears, while in the pig the latter is either the independent precursor or the contemporary of Lateral 2, while Ventral 2 is not formed until after the other two branches are well developed.
At this stage the following divisions have appeared in the primitive bronchial tree:
TBACHEA.
Lateral 1.
Right stem bronchus. Left stem bronchus.
L. 2. L. 2.
V. 2.
D. 2.
In a reconstruction of the bronchial tree of a pig 12 mm. (Pl. I, figs. 13, 14) the trachea and stem bronchi have increased considerably in size. At the same time, Lateral 1, the tracheal bronchus (Pl. I, figs. 13, 14, L. 1) has grown further lateralwards. Its terminal bud beyond the constriction near the point of origin bends somewhat ventralwards in conformation to the topography of the environment of the thoracic cavity at this level. Its general course after its origin is dorsalwards causing its lower extremity to overlap the upper part of L. 2 (Pl. I, figs. 13, 14, L. 2). The asymmetrical characteristics of the two-stem bronchi are also maintained, the right extending lower and nearer the midline than the left, which projects more lateralwards. They also bend slightly dorsalwards. It is probable, however, that the asymmetry of this specimen is extreme, as I possess other specimens at this age in which the two sides, while markedly asymmetrical, are more nearly enanteomorphic than this one. In order to control this specimen, it was reconstructed a second time with exactly the same results. Of the two second lateral bronchi (Pl. I, figs. 13, 14, L. 2), the right extends a little farther lateralwards and ventralwards than the left, its growth being influenced at this stage by the presence of L. 1 above and behind it. The left, however, with practically unobstructed environment grows lateralwards and dorsalwards and upwards at this period. Both are terminated by the end buds, which like that on L. 1, are in a stage preparatory to division. On the right side, Ventral 2 (Pl. I, fig. 13, V. 2) the Bronchus infracardiacus has developed to a button-like bud on the ventral portion of the stem bronchus separated from it by a sharp constriction at the base. It is not so well developed as the two second lateral bronchi or L. 1. On the corresponding portion of the left stem bronchus, no analogous branch has appeared. It remains, in fact, naked through the whole future development of the tree. Neither is there in the pig, at this or later stages, a branch which forms at this point and subsequently wanders up on left L. 2, as d’Hardiviller suggests, to form the so-called cardiac bronchus of Hasse. On the lateral sides of both stem bronchi, buds forming Lateral 3 (Pl. I, figs. 13, 14, L. 3) have appeared. These extend directly lateralwards for a short distance to terminate in swollen bud-like extremities, while the portion near the stem bronchus has a definite constriction. Of the two, the right is slightly larger than the left. From this point on, the stem bronchus continues caudalwards to terminate in the enlarged end buds. On the right side, the axial bronchus extends considerably lower than on the left. On the dorsal side of the stem between L. 2 and L. 3, appears on each side, the bud representing Dorsal 2 (Pl. I, fig. 14, D. 2). That on the right side appears before the left and is a trifle more developed. The left, however, is quite apparent. It is also possible that either of these buds may not be formed, in which case this area of the stem remains naked throughout life. This state of things, while occurring seldom, is found oftener on the right than on the left side and the cause may possibly be due, in this particular instance, to the presence of the rapidly growing Ventral 2, together with the presence of L. 1 above, or otherwise simply to the general tendency for the tree to vary within wide limits. As in the case of the ventral and lateral group, the position of these dorsal buds may vary from complete suppression to a position on the stem at the level of L. 2, or to one opposite Lateral 3. The usual situation is about midway between the second and third lateral branches. These buds are the same as Narath’s Dorsal 2 and Aeby’s Dorsal 1. Our results agree with Aeby’s designation as Narath, in considering Lateral 1 and a dorsal branch, was forced accordingly, to change the denomination of his dorsal series. Like Ventral 2, I have designated the first dorsal bronchus as D. 2, simply to keep it in harmony with the lateral series.
At this period the following branches of the bronchial tree have developed: TRACI-IEA.
L. 1.
Right stem bronchus. Left stem bronchus.
L. 2. L. 2.
V. 2.
D. 2. D. 2.
L. 3. L. 3.
13.5 mm. (Pl. II, figs. 15, 16). At this stage the trachea is slightly larger and somewhat longer than in the preceding embryos. On its right side passing dorsolaterally is found L. 1 (Pl. II, figs. 15, 16, L. 1) which has undergone division and yielded two practically equivalent branches, one of which passes downwards and dorsalwards (Pl. II, figs. 15, 16 dvl) and the other lateralwards and slightly upwards (Pl. II, figs. 15, 16 vs). These primary subdivisions, terminating in rounded buds, represent in the adult the dorsoinferior and the ventrosuperior branches of L. 1. At this stage, the two halves of the lung are much more symmetrical than we have seen them in any of the preceding reconstructions. The trachea and two main bronchi denuded of their side branches, now have more or less of a wish-bone shape. The trachea passes ventralwards to the origin of the stems and then, as the two axial bronchi diverge from the point -of union, they also pass somewhat dorsally and reach their maximum point of separation at the level of the third lateral bronchi. From this point, as the end buds are approached, they again converge towards the median line. The right is only slightly larger and more developed than the left. At the same time, there has been a more symmetrical readjustment of the two second lateral bronchi, making them both with reference to their direction and the distance which separates them from the trachea practically mirror images of each other.
L. 2 on the right side passes laterally and somewhat superior, undergoing like the tracheal bronchus a division into two practically dichotomous branches. Of these, one branch, which will continue as the main bronchus (Pl. II, figs. 15, 16 lt) lies ventralwards, while the other is directed dorsally and slightly inferior._ The latter is the dorsal inferior branch (Pl. II, figs. 15, 16 di) of the right L. 2 in the adult, andlits downward course is due, as we shall see later, to the presence of L. 1 above, which prevents its growing upwards to the apex of the lung like the corresponding branch of the left side (Pl. II, figs. 15, 16 up). In comparing the growth of the three first divisions of the bronchial tree until they have reached their present development, it is possible to note in the progress of L. 1 and L. 2 on each side their passage through practically the same stages simultaneously. If the apical branch of L. 2 on the left side is equivalent of L. 1 or the tracheal bronchus as Willach, Narath, and others suggest, it is diificult to explain the tardy appearance of the left element and to give a reason why the right should be so well developed. As a matter of fact, this apical branch of the left Lateral 2 is not the homologue of L. 1, but of the dorsoinferior branch of right Lateral 2, a branch, which, in the adult lung, is practically but not quite as well developed as the apical branch itself. The difference between the two lies in the different nature of the environment in which they grow. Of equivalent age and value in the bronchial tree, the dorsoinferior branch on the right side, influenced by its space relationships and the presence of L. 1 above is forced to grow downwards and backwards, while on the left side, the corresponding branch, unobstructed through the absence of L. 1, mounts upwards to the apex of the lung to supply the territory through which the tracheal bronchus runs on the opposite side. This power of substitution, which the bronchi possess is not confined to this branch alone, but may take place in many other parts of the tree, as we shall see in the later stages. In my corrosions, I have never found an instance of the suppression of L. 1 in the pig. Narath, or (Pl. VII, fig. 5), however, shows a case in the human lung which indicates how, under these circumstances, this dorsoinferior branch of right L. 2 with an unobstructed environment may take a course almost exactly like the corresponding branch on the opposite ide.
Arising as in the preceding stages from the axial bronchus between L. 2 and L. 3, Ventral 2 on the right side has increased considerably in length and passes ventralwards, medianwards, and caudalwards. At its terminus there is a definite bud. The corresponding portion of the stem bronchus on the left side, however. remains nude. In seeking for an explanation of the cause for the extreme development of Ventral 2 on the right side and its usual absence on the left, I have been impressed with the extreme adaptability of the lung to its environment and the way in which the bronchi follow mechanical principles in growing along the lines of least resistance. We realize, of course, the fact that the lungs are relatively late accessions to the animal economy, that they also, excepting possibly in marsupials, are functionless until the period of birth. It is natural, therefore, to find them secondary to and influenced by such organs as the heart and liver, as well as the chest wall by which they are surrounded. These, moreover, have chronologically the developmental precedence and are of definite functional use during the embryonic life of the organism. For the suppression of left V. 2 there is an explanation as we shall see in the chapter on the development of the pulmonary vessels and we may look upon the hyperdevelopment of right V. 2 as an eflort to fill up the space which exists especially in quadrupeds between the heart and diaphragm in the region of the median plane.
Dorsal 2 (Pl. II, fig. 16, D. 2), situated between L. 2 and L. 3, shows a slight growth over the preceding stages, but still persists simply as a.
slight projection from the axial bronchus. The third lateral (Pl. II, figs. 15, 16, L. 3) has increased in size over thecorresponding branch in a younger embryo, and now possesses a more distinct terminal bud.
There is, however, no indication of division as yet. On the ventral side of the axial bronchus, just beneath L. 3, there appears a slight swelling, indicating the origin of the third ventral bronchus (Pl. II, fig. 15, V. 3). Directly behind it, on the dorsal surface of the stem, is a protuberance showing the point of origin of Dorsal 3 (Pl. II, fig. 16, D. 3).
Below these two branches, there is on the lateral side of the axial bronchus a bud indicating the point of origin of Lateral 4 (Pl. II, figs. 15, 16, L. 4), while the axial bronchus continues downwards and ends in a terminal swelling on which some signs of the origin of L. 5 (P1. II, figs. 15, 16, L. 5) are already shown.
At this level, an evagination (Pl. II, figs. 15, 16 MS) appears on the inner side of the end bud pointing medialwards and slightly dorsalwards just opposite the bud of Lateral 5. This is the first one of the medial series to appear on the reconstructions. They are, however, extremely variable both in their constancy and origin. In some trees they are entirely absent, in others they may occur with great regularity, but never in my specimens, which included sections and corrosions of over one hundred lungs, do they occur higher than a short distance above -the level of Lateral 4. They may exist only on one side or else on both. Like the other series, they arise as lateral outgrowths of the bronchial stem, not as secondary derivations of the dorsal series, according to the processes described by either Narath or Robinson. It is interesting, moreover, to note the relation of this group to the oesophagus. In the higher levels where the oesophagus lies between the stem bronchi, no medial bronchi occur, in the lower levels, however, as the oesophagus passes ventralwards to the stems, leaving the medial surfaces of the lung free, these branches are produced. Text fig. 26 shows these conditions well. The edge of the oesophagus is seen in cross-section, while from the median wall of the end bud below it an evagination which will form a Medial 4 or 5 is clearly seen (fig. 26 M). This would seem to indicate another adaptation on the part of the tree, to its space relationships.
On the left side, L. 2 (Pl. II, figs. 15, 16, L. 2) is directed lateralwards and slightly dorsalwards. Like the corresponding bronchus on the right side, there has been a dichotomous division, which has yielded two branches, one directed dorsally and superior (Pl. II, figs. 15, 16 ap) and the other lateral and ventral. The latter is the continuation of the main bronchus, while the former constitutes the apical branch, or Bronchus ascendens of His, of L. 2 on the left side. Owing to the unobstructed possibility of its growth upwards, inasmuch as there is on the left side no L. 1, this branch, as we have seen, grows in a slightly different direction from the corresponding division of the same lateral bronchus on the right side, but, for the reasons given above, it must be viewed as distinctly homologous with the dorsoinferior branch of right L. 2. This branch and its relationships may be seen in many of Narath’s illustrations, from which the nature of its origin is as clearly shown as in the pig’s lung. On the left side, there is no V. 2, but between L. 2 and L. 3, the Dorsal 2 (Pl. II, fig. 16, D. 2), which already appeared in the earlier stages, is now well marked.
Lateral 3 (Pl. II, figs. 15, 16, L. 3) is directed laterally and possesses a distinct bud at the end. It is also directed slightly dorsalwards, occupying a plane almost identical with the second lateral branch above. On the ventral surface of the axial bronchus, just below thepoint of origin of the third lateral, a small projection indicates V. 3 (Pl. II, fig. 15, V. 3), while behind the stem bronchus, but somewhat lower, a similar projection marks the origin of the D. 3 (Pl. II, fig. 16, D. 3). The fourth lateral bronchus (Pl. II, figs. 15, 16, L. 4) «exists at this stage simply as a slight projection from the lateral wall of the axial bronchus as it continues downwards and ends in a terminal bud. The following is a tabulation of the tree in a pig of this age: TBACHEA.
L. 1.
(2) DI.
(2) VS.
Right bronchus. Left bronchus.
L. 2. L. 2.
(2) DI. (2) Ag).
(2) LI. (2) LI.
V. 2.
D. 2. D. 2 L. 3. L. 3 V. 3. V. 3 D. 3. D. 3 L. 4. L. 4 L. 5.
M. 5.
In a pig 15 mm. long (Pl. II, figs. 17 and 18) the trachea has increased in size and passes somewhat ventralwards to the point of bifurcation. On the right side and directed slightly dorsal and inferior, is Lateral 1 (Pl. II, figs. 17 and 18, L. 1). A short distance from its point of origin, the ventral superior (Pl. II, figs. 17, 18, vs) and dorsal inferior (Pl. II, figs. 17, 18, di), branches are seen. These, in turn, now give rise to secondary branches. On the dorsal inferior branch, the first division (Pl. II, fig. 18, d) is directed dorsally and somewhat medially This is the first main dorsal branch of the dorsoinferior in the adult lung, The other division continues on as the stern branch. The ventral superior bronchus passes laterally and superior. It now possesses a branch (Pl. II, fig. 18, d) passing dorsally and slightly upwards. This is the dorsal branch of the ventrosuperior division of L. 1, and is found usually in the adult lung. The more general symmetry of the two main divisions of the trachea noted in the last reconstruction persists, the trachea passing downwards to the point of division and the right and left bronchi, as in the last stage, form with it a structure suggestive of a wish-bone. The axial bronchi bend laterally, dorsally, and medially, their point of widest divergence being now opposite the fourth lateral bronchi, a relation which persists in adult life, and with which the oesophagus, passing ventralwards at this level, probably has something to do. On either side, the second lateral bronchi pass lateralwards, then bend slightly dorsalwards and finally at their tips begin to bend ventralwards again. This indicates the first appearance of the folding of the lung wings around the heart and liver, a process which is naturally directed largely by the form of the chest wall and shows another adaptation of the bronchi to the space in which they have to grow. As yet, however, the remaining lateral bronchi have not developed sufficiently to bend towards the ventral side of the body. On the right side, L. 2 has increased considerably in length, but possesses no more branches than the reconstruction of the preceding stage.
The dorsal inferior branch, however, is considerably longer, and now grows distinctly downwards and lateralwards. Owing to the presence of Lateral 1, with the Lobus superior above and a consequent lack of space, this branch does not grow as rapidly as the relatively unobstructed corresponding branch on the left side, which, at this stage, is somewhat further advanced in its development. V. 2 (Bronchus infracardiacus) passes from its point of origin on the ventral side of the axial bronchus between L. 2 and L. 3, downwards, ventralwards, and medialwards. It is divided into branches of equal size, the first passing somewhat inferior (Pl. I1, fig. 17, 13) and somewhat lateral, forms the inferior branch of the infracardiac bronchus in the adult. The other division passing more medialwards, is the continuation of the main bronchus. From the dorsal side of the stem, D. 2 (Pl. II, fig. 18, D. 2) arises and subdivides into two short branches, the upper and median of which forms the median branch (Pl. II, fig. 18 D. 2, m) of this trunk, while the other continues as the main bronchus. L. 3 (Pl. II, figs. 1'7, 18 L. 3) passes lateralwards and slightly dorsalwards and, while considerably longer than in the preceding stages, it possesses as yet no secondary divisions. V. 3 on the right side is, in this specimen suppressed. It is noteworthy that next to Ventral 2 of the right side, this element of the ventral series is most often missing, a fact which may easily be accounted for by the hyperdevelopment of Ventral 2, which does not, as a rule, leave much territory in this region to be supplied by a ventral bronchus in this segment of the tree.
Dorsal 3 (Pl. II, fig. 18, D. 3) has grown considerably in size and now possesses a terminal bud. The fourth lateral (Pl. II, figs. 17, 18, L. 4) shows a marked growth and is provided with an end bud, while between it and L. 5, on the ventral side of the axial bronchus a small projection indicates the fourth ventral bronchus (Pl. II, figs. 17, V. 4). Immediately opposite it, D. 4 (Pl. II, fig. 18, D. 4), arises as a small bud from the dorsal aspect of the axial bronchus, while Lateral 5 (Pl. II, figs. 17, 18, L. 5) originates from the outer side of the stem as a small bud—like projection. From this point, the axial bronchus passes downwards and terminates in a slight end bud. On the left side, Lateral 2 shows a marked growth of its apical branch (Pl. II, figs. 17, 18, up), which passes upwards and dorsalwards and terminates in two branches, one of which passes dorsally and inferior and indicates its first dorsal branch (Pl. II, fig. 18, L. 2, d), while the other continues upwards as the extension of the stem of this bronchus. Near the extremity of L. 2 another branch is given oif, which extends ventralwards and inferior (Pl. II, fig. 17, L. 2, vi). This corresponds to the ventroinferior division of the bronchus in the adult lung. As there is no ventral bronchus between L. 2 and L. 3 on the left side, the axial bronchus remains at this point perfectly smooth. The second dorsal bronchus (Pl. II, fig. 18, D. 2) of this embryo is placed somewhat lower than the corresponding branch of the opposite series and arises just above the point where Lateral 3 originates. Like its homologue, it shows a subdivi sion into two secondary branches, one of which is the regular medial branch (P1. II, fig. D. 2, m), while the other forms the stem of Dorsal 2.
TEXT fiG. 14. Section through the left lung of a pig 14.5 mm. long, showing the median evagination of the end bud to produce Medial 5. V = Ventra1. D = Dorsal. M = Media1 5. S = 1umen of end bud.
The third lateral bronchus (Pl. II, figs. 17, 18, L. 3) grows lateralwards and dorsalwards, and is not provided with secondary branches at this stage. Appearing as a small bud from the ventral aspect of the axial bronchus, a short distance above L. 4 is Ventral 3 (Pl. II, fig. 17, V. 3), while at a point about opposite this branch and a little above, Dorsal 3 (Pl. II, fig. 18, D. 3) also originates as a small bud from the posterior surface of the stem bronchus, approximately midway between L. 3 and Lateral 4. The latter (Pl. II, figs. 17, 18, L. 4) is somewhat shorter than the third, and has no secondary divisions. Ventral 4 (Pl. II, fig. 17, V. 4) appears as a Very faint swelling of the ventral aspect of the axial bronchus below L. 4. In a corresponding position on the opposite side of the main bronchus Dorsal 4 appears (Pl. II, fig. 18, D. 4) also in the form of a slight evagination from the stem. Lateral 5 (Pl. II, fig. 17, L. 5) is merely indicated by a slight swelling on‘ the side of the terminal bud of the axial bronchus. About opposite it on the inner side of the stem bronchus, is an evagination marking the anlage of a bronchus of the medial series (P1. II, figs. 17, 18, M. 5) like that seen on the right side at a similar point on the tree in the reconstruction of the preceding stage. fig. 14: shows a section through the end bud where this element is in process of formation. The numerous karyokinetic figures and the definite extension of the evagination from the median portion of the lumen of the bud (fig. 14, S’) is clearly shown. This picture, when compared with the reconstruction and Text fig. 26, indicates that there is no essential diiference in the method of formation of these branches of the stem. Like the dorsal, ventral, and lateral elements, they are products of monopodial growth.
Following is a tabulation of the derivatives ofithe bronchial tree at this stage: TBACHEA.
L. 1.
(2) DI.
(3) D.
(2) VS.
(3) D.
Right bronchus. Left bronchus.
L. 2. L. 2.
(2) D1. (2) Ag).
(3) D.
(2) VI.
V. 2.
(2) I.
D. 2. D. 2.
(2) M. (2) M.
L. 3. L. 3.
V. 3. V. 3.
L. 4. L. 4.
D. 4. D. 4.
V. 4. V. 4.
L. 5. L. 5.
M. 5.
In a pig 18.5 mm. long (Pl. II, fig. 19, Pl. III, fig. 20) the trachea is only a little larger than in the preceding embryo. It still passes slightly ventralwards from the upper end to the point of bifurcation.
On the right side passing downwards and slightly dorsalwards, one finds Lateral 1 (Pl. II, fig. 19; P1. III, fig. 20, L. 1), which divides almost at right angles into its main divisions, the dorsoinferior (Pl. II, fig. 19; Pl. III, fig. 20, L. 1, di) and ventrosuperior (Pl. I, figs. 19; Pl. III, fig. 20, L. 1, vs) branches. The dorsoinferior passes downwards and dorsalwards and terminates in the neighborhood of I). 2, a relationship which persists to the adult stage as its further growth downwards is now checked by the series of dorsal bronchi below. This branch shows new divisions over the preceding stage as we find besides the dorsal branch, which passes dorsalwards and medialward, a lateral branch (Pl. III, Fg. 20, l) arising about the same level, which passes laterally and dorsally. Both of these divisions terminate in end buds. The main stem of the bronchus continues downwards to its termination, which is marked by slight end swelling. The ventrosuperior or apical branch (Pl. I], fig. 19, Pl. III, fig. 20, 123) of L. 1, extends further cephalad than in the earlier stages. Besides the dorsal branch indicated in the preceding reconstruction, which shows signs of division, a lateroinferior branch (Pl. II, fig. 19; Pl. III, fig. 20, Zi) is given off somewhat further on, which passes at this time downwards and slightly outwards, and forms the first lateroinferior branch on this bronchus of the adult tree. The main stem continues upwards and ends in a terminal bud. The trachea and the stem bronchi still preserve the characteristic wish—bone appearance noted in the two preceding reconstructions. The two axial bronchi I bending lateralwards, dorsalwards, and medialwards, the point of widest separation being, as in the earlier stages, about the level of the fourth la.teral bronchi. In the preceding reconstruction, the beginning of the ventral growth of the two wings of the lung were apparent on Lateral 2.
This action is now also shown on the third lateral branches. The first pair, however, curve around the heart, while those of the lower series follow the chest wall and the curvature of the diaphragm over the liver. The fourth, fifth, and sixth lateral divisions still pass outwards and slightly backwards without showing this bending at the extremities.
On the right side, the second lateral bronchus arises about the point of bifurcation of the trachea, and passes slightly ventralwards, then runs upwards, slightly dorsalwards, and again ventralwards, preserving its course practically in one horizontal plane. In this specimen the first branch is a ventroinferior (Pl. II, fig. 19, L. 2, vi), which extends downwards and ends in a bud, while the dorsoinferior branch (Pl. II, fig. 19; Pl. III, fig. 20, L. 2, di), which is scarcely larger than the preceding stage, is the second branch of Lateral 2. This condition indicates one of the very important factors in the growth of the bronchi, namely the ability of either branch after a division to continue on as a stem. In nine out of ten cases, the ventral fork, after the first division of Lateral 2, produces the main trunk, leaving the dorsal fork as the large dorsoinferior branch, which is the equivalent of the apical branch on L. 2 of the left side. In this specimen, however, the ventral fork becomes the ventroinferior branch and the dorsal fork continues as the main bronchus, giving rise to the dorsoinferior branch only after undergoing another subdivision. In a much smaller percentage of lungs, the same thing happens on the left side, the ventral fork giving rise to a ventroinferior branch, while the dorsal grows on as the stem, producing the apical or stem only after passing through another division at the end. When this state of affairs occurs, we have the so-called “ cardiac bronchus of Hasse,” which d’I-Iardiviller believes is formed on the stem bronchus in the space for left V. 2, and then wanders up to Lateral 2. Of course in some animals Ventral 2 is formed regularly on the left side, and in others as a variation which establishes the symmetry of this segment of the tree.
In the pig, however, owing to the relations of the pulmonary vein to this part of the stem (see chapter on pulmonary vessels), I have never seen a left Ventral 2. This power of the bronchi gives us a suggestive insight into the adaptations of the growing branches. The selection of the division to continue as the stem is probably governed largely by the physical environment in which the branches find themselves. As the conditions are usually the same, the same branches ordinarily become the stem, but if these are changed, what generally forms the stem is shunted off to become a side branch of relatively small size, while the division which usually constitutes the side branch, grows out as the stem and produces a numerous progeny of lateral divisions. In other words, the extent of the growth of a branch depends to some degree upon the nature of its physical environment. As I have stated above, owing to the generally fixed conditions, the major branches, especially such important ones at Lateral 2, have ordinarily a fixed type of division, but further out on the laterals or in the lower divisions, like Lateral 4 or 5 for example, this interchange of forks frequently takes place, as almost every specimen shows variations in the order of the branching.
The next division of the L. 2 is the ventrosuperior (Pl. II, fig. 19; Pl. III, fig. 20, L. 2, s), projecting from the main bronchus just external to the dorsoinferior branch, while a short distance lateralwards and dorsalwards is given off a dorsosuperior branch (Pl. II, fig. 19; Pl. III, fig. 20, L. 2, ds), which already shows indications of division. These branches represent apparently branches of the second order, but in reality, after a dichotomous division, each segment of the stem between the successive branches is equivalent in its order to that of the last lateral division. In the adult lung these branches are all easily recognizable.
Ventral 2, the infracardiac bronchus, has grown markedly, and presents a long inferior branch (Pl. II, fig. 19, V. 2, i), which passes downwards and ventralwards and is indicated in the architectural history of the younger stages. The next division is a small bud from the upper portion of V. 2. (Pl. II, fig. 19, V. 2, vs) which has a ventrosuperior direction and is found in specimens of the adult tree. This branch is small and at this stage consists simply of a slightly marked bud from the main bronchus. In most of the corrosions I have made of the lungs of older embryos it always shows by its flattened spreading branching that it is more or less influenced by the presence of the heart above it. The ventroinferior branch (Pl. II, fig. 19, V. 2, vi), which is the next in order, is a slight bud, passing downwards and slightly ventralwards, and which, it may be worth while observing, with the inferior branch, sometimes substitutes for Ventral 3, when it is suppressed. After this branch, the main bronchus continues on to terminate in slight end swelling.
Here we are able to observe again the mechanical influence of environment on the growth of a bronchus. The inferior group of branches of Ventral 2 have space in which to grow and are accordingly of exaggerated size in comparison with the superior group, which cannot attain such extensive development, owing to the presence of the heart above them. In this bronchus, as well as in the laterals, we also have the possibility of propagation of the stem through either branch of a dichotomous division, as I have a number of specimens on which the ventrosuperior division arises before the inferior, indicating in these specimens, the use of the latter as the stem with the inferior branch arising from a subsequent forking. Right Dorsal 2 (Pl. III, fig. 20, D. 2) of this specimen has not developed as far as the corresponding bronchus in the preceding stage, the terminal bud merely suggesting an approaching division, which was already well advanced in the bronchial tree from a 15 mm. pig. Such variations, however, are not uncommon. The third lateral bronchus (Pl. II, ~ fig. 19 ; Pl. III, fig. 20, L. 3) passes outwards and slightly ventralwards. From its dorsal aspect, a dorsal branch (Pl. II, fig. 19; Pl. III, fig. 20, L. 3, d) originates, which terminates in the swelling already showing signs of division. The third Ventral bronchus (Pl. II, fig. 19, V. 3) arises from the ventral aspect of the stem, between Lateral 3 and 4 and grows downwards, apparently influenced by the marked development of Ventral 2 above it. Dorsal 3 (Pl. III, fig. 20, D. 3), passes dorsalwards and lateralwards, and has a Well-marked median branch (Pl. III, fig. 20, D. 3, m) which terminates in a large bud, while the main bronchus points somewhat dorsally and laterally. Lateral 4 (Pl. II, fig. 19; Pl. III, fig. 20, L. 4) has a definite ventral bud and at its ends is undergoing division. The fourth ventral bronchus (Pl. II, fig. 19, V. 4) is somewhat smaller than the V. 3, and appears as a constricted button-like bud from the ventral aspect of the axial trunk, while Dorsal 4, arising at a somewhat higher level on the opposite side of the stem ends in a relatively large bud, which is as yet undivided. From the lateral aspect of the axial bronchus Lateral 5 (Pl. II, fig. 19 ; Pl. III, fig. 20, L. 5) takes origin, and ends in a terminal bud without division.
D. 5 (Pl. III, fig. 20, D. 5) is the smallest of the dorsal branches on this side, and appears simply a pedunculated projection from the dorsal aspect of the main stem, while the fifth ventral bronchus is present solely as a slight elevation or projection (Pl. II, fig. 19, V. 5) from the ventral wall of the axial bronchus which, continuing caudalwards, ends in a terminal bud.
On the left side Lateral 2 (Pl. II, fig. 19 ; Pl. III, fig. 20, L. 2), which was practically symmetrical with the corresponding branch on the right side in a pig 13.5 mm. long has now, in the rapid development of its main branch, lost even more than in the preceding stage its symmetrical relationships with right L. 2. The ventrosuperior or apical branch (Pl. II, fig. 19; Pl. III, fig. 20, L. 2, up) is markedly increased in size, and now arises from the more superior aspect of the bronchus and passes superiorly and slightly dorsalwards. Its termination has reached a height equal to the point of origin of the tracheal bronchus on the right side. From its dorsal aspect, the first dorsal branch (Pl. III, fig. 20, L. 2, cl) is derived, which is now subdivided into two regular buds. A little higher, the lateral branch (Pl. III, fig. 20, L. 2, Z‘) is seen, while the apical end of the bronchus is in the stage of division. Further lateralwards, on L. 2 a dorsosuperior branch (Pl. III, fig. 20, L. 2, ds) originates, which has a marked bud and is in process of division, while the next is an inferior or ventroinferior branch (Pl. II, fig. 19; Pl. III, fig. 20, L. 2, ml) existing simply as a small pedunculated projection from the under surface of the bronchus. Lateral 2 terminates in a bud, which has undergone definite division, but the resulting branches are not yet sufficiently characteristic to be placed with reference to the adult tree.
Inasmuch as Ventral 2 on the left sideis always missing on the pigs lung, that aspect of the main bronchus remains perfectly smooth. At this period, however, the Vena pulmonalis already overlies this portion of the axial stem, but, for the sake of clearness in the illustration, it has been placed in approximately the median plane. Dorsal 2 (Pl. III, fig. 20, D. 2) arising just above L. 3 passes dorsalwards, and has two marked bud-like projections, one of which represents the median branch (Pl. III, fig. 20, D. 2, m), usually the first branch of the dorsal series, which is already indicated in the preceding construction. Lateral 3 (Pl. II, fig. 19; Pl. III, fig. 20, L. 3) passes lateralwards and slightly ventralwards.
It has a well-marked dorsal (Pl. III, fig. 20, L. 3, d) and somewhat further out a ventrosuperior branch (Pl. II, fig. 19; Pl. III, fig. 20, L. 3, vs), both of which are represented in the adult lung. The continuation of the bronchus ends in a bud, which is already undergoing further division. At a point just above the fourth lateral, Ventral 3 (Pl. II, fig. 19, V. 3) arises, and ends in a slight terminal swelling. Dorsal 3 (Pl. III, fig. 20, D. 3) is considerably smaller than D. 2, and also smaller than the corresponding branch on the opposite side, but is already divided into two buds, one of which represents the median branch of this bronchus, while the other forms the stem. Such variations in size as are shown in this instance, however, occur very frequently.
Lateral 4 is somewhat shorter than L. 3, and has a well-marked ventral and a less marked dorsosuperior branch. The fourth ventral bronchus (Pl. II, fig. 19, V. 4) is slightly smaller than the third and arises from the corresponding position in this interspace, while D. 4 (Pl. III, fig. 20, D. 4) is considerably longer than the third, and ends in a bud which is not yet divided. Lateral 5 (Pl. II, fig. 19; Pl. III, fig. 20, L. 5) terminates in an undivided bud, and V. 5 (Pl. II, fig. 19, V. 5) consists simply of a slight bulging of the epithelial wall of the axial bronchus. Similarly the fifth dorsal (Pl. III, fig. 20, D. 5) is merely suggested by a faint projection from the epithelial tube.
Lateral 6 (Pl. II, fig. 19; Pl. III, fig. 20, L. 6) is the smallest of the lateral series and ends in a slight swelling, while the axial bronchus continues downwards, terminating in an end bud. At this point the division of the stem is practically dichotomous. This specimen has no medial bronchi and is especially characterized by the lack of variations, for all of the bronchi, excepting the medial group, are present in almost schematic order. The entire absence of the medial group, however, must be regarded as exceptional for most trees, either on one side or both, have medial branches in some of the interspaces below the level of Lateral 4.
While we have seen in the reconstructed series, examples of variations caused by the suppression of either a dorsal or ventral bronchus, another type occurs, not represented «here, of which I have several specimens in my corrosions of the embryonic lung, namely, a reduplication of either the dorsal, ventral, or the medial bronchi in any one interspace. This may or may not be accompanied by a simultaneous suppression of one of the adjacent elements of the same series. Following is a tabulation of the branches of a tree in an embryo 18.5 mm. long.
TRACHEA.
L. 1.
(2) DI.
(3) D-L.
(2) VS.
(3) DS-LI.
Right bronchus. Left bronchus.
L. 2. L. 2.
(2) VI.
(2) DI. (2) Ap.
(3) D-L.
(2) LI. (2) LI.
(3) DI—S-D8. (3) DS-VI.
V. 2.
(2) I.
(2) VS.
(2) VI.
D. 2. D. 2.
— (2) M.
L. 3. L. 3.
(2) D. (2) D.
(2) VS.
(2) I.
V. 3. V. 3.
D. 3. D. 3.
(2) M. (2) M.
L. 4. L. 4.
(2) V. (2) V.
(2) DS.
D. 4 D. 4 V. 4. V. 4 L. 5. L. 5 D, 5 D. 5 V. 5 V. 5 L. 6 Owing to the increasing complexity of the tree, it becomes almost impossible to reconstruct it by Born’s method after this stage. At the same time I have not succeeded in getting good celluloid corrosions younger than 4. cm. pigs. This gap, however, has been partially bridged by drawings of the serial sections of the lung of a 23 mm. pig, aided by specimens cleared in oil of cloves, or injected and subsequently cleared according to the suggestion of Hochstetter, 98. By these methods, it is possible to follow the main divisions of the ramifications considerably beyond that of the last reconstruction. With reference to the smaller buds, however, it is impossible either in sections or in clear specimens to determine definitely their course and final relationships. Nevertheless, as shown in these specimens, the bronchial tree evolves along the same lines. The tendency for the tips of the wings of the lungs to fold ventralwards around the heart and liver also becomes more exaggerated than in the case of the lung of a 18.5 mm. pig.
With the exception of the smaller buds, following is a tabulation of the main branches of the lung at this age.
TRACHEA.
L. 1.
(2) D1.
(3) D-L-M-L.
(2) VS.
(3) DS-LI-LI-D.
Right stem bronchus. Left stem bronchus.
L. 2. L. 2.
(2) DI. (2) Apical.
(3) D-I-D. (3) D-L-M-D.
(2) LI. (2) LI.
(3) VI-DS—I. (3) DS-VI-DI-DS.
V. 2.
(2) I.
(3) D1.
(2) VS.
(2) VI.
D. 2. D. 2.
(2) M. (2) M.
(3) S.
(2) L. (2) SD.
(2) M. (2) L.
L. 3. L. 3.
(2) V. (2) D.
(2) D. (2) V.
(2) SV. (2) I.
(2) D. (2) S.
V. 3. V. 3.
(2) L. (2) S.
(2) M. (2) M.
D. 3. D. 3.
(2) M. (2) M.
(2) L. (2) L.
(2) M.
L. 4. L. 4.
(2) V. (2) D.
(2) V.
(2) D. (2) S.
(2) V. (2) D.
(2) V.
D. 4. D. 4.
(2) M. M.
(2) L. (2) L.
V.-1. V. 4.
(2) M. (2) M.
L. 5. L. 5.
(2) V. (2) D.
(2) D8. (2) V.
(2) I. (2) DS.
D. 5. D. 5.
(2) M. (2) M.
V. 5. V. 5.
M. 5. M. 5.
L. 6. L. 6.
(2) V.
(2) D.
D. 6. D. 6.
V. 6. V. 6.
In a pig 5 cm. long, the bronchial tree can be studied by celluloid corrosions (Pl. IV, fig. 21), but perfect specimens of the air passages in these small embryos are extremely difficult to obtain. The main features of the tree remain practically the same as in the earlier stages, save that it has increased markedly in the complexity of its branching. The trachea with its main bronchi maintains the wish-bone appearance observed in the reconstructions of younger embryos, but a marked difference is noted in the lateral bronchi, which now ‘bend sharply ventralwards as the lung folds around the heart and liver, following the curve of the thoracic wall. The first lateral bronchus, while showing the chief characteristics observed in the younger stages, has a more complicated system of branches. It extends lateralwards and posterior, and divides into its two main branches, the dorsoinferior and ventrosuperior. The former runs dorsalwards, ventralwards, and posterior, while the latter branch passes anterior, ventralwards, and slightly medianwards. The main branches of the dorsoinferior bronchus are, at this stage, seven in number, and extend dorsally, laterally, and medially. Their serial arrangement may be determined from the tabulation at the end of this section. There are five main branches of the ventrosuperior or apical division, which have chiefly a dorsosuperior and a lateroinferior course.
Lateral 2 on the right side shows a marked increase in the complexity of its large dorsoinferior bronchus, which now shows six subdivisions.
The lateroinferior branch which serves as the continuation of the main bronchus, runs lateralwards, ventralwards, and slightly posterior. This has five main divisions, which have, in general, a ventroinferior and dorsosuperior course. V. 2, the Bronchus infracardiacus, passes medianwards, ventralwards, and slightly posterior. The main divisions noted in the earlier stages show an increase in their branching. Dorsal 2 extends in a dorsoposterior direction and its main branches radiate medialwards, lateralwards, and superior. The third lateral bronchus passes lateralwards, ventralwards, and slightly posterior. Its branches run ventrally, dorsally, and in a ventrosuperior direction. V. 3 bronchus in this specimens is not present. Dorsal 3 has four main branches, which have the same general direction as the second dorsal, namely, median, lateral, and superior. The fourth lateral bronchus has, at this stage, six main divisions, extending superiorly, laterally, and medially. D. 4 runs lateralwards, ventralwards, and slightly posterior, and has seven main branches passing in a ventral, dorsosuperior, and dorsoinferior direction. In this tree there is a median branch, M. 5, rising from the main bronchus opposite L. 5, the branches of which run in an ventrosuperior and a dorsoinferior direction. This bronchus is fairly constant, and is met with frequently in corrosions of older lungs. Its origin has been traced in the series of reconstructions of embryonic lungs from a medial evagination of the wall of the stem bronchus. D. 5 passes dorsvalwards and slightly inferior. It has three main divisions extending medially, laterally, and inferior. The Ventral 5 runs ventralwards, medialwards, and slightly posterior, and has a medial and a lateral branch. Lateral 6 passes lateralwards, posteriorly, and to a slight degree ventralwards. It is, as yet, not long enough to show the ventral curvature, which is more marked in the lateral branches of the higher orders. Its branches, at this stage, run chiefly ventralwards and dorsalwards. Dorsal 6 projects dorsally and slightly posterior and has a single median division, while Ventral 6 as yet, has no branches.
Lateral 2 on the left side, owing to the further apical growth of its main division which passes up to the apex of the lung varies even more than in the preceding stage from the corresponding branch on the right side. This bronchus supplies the apical region of the left lung, which, in general, is taken by L. 1 and L. 2 on the opposite side, although the total volume of lung tissue is not nearly as great as that combined in the territory tributary to right L. 1 and L. 2. The apical branch grows almost directly superior, and has six main branches that run chiefly in dorsal, lateral, and medial directions. Its first main dorsal branch extends dorsalwards and slightly posterior, and bears a strong resemblance to the series of dorsal bronchi from the stem bronchus. Its branches run medially, laterally, and dorsoinferiorly. The continuation of the main bronchus, the lateroinferior branch, corresponds in its course practically to the main branch of the opposite side. It possesses seven main divisions, which run dorsosuperiorly, ventroinferiorly, and dorsoinferiorly.
There is, as usual, no Ventral 2 on the left side. Lateral 3 runs laterally, ventrally, and slightly posterior. At this stage it has seven main branches, which pass dorsally, ventrally, superior, and inferior. While the remainder of the branches on the left side below this point show many asymmetrical arrangements from the corresponding divisions on the right, the architectural characters are sufficiently similar to avoid a repetition of the description. The main idea of these tabulations is to show the successive appearance of the chief bronchi of the adult lung and to indicate how the divisions are adapted to the space relationships to which the growing tree must adapt itself. It is not to be supposed that simple mechanical conditions govern entirely the growth of the bronchi, as its chief architectural features are undoubtedly phylogenetic.
This much, however, is certain, that there remains always a considerable adaptability on the part of the growing branches, which is shown in their substitution power when one of‘ the usual elements is suppressed, and apparently by the ability of either fork from a division to serve as the stem.
Following is a tabulation of the branches of the tree at this stage:
TRACHEA.
L. 1.
(2) D1.
(3) D-L-M-L-D-L-M.
(2) VS.
(3) DS-LI-LI-D8-LI.
L. 2. L. 2.
(2) DI. (2) Apical.
(3) D-I—D-V-DI-D. (3) D-L-D-M-V—D.
(2) LI. (2) LI.
( 3) VI-DS-I-DS-VI. ( 3) DS-VI-DI-Ds-VI-D-I-D—S.
V. 2.
(2) I.
(3) D1.
(2) VS.
(3) I.
(2) VI.
(3) LI-VI.
D. 2.
(2) M.
(3) S.
(2) L.
(3) S-D-V.
(2) M.
(2) S.
L. 3.
(2) V.
(2) SV.
(3) DS-V.
(2) V.
(2) SV.
( 3 ) D-V-S.
(2) V.
V. 3 suppressed.
D. 3.
(2) M.
(2) L.
(2) M.
(2) S.
L.4 .(2) V.
(3) S-M.
(2) D.
(3) MS.
(2) V.
(2)178.
(3) D-V-S.
(2) D.
(2) V.
D. 4.
(2) M.
(2) L.
(2)1.
(2) S.
D.2 L.3 (2) M.
(3) SL-IV.
(2) SL.
(3) S-LI—D.
(2) L.
(2) M.
(2) S.
(2) D.
(3) SM-IM-L-M.
(2) V.
(3) S-M.
(2) I.
(3) D.
(2) S.
(3) D.
(2) S.
(3) D-V-S.
(2) DI.
(2) V.
V. 3. suppressed.
D. 3.
(2) M.
(2) L.
(2) M.
L. 4.
(2) V.
(3) S-M.
(2) D.
(3) S-M-L.
(2) LS.
(3 ) D-V-D-V-S-I.
(2) D.
(2) V.
(3) M-L.
(2) S’.
(3) V-D.
(2) V.
(2) D.
D. 4.
(2) M.
(2) L.
(2) I.
(2) S.
(2) M.
(2) L.
V. 4. V. 4.
(2) M.
(2) S. (2) L.
(2) L. (2) M.
(2) M. (2) S.
(2) L.
(2) M.
M.4between L. 4 and L. 5.
L. 5. L. 5.
(2) V. (2) D.
(3) ‘S’-M-L. (3) L-M-L.
(2) D8. (2) V.
(3) S-M. (3) L-M-L.
(2) DI. (2) D.
(2) V. (2) V.
(2) S. (2) S.
(2) DI. (2) V.
(2) V.
M. 5 opposite L. 5.
D. 5. D. 5.
(2) M. (2) M.
(2) L. (2) L.
(2) I. (2) MS.
(2) L.
V. 5. V. 5.
(2) M. (2) M.
(2) L. (2) S.
L. 6. L. 6.
(2) V. (2) D.
(2) D. (2) V.
(2) V. (2) I.
(2) D.
D. 6. D. 6.
(2) M. (2) M.
V. 6. V. 6.
(2) L.
(2) M.
In the study of the further development of the bronchial tree, I have made corrosions of the lung in a series of pig embryos of increasing age increments represented by a centimeter of growth up to and beyond the time of birth. From this series of corrosions it would be possible to tabulate the history of each bronchus until the full growth is attained.
The results would be too detailed, however, to be of any value. Moreover, theiwide range of variation of the branches destroys the absolute sequence of the branches in a successive series giving the formulae only an average relative value. Those which have preceded are, however, suf‘ficiently constant to serve as a general guide to the direction taken by the main branches of the adult tree.
It may be well, however, to show pictorially the subsequent evolution of the tree without taking up the details of the branching, as a good corrosion of the bronchial system holds the general form of the lung quite as well as a hardened specimen of the lung itself. The tree of a pig '7 cm. long is shown in Pl. IV, fig. 22. Besides the increasing complexity of the branching, one notes the ventral curvature of the lateral bronchi parallel with the chest wall. This is most marked in Lateral 2, less so as we proceed to Lateral 6. There are some peculiarities on this tree which are of great interest, for Ventral 3 on the left side is suppressed and in its place a prominent division of the second ventral or infracardiac branch has grown medianwards to take its place. A branch from Lateral 3 also runs to this region, giving an appearance as though it might be a ventral bronchus which had not left the lateral series. It is, however, a simple substitutive process on the part of the lateral branch for an element which has not developed in the earlier stages. This specimen also shows an instance where the dorsal fork of the first division of Lateral 1 continues as the stem, leaving the ventral fork, which usually serves that purpose, as a ventrosuperior branch, while the large dorsoinferior branch which is usually comparable to the apical branch on the opposite side rises from the next division. A median bronchus occurs on the left side opposite Lateral 4. On the right side, median divisions are not present.
In the corrosion of a tree from the lung of a pig 18 cm. long (Pl. IV, figs. 23, 24) a. number of interesting features may be observed, which serve to illustrate some of the developmental characteristics of the growing bronchial tubes. In the first place, we ordinarily have five paired lateral bronchi, while in this specimen there are but four. This indicates the suppression of the last of the lateral elements which is compensated for by an hyperdevelopment of Lateral 5 to supply the region usually tributary to Lateral 6. Accordingly the terminal forking of the stem bronchus, which usually occurs between Lateral 6 and the continuation of the stem, takes place in this instance between it and Lateral 5 (Pl. IV, figs. 23, 24). While this tree shows the suppression of one of the lateral branches, I also have some specimens which present a series of six paired lateral bronchi below L. 1, indicating a possible variation in these elements between these limits with 5 as the average. Ventral 3 is suppressed on both sides, on the right it is substituted for by inferior branches of Ventral 2 and partly by one of the branches of the first ventroinferior division of Lateral 4. On the left side, the ventroinferior divisions of Lateral 3 and Lateral 4 send branches to this region. Median 4 occurs on both sides opposite Lateral 4.
It is particularly interesting to note the efiect of the presence of median branches upon the dorsal series. Where median bronchi are present the median branches of the adjacent dorsal elements are very small and poorly ‘developed, owing to the usurpation of their territory by this series. This naturally gives rise to the pictures which make it appear as though the median series might be transplanted elements from the dorsal bronchi. This relationship, however, is only another indication of the adaptability of the branches of the tree, for in this instance, had the median branches been suppressed, the median branches of the neighboring dorsal series would have grown over to occupy the territory in which the former are found.
In this specimen the ventral curvature of the lateral series is much more marked than in the preceding stage and now aifects, to some extent, the whole lateral series, although Lateral 5 bends slightly, while Lateral 2 (Pl. IV, fig. 23) shows an extreme ventral curvature, a characteristic which is progressively diminished until Lateral 4 is reached. This unequal bending has a marked effect on the stem bronchus and its other branches, and is responsible for the characteristic spiral-like insertion of the lateral and dorsal series upon the stem of adult lungs which has been observed but not explained by most of the investigators since Aeby. As the lateral bronchi turn ventrally more rapidly in the upper than in the lower series, the stem bronchus and its branches twist with them. Thus in the adult lung Lateral 2 appears to rise on the ventrolateral aspect of the stem and each successive element of the lateral series is inserted slightly more lateralwards. Similarly, on the adult tree, Dorsal 2 appears to originate somewhat on the dorsolateral surface of the stem, and the succeeding elements are successively inserted more directly dorsalwards. The spiral line connecting the origins of these two series of bronchi simply represent the degree of torsion of the stem bronchus as the lateral bronchi, in following the curvature of the chest wall, bend around the heart and liver. This is also nicely shown by the course of the pulmonary artery which, naturally, is mechanically influenced by the twisting of the stem bronchus as it is held in the angle formed between the lateral and dorsal series of bronchi. It is, of course, this secondary relationship of the lateral bronchi which led Aeby to term them ventral. In their origin, however, they are, as we have seen, distinctly lateral, and I have applied to them, therefore, the genetic nomenclature.
The condition of the tree a few days after birth is shown in P1. IV, fig. 25. In order to show the three chief series of bronchi in a single illustration, Ventral 2, the Bronchus infracardiacus, has been broken off near the root. The tip of the ventrosuperior branch of the tracheal bronchus, owing to an accident, was also broken and should extend upwards and ventralwards for a considerable distance. Although the general form of the tree has not changed to any marked extent, besides the increase in the branching, the second laterals extend far ventralwards so as to embrace the heart. The effect of the presence of the heart on the tree, as in earlier stages, is shown particularly well by the direction of the branches of the tracheal and second lateral branches. The portions of these bronchi, which come in relation to the heart are nude, their branches extend so as to occupy the remainder of the chest cavity in their neighborhood, a relationship, which may also be seen by an inspection of the tables in the younger stages. Below Lateral 2, however, owing to a freer environment, the bronchi show the power of branching in any direction. In this specimen a few interesting variations are shown, one of which is of particular importance for comparison with the conditions shown in the preceding stage, namely, in the presence of seven lateral bronchi on the right side and five on the left. On the right side the whole ventral series is present, while on the left, two ventral bronchi occur between Lateral 5 and Lateral 6, a fact which would be difficult to explain if we viewed these branches as derivations of the lateral series since the entire group is complete from Ventral 3 down. Dorsal 3 on the right side is hyperdeveloped, while Dorsal 4 is quite small, a not unusual variation. None of my other specimens show such a marked development of the medial bronchi as Medial 4, 5, and 6, present on the right side, as well as an element of this series opposite Lateral 5 on the left side.
Relations on the Blood-Vessels to the Bronchial Tree
In tracing the angiogenesis of the vascular system in the submaxillary gland and the suprarenal body, the author, oo, 02, 03, showed that some of the mechanical principles, which Thoma, 93, in his wel1—known researches found were involved in the development of the blood-vessels in the Area vasculosa of the chick, might be applied to vascular systems developing in three dimensions in the growing organs of mammals.
Thoma found in the chick, that arteries and veins are originally simple capillaries. The subsequent transformation of the latter into arteries on the one hand and veins on the other, is due to their fortuitous location with reference to the primitive aortas and the venous ostia of the heart.
Their growth in size bears a definite relationship to the velocity of the current in them, while their arterial or venous nature is determined by the character of that current, a high pressure pulsating column of blood giving rise to an artery, a low pressure constant current forming a vein.
The nature of the current depends, naturally, mechanically upon its position on the arterial or venous side of the capillary plexus. In considering the problems of angiogenesis in mammals, I called attention to the fact that Thoma’s principles do not explain all the facts of vascular development nor do they entirely accord with them. For example, the statement that a new growth of blood-vessels follows a rise of blood pressure in a capillary area must be considered only an hypothesis and not a demonstrated fact, for this would make the vascular system the stimulus for the new growth of cells, while it is much more probable that cells give the stimulus for the production of new capillaries. It is, of course, obvious that the principal factors that govern organic growth are resident in the cells rather than the blood-vessels as is indicated by their behavior in the embryo before the vascular system is laid down.
In tracing the development of the intrinsic vascular system of the mammalian lung, it is also obvious that the vessels follow the same histomechanical and histogenetic principles which are active in forming the vascular systems of such organs as the G1. submaxillaris and the G1. suprarenalis. Different conditions in the chief cells of the lung, namely, those of the bronchial tree, and different relations of the arterial supply and the venous drainage, give rise to different relationships on the part of the arteries and veins in the pulmonary apparatus. In the suprarenal body, we have the formation of a blood vascular system with a wellmarked capsular plexus from which the blood supply of the organ is derived, and in the submaxillary gland an organ, where the bloodvessels, as in the lungs, accompany the ducts. In the latter instance, however, the conditions are such as to give rise to a venous system where the blood is drained by Venae comites of the main arteries, while in the pulmonary circulation, a relationship exists in which the arteries and veins are separated from each other by means of the bronchial tubes.
According to the studies of Bremer, 02, which have also been confirmed by Sakurai, 04, the pulmonary arteries in the pig appear to originate symmetrically from the pulmonary arches like those of other mammals. At first they remain comparatively parallel and later (7-8 mm.) bend towards each other, sending out at the same time small branches which finally fuse into transverse anastomoses which yield ultimately a common trunk with two origins above and two main pulmonary arteries below.
Bremer suggests that the bending of the arteries towards each other may be caused by the growth of the right and left auricles. This state of affairs occurs in the pig 11 mm. long. Later, the upper part of the right artery degenerates, and, with it, finally the right pulmonary arch.
Thus we have the next stage where both arteries arise as a common trunk from the left pulmonary arch.
In the earlier pig’s embryo (5 mm.), the arteries arising from the pulmonary arches on each side may be followed caudalwards a short distance from their origin on the arches, but only in particularly good specimens, as they are soon lost in the irregular capillary plexus surrounding the head gut to which, in their course, they give off frequent branches. At the same period, it is also possible to note the ingrowth of the pulmonary vein from the yet undivided portion of the auricle. It may be seen in a few sections running dorsalwards in the Mesocardium posterior towards the pulmonary anlage, which is, as yet, only partially separated from the oesophagus. It is asymmetrical as it lies slightly to the left of the medial plane. Its branches connect with the capillary plexus about the head gut and pulmonary anlage, establishing a venous outflow on the ventral side of the respiratory apparatus. Concerning the early appearance of the Vena pulmonalis in the pig, my observations are in accord with those of Narath on the rabbit for in these animals, the Vena pulmonalis is apparently evident at a much earlier stage than His, 87, or Schmidt, 70, were able to observe it in man.
At 6 mm. after the formation of the primitive lung sacs is well under way, the pulmonary arteries may be seen (Pl. 1, figs. 5, 6 ad. as.) running in approximately parallel courses until they diverge and are lost behind the right and left bronchi in the capillary plexus about the primitive lung sacs. Their course, however, on the two sides is different owing to the horizontal position of the left stem bronchus, the artery on that side (Pl. I, fig. 5as) is forced to turn dorsalwards in order to pass behind the left sac sooner than the right pulmonary artery, which maintains its more ventral course and, finally, at a lower level descends behind the right stem bronchus.
The factors which determine the course of the pulmonary artery in passing behind the lung sacs are, first of all, the ventral position of the venous outlet into the Sinus venosus, leaving the arteries to develop from behind. That is to say, with the increasing size of the right and left stem bronchi and the consequent enlargement of the capillary plexus about them, it is natural, with the venous outlet already established on the ventral side of the sacs, that the capillaries on the dorsal side should enlarge into arteries. Furthermore, after its origin and partial separation from the oesophagus, the terminal part of the entire pulmonary apparatus extends somewhat ventralwards from the head gut making it additionally easier for the arteries to form on the dorsal than the ventral surface of the anlage. These factors are responsible for the course, which the arteries and veins take With reference to the bronchial tree, while the asymmetry of the stem bronchi appears to cause the chief difference in the course of the arteries on the two sides. It is, furthermore, possible that some of this irregularity is also due to the medial
SCHEMA A.
Schema to show the origin of the relations of the pulmonary vessels to the lungs.. LA=Lung anlage. AP=Arteria pulmonalis. VP=Vena pulmonalis. L. 1 = Site of origin of_ Lateral 1 the “ eparterial bronchus.” L. 2 = Site of origin of Lateral 2, the first bronchus in the “ hyparterial. region.” L: Liver anlage.
bending of the right artery in preparation for its transfer from the right to the left pulmonary arch according to the suggestion of Bremer, although in Bremer’s descriptions, with which my specimens agree, this actual transfer is made at a much later period, and I am accordingly inclined to minimize the possible influence of this factor. It is also worthy of note that We have no crossing of the bronchi by the arteries in the sense of Aeby. As they run down, they gradually turn dorsalwards to take up a position behind the primitive sacs and are lost in the capillary plexus, which surrounds them. The pulmonary vein, scarcely longer than in the preceding stage, through the further growth of the auricular septum now empties into the left auricle.
In a pig 7.5 mm., the arteries (P1. I, figs. 7, 8 ad. as.) maintain the same relationship as those in the preceding stage, namely, the right lies more ventral than the left and also somewhat nearer the median line. Behind it, however, the evagination for the formation of Lateral 1 has appeared. At this time, the artery consists simply of an endothelial wall supported by the surrounding mesoderm. Situated some distance from the trachea, it is absolutely impossible that such a structure should have a determining influence upon either the production or position of this or other branches of the bronchial tree. Furthermore, it is now well known that such vessels do not influence mechanically the growth of organs which they supply, but follow the developmental processes which are inaugurated in the chief cells of the organ itself acording to definite histodynamic and histomechanical principles.
By a glance at the schema which elucidates this point, we see how the two factors outlined above have worked to bring about the relationship of the artery to the primitive lung sacs. After its origin during the production of the primitive lung sacs, the lung anlage (Schema LA) extends ventralwards. The Vena pulmonalis (Schema VP) in growing in from the auricle has established the venous outflow ventral to the anlage, leaving the pulmonary arteries (Schema AP) to form on the dorsal side of the primitive stems. This relationship occurs, however, before there is the slightest indication of the presence of any of the main bronchi. Later as they appear, Lateral 1, the so-called “ eparterial bronchus” (Schema L. 1) develops behind the artery and Lateral 2 (Schema L. 2) in front of it. Sometimes Lateral 1 is higher up, Where it appears on the trachea, sometimes lower down where it forms on the stern, often where it forms on both sides, the left is lower than the right. The most important element in determining the position of Lateral 1 is the point at which the trachea separates into the two stems.
As we have seen, when this is high, taking Lateral 2 on each side as the fixed topographical point, Lateral 1 is on the stern; when it is low, as in the pig, Lateral 1 forms on the trachea.
It is also important to observe that the relationship between the Arteria pulmonalis and Lateral 2 is not “eparterial” as Aeby suggests; the artery in the embryo simply runs ventralwards to Lateral 1 and then passes gradually behind the stem. The “eparterial and hyparterial” topography of the bronchi is due to the descent of the heart in the later stages of embryonic life and to the degeneration of the Ductus arteriosus after birth when the entire circulation from the right ventricle, consequently, is transferred from the systemic into the pulmonary system.
Until this occurs, the pulmonary arteries do not even approximately cross the stem bronchi as Aeby suggests. Apparently, as we shall see later, he recognized this fact. Furthermore, my observations in older stages are in accord with the findings of Zumstein and Narath, who hold that, in the sense of Aeby, a true crossing on the part of the artery never exists.
It seems to me important, therefore, for a logical conception of the architecture of the bronchial tree, that the terms “ eparterial and hyparterial ” or, at least, all that they imply should be abandoned.
The pulmonary vein (Pl. I, fig. 7 12) is seen at this stage with two small tributaries, one from the head and another from the caudal region running in the Mesocardium posterior. They are in connection with other dilated capillaries which may be seen in the neighborhood of the lung sacs, but the latter have not become large enough as yet to form definite veins. The vascular apparatus of the lungs, then, at the period of the formation of the two lung sacs, consists in two small asymmetrical arteries passing down behind the primitive stem bronchi ending in an irregular capillary plexus about the dilated epithelial tubes from the ventral side of which run enlarged capillaries emptying into the pulmonary vein in the Mesocardium posterior.
No particular change is observed in the next older embryo 8.5 mm. in the relationships of the arteries (Pl. I, figs. 9, 10 ad. as.). With the lengthening of the stem bronchi, however, owing to the increased capillary field about the bronchi, the right and left pulmonary veins (Pl. I, fig. 9 12) may be seen emptying into the common trunk which, in turn, now opens into the left auricle. In a pig 10 In. long, the pulmonary arteries maintain their general relationship to the trachea, the right passing ventral to Lateral 1 (Pl. I, figs. 11, 12 ad). Continuing downwards, they gradually extend behind the stem bronchi giving off branches to the irregular capillary plexus which surrounds the primitive tree, elements of which may be seen, here and there, in Well-prepared crosssections of the lung. As a rule, the arteries lie on the dorsolateral aspect of the stem. At this stage, it is quite evident that the three first branches of the tree, practically in the same period of development, are growing without reference to the arteries as they are surrounded only by a capillary plexus derived from branches of the arteries and from which dilated capillaries empty into the Veins. As they increase in size, the arteries and veins, which follow the various ramifications of the tree are formed from the capillary plexus according to the regular histomechanical laws. The two main tributaries of the vein (Pl. I, fig. 11 11) 5 forming the right and left stem veins, run on the ventromedial aspect of the stem originating from the plexus about the main bronchi. In this way, we have established the regular alternation of artery, bronchus, and vein which persists throughout the life of the tree, although it will be remembered that this relationship is due primarily to the position of the vein with reference to the anlage.
At 12 mm. (Pl. I, figs. 13, 14) the vessels have followed the natural growth of the bronchi. From the capillary plexus on the dorsal surface of Lateral 2 on each side, the artery to that branch is formed. The vein (Pl. I, fig. 13) by the rapid development of Ventral 2 is pushed somewhat medialwards at this point. With the marked development of Lateral 1, the tracheal bronchus, in a pig 13.5 mm. long, a branch (Pl.
II, fig. 15) is given off from the right pulmonary artery, which runs up along the ventral surface of the bronchus to end in the plexus about that branch. Continuing downwards, the arteries (Pl. II, fig. 16) on both sides run on the dorsolateral aspect of the stem. The branches to Lateral 2 have increased somewhat in length, and from the right pulmonary artery a new branch is formed, which, passing under the root of right Lateral 2, ends on the lateral and under aspect of Ventral 2, the Bronchus infracardiacus. The artery still maintains its position with reference to the stem, which causes it to lie in the angle between the lateral and dorsal bronchi. Thus, the artery itself, however, is not responsible for the division of these two groups from the stem as Aeby implies when he says in speaking of Lateral 1, “In ihm hat offenbar die Scheidung des hyparteriellen Gebietes in zwei streng geschiedene Bezirke noch nicht stattgefunden, ein Thatbestand, der wohl damit in Verbindung gebracht werden darf, dass die Lungenarterie nicht sondernd einzugreifen vermocht hat.” Should we still suspect a causal relationship here, it is only necessary to glance at the ventral bronchi, particularly Ventral 2, to see an element not only originating from the stem away from the influence of the artery but also with its growth, developing from its capillary plexus an artery which passes around the stem and rests on its lateral side. Interesting changes, at the same time, are occurring in the veins (Pl. II, fig. 15).
From the tracheal bronchus, a branch may be observed passing down to the common pulmonary vein running still more ventral than the artery to Lateral 1, another one of the final adult relationships in the pig’s lung.
Here, however, We have an exception to the general relationships of the vessels to the bronchi due to the more ventral position of the veins and the failure of right pulmonary artery to form behind Lateral 1, which, in this particular instance, gives us a Vena comes to the artery to the tracheal bronchus instead of the usual alternation found in other portions of the tree. On the ventral surface of Lateral 2, veins originate, which empty into the right and left pulmonary veins, while medialward and above Ventral 2 lie the vein of that bronchus which joins the right pulmonary just below the tributary from Lateral 2. In this stage, either owing to the hyperdevelopinent of Ventral 2, or the increasing asymmetry of the heart, or both, the pulmonary veins are shifted somewhat to the left, causing them to lie somewhat beyond the median line. At the same time, the veins in these young stages are frequently reduplicated as the final channels are not always definitely selected. In order to show the different branches of the tree without extra illustrations, in this and the succeeding reconstructions, the pulmonary vein has been kept in the median line, and only the chief channels are shown in the case of reduplication, which is a frequent occurrence.
In a pig 15 mm. long, the pulmonary artery (Pl. II, fig. 17) on the right side still has a more ventral and medial position than that on the left, a fixed relationship from embryos 12 mm. in length as the arteries both rise from a common trunk originating from the left pulmonary arch.
Just below the point of origin of Lateral 1, the artery to that trunk is observed (Pl. II, fig. 17), which passes up and divides with it into its ventrosuperior and dorsoinferior branches. The two pulmonary arteries bending dorsalwards pass back of the right and left bronchi, giving off the branches to the second lateral bronchi, which lie on their dorsal and superior surfaces. On the right side, the artery to the second ventral bronchus (Pl. II, fig. 17) has increased in length with the growth of that branch, while arteries to the second dorsal bronchi (Pl. II, fig. 18) are observed passing along their lateral walls. From this point, the pulmonary arteries continue on in the angle between the dorsal and lateral bronchi, giving ofl branches to the third and fourth lateral elements (Pl. II, fig. 18) on each side which lie above and behind them.
From the capillary plexus around the termination of the right and left stem bronchi, the beginnings of the pulmonary veins (Pl. II, fig. 17) are seen as in the preceding stage. From the fourth lateral and third lateral branches on either side, veins are formed which lie below and in front of these bronchi and pass in front of the stem bronchi to empty into the pulmonary veins, which lie upon their median and, ventral aspects. The vein from the second ventral bronchus (Pl. II, fig. 17), as in the younger stage, is placed medially to it and empties into the right pulmonary at the base of the third lateral bronchus. The veins from the second laterals have increased considerably in length, and lie on the ventral aspect of these divisions, while the Vena pulmonalis, formed by the confluence of the two right and left veins, lies ventral to the trachea just below the point of bifurcation. On the right side, the vein from Lateral 1 passes downwards and medianwards to empty into the Vena pulmonalis at a point just above the confluence of the two vessels which accompany the stem.
In a pig 18.5, mm. long, the relationships of the pulmonary arteries to the trachea (Pl. II, fig. 19) remain the same. Just above the point of bifurcation, they pass gradually behind the main bronchi to take up their dorsolateral position. No marked changes are observed in the arteries to Lateral 1, save in an increase in length. The second lateral branches present no changes, except on the left side where a branch runs up on the dorsolateral aspect of the apical division of Lateral 2 (Pl. II. fig. 19). The artery to Ventral 2 arising just beneath the Lateral 2 on the right side and passing around the stem and under the root of Lateral 2 to run along the outer aspect of the second ventral bronchus, now shows a secondary branch which follows the inferior division (Pl. II, fig. 19) of Ventral 2. Small arteries are given off to right and left Dorsal 2 which run along their lateral superior aspect. On either side, branches to Lateral 3 (Pl. III, fig. 20) run from a point just below the origin of the arteries of Dorsal 2. Beneath the third lateral bronchi, arteries arise which pass around the axial bronchus, and run lateralwards to Ventral 3. Below this level, branches are given off on both sides successively to Dorsal 3, Lateral 4, Ventral 4, Dorsal 4, and Lateral 5 (Pl. III, fig. 20). The pulmonary veins (Pl. II, fig. 19) lie medialwards and ventral to the main bronchi. Besides the branches from the lateral bronchi, which have been observed in the preceding stages, venules, lying on the medial surface of the dorsal bronchi, pass around the median aspect of the main bronchus and empty into the pulmonary veins. Similar veins from the ventral bronchi run along their median aspect, and empty into the Venae pulmonales on both sides.
Otherwise, there are no marked changes in the venous system at this stage save that the veins from the Lateral 2 and Lateral 1, on the right side now empty into the Vena pulmonalis by a common trunk. The second lateral vein on the left and with it a vein from the apical branch, which joins it about the root of Lateral 2 empties into the main pulmonary vein at a level somewhat higher up than the one which accompanies the left stem bronchus. The two veins from the stems join about the point of origin of the main bronchi and are continuous with the Vena pulmonalis above. From the infracardiac bronchus, a vein empties into the right stem vein just above the level of L. 3.
At this stage the main characteristics of the pulmonary vessels are established for life. The arterial branch to Lateral 1 runs upwards from the right pulmonary artery along the ventral surface of the bronchus and then follows the main divisions of the bronchi. Both arteries pass down behind the stem, lying on their dorsolateral surface in the angle between the dorsal and lateral bronchi. From it, three series of vessels arise, namely, those to the lateral bronchi, which run on the dorsosuperior surfaces ; those to the dorsal bronchi, which pass backwards from the stem artery on the laterosuperior aspect of the bronchus; and those to the ventral bronchi, which pass lateralwards around the stem bronchi to the lateral surfaces of the ventral group. Owing to the suppression of median bronchi on the tree of the 18.5 mm. embryo, the origin of the vessels to the median bronchi will be studied later in the corrosions of older embryos.
The veins have two chief branches accompanying the stem bronchi on their ventromedial surfaces. They receive as tributaries, veins from the lateral bronchi, which run along their ventroinferior surfaces and join the stem vein by passing above the corresponding ventral elements.
Branches from the dorsal series of bronchi run along the medial surface of the bronchi across the median aspect of the stem to empty into the veins on either side. A series of tributaries are also derived from the ventral bronchi, which, after a short course on the medial aspect of these bronchi, terminate abruptly in the stem veins. The vein from L. 1 lies ventral to the corresponding artery and empties into the vein of Lateral 2 in the Vena pulmonalis. Thus we have the veins from the upper and middle lobe emptying together into the main Vena pulmonalis on the right, while the single vein from the upper left lobe joins the main trunk on the opposite side. Below, the veins accompanying the stem fuse just below the division of the trachea and empty at this point into the Vena pulmonalis. The moving of the veins towards the left, due up to this time to the asymmetry of the heart and the hyperdevelopment of Ventral 2, is now somewhat exaggerated by the development of the inferior Vena cava on the right side of the infracardiac lobe, which also presses this structure to the left and, accordingly, must be looked upon as a factor in increasing the asymmetrical position of the pulmonary veins.
The next period of growth in the vascular system can be easily followed in specimens of the entire embryonic lung which, after fixation in some fluid like formalin or corrosive acetic to preserve the blood in the larger vessels, are subsequently cleared in oil of cloves or creosote. If the vessels are not too full both series are easily traced, but, in any case, the veins stand out distinctly. Owing to the complicated structure of the tree, however, the exact relationships of the arteries and veins to the bronchi are best seen in double corrosions in which, either the bronchi and arteries or the bronchi and veins are injected, or else, in triple injections where all three systems are filled with different masses. Preparations with the artery and veins filled with one color and the bronchi another, are relatively easy to obtain, but the more instructive triple injections are extremely difiicult to make. The changes gradually taking place with the growth of the tree, may be followed step by step in these cleared and corroded specimens, but they need not be described in detail until they are more exaggerated, as shown, for example, in triple corrosions of a pig 15 cm. long. Owing to my inability to find an artist who could draw these complicated structures, the reader may perhaps find it convenient to follow the following descriptions by means of the metal corrosions shown in Pl. IV, figs. 23, 24. The common pulmonary artery now divides to the left of the trachea a short distance after its origin from the pulmonary arch. The branch to the tracheal bronchus is given off from the right pulmonary artery at the left margin of the trachea and, after crossing ventralwards to it, divides with Lateral 1 into a dorsoinferior and a ventrosuperior branch. The latter passes ventralwards to the tracheal bronchus, and, at its point of division, mounts up over the ventrosuperior branch and comes to occupy a position dorsal, slightly medial, to this bronchus. The dorsoinferior branch passes beneath, and runs dorsal to the dorsoinferior bronchus. The right pulmonary artery then passes downwards in front of the trachea, and turns back and out to occupy a dorsolateral position to the axial bronchus. J ust above the second lateral bronchus, the branch to that division of the tree is given oif, which courses a little above and behind the bronchus sending ramifications to accompany its side bronchi. The dorsoinferior branch crosses behind the main bronchus, and runs dorsal to the branch which it supplies, leaving that structure between it and the corresponding vein.
In the remainder of its course, the second lateral branch lies dorsal to the bronchus with the bronchus between it and its accompanying vein.
The branch to Ventral 2 originates just below Lateral 2 and, passing underneath its root, winds around the axial bronchus to gain the lower and lateral aspect of the Bronchus infracardiacus, which it accompanies in its ramification. The dorsal branch to Dorsal 2 runs on the lateral surface of the bronchus and is given olf from the right pulmonary artery near the origin of the bronchus. The third lateral branch lies dorsalwards and sligthly superior to Lateral 3, and ramifies with its branches.
The branch to the third ventral bronchus arises in a manner similar to that of the second, and winds underneath the third lateral bronchus around the stem to the lateral aspect of Ventral 3. The artery corresponding to Dorsal 3 has a similar distribution to the one above. The fourth lateral lies above and behind the bronchus, while the fourth ventral passes in a similar manner to those supplying the same series of bronchi in the upper part of the tree. The fourth dorsal runs backwards just lateral to the bronchus, maintaining, in general, this position as it ramifies. In cases where there are median bronchi, as in this specimen, the . artery passes medianwards around the dorsal surface of the stem and is placed dorsal to the bronchus during its ramification. The fifth lateral, ventral, and dorsal have corresponding positions to those of the higher orders, and occupy the same relative positions. On the left side the pulmonary artery passes down without crossing the left bronchus at all to take its dorsolateral position to the stem. Just above the point of origin of left Lateral 2, the corresponding artery arises, and after passing a short distance dorsosuperior to the bronchus, almost immediately divides, sending a branch to the apical bronchus which continues upwards, placed laterally and dorsally to it. The remainder of the arteries on the left side have the same course as the corresponding branches on the right. In this description, I have followed strictly the typical specimens, although it is well to bear in mind that here, as in other parts of the vascular system, frequent variations are encountered. The veins still unite to empty into the left auricle through a common Vena pulmonalis. Branches from Lateral 1 and 2 form a common, large venous trunk on the right side, emptying directly into the Vena pulmonalis, while the vein from the left Lateral 2 joins the latter at a corresponding level on the opposite side. Below, the veins accompany the stem bronchi and their tributaries form a common trunk at the level of Lateral 3, which, crossing the ventral part of the stem bronchus between Lateral 2 and 3, empties into the pulmonary vein from below. The further growth of Ventral 2 on the right has gradually pushed the veins from the lower portion of the bronchial tree much more to the left, so that the large common trunk from the portion of the tree below Lateral 2 lies directly over the left axial bronchus at a point where the second ventral bronchus on that side would originate if the latter were present. It is this fact, as We have pointed out above, which has such great significance in explaining the suppression of that branch. From the ventrosuperior branch on the tracheal bronchus, the vein lies ventral and medial to it, receiving tributary vessels placed somewhat below the side branches of this bronchus. The vein from the dorsoinferior branch of Lateral 1 is placed ventralwards to that branch, and passes upwards to join the main trunk at a higher level. The main vein from Lateral 1, then passes down ventral to the artery and bronchus to form a common trunk with that from Lateral 2 as we have described above. The latter is placed above and ventralwards to the bronchus, receiving tributaries from its side branches. The main dorsoinferior branch of Lateral 2 lies ventralwards to its bronchus, while the corresponding artery is placed dorsalwards and above. This vein crosses behind Lateral 2 to join the main venous trunk, which accompanies Lateral 2 until, in common with the vein to the tracheal bronchus, it empties into the common pulmonary.
The veins from Lateral 3, 4, and 5 have shifted now so that they lie a short distance ventralwards from the corresponding bronchi. They pass medialwards under the ventral bronchi and empty into the right pulmonary stem vein; those from Ventral 3, 4, and 5 lie medial to the respective bronchi and run dorsalwards to the stem vein. Two veins now accompany Ventral 2, one above running medialwards and upwards and emptying into the large trunk formed by the fusion of the two stem veins, and another lying behind the branches of Ventral 2 which passes upwards and joins the common vein from the lower part of the tree on its right side at the point of junction of the veins from the right and left stems.
From Dorsal 2, 3, 4, 5, and 6 the veins, lying medial to their stems, ‘run ventralwards past the stem bronchus to empty into the large stem veins opposite their corresponding branches. The veins from the medial branch lie ventralwards to them and pass lateralwards to the stem vein.
The relationships of the veins on the left side of the tree below Lateral 2 are, with the exception of those from the Lobus infracardiacus, similarly arranged to those on the right.
Throughout the whole tree to this stage, we note with the single exception of Lateral 1 the constant relationship, which was indicated in the earlier embryos, of the regular alternation of artery, bronchus, and vein. In the earlier stages, the vessels were placed relatively close to the bronchi; but with the increasing age of the embryo, the position of the artery and vein has gradually shifted giving them a position at some distance from it. In some cases, this wandering may be so marked, especially below Lateral 2, that the main veins and their chief branches may occupy a position approximately midway between the adjacent bronchi.
The arteries, however, always lie closer to the air passages.
In the first part of embryonic life, the left pulmonary arch with a portion of the right connects the right ventricle and the aorta, and the pulmonary arteries, after the manner described by Bremer, finally take origin from the left by a common stem arising from its under surface.
The aortic arch lies above, and both arches are situated superior to the point of origin of Lateral 1, the tracheal bronchus. This relationship and the subsequent behavior of the two arches as the heart moves down affords us, I believe, some suggestive hints to explain the suppression of Lateral 1 on the left side and its unusual low position in those animals in which it is present. Through all of the stages we have followed hitherto, both the aortic and pulmonary arches, and the origin of the pulmonary arteries lie well above the origin of Lateral 1. As shown by a corrosion of the bronchi, arteries and veins in an embryo 15 cm. long, the pulmonary arch is exactly opposite the site of origin of Lateral 1, while the aortic arch is still higher. At the age represented by a pig 20-21 cm. long, the heart and vessels have descended further caudalwards, leaving the pulmonary arch well below the root of Lateral 1 and the aortic arch exactly at its level. At the time of birth, both arches have descended still more and pass dorsalwards in the interval between the trachea, the stem bronchus, and the apical branch of Lateral 2 (cf. Pl. IV, fig. 25). Now, had a symmetrical branch to Lateral 1 developed on the trachea, it is obvious that the descent of the great vessels and heart would have been prevented. Instead of reaching their final resting place just above the division of the trachea, they would have been left hanging above the level of Lateral 1. It is thus reasonable to suppose that the failure of this branch to form is due to a phylogenetic provision on the part of the tree to leave a passage for the descent of the heart and its great vessels.
A similar state of affairs is met with in the suppression of Ventral 2 on the left side. As the pulmonary vein forms approximately in the median line in the younger stages, the hyperdevelopment of right Ventral 2, the development of the inferior vena cava on the right side, and the shifting of the origin of the pulmonary vein from the site of its formation near the center of the undivided portion of the auricle to the left auricle, together with the increasing asymmetry of the heart, tends to carry the vein to the left. From its primitive approximate midline position in the earlier embryos, it is found with the increasing age of the embryo gradually passing to the left. In a pig 15 cm. long, we have the vein for the entire lower segment of the tree lying over the portion of the stem where left Ventral 2 should develop. Later still, in an embryo 20-21 cm. long, the descent of the hearthas changed once more these relationships leaving this area of the stem bronchus covered by the root of the pulmonary vein as it empties into the left auricle. As in the case of Lateral 1, this suppression represents a provision on the part of the tree to leave a space for the pulmonary veins.
We are forced, however, to consider those animals in which these commonly suppressed elements are present. In these relationships we can see a reason why no Lateral 1 ‘and Ventral 2 should form, but ‘whether this stands absolutely in the relation of cause and effect, it is impossible from my material to say, as it is conceivably possible, although less probable for the condition to represent an adaptation on the part of the vessels to the use of unoccupied space. For either its absolute afl‘irmation or disproof, therefore, a series of animals, in which these elements occur, must be examined from this standpoint during their developmental stages. This much may be said, however, in all of the lungs objectively pictured by Aeby, Huntington, and Narath where Lateral 1 is present on both sides, the one on the left is usually lower than the corresponding branch on the right. In the instances where they are on the same level, both are so low that they do not interfere with the descent of the heart and great vessels. Similarly, a bronchus that is not situated on the left stem in the segment between L. 2 and L. 3 cannot be considered as the homologue of V. 2, the Bronchus infracardiacus.
All other cases are substitution branches of the lateral bronchi or the stem. In the lungs which have been well pictured in the literature, where a real Ventral 2 occurs on the left stem, they are usually small and poorly developed and would not materially influence the migration of the Vena pulmonalis. It is also possible in these cases, as the veins are never drawn, that the latter have difierent relationships from those Influence of the Vessels upon the Architecture of the Bronchial Tree.-— After following the development of the vascular system, we may consider now the possibility of the influence of the vessels upon the architecture of the bronchial tree. Concerning the general asymmetry of the lungs, many of the older investigators have looked upon the heart or the great vessels as being responsible for this irregularity. Thus Bichat, 29, and Riidinger, 73, thought the left bronchus owed its greater length to the asymmetry of the heart, while Meyer, 61, looked upon the aortic arch as the factor which drew it out to greater length. In reviewing these statements, Aeby felt these authors passed over the most weighty relationship in overlooking the crossing of the bronchi by the arteries at a particular point on the stem to run down on its dorsal surface. This crossing enables the artery in the “hyparterial” to divide the side bronchi into a dorsal and ventral series, while the “ eparterial ” bronchi, situated above this separating influence of the artery, have their dorsal and ventral branches arising from a common stem. In quoting K6lliker’s observations on a 35-day human embryo, Aeby calls attention to the origin of the pulmonary arteries in the embryo above the lungs, and states: “Ein spater eparterieller Bronchus muss somit so lange hinter ihr liegen, als nicht in Folge des hohern Aufsteigens des Organs eine bogenfiirmige Ablenkung derselben iiber den ersten Ventralbronchus hinweg nach vorn hin stattgcfunden.” While Aeby looked upon the lungs instead of the heart as the movable factor in establishing the adult relationships of the arteries to the tree, he recognized notwithstanding this misinterpretation, the necessity of the embryological topography of the “ eparterial ” or first lateral bronchi to produce the conditions which we find in later stages. It is clear from the above account of the development of the pulmonary arteries that these delicate vessels which regularly follow the growth of the bronchi and do not, in fact, appear in any part of the lung until after the respective branches which they supply are present, have no formative influence on either the structure or relationships of the bronchi, but are simply passive followers of their development produced by histomechanical principles from the capillary plexus which surrounds them. finally, a crossing of the stem bronchus by the artery does not occur until after birth when all of the bronchi are laid down, and even then, in the strict sense of Aeby, does not exist as Zumstein and Narath have already shown. It is thus most difficult to determine just what led Aeby to lay such stress upon the adult relationship of the artery to the stem when he obviously, as the above quotation shows, clearly recognized that it was not associated with the earlier formation of bronchi, but was due, as he supposed, to the later ascent of the lungs. Furthermore, the pulmonary artery is not responsible for the dorsal and ventral divisions of the stem bronchi as We have ventral and medial elements also arising from the stem away from any possible influence of the artery.
Muller, 98, brings forward an interesting suggestion with reference to the effect of the pulmonary arteries on the tree dependent upon the descent of the heart in mammals which have had the form of their chest Wall altered by their life in water. The pulmonary arteries, according to Miiller, following the descent of the heart tend to drag the “ Ventral bronchi” caudalwards, leaving the dorsal bronchi free and uninfluenced by the arteries to wander up on the stem bronchus or trachea to form the so-called “eparterial” bronchi. This ingenious suggestion is not borne out, however, by the facts of embryology, for as we have seen, all the bronchi are well formed before the heart in its descent reaches a level Where the pulmonary arteries could exert such a traction upon the lateral bronchi.
Huntington, 98, says: “If We seek for an explanation of the cause which leads to the migratory changes of the cephalic bronchus (Lateral 1), I admit that we enter the realm of pure hypothesis. At the same time, the very general development throughout the marnmalia of this type, with the resulting greater respiratory area of the right lung, may, I think, not improperly be referred to the development of the mammalian form of the systemic and pulmonary arteries. On the left side, the greater quantity of blood thrown from the right ventricle into the left pulmonary artery passes through the Botallian duct directly into the aorta, only a small portion traversing the left pulmonary circulation.
On the right side, however, with the early obliteration of the dorsal segment of the fifth arch, all the blood entering the right pulmonary artery is forced to traverse the entire pulmonary circulation returning to the left auricle by the pulmonary veins.” This explanation, according to Bremer’s description of the development of the pulmonary arteries, could not account for the increased size of the right lung, especially in the pig where all of the‘ blood to the lungs is forced to pass through the left pulmonary artery after the establishment of the transverse anastomoses and the subsequent degeneration of the proximal portion of the right pulmonary artery.
We may say then in conclusion, that there is one simple possible explanation for the general asymmetry of the mammalian lung which lies in the asymmetry of the anlage. Owing to the fact, however, that the pulmonary anlage in lower animals is frequently symmetrical, it seems more probable to look upon this characteristic as an adaptation on the part of the pulmonary apparatus to its environment which may reach such extremes as we find in the lung of the snake. It is more probable then, that, with the necessity of an increased respiratory surface as we ascend the animal scale, the asymmetrical heart and the development of its adult form gives us adequate ground for a normal asymmetry of the respiratory apparatus, especially as the heart and liver, forming the principal environment of the lungs, have phylogenetic precedence and are of more physiological importance during intrauterine life. In its final form, this asymmetry consists, in the vast majority of lungs, in a suppression of left Lateral 1 to leave space for the descent of the aorta and pulmonary arch with the heart and a suppression of left Ventral 2 to provide room for the pulmonary veins from the lower lobes. In animals, however, where these branches are formed they are so placed that they do not interfere with either of these features of the development of the vascular system.
The relation of the mesoderm to the primitive tree has been described in connection with the appearance of the bronchi, largely because it arises from the general mesoblast of the head gut and takes part in the separation of the pulmonary anlage from the (esophagus. The mesoderm, it will be remembered; shows the influence of the first irregularity of the early branches of the tree and forms two indefinite unequal rounded projections into the primitive coelom on either side. These are the anlagen of the two lung wings. On both sides the Recessus pleuroperitonealis projects upwards and somewhat medialwards to the bronchi; the left, however, is very poorly developed. Ventralwards the mesoderm continues forwards into the Mesocardium posterior.
TEXT fiG. 15. Outline drawing of the lungs of an embryo pig 10 mm. long.
Ventral view. (figs. 15-19-24, inclusive, drawn with a camera lucida from cleared preparations.) L. 1, L. 2 = Swelllngs, limited by shallow grooves, over Lateral 1 and Lateral 2. ST: Mesoderm over the caudal portion of the stem bronchi. Also L. 1=Lobus superior. L. 2=Lobus medius (right) ’ and Lobus superior (left). ST=Lobus inferior.
File:Flint1906 textfig16.jpg
TEXT fiG. 16. Outline drawing of the lungs of an embryo 12.5 mm. long.
A. Ventral view. B. Dorsal view. L. 1, L. 2, L. 3, V. 2, and ST=Swelllngs over the several bronchi and the stem designated by these abbreviations. At this stage the anlagen of the lobes are complete. L. 1 =Lobus superior, L. 2 =Lobus medius (right), Lobus superior (left). V. 2=Lobus infracardiacus. L. 3 and ST=Lobus inferior.
At 10 mm. the two simple lungs are quite asymmetrical (fig. 15).
Increasing in size with the growth of the bronchi, they also follow their asymmetrical development. The fain swellings observed in the preceding stage have become so exaggerated that we have on the surfac of the lung marked rounded elevations indicating the presence of Lateral 1 (fig. 15, L. 1) on the right side, and Lateral 2 on both sides (fig. 15, L. 2).
These projections are limited by shallow groves. From above downwards, the trachea and hence the mesoderm extends ventralwards until the point of bifurcation is reached, when, following the course of the stern bronchi, it passes dorsalwards on either side of the oesophagus.
At 12.5 mm. (fig. 16) these characteristics are exaggerated. On the right side, high up, we have the projections over the bronchi, which have been found before this stage. They have increased in size with the growth of their respective elements ; also there is now a well-marked projection over the newly-formed V. 2 (fig. 16, V. 2) and a less apparent swelling, the bud representing Lateral 3 on each side (fig. 16, L. 3).
TEXT fiG. 17. Outline drawing of the lungs of an embryo pig 13.5 mm. long. A. Ventral view. B. Dorsal view. The letters represent the mesodermic swellings over the bronchi designated by the abbreviations. Designations the same as in fig. 16, except that L. 3 and all swellings below that order unite in the pig to form the Lobus inferior.
The furrows have deepened, and the lower part of the wings below Ventral 2 now embraced by the Wolffian body and chest wall dorsally, the heart, liver, and diaphragm ventrally, and the mesoderm of the oesophagus medially, have already in cross-sections an irregular prismatic form. At this stage we may say, the anlagen of the lobes are complete. From each of these main projections, a lobe is produced and the shallow grooves deepen with the further growth of the lungs to form the interlobar fissures. That is to say, on the right side the swellings over Lateral 1, Lateral 2, Ventral 2, and the stem produce respectively the Lobus superior, Lobus medius, Lobus infracardiacus, and Lobus inferior, while Lateral 2 and the stem bronchus produce the Lobus superior and Lobus inferior on the left. At 10 mm. the swelling over L. 1 is practically in the same lateral plane as L. 2, while at 12.5 mm. it is crowded slightly dorsalwards by the further growth of the latter.
In a pig 13.5 mm. long (fig. 17), the characteristics of the lobe formation are intensified. On the right side, the upper lobe containing Lateral 1 is pushed still more dorsalwards, while the middle lobe containing Lateral 2 is, at the same time, forced slightly ventralwards by the antagonism in the growth of their two main bronchi. The Lobus infracardiacus, containing Ventral 2, extends downwards and medialwards, while the lower lobe extends more caudalwards and is now, through its whole extent, distinctly prismatic in cross-section. On the left side, the Lobus superior, owing to its more unobstructed environment, extends somewhat higher than its homologue, the Lobus medius, on the right side. The Lobus inferior is not quite so large or well developed as the corresponding right lobe. The primary fissures between the several lobes have deepened and now extend well into the substance of the lung. With the division of Lateral 1 and Lateral 2 on each side, the secondary branches also raise secondary projections on these surfaces of the lobes between which are slight secondary furrows. Similarly the Lobus inferior on each side shows slight swellings limited by shallow grooves over L. 3 and L. 4. In the pig, these swellings and grooves, however, under ordinary circumstances, never lead to a separation of the lung substance into extra lobes.
TEXT fiG. 18. Outline drawing of the lungs of an emtryo pig 14.5 mm. long. A. Ventral view. B. Dorsal view. Designation of lobes as in fig. 16.
Fig. 18 shows the lungs of an embryo 14.5 mm. long. The Lobus superior on the right side (fig. 18, L. 1) is now pushed dorsalwards by the presence of the heart and the Lobus medius (fig. 18, L. 2), so that its caudal portion now lies above the series of swellings over the dorsal bronchi (fig. 18 B, D. 2). On the left side, the Lobus superior now shows a dorsoapical swelling over the apical branch of L. 2 (fig. 18, L. 2), which indicates the beginning of the portion of the left upper lobe, which substitutes for the Lobus superior on the right side. The fissure between L. 2 and L. 3 on each side deepens, while the Lobus inferior on both sides shows a series of projections over the several branches of the stem. On the ventral surface, V. 3 is indicated; on the lateral border, L. 3, L. 4, and L. 5; while, on the dorsal border, swellings for D. 2, D. 3, and D. 4 are present.
TEXT fiG. 19. Outline drawing of the lungs of an embryo pig 18.5 mm. long. A. Ventral view. B. Dorsal view. The abbreviations on the swellings represent the order of the bronchi beneath. Designations as in fig. 16.
In a pig 18.5 mm. long (fig. 19), the right Lobus superior containing Lateral 1, projects upward some distance beyond the tip of the upper lobe on the left side. The fissure separating it from the Lobus medius has deepened. Its lower portion now passes behind the medial lobe, although the two are united at their roots, that is to say, the ventromedial aspect. The Lobus infracardiacus projects ventralwards and medialwards until it extends over the median line above the oesophagus.
The lower lobe on the right side shows projections along the lateral border for L. 3, L. 4, and L. 5, and, on the dorsal border, for D. 2, D. 3, and D. 4. The ventral surface, likewise, has very slight swellings for V. 3 and V. 4. The latter, however, are very faint and are separated from the rest of the lobes by very shallow grooves. On the left side, the Lobus superior (fig. 19, L. 2) is separated from the lower lobe by a deep cleft, while the development of the apical branch of L. 2 has pushed up with it a segment‘ of this lobe which also grows backward until it lies above the series of dorsal swellings (fig. 19 B) and bears a marked resemblance to the Lobus superior on the opposite side. Excepting for the Lobus infracardiacus, the lower lobe has characteristics practically homologous to the corresponding lobe of the right side. The dorsal flexion of both lower lobes still persists and the lateral tips or margins of the median lobes now begin to show, at their lateral extremities, a slight bending ventralwards as they begin to fold around the heart.
As the lung continues to grow, with the successive appearance of new branches, new elevations are formed on the surface of the primitive lobes until finally, as Narath describes, they have an appearance like the surface of a mulberry. The primitive lobes, however, keep their independent character and alter in form by two chief factors, namely, the intrinsic growth of the lung itself, and the change in its environment formed by the chest wall, heart, liver, and diaphragm. N arath has given as the cause of the lobe formation, the extremely rapid growth of the first branches of the tree, while, the later branches of slower growth fail to form furrows in the mesoderm deep enough to subdivide the lung further.
With this view, I am in complete accord, but it ought, it appears to me, to be extended to include the character of the mesoderm. In the early stages, this is in extremely plastic form, which easily moulds itself to the pressure of the growing bronchi beneath. Up to 10 mm. there is scarcely any differentiation in the mesoderm into distinctly fibrillar and cellular portions, while at 12 mm. this change is inaugurated and fibrils appear particularly in the region of the root of the primitive lung. At 20 mm. the whole mesodermic portion is composed of young connective tissue with well-marked fibrils. As the mesoderm differentiates, therefore, it becomes firmer and is less easily influenced by the growth of the young bronchi.
Fig. 20 is an outline drawing of the-lateral and diaphragmatic aspects of the lungs of an embryo 19 cm. long. At this time, all of the important adult topographical features of the lungs are present. A., shows well how the right Lobus superior has grown down and back into the dorsal area, moulding itself even more than in an embryo 18.5 mm. long (fig. 19.) to that portion of the thoracic cavity and extending now up over the base of the heart beyond the midline making the sum of lung tissue in L. 1 and L. 2 considerably greater than that in L. 2 on the opposite side.
TEXT fiG. 20. Outline drawings of the lungs of a pig 19 cm. long. A. Right side. B. Left side. C. Diaphragmatic surface. At this stage, the surface of the lungs is smooth. The topography of the bronchi beneath, taken from corrosion specimens of the same age, is indicated by dotted lines and letters.
Owing to this growth, the Lobus medius is pressed ventralwards, its dorsal segment lying in the angle between the Lobus superior and the Lobus inferior. It may be interesting to note, that the portion of the lobe which lies in this angle, is supplied by the large dorsoinferior bronchus. It is, therefore, ontogenetically equivalent (vide Pl. II, figs. 15, 16) to the apical segment of the Lobus superior on the other side. Nothing could indicate clearer the adaptation of the growing bronchi to their environment, or the possible influence of environment upon the branches of the tree. The tips of the Lobus medius have grown around the heart until they have almost met in the midline. On the undersurfacc, the unpaired Lobus infracardiacus (fig. 20, V. 2) is clearly seen particularly in its relationship to the Vena cava inferior.
With the increase in size between this and the last stage, the swellings over the various bronchi have disappeared and the surface of the lobes become smooth. The topography of the Lobus inferior on both diaphragmatic and lateral surfaces is indicated on the surface of the lungs by dotted lines. By a comparison with fig. 19, the origin of these topographical relations are clear.
With the further development of the pigs lung which has been described by Narath, I cannot agree. In the account of the form relationships, his work is accurate, but in the interpretation of the relative significance of the different parts of the lung and the equivalent values of the lobes on each side, our results differ chiefly with our derivation of the principal bronchi. That is to say, according to his view the Lobus superior and the Lobus medius on the right side are equivalent to the Lobus superior on the left. They are almost or completely separated through an accessory fissure, making the Lobus superior correspond to the dorsal or apical area in his preparations and equivalent to the cephalic or apical projection of the Lobus superior of the left lung. The latter, as we have seen, is only a secondary substitution product of a branch of left L. 2, ontogenetically equivalent to the region of the Lobus medius on the right side which is supplied by the large dorsoinferior bronchus.
On the other hand, the right Lobus superior, supplied by L. 1, is totally unrepresented in the left lung. This unpaired lobe, therefore, and also the cephalic portion of the upper lobe on the left, properly belong not to the dorsal area, as N arath suggests, but to our lateral and his ventral region. The fissure between Lobus superior and Lobus medius on the right would be primary and not accessory in the sense of Narath.
In recapitulating the development of the lobes, we may say, then. that the mesodermic portion of the lungs, derived from the general mesoderm about the head gut, is pushed out by the growing bronchi to form irregular asymmetrical swellings in the coelom. These are the anlagen of the primitive wings of the lungs. With the appearance of L. 1 on the right side of the trachea, and L. 2 on each stem bronchus, primary swellings are formed in the two wings over these bronchi, giving rise to the simplest form of the Lobus superior, Lobus medius on the right side, and the Lobus superior on the left. The remainder of the mesoderm about the stem bronchi form the anlage of the Lobus inferior on each side. With the appearance of V. 2, the Bronchus infracardiacus, on the right, a swelling forms over it yielding the anlage of the Lobus infracardiacus. These swellings are at first surrounded by shallow grooves, which, with the rapid growth of the bronchi beneath, develop into the fissures separating the various lobes. With the further growth of these chief bronchi and the appearance of the series of bronchi on the stem, a series of swellings and fissures are formed over and between them.
These are equivalent, in all senses except in age and size, to the earlier fissures and swellings, but, under ordinary circumstances, never deepen into distinct lobes. This is partly due to the more rapid growth of the ‘first bronchi, to the gradual increasing density of the mesoderm, and, lastly, to the environment of the several lobes of the lung. That is to say, the Lobus superior with L. 1 has the territory between the chest wall and the upper part of the heart on the right side. The right Lobus medius and the left Lobus superior, with L. 2, have the large space between the chest wall and the angle formed between the heart and liver on each side. It is important, however, to note on the left side, owing to the absence of L. 1, the Lobus superior sends up the apical segment of the lung containing the left Bronchus ascendens. The Lobus infracardiacus, with V. 2, grows out into the space left between the heart and liver and the two lower lobes, while the Lobus inferior on each side lying in the more or .less triangular space between the chest wall and liver and diaphragm becomes prismatic in cross-section and grows caudalwards and lateralwards to fill up the rest of the pleural cavity.
In the pig, then, we have a series of primary projections limited by a series of fissures some of which give rise to the permanent pulmonary lobes. Those projections and fissures which take part in the lobe formation in the pig, it is well to observe, are the first to form, but in other animals these same conditions do not appear to obtain. In Hystrix cristata, for example, not only the primary fissures between practically all of the principal bronchi may give rise to a series of lobes, but these may even be subdivided by the secondary fissures formed by the secondary branches of these elements, while in other animals, as for example man, the deepening of the fissures about V. 2 usually do not produce a separate lobe, leaving this region of the lung included in the right Lobus inferior.
Between these forms we have extensive individual and general variation.
The drawings in fig. 20 may be used conveniently to explain the lobe production in all mammals; A represents the conditions in animals where L. 1 is present on one side or both; B, the conditions where L. is absent on one side or both; 0 represents lungs where a Lobus infracardiacus is present, and by eliminating this lobe and altering the topography of the ventral bronchi, it may be used for lungs where V. 2 is either absent or included in the Lobus inferior. For example, B represents the conditions found in Hystrix cristata in both lungs where not only all of the primary bronchi in that animal have produced lobes, but some of them are still further partially subdivided. There is also a type of lung represented by Phoca vitulina where L. 1 is present on both sides, but L. 2 in this species is thrown into the Lobus inferior. For this state of affairs A would suffice if the permanent fissure between L. 2 and L. 3 were replaced by a dotted line. The suppression of the lobes indicated in Phoca vitulina may involve all fissures giving us a lobeless lung like those of Delphinus delphys and Pitheeus satyrus.
It is, of course, clear from the above description how we regard the equivalent values of the lobes on the two sides, but they may be simply stated in two simple formulae of equivalence which will fit the lungs of most animals depending upon the presence of L. 1 and V. 2 on one or both sides. Type 1 includes the great majority of mammalian lungs.
Type 1. Type 2.
L. 1 present only on the right side. L. 1 present or absent on both sides.
Right Side. Left Side. Right Side. Left Side.
Lobus superior : 0. Lobus superior = Lobus superior.
Lobus medius : Lobus superior. Lobus medius = Lobus medius.
Lobus inferior :: Lobus inferior. Lobus inferior =Lobus inferior.
01‘ 01‘ Lobus inferior + V. 2 :: Lobus in- Lobus superior = Lobus superior.
ferior + V. 2 or O. Lobus inferior = Lobus inferior.
While lobe production in the lungs is obviously dependent on the growth of the bronchi in the majority of instances, the number of lobes is apparently without definite morphological significance. It may vary in animals from multilobed lungs like those of Hystrix to lobeless lungs like those of Pithecus satyrus. The common relationships, however, are expressed in the types given above.
The Organogenesis of the Lungs
In turning to the organogenesis of the lungs from the period of the formation of the Anlage until the adult stage is reached, the first interest settles in the chief cells of the bronchi and the pulmonary connective tissue. Both of these structures have been followed up to the age represented by a pig 10 mm. long, in the chapter on the development of the bronchi. From this time, it is more convenient to consider these stages by themselves. In the description of the differentiation of the framework, I have taken as a basis the work of Mall, 02, who has described in the pig the origin of the connective tissues from a common mesodermic syncytium. By a differentiation of this syncytium into an endoplasmic and exoplasmic portion, the connective tissues are produced. The former remains as the protoplasm about the connective tissue cells, the latter forms the various fibrils. The author, 03, has traced the development of the framework of the submaxillary gland in the pig, where, in the earlier stages, the process of differentiation is the same as in the lungs. By way of review, suffice it to say that the syncytium forming the primitive framework of the lungs differentiates slowly until 10 mm. is reached when, in the neighborhood of the root of the lung and the Mesocardium posterior, the fibrils begin to appear and the cells become more isolated from each other. About the young bronchi, however, they are still in close apposition during the formation of the reticulated membrane about the tubes, which, in Mallory preparations, may be seen as a dark blue line.
TEXT Fig. 21. Longitudinal section of the left lung of an embryo pig 13 mm. long. fixed in Zenker’s fluid and stained by Mallory’s FuchsinAnilin blue method. X70. b=Stem bronchus. p:p1eura.. a=Young connective tissue. 0: syncytium. m : evagination forming medial bronchus.
At 13 mm. (fig. 21) these conditions are well shown. The stem bronchus (fig. 21 b) and its chief lateral branches is seen in longitudinal section lined, by an epithelium consisting of a row of inner columnar cells with smaller polygonal cells beneath them. The epithelial tube is surrounded by a simple reticulated membrane which is in process of formation. Above, at the root of the lung (fig. 21 a), the transformation of the exoplasm into young connective-tissue fibrils has taken place, while in the lower portions of the Lobus inferior (fig. 21 0), the framework consists of a mass of anastomosing syncytial cells without any particular differentiation. About the basement membrane, the cells are thickly packed and under the primitive pleura (fig. 21 p) the epithelium of which has begun to flatten, we have a distinct blue line indicating the formation of a membrana propria.
Tnxr fiG. 22. Section of the lung of an embryo pig 3 cm. long. Same preparation as used with tissue shown in fig. 21. X 70. pzpleura. a: connective tissue. b zbronchus.
In a pig 30 mm. long (fig. , the framework of the entire lung shows a differentiation into primitive fibrils. The young fibrils are more distinct and less granular, while the spaces between are larger than in the preceding stage. With the differentiation, the relative quantity of endoplasm has diminished in the loose part of the syncytium, leaving in some places isolated connective—tissue cells (fig. 22 c), or in others they are multipolar in appearance with branching and sometimes anastornosing processes. Immediately about the trachea and large bronchi, the cells are closely packed together preparatory to the production of the various coats of these structures. The basement membrane is distinctly fibrillated as is seen at points where the plane of section is tangential to the bronchi. About the larger bronchial elements a group of elongated fusiform cells having a distinctly circular arrangement may be noted, representing the earlier stages of the production of the muscular coat.
TEXT fiG. 23. Section of the lung of an embryo pig 5 cm. long. Same preparation as used with tissue shown in fig. 21. X 70. p=pleura. a: connective tissue. 2) zbronchus.
The epithelium in all the large and in the majority of small bronchi still consists of two layers of cells, the inner columnar, the outer polygonal in form. But in the youngest branches of the oldest bronchi, namely Lateral 1 or 2, there is now a reduction to a single layer of columnar cells (fig. 22 b). Cilia. are as yet invisible in these specimens, but the cuticula at the inner margin of the cells is already differentiated. At the root of the lung, a few dilated lymphatics may be noted near the bronchi and pulmonary vessels; they have not, however, grown beyond this point into the substance of the lung wings.
Embryo 5 mm. long (fig. 23). The general framework (fig. 23 a) of the lung at this period has undergone a further difierentiation over the preceding stages, consisting in an increasing density and complexity of the young fibrils, which now possess a more distinctly flbrillar appearance, while the quantity of endoplasm about the connective-tissue cells has slightly diminished, except in the immediate neighborhood of the larger bronchi. The pleural epithelium (fig. 23 p) is much more fiattened and the nuclei of the individual cells consequently further apart.
As shown by points Where the plane of section falls tangential to its surface, the basement membrane beneath this epithelium is distinctly reticulated. About the larger bronchi, there is a distinct circumferential arrangement of the exoplasmic fibrils in which are imbedded a great many cells. The basement membrane is slightly thickened and just beneath the latter there is now a well-marked layer of fusiform cells with elongated nuclei running circularly about the bronchial tube. External to this stratum, is a looser circular arrangement of the exoplasmic fibrils as well as the cells embedded in it. When the bronchi are cut longitudinally, these circumferential lamellaa of cells and exoplasm run parallel to the long axis of the tube. The epithelium, as in the preceding stages, shows a distinct division into two or three layers, with the nuclei situated approximately in the middle of the cell. The thickening on the edge of the cell lining the lumen is apparent, although cilia are as yet unformed. As the branches of the tree are followed towards the periphery, the layers of circularly directed syncytial cells disappear and we have simply the primitive basement membrane with the connectivetissue cells immediately about it. In the most terminal parts of the air passages, the double layer of epithelium has been replaced by a single layer of lower columnar epithelium (fig. 23 b). All of the bronchi from the first to last possess marked lumina. From the root of the lung, the lymphatics have now grown some distance into its substance. They have thin walls composed of young fibrils lined by endothelium with occasional valves. They are confined, however, to the immediate neighborhood of the main bronchi and their chief subdivisions.
TEXT fiG. 24. Section of the lung of an embryo pig 7 cm. long. Same preparations as used with the tissue shown in fig. 21. X 70. =p1eura. b zbronchus. a=connective tissue. lzlymphatics. This stage shows the beginning of the lobulation.
Pig '7 cm. long (fig. 24). A number of interesting changes have taken place in the evolution of the lungs since the last stage other than in a further differentiation of the framework, which at this time is considerably denser. The circularly arranged fusiform cells noted in the earlier stages about the main bronchi are collected into bundles to form the muscular layer outside of the mucosa, while still external are stages in which the chondrification of the syncytium is progressing as the latter ‘passes over into the precartilage stage at the periphery, and into young cartilage in the center to form the simple chondral rings of the trachea and larger bronchi. The epithelium of the latter is sometimes thrown into folds, is cylindrical, and composed of a. double layer of cells. As one follows the branching to the end buds, it first becomes single layered and then of a low columnar type (fig. 24 b). Chondral rings and bronchial cartilages are present only around the trachea and the upper part of the stem bronchi; the muscular coat, as one passes peripheralwards, thins out until it first consists only of a single layer of cells, and finally at the smaller branches and end buds is replaced by the young connective tissue, which, in the latter region, is engaged in the formation of the reticulated membranes.
The most interesting change, however, lies in the further growth of the lymphatics, which, in the earlier stages, are found in the root of the lung in the neighborhood of the pulmonary vessels and large bronchi.
As they grow in, they accompany these structures for a distance, then, approaching the end branches, they leave them and run in a plexiform manner midway between the bronchial tubes (fig. 24 Z) until they reach the pleura (fig. 24 p). This gives the lung now an indefinitely lobu— lated appearance, in which the periphery of the simple lobule is indicated by the lymph vessels and the center by the bronchi. The lymphatics are lined by flattened endothelium, their walls are formed by the young connective-tissue fibrils, and, here and there, valves are beautifully shown, which, in general, point away from the pleura. The pleural epithelium (fig. 2419) is much flattened and now rests upon a thickened layer of young connective-tissue fibrils.
Pig 13 cm. long (fig. 25). At this stage, we have the whole lung subdivided into a series of connective-tissue lobules with essentially the same characteristics as those shown in the preceding stage, namely, a peripheral plexus of lymph vessels with the bronchus in the center. The growth is centrifugal in so far as the bronchi are concerned and, in this sense, the lung at this stage may be compared in some respects with the younger stages of the salivary glands for example, Where similar lobules without peripheral lymphatics are also formed from a centrifugal growth of the ducts. The framework at this stage (fig. 25 a) is considerably thicker than in the preceding embryo, the fibers denser and, at the same time, there are more connective-tissue cells. Under the pleura (fig. 25 p) and in the interlobular spaces, the fibrils are gathered into slight trabeculse, which limit small spaces in the connective-tissue netWork.
The larger bronchi show an increase in the characteristics indicated in the last stage. The epithelium is thrown out into longitudinal folds, which are accompanied by folds of the basement membrane and submucosa. This is composed of trabeculae formed from the young connective-tissue fibrils. In the young submucosa, the simple muscle bundles lie, and still external to the muscularis the cartilagenous rings are in process of formation. Proceeding peripherally, the bronchi grow essentially younger and the epithelium is first reduced to a single columnar layer, which then becomes lower until, in the lobules, it forms a lower columnar epithelium. Still further out in the growing terminal buds (fig. 25 c), it now has a distinct cubical form. About these, the membrana propria is formed from the connective tissue of the lobule.
TEXT fiG. 25. Section of the lobule of the lung of a pig 13 cm. long Same preparation as used with the tissue shown in fig. 21. X 70. p =p1eura. azconnective tissue. bzbronchus. c: end bud. lzlymphatics.
The lymphatics (fig. 25 Z), forming a plexus around the bronchial veins and arteries at the root of the lung, accompany them towards the periphery, giving off branches to the interlobular spaces en route. Their walls, owing to the increasing differentiation of the framework, are thicker. On reaching the periphery of the lung, they leave these structures and pass out as in the preceding stages to the pleura. They have a plexiform arrangement and may (fig. 25) be traced at times into the substance of the lobules. This course may also be observed in the deeper lobules of’the lung as well as those on the surface under the pleura.
In the period of embryonic life between pigs 13 and 19.5 cm. in length there are no marked changes of the relationships we have thus far described. In the larger bronchi, a gradual development has occurred.
The epithelium now possesses well-marked cilia springing from the cuticular border of the inner layer of epithelium, between the elements of which, goblet cells appear here and there, partly filled with mucous.
These are clearly seen first in the stem bronchi of pigs between 15 and 17 cm. long. The folds, which have already been described running longitudinally with the bronchus, now look in cross—sections like regular papilla with a core of submucosa. That they are regular structures of the bronchi and not shrinkage products is shown by the impressions they leave on corrosion specimens which are injected under considerable pressure as well as their appearance in distended lungs. The muscularis mucosae is more developed and the bronchial cartilages are well formed. In general, the relations of the lymphatic system has not changed; lymph glands may be observed forming in the neighborhood of the root of the lung, and large bronchi in pigs as young as 12 cm. They naturally increase in size and number with the age of the embryo. With the other changes, there has been a gradual flattening of the epithelium in the growing ends of the tree, until, in an embryo 18 cm. long, the end buds are lined by a very flat form of cubical cells with spherical nuclei. The cytoplasm, which in the earlier stages was granular, is now clear and transparent.
At 19 cm. (fig. 26), some notable changes have been inaugurated in the structures. The ciliabed epithelium of the stem bronchi possesses a great number of goblet cells. In the submucosa, the muscularis has gathered into distinct bundles, while from the fundus of the crypt-like invaginations between the mucosal folds appears an ingrowth of glands, containing partly serous cells and partly mucous cells which penetrate sometimes as far as the muscularis and sometimes between its bundles into the submucosa between it and the bronchial cartilages. In general, the relations of the lymphatic system (fig. 26 Z) have not changed, but the connective-tissue lobules (fig. 26) containing the growing ends of the bronchial tree have increased considerably in size. The framework (fig. 26 a) is denser around the end buds (fig. 26 c), which, while still lined by flat cubical epithelium, now show a dilatation of their lumina preparatory to the formation of the respiratory lobules of Miller.
TEXT fiG. 26. Lobule of the lung from a pig 19 cm. long. Same preparation as used with the tissue shown in fig. 21. X 70. b=bronchus. p=p1eura. Z: lymphatics. c : end buds. a : connective tissue.
In pigs about 22 cm. long (fig. 27), the chief changes are in the grow which now have an extremely complicated contour and show widely dilated lumina. As they begin to pack together in the lobule, the connective tissue is compressed between them, and its nuclei in consequence appear more numerous. From the low cubical
tissue shown in fig. 21.
torious.
TEXT fiG. 27. Section of lobule of the lung of a pig 22 cm.
X 130. l = lymphatics. 1: ductulus alveolaris. p : pleura.
long. Same preparation as used with the b = bronchiolus. M = bronchiolus respirating end buds (fig. 27 i)
TEXT fiG. 28. Section of a lobule of the lung of an embryo pig 27 cm. long. Same preparation as used with the tissue shown in fig. 21, except that the lung was distended with the fixing fluid. X 130. b=bronchio1us. br= bronchiolus respiratorius. izductulus alveolaris. azatria. Z: lymphatics.
epithelium of the smaller bronchi (fig. 27 b) the transition is easy to follow over into the irregular flattened epithelium that now lines the young respiratory lobules. The nuclei are pressed against thesides of the lobules and the relatively slight amount of clear cytoplasm extends between them. The Bronchioli respiratorii (fig. 27 br) are now readily recognized leading off from the bronchioli (fig. 27 b). They open into the dilated Ductuli alveolares (fig. 2713) from which the primitive Atria may be seen as lateral outgrowths.
Shortly before birth, in a pig 27 cm. long (fig. 28), the framework of the lung at the root, between the lobules and under the pleura, consists of definite trabeculae composed of fibrils in the meshes of which lie the connective-tissue cells. In the neighborhood of the root, the trabeculse are thick and firm and thin out as the periphery is reached. The structure of the stem bronchi is on the same plan as in the earlier stage, but the epithelium submucosa, muscularis, and cartilages are more developed.
As the periphery is approached in this, as in the younger stages, they become essentially younger in structure, loosing first their cartilages, then the muscularis, and finally, before terminating, have only a thickened basement membrane which contains connective-tissue cells (fig. 28 b).
The respiratory lobules are now fully formed, but are not as large or as complicated as in the stages after birth. In this section there are two Bronchioli respiratorii (fig. 28 br) from the ends of which the Ductuli alveolares (fig. 28 45) lead. These terminate in dilated Atria (fig. 28 a) on the walls of which the Sacculi alveolares are now indicated as slight irregular outgrowths. While complete corrosions of the lungs in which the respiratory lobules are injected are of great service in interpreting the pictures found in sections, I have feared to trust these preparations for an exact description of the growth of these structures, owing to the possibility of artefacts. The nuclei of the respiratory epithelium now project often into the lumen of the air spaces. In general, the cells are extremely flattened and the nuclei elongated. A flat sheet of protoplasrn extends out from either pole of the nucleus resting upon the mebrana propria. Here and there, where capillaries project into the lumen of the air passages, the nucleus lies in the angle formed by the capillary and the basement membrane with the protoplasmic portion of the cell projecting up over the capillary, like a non-nucleated plate.
Adjacent Lobuli respiratorii impinge on each other, pressing the loose connective tissue, which has hitherto existed between the lobules into a thin membrane in which the capillaries run. This interalveolar membrane now consists of the membrana propria of the adjacent lobules, together with the interalveolar connective tissue. The lymphatics in the various parts of the lung still show essentially the same relationships.
After birth (fig. 29) the development of the lung has advanced along the same lines followed in embryonic life. The chief changes occur in the respiratory lobules. The bronchiolus (fig. 29 b) is clothed by cubical epithelium surrounded by a well-marked basement membrane about which are numerous connective-tissue cells. There is as yet, however, no differentiation of this layer into muscle fibers. From this arise the short Bronchioli respiratorii (lhg. 29 221') where the cubical epithelium flattens as the passages run into the Ductuli alveolares (fig. 29¢).
TEXT fiG. 29. Sections of a portion of the lobule of the lung of a. pig, two day old. Same preparation as used with tissue shown in fig. 28. X 130.
Z: lymphatics. b : bronchiolus. D1“ = bronchiolus respiratorius. i : ductulus alveolaris. a—_-atria. sazsacculi alveolares. czalveoli pulmonaris.
From these structures, the Atria (fig. 29 a) are formed, which in turn produce the Sacculi alveolares (fig. 29 Sat). The air sacs which Were only indicated in a pig 27 cm. long are now distinctly seen. It is possible that they are even more developed before birth than is shown in fig. 28, as I have frequently found embryos in utero 29 cm. long.
Unfortunately, I have been unable to obtain good sections from specimens of this age. This makes, however, no essential difference as the whole respiratory lobule is produced before the pig is born. Following the use of the lungs for respiration, there is a dilatation of the various structures of the lobule (cf. figs. 28 and 29) which is accompanied by a still greater flattening of the connective tissue between the alveoli, yielding practically a single membrane containing the blood—vessels between the two layers of respiratory epithelium. This, however, as we have seen, ontogenetically consists of the two basement membranes and the interalveolar framework of the adjacent alveoli. The larger connective—tissue lobules still retain their general relationships, increasing in size with the growth and dilatation of the respiratory lobules of Miller. The lymphatics (fig. 29 Z) still have their regular relationships.
In a half-grown pig, one observes the thickening of the framework, which in the main septa at the root and under the pleura is now made up of well-formed‘trabecu1a=,, consisting of connective—tissue fibrils. The bronchi have developed peripheralwards taking on an older type, 73. e., adding muscular layers, submucous glands, and bronchial cartilages, which may be traced as far as the larger intralobular branches. From this point peripheralwards, gradually thinning, the muscle layer extends to the opening of the atria. in the Ductuli alveolares. The lymphatics in the interlobular septa are difficult to see as they are pressed together by the growth and distension of the connective—tissue lobules. No marked changes occur between this and the adult stage, save that the lobules are sometimes less apparent owing to their larger size and the fact that the septa may become thinned out in the later stages of growth.
They may be demonstrated as definite anatomical structures in the pig by thick sections stained by Mal1ory’s method or better still by complete Wood’s metal injections. When a lung has been distended for a short time with air to its maximum, Wood’s metal will pass into all the individual alveoli. After digestion, we have a cast of granular appearance which maintains absolutely the form of the lungs. This may now be broken up into the lobules, as the splitting always occurs along the septal lines and, thus, the entire connective-tissue lobular system may be revealed. It should be observed that the lobules may become compound through a failure of the septa to persist, a process similar to that which takes place in the submaxillary gland where the whole series of primitive lobes, which are first formed in the embryo and separated by well-marked septa, disappear and are indicated in the adult only by irregular septa, Without distinct relationships, passing in from the capsule. Usually, however, these lobules in the pig’s lung not only persist, but may be easily demonstrated by any of the ordinary connective-tissue stains, Recapitulation of Orgamgenes-1L9.—In recapitulating the growth of the main structures of the lungs, we have stem and main bronchi originating in the primitive lung sacs as an epithelial tube with a double layer of epithelium, the inner of which is columnar, while the outer is composed of smaller polygonal cells. This simple tube is surrounded by a membrana propria formed by a deposit of fibrils from the exoplasm of the connective-tissue syncytium. As the bronchi grow, a layer of spindle cells difierentiates from the rnesoderm, which is transformed into the muscular coat of the bronchi. Later still, a chondrification of the perimuscular syncytium takes place from which the cartillaginous rings of the trachea and the bronchial cartilages are formed.
With these changes the connective-tissue fibrils become grouped into trabeculae about the bronchi and in the submucosa. Later, the mucosa is thrown into a series of longitudinal folds, while from the cuticular border of the inner row of cells, cilia develop. From the bottom of the crypt-like invaginations formed by the longitudinal folds of epithelium, glands begin to grow down into the submucosa, which sometimes pass between the developing muscle bundles into the deeper layers of this coat. As this process takes place, there is a differentiation of some of the epithelium into goblet cells, a process which also takes place in the glands, giving rise to a series of submucous glands with partly serous and partly mucous cells. While these changes are taking place in the mucosa, the cartilages are also growing, and with them, a further differentiation of the framework into distinct fibrous trabeeulaa. As we follow the bronchi peripheralwards, they become simpler and essentially younger in structure and yet develop their adult characteristics in precisely the same way. The epithelium soon becomes single layered of a columnar type, and then of a distinct, flat, cubical form. The Lobuli respiratorii begin to develop in pigs about 19 cm. long by a slight dilatation of the growing ends of the bronchi. These represent the bronchioli. Later, the Bronchioli respiratorii are formed which have a progressively flattened epithelium, running over into Ductuli alveolares.
These are present at the age represented by a pig 22 cm. long. Subsequently, Atria, Sacculi alveolares, and Alveoli pulmonis form in the prenatal period, all of which have the characteristic flattened respiratory epithelium. After birth there is a dilatation of the lobules and a further flattening of the epithelium occurs, and before the pig is half grown, a muscle layer develops about the air passage as far as the Atria, where it stops in sphincter-like bands.
The framework of the lung develops from a general syncytium forming the mesodermic anlagen of the two lung wings. By a gradual differentiation of connective-tissue fibrils from the exoplasmic part of the syncytium, the framework becomes denser and, finally, at 8 cm., a suggestion of lobulation is obtained about the end branches of the growing bronchi. Within the lobules the framework differentiates as the embryo grows, forming simultaneously basement membranes for the young bronchial buds. At the same time, the interlobular fibers, and those beneath the pleura, unite to produce trabeculae. As the lobulii respiratorii towards the end of foetal life begin to impinge on each other, the interalveolar framework and the two adjacent basement membranes are pressed together into a single wall or septum in which the blood-vessels run. These lobules remain until adult life, and correspond in the pig apparently to those described by Laguesse and d’Hardiviller, 98, and Councilman, or, in the human lung. Noteworthy, however, is the fact that they may become compound by the loss of the interlobular septa and the subsequent confluence of several adjacent lobules. This usually takes place at the base leaving the periphery of the compound lobule separated by partial septa.
The lymphatics appear at the root of the lung in an embryo 4-5 cm. in length. Accompanying the bronchi and vessels, they gradually grow in for some distance and until the smaller air passages are reached, they leave these structures and grow towards the pleura in the inter spaces between the smaller bronchi, aiding in the differentiation of the connective—tissue lobules. The reason for this course is not entirely clear, , but it may be due to the increasing density of the framework about the bronchi, which forces the later-appearing lymphatics into the interlobular spaces as a Locus minoris resistentiw. Upon reaching the pleura, they turn and form a plexus in the subpleural connective tissue. Here and there they may be seen penetrating into the lobules, but cannot be followed for. any distance in them. At 23 cm. the first evidence of the submucous lymphatic plexus is seen in the stem bronchi. It may, however, be found earlier, but the vessels are difficult to follow in uninjected specimens.
It would seem, thus, that we have in the pigs lung, besides the lymphatic plexuses accompanying the bronchi, arteries and veins, an interlobular system which Miller has been unable to find in the human lung.
Injections pointing to such a relationship he has interpreted as artefacts. If ,Miller’s conclusions prove to be correct, then the lymphatics of the human lung must develop so far as the interlobular septa are concerned in some other way.
In following the organogenesis of the lungs in the pig, one finds at no period in their life history, openings, or fenestrae, which suggest a communication between adjacent respiratory units. They form, as we have seen, independently at the growing ends of the tree and as they approximate each other, it is always possible to demonstrate the interlobular or interalveolar framework Without interruptions suggestive of fenestraa offering a communication between adjacent alveoli. Furthermore, in all my corrosions, many of which are complete enough to fill completely the Alveoli pulmonis and maintain the entire form of the lungs, no instance was found of an interalveolar communication. Ruptures frequently occur forming irregular extravasations, but in the most complete injections, one is always able to isolate completely the individual Lobuli respiratorii. The results of this paper, then, support the conclusions of Miller, Laguesse, and Oppel, and are not in accord with the views of Hansemann, Zimmermann, Merkel, and Schulze with reference to the presence of these foramina in the walls of the alveoli of the mammalian lung.
Discussion of the Literature
The Anlage of the Lungs
As in the case of the early stages of the amphibian and reptilian lung, there is a general agreement among most authors who have worked upon the mammalian lung that the respiratory apparatus arises from an unpaired anlage, which the majority regard as asymmetrical. Of these investigators, His thinks the future asymmetry of the lungs is to be sought in this characteristic of the anlage, while Minot looks upon the asymmetry of both anlage and lungs as secondary to changes taking place at this time in the heart. Fol believes the anlage is paired and regards it, moreover, like Gotte and Weber and Buvignier as associated with the gill pouches. The anlage, in the pig, arises from the ventral portion of the head gut as a ventral groove with a more marked projection at the caudal extremity, which becomes separated from the dorsal segment of the gut by two longitudinal fissures, along the line of which the final separation occurs. The upper part of the anlage gives rise to the trachea, the lower to the lungs. If the pulmonary apparatus in mammals should finally be shown to have a serial relationship with the gill pouches, all trace of the process is certainly lost in the pig. From the first, the anlage is asymmetrical. Whether this is a characteristic of the respiratory apparatus or is due, as Minot suggests, to the influence of the heart, it is impossible, from my material, to say. Suggestive, however, is the fact that the pulmonary anlage in many of the lower animals is symmetrical.
The Growth of the Bbonchial Tree
Few of the many characteristics of the bronchial tree have given rise to more discussion than the method of its growth. Between the two extremes of dichotomy and monopody, most of the possible intermediate processes have been described. A special review of the literature on this point seems desirable to see what harmony can be drawn from the difierent observations. So far as possible when space permits, the process will be described in the words of the various contributors to this field.
If we recapitulate the history of the several series of bronchi it may be said that all of the chief bronchi are produced in the same manner, that is to say by monopodial growth. Even the formation of the stem bronchi from the pulmonary anlage does not differ in any material way from the subsequent formation of the products of the stems themselves. As the tree grows, there is no definite division of the end bud as the main branches are outgrowths of the walls of the trachea or the two stem bronchi. In the pig, the trachea produces only a single element, namely, Lateral 1. The process of growth is successive, that is to say, the elements are produced one after another from above downwards, recapitulating the manner of growth shown in simpler animals like the reptiles, for example. When a new element is about to be formed, one notes an increase in the number of karyokinetic figures in the epithelium in the region of the new branch. The basement membrane becomes much less distinct and the connective-tissue nuclei in the surrounding mesoderm are more closely packed together. In this region, a slight bulging of the epithelial wall is then noted, as is shown, for example, in fig. 12, which increases in size until a small elevation is raised on the surface of the stem. This subsequently grows, yielding a rounded projection on the stem, which gradually emancipates itself and gives rise to a new bronchus.
The process is essentially the same Whether it occurs either in the neighborhood of the terminal bud, higher up on the stem, or on the trachea.
In general, we may say that the lateral and medial bronchi are produced nearer the terminal end of the main bronchus, while the dorsal and ventral elements are produced somewhat higher up from the stern, often where the latter has regained its cylindrical form.
If Narath’s interpretation of the bud as reaching up to the last apparent lateral branch is allowed to stand, then all of the branches except the tracheal bronchus must be considered in the sense of Narath as lateral productions of the end bud. Narath’s distinction, however, does not seem to be well made for, in the pig’s lung at least between the last lateral branch and the tip of the stem bronchus, there is always a considerable portion of the main stem which has a definite cylindrical form and terminates in a distinct dilatation at the end. Much as Narath’s view would tend to simplify the question, there is little justification, therefore, in looking upon the entire distal part of the stem bronchus as the terminal bud. On the other hand, there is no essential difference in an evagination taking place at the bud and in one taking place on the stem.
It may be well to notice certain differences in the behavior of the stem at difierent periods in the life of the organism as well as difierences between different species. For example, in the pig, the stems seem relatively more irregular and dilated in size in embryos between 10 and 13 mm. long, but on the whole are fairly cylindrical throughout the growing period. On the other hand, in some species the stems, particularly at the growing ends, are quite irregular in shape and may be considerably dilated, suggesting somewhat pictures corresponding to the growing lungs of reptiles.
After the formation of the chief branches has occurred, the primitive monopodial system may persist for a few generations on the side branches.
The principal method of division is, however, by dichotomy equal and unequal. Apparently the selection of the method depends somewhat on the physical conditions of the space in which the bronchi are forced to divide. In the case of the first divisions of Lateral 1, of Lateral 2 on each side, and Ventral 2 on the right side, the division is of practically equal dichotomy, as they have a relatively free space about them. When, however, the direction is more or less controlled by the limited environment of the bronchi, it becomes unequal, one fork growing on so rapidly to become the stem, that the other is left either as a small bud or a small side branch, which develops further when the space relations permit.
Later still when the total volume of the lung is such that each bronchus is more or less equally surrounded by mesoderm, the dichotomy is equal, although of the two forks resulting from a division, one becomes the stem and the other is shunted off as a side branch. The point, however, where monopody ceases and dichotomy begins is apparently diiferent in different species and may be different in different parts of the lung. In the pig it is below Lateral 6 while, in man, according to His, the transfer is made at Lateral 4. It must be remembered in this connection, however, that the space relations in this region of the human lung are quite difierent from those in the pig owing to the different position of the heart, diaphragm, and liver.
The bronchi, apparently, show great adaptability both in the power and direction of their growth. This interesting characteristic is best shown when one of the chief bronchi are suppressed. Adjacent branches, while still rooted firmly at their point of origin, then grow into the area of the lung usually supplied by the suppressed element, a process which, taken in connection with the extreme variation of the point of origin of the bronchi, give rise, in the adult tree, to the series of pictures which suggest a wandering of the branches. In my whole series of specimens numbering ten reconstructions and many cleared specimens 3 to 18.5 mm., and about 100 corrosions of pigs from 4 cm. to the half-grown stage, I have never found any evidence which pointed to a wandering of any elements of the tree. The bronchi remain attached to their stems where they are formed, although their branching is controlled to a great extent by the space in which they have to grow. When this is altered by the suppression of one of the usual elements, adjacent branches show a power of substitution which is perhaps best exemplified in the fate of the two dorsal forks of the first division of the right and left ‘Lateral 2.
On the right side, this branch, owing to the presence of the Lateral 1 above it, is forced to grow downwards and posterior to form a dorsoinferior branch of Lateral 2, while on the left side, this same fork, unobstructed by the absence of Lateral 1, grows upwards to substitute for the suppression of the lateral element above.
In turning to the literature we find that between such outspoken descriptions as those [of d’Hardiviller for example, on the one hand, and Justesen, on the other, it is not difiicult to diflerentiate, but in the cases where terms like sympodial dichotomy and monopody with acropetal development of the lateral buds are used, it is not always easy to determine whether the authors have not been describing the same process with different words. At the outset, therefore, it may be well to state that by monopody we understand lateral outgrowths from the wall of the bronchus whether they occur on the side of or above the terminal bud, and by dichotomy we understand an undoubted division of the terminal bud. In equal dichotomy the two divisions grow for a time equally but later may give rise to a system of monopodial appearance by the selection of one branch to continue as the stem, while in unequal dichotomy the two buds develop unequally from the first. In the case of dichotomous divisions, however, it is obvious the portion of the stem between two side branches is genetically equivalent to the side branch of the lower order.
Since one can explain theoretically the entire bronchial tree equally well by either a monopodial or a dichotomous process of growth, it is not surprising to find different views among those who have studied only the finished bronchial system. This is well shown among modern investigators in the work of Aeby, 80, and Ewart, 89, the former of whom believed in monopodial growth from first to last, while the latter says “ Dichotomy is the alpha and omega of bronchial division.” Huntington, 98, also in working upon comparative material of adult stages finds a double system primarily dichotomous with a subsequent monopodial type of branching in the development of the stem bronchus. In a system thus capable of two explanations, obviously, the only observations which will really aid in solving the question come from those who have studied the lungs during the process of their growth.
If we turn to this series of investigations we find Kiittner, 76, stating that “Das Wachsen ist monopodisch, d. h. das Epithelrohr wachst an seinem Scheitel ungetheilt fort, wahrend seitliche Sprossen am Stamm desselben hervortreten und mit ihrer Langsaxe zu der des erzeugenden Rohres rechtwinkelig gestellt sind.” Furthermore, he states that these buds grow and divide rapidly, giving rise to so many more lateral branches than the principal axis that it is difficult in the adult tree to recognize its primitive monopodial character.
Cadiat, 77, describes the process as follows, and it is important to remember he is speaking of solid buds: “ Il est facile de comprendre maintenant comment se produisent les ramifications bronchiques. Un premier bourgeon se forme plein et se développe en longueur, l’ampoule se produit a l’eXtrémité. Alors son evolution est arrétée; sur les parois naissent des bourgeons secondaires qui se terminent de meme, et ainsi les canaux bronchiques vont sans cesse en se multipliant, mais toujours dans des directions différentes.” Stieda, 78, states: “Zuerst ist der Canal einfach, dann theilt er sich in Aeste, welche sich abermals theilen, so dass sowohl durch fortgesetzte Theilung des auch durch seitliche Sprossenbildung im epithelialen anfangs noch leicht iibersehbaren Canalsystem ensteht, dessen blinde Enden etwas leicht erweitert sind.” Ktilliker, 79, describing a 12-day rabbit embryo, says: “Das innere Epithelialrohr, das nun Bronchus heissen kann, hat in jeder Lunge drei Ausbuchtungen und Werden Von nun an mit dem Grosserwerden des Organes die Verastelungen bald so zahlreich, dass dieselben nur schwer Schritt fiir Schritt zu verfolgen sind.” Further, in speaking of the increase of the bronchi in man and animals, he says in general: “ Das innere Epithelialrohr hohle Aussackungenen oder Knospen erzeugt, welche, rasch sich vermehrend, bald in jeder Lunge ein ganzes Baumchen von hohlen Kanalen mit kolbig ansgeschwollenen Enden erzeugen, von welchen aus dann durch Bildung immer neuer und zahlreicher hohler Knospen endlich das ganze respiratorische Hohlensystem geliefert wird.” His, 87, in working on the development of the human lung, describes the process of growth as follows: The first branches as far as Lateral 4 arise by rnonopodial division, which he describes in the following terms: “An keiner Stelle findet sich eine Andeutung, als ob aus den einmal cylindrisch gewordenen Wurzelrohren Seitensprossen zu entstehen vermochten. Die einzige Productionstiitte neuer Formbestandtheile sind die Endknospen, und zwar erfolgt die Umgestaltung auf dem Wege dichotomischer Theilung. Die Knopsen verlieren ihre kugelige Grundform, indem sie an der Anheftung gegeniiberliegenden Seite sich abplatten "und zugleich in transversalem Sinne strecken. Bald tritt eine trennende Furche auf, Wodurch die urspriingliche einfache Knospe in zwei getrennte Verwolbungen auseinander geht. Allmahlich emanzipiren sich diese letzteren und bekommen auch ihrerseits cylindrische Stiele, woraufhin derselbe vorgang Von Neuem Platz greifen kann.” In summarizing the process he continues: “ Nach erfolgter Trennung der beiderseitigen Anlagen bildet eine jede derselben einen gebogenen und zugleich bimformig ausgeweiterten Schlauch, mit einzelnen scharfer markirten Vor treibungen. Aus diesen treten die primiiren Seitensprossen als monopodische Bildungen im Sinne von Aeby hervor und ihre fiir beide Seiten asymmetrische Anlage bestimmt auch die Differenzen spiiterer Ausbildung. Der weitere Verzweigungsmodus bleibt nun wahrend geraumer Zeit der dichotomische. Zuletzt tritt aber ein Zeitpunkt ein, W0 die Endknospen aufhoren sich dichotomisch zu theilen und W0 sie wieder in ein System mehr oder minder ausgiebiger Seitenknospen auslaufen.” In mouse, mole, and pig, Willach, 88, describes the process as follows: “ Ich glaube vielmehr, dass beim Menchen, wie bei den Saugethieren, die Sprossung eine sogen monopodische ist, welche darauf beruht, dass das Mutterrohr vor seinem kugeligen Endblaschen eine Verengerung seines Lumens erfahrt, wahrend das Lumen des Endblaschens sich erweitert und seitliche Ausbuchtungen treibt, jene Knospen, die wieder zu Rohren werden, und das Mutterrohr Weiter fortwachst. Das Tochterrohr ist enger als das Mutterohr.” The growth process is described by Robinson, 89, in these words: “ In the rat and the mouse, the ramification of the bronchi is produced principally by dichotomy. The germ of each bronchus, as it grows outwards and dorsally, becomes expanded at its termination; this expansion is gradually constricted into two portions of unequal size, that is the dichotomy is in the form described by botanists as unequal or sympodial.” Further he states: “Although most of the branches are produced by dichotomous division of terminal expansion, certain of the dorsal branches arise as hollow buds from the wall of the stem bronchus after it has assumed its cylindrical form, and these buds are interpolated between preexistent branches.” He describes the origin of our median bronchi in the rat as follows: “ The second dorsal branch immediately after its origin is similarly divided, and the constriction passes rapidly towards the axial stem, until its apex reaches the level of the circumference of the main bronchus. Thus, from the dorsal bud, a. dorso-internal (median) branch is formed.” Robinson apparently does not believe that the branches are successive in their formation.
Minot, 92, states that “ the branching occurs in a highly characteristic manner, for the stem always forks, but the forks develop unequally, one (terminal bud) growing more rapidly and becoming practically the continuation of the main stem, while the other (lateral bud) appears as a lateral branch. Speaking in general it may be said that the ventral fork serves as the stem. In consequence of this method of growth the adult lung consists of main stems with lateral branches. . . . But it is erroneous to suppose, as did Aeby, that the system of growth is strictly monopodial, it being in reality a modified dichotomous system. The branches all arise by terminal forking, never as outgrowths from the side of a stem.” d’Hardiviller, from his studies on the rabbit and sheep, announces the following law of development: “ Toutes les bronches primaires, principales ou accessoires, naissent en divers points des bronches souches par ramification collatérale, le bourgeon terminal des bronches souches ne prenant aucune part a leur formation.” These principal branches then, according to d’Hardiviller, give rise to secondary branches by the production of lateral buds as well as by equal and unequal dichotomy. d’Hardiviller does not believe that all branches of the stem are successive in their formation.
Nicholas and Dimitrova, 97, in the sheep, describe the growth of the main bronchi as lateral buds which appear successively on the terminal portion of the stem bronchus.
The results of J ustesen, 00, contained in an extensive paper devoted entirely to the method of growth of the bronchial tree, may be given in one sentence, “Die Bronchialverzweigung ist also eine dichotomische,” in which process he would include all branches of the tree from first to last.
The process of growth of the bronchial tree according to Narath, 92, 96, or, is a rather complicated process. He looks upon the primitive lung sac as the first production of a stem bud. When a side branch is produced from the end bud a slight swelling is observed on its lateral side, emphasized by the occurrence of mitosis in this region. In consequence of the greater pressure at this point, the end bud bends slightly in the opposite direction, that is to say, medialwards. As the new bud grows, this process continues until there is a distinct kink in the axis of the stem opposite the new element. As it increases in size, the side bud takes first, the form of a cone—like projection with a rounded summit, as the stem bud grows on, then the epithelial wall about its base sinks somewhat towards the axis of the stem, until the daughter bud is isolated from the stem and then grows on. It is important to note, furthermore, that Narath considers the end bud the entire terminal part of the stem up to the last Well-formed lateral branch.
In reference to the origin of the dorsal bronchi, Narath states from his observations on the rabbit, that they are produced Without participation of the stem bud and that they appear later than the corresponding lateral bronchi. Furthermore, the comparative anatomy of the tree suggests to him that the dorsal series are primarily side branches of the lateral bronchi which, in course of ontogeny or phylogeny are placed back on the stem. In support of this view, he finds the dorsal buds arising at the same level as the lateral and, apparently, in communication with the contour of the latter. Then, he continues, if lateral bronchi are able to give up dorsal branches to the stem, this process repeats itself with the latter series in giving rise to the median bronchi. While he is not absolutely certain that this process takes place in the origin of the dorsal elements, he states that it can be proved with certainty in the formation of the medial series. He shows a schematic series of drawings of the median branches of D. 2, D. 3, and D. 4 in their different stages, giving an apparent transplantation of this median branch upon the stem bronchus. Like the median series, Narath also believes that the ventral bronchi (the Ventro-accessory of Aeby) are branches which are given up from the lateral branches to the stem. In one rabbit embryo Naratli was able to Show a relationship between Ventral 1, the infracardiac bronchus, and Lateral 1. He says further: “Der Zusammenhang der Knospen ist ein primares Verhaltnis und kein sekundares.
Und wenn weiter eingewendet werden sollte, die Knospen hangen deswegen so innig zusammen, Wei bei der erwachsenen Lunge die Bronehien so enge beisammenstehen, so wiirde ich auch wiederum gerade diesen Befund bei der erwachsenen Lunge als fur die Aeby’sche Ansicht sprechend verwerthen.” In a word, while not absolutely pledging himself to this view, N arath believes that there are but one primary set of bronchi, namely the lateral, and that the other three series, the dorsal, ventral, and medial originate either directly from these branches as in the case of the dorsal and ventral groups, or the median branches of the dorsal series as in the case of the median bronchi, and are then given up on to the stem bronchus.
Moser, 02, says for the vertebrate lung in general that “ Das Verzweigungssystem der Kanale innerhalb der Lunge ist stets und ausschliesslich ein monopodiales.” It must be remembered, however, that Moser’s material on the mammalian lung was very limited and confined to older embryos which were studied by means of sections instead of corrosions and reconstructions. Some criticism might be made of her comparative material especially in view of the more exact methods used by Hesser in the same field.
Blisnianskaja, 05, in the human lung states that “Die Bronchialverzweigung geschieht nacht dem dichotomischen Typus, der durch ungleiches Wachstum der Gabeliistc ein monopodisches Aussehen erhalt.” Hesser, 05, in his important work on the reptilian lung states that “ausser allem Zweifel, bei niederen wie bei hoheren Reptilien die erste Aste aus dem Stammbronchus monopodial angelegt werden. Tarentola, Anguis, Chrysemys u. a. zeigen dies unzweideutig. Die Bronchien haben eine ansehnliche Lange erreicht, bevor noch Seitenaste auftreten, und wenn die erste Knospe sichtbar wird, tritt sie aus der Seite des Bronchus hervor, und zwar in einer bedeutenden Entfernung von dessen kaudalem Ende.” In speaking of the further growth of the branches, he continues.
“ Denn dadurch, dass das Langenwachstum der Aste nicht porportional zur Vermehrung der Anzahl ihrer Knospen ist, geht die Monopodie allmahlich in Dichotomie iiber. . . . Also besteht zwischen Monopodie und Dichotomie nur ein gradueller, aber kein wesentlicher Unterschied, und es wiirde daher kein Erstaunen hervorrufen diirfen, wenn in der Architektur des Bronchialbaumes sowohl die eine wie die andere Weise zur Anwendung gekommen ist.” If we attempt to tabulate these views on the growth of the bronchial tree, the results may be placed in three main divisions as follows:
- Dichotomy. Older authors, Ewart, Minot, J ustesen, Blisnianskaja.
- Monopody. Kiittner, Cadiat, Kolliker, Aeby, Nicholas’ and Dimitrova, Willach, Narath, Moser.
- Monopody and Dichotomy. Stieda, His, Robinson, Huntington, d’Hardiviller, Hesser, Flint.
It is also possible to subdivide them still further in the following way:
- Dichotomy. Older authors, Ewart, Justesen, Minot.
- Unequal Dichotomy. Robinson (?), Blisnianskaja.
- Monopody. Aeby, Moser.
- Monopody with participation of the end bud. Willach, Narath, Nicholas, and Dimitrova.
- Mixed Monopody and Dichotomy simultaneously. Stieda, Robinson.
- Monopody and Dichotomy successively. His, d’Hardiviller, Huntington, Hesser, Flint.
While we have already called attention to those who have only studied the branching from the finished tree, to which class belong Aeby, Ewart, and Huntington, there is still a group, in the series of authors given above, who have not followed the lungs through the development of the stem and its chief branches in mammals, that is to say, their material consisted of embryonic stages after the formation of the principal bronchi was complete. The observations of these investigators are only important for the specific fields in which they worked, for it goes without saying, as His has suggestively remarked, the conditions which govern the form development of a growing part need not necessarily remain the same through the different phases of its evolution. It may change its character either once or more than once.
Thus for a series of animals covering amphibia, reptilia, birds (Moser, Hesser, Schmalhausen), man (His), rats and mice (Robinson), mouse, mole (Willach), rabbit (d’Hardiviller), sheep (Nicholas and Dimitrova), rabbit, echidna, cat (Narath), pig (Flint), we have a general agreement, that the stem and its principal branches are produced by monopodial growth. I have placed Robinson in this group, partly because he believes some of the chief branches are monopodial in nature, but largely because, notwithstanding his own use of the term “ sympodial dichotomy,” his own description of the process of division appears to me to be esentially of a monopodial character. Against these views we have the outspoken description of Minot for dichotomy, in the human lung, as well as that of Blisnianskaja. The latter does not describe the process in detail and her illustrations appear to me to be capable of a monopodial interpretation, especially in View of the careful work of His on the same material. It is also noteworthy that she quotes the statements of J ustesen in supporting her ideas on the sympodial development of the chief divisions of the stem. It may be recalled, however, that this author did not possess in his material stages which showed the development of these particular branches.
While it is possible to draw much harmony from the verbal descrip tions of the process of division which I have given above, there are, of course, many exceptions and different complexions to these views. Since, in my opinion, it makes little difierence whether the monopodial outgrowths take place from the end bud or from the stem a little higher up, we may justifiably say that among those who have studied the production of the chief bronchi of the vertebrate lung, the following stand for an absolute monopodial system: Moser, Hesser, Schmalhausen, His, Wilach, Robinson (?), d’Hardiviller, Nicholas and Dimitrova, Narath, and Flint. This series includes obviously all who have worked on the development of the lung during this period except Minot, Blisnianskaja, and Robinson, whom I have placed in both lists. Of these authors, Willach, Narath, Minot, and Blisnianskaja believe that our Lateral 1, the so—called “Eparterial or tracheal bronchus,” is a derivation of our Lateral 2, which wanders up on the stem bronchus or trachea, the others look upon it as an independent and unpaired element. Narath and Blisnianskaja regard the other chief bronchi as secondary derivatives of the lateral group as “ accessory ” in the sense of Aeby. Willach believes the ventral and median groups as accessory, that is to say, derived from the lateral and dorsal bronchi respectively, while Robinson thinks the chief bronchus of the ventral series, Ventral 2 (the Bronchus infracardiacas) is ontogenetically independent, but phylogenetically accessory.
The latter describes the origin of the medial bronchi, his dorsointernal group, from the dorsal by a process of progressive splitting of the first medial branch of the dorsal bronchi until it comes to have an independent origin on the stem, a view which is advanced in greater detail by Narath.
All of the arguments of N arath and Blisnianskaja concerning the derivation of the ventral, dorsal, and medial series either primarily or secondarily from the lateral bronchi are quite unconvincing, for like the support, which Narath brings from the comparative anatomy, the facts are capable of a simpler explanation, 13. e., a wide variation in the position of the buds and the power of one bronchus substituting for another. These two factors which I have followed in detail in the pig’s lung, will explain all of the conditions in the adult tree which led first Aeby and then Narath and their followers to look upon the ventral and medial groups as derivatives of the lateral series. It may also be Well to call attention to Hesser’s pointed criticism of Narath’s view when he remarks that the lateral buds of Narath when they have only reached the development of a low round cone with a broad base, represent the anlagen of four different branches, namely the dorsal, lateral, ventral, and medial bronchi which must isolate themselves and take their places on the stem. And lastly, we cannot help noting the lack of the one convincing argument which should come from comparative anatomy consisting in a primitive lung that possessed only lateral bronchi.
Furthermore, the series of schematic figures, which Narath gives to show the origin of the medial from the dorsal bronchi are objectively correct and agree with the conditions found in the pigs lung not only in the embryonic, stages but in the adult tree as well. He finds the first median division of the dorsal bronchi as one descends from D. 2 to D. 5, is placed successively nearer the stem bronchus until, at the latter point buds are seen on the dorsal and medial sides of the stem. He interprets this condition as indicating a wandering of this median branch to the stem. As a matter of fact, however, this is the normal relationship for the grown lung, and, as I have pointed out above, the medial series do not occur higher than Lateral 4. It is scarcely justifiable, therefore, to interpret the successive change in the insertion of this median branch, together with the appearance of the medial buds in their usual position as evidence of wandering on the part of the median bronchi.
In reference to the further division of the tree after the principal branches are laid down, Moser, Willach, Narath, Cadiat, Kiittner, and Kolliker believe in a monopodial propagation, while His, Minot, d’Hardi-.
viller, Hesser, and Flint believe in the dichotomous form either equal, unequal, or both.
Aeby’s Eparterial and Hyparterial Theory
The substance of Aeby’s views with reference to the influence of the pulmonary artery upon the bronchial tree has been given in the abstract of his monograph. This theory, which has influenced, more or less, the work of all subsequent investigators has been accorded a varied reception.
His, Willaeh, Robinson, d’Hardiviller, and Miller, either actively or passively, support the views of Aeby, while Ewart, Zumstein, Narath, Minot, Huntington, Justesen, and Merkel have abandoned them. In some cases it is difficult to ascertain just what position an author takes concerning the theory for some of them use indiscriminately the terms hyparterial and eparterial in describing the tree. These terms, of course, may have only a simple topographical significance, as in the case with Huntington, without implying the meaning which Aeby attaches to them. Of all the authors who are considered as supporting Aeby’s theory His, alone, is outspoken in his belief that the eparterial bronchus is a dorsoventral bronchus which if it were in the hyparterial region would divide into dorsal and ventral branches. Willach, who first describes the eparterial branch as arising from the first ventral bronchus, apparently accepts the theory, although Narath, a few years later advocating the same view, states that this single fact is sufficient to disprove Aeby’s hypothesis once and for all. Zumstein attacked the theory from another point of view, namely, by failing to find in corrosion specimens the relationship, which Aeby describes, and by noting variations in the pulmonary artery which, apparently, had no influence on the architecture of the tree. In these observations Zumstein is supported by Narath, who also describes such specimens. Both observers also call attention to the fact that, at the time the primitive bronchi are formed, the pulmonary artery is a fine, delicate vessel which would have no influence on the larger, firmer epithelial structures. Huntington attacks the theory from another point of view in looking upon the wandering of bronchi as the chief factor in the formation of the eparterial bronchi to which the relationship of the artery is simply secondary and topographical.
From the results recorded in this paper, it would appear that the relationship of the arteries to the tree and the differentiation of two sets of bronchi with diiferent relationships to the pulmonary arteries are primarily due to the topography of the anlage with respect to the Vena pulmonalis and the projection of the anlage ventralwards from the head gut.
In consequence, the arteries form behind the primitive stems before any of the side branches are produced. Later the first lateral bronchus develops above and behind the artery, while the remainder of the series are formed below and in front of it. As the heart descends, the topography of the arteries to the stems changes, but in no way and at no time have the arteries a fundamental influence in differentiating two segments of the tree. On account of the association of this influence with the terms “ eparterial ” and “ hyparterial ” it is, perhaps, well to abandon them as Zumstein and Narath have suggested. However, this much is certain: The theory ought not to be abandoned without an acknowledgment of our indebtedness to it. That the theory would stand or fall from the results of embryological research, Aeby clearly recognized, much more clearly apparently than some of his critics. As a working hypothesis, his view was generally accepted from the time of its publication until the appearance of Narath’s paper.
1s'r LATERAL BRONCHUS. “ EPARTERIAL ” BRONCHUS or AEBY.
This, Aeby regards, as a dorsoventral bronchus which lies above the pulmonary artery and, therefore, not under its influence. If it were in the hyparterial region the artery would divide it into dorsal and ventral bronchi, a View in which Aeby is supported by His. It is an independent structure; it may be either paired or suppressed. These characteristics form the basis of Aeby’s classification of the mammalian lungs. Willach first proposed the idea that this was a branch of the‘1st ventral bronchus, while Robinson, like His, believes it is an unpaired and independent branch. Zumstein in abandoning the eparterial theory terms this the first lateral bronchus. Narath uses the expression Apical bronchus and takes the same View as Willach inasmuch as he considers it a branch of the 1st ventral bronchus. The former, however, goes further in regarding this element as a definite dorsal bronchus. This is compatible with his tentative view of the whole series of dorsal bronchi arising probably primarily from the ventral group. Minot supports Willach, while d’Hardiviller thinks it is an independent element arising from the trachea in sheep and the stem bronchus in rabbits in which view he is upheld by Nicholas and Dimitrova so far as his observations in the sheep are concerned. Justesen, Merkel, and Blisnianskaja follow Willach. The unique and remarkable observation of d’Hardiviller, who states that in the rabbit there exists primitively an eparterial bronchus on each side, is the only suggestive evidence of the degeneration of an eparterial bronchus taking place during the ontogeny of the embryo. For a time each develops symmetrically and then later the left atrophies and disappears. Upon this observation d’Hardiviller concludes that Aeby, His, Robinson, Narath, Nicholas, and Dimitrova are mistaken in stating no bronchus arises at this level on the left side, and, believes in consequence, Aeby’s classification of mammalian lungs is only of secondary value. In certain species they may both develop, in others the left only may atrophy, while in still others both may undergo the atrophic changes leaving the tree consisting only of a symmetrical hyparterial system. This observation of d’Hardiviller has only received a single supporting observation in the whole literature and that is by Bremer in the opossum lung. Bremer finds in embryos of 12.5 mm. what he calls an eparterial bronchus on the left side. In his specimens, 14 cm. long, this is absent and, therefore, he presumes the bronchus has degenerated between the two stages he has been able to observe. Narath, in the possession of two adult rabbit lungs with left eparterial bronchi as variations, is inclined to believe d’Hardiviller is dealing with an abnormality, and, furthermore, in view of the unique nature of the observation, adds that absolutely indisputable histological preparations must be produced to show the degeneration of a bronchial bud which has once been formed. This criticism of Narath would, in part, apply to Bremer’s observation. The production of the bronchial tree in Echidna, according to Narath, follows the same principles which we observe in other mammals and the lung of the adult is not differentiated from that of placentalia. Moreover, the vessels and their relationships undergo no further changes while the young are in the pouch either in respect to the artery or the veins. It is thus hardly possible in these observations of d’Hardivi1ler and Bremer that we are dealing with a true regressive process. In fact, it is more probable that in both cases we are either dealing with a variation or a dorsal bronchus which is placed higher up than usual upon the stem bronchus. This assumption is made quite probable by Bremer’s statement that his left eparterial bronchus did not supply the apex of the lung.
This bronchus is undoubtedly one of the lateral series as Zumstein and Nicholas and Dimitrova hold. It, like the remainder of the lateral series, originates from the lateral wall of the trachea or the stem. The fact that it is usually unpaired and has a difierent topography to the pulmonary artery does not separate it from this group. It is true, the bronchus originates a little more dorsalwards than the remainder of the series, but this is due partly to the different space relationships in the upper part of the thorax and partly, to the ventral torsion of the lower lateral bronchi, which exaggerates the slight difference that occurs between Lateral 1 and the remainder of the series in the embryo.
Inasmuch as a bronchus corresponding to Lateral 1 has never been described in Reptilia or Amphibia, it must be regarded as peculiar to mammals. The great rarity in the occurrence of paired first lateral bronchi suggests that no more morphological significance can be laid on its presence on both sides than its absence. The unpaired Lateral 1 on the right side must be regarded as the normal condition for mammalia, due to a phylogenetic provision for the descent of the heart and great vessels through the suppression of the element on the left side. In cases where it is formed bilaterally, no instance of a left Lateral 1 on the trachea has yet been described. As Narath shows, it is always somewhat lower on the left side than the right when the element is bilaterally present. From Narath’s tables, the bronchus is unpaired on the right side in 199 species, is bilateral in 15 species, and is absent on both sides regularly in 3 species. These three types, apparently, obey no definite law; in the same order of animals, all three types may be found in nearly related species.
In some instances, Lateral 1 arises from the trachea, in others from the stem bronchus. When, however, we observe the conditions in those animals where it is formed on the trachea, we find the bifurcation occurs near the second pair of lateral bronchi. On the other hand, where Lateral 1 is produced on the stem, the division of the trachea takes place high up, throwing it on to the main bronchus. Its dorsal character, in which Narath believes, is, however, secondary, as its lower branches are forced backwards by the presence of L. 2 below it and the relatively free. space beside the vertebral column just above the dorsal bronchi.
Apical Bronchus Of Willach and Narath
So general is the acceptance of the view that Narath is the author of this idea, it may be well to quote his own words in which he gives the credit to Willach: “Ich bin ganz der Meinung Willach’s, dass der apicale Bronchus ursprunglich ein Seitenast des 1. ventralbronchus sei, der auf den Stammbronchus geriickt ist.” Willach explains himself thus: “Man konnte also den Von Aeby als eparteriell bezeichneten Bronchus als Nebenbronchus zum ersten Ventralen derselben Seite im Sinne Aeby’s auifassen, der, wenn bronchial, an den Stammbronchus, wenn tracheal, an die Trachea abgegeben worden ist.” Further, he says: “Andrerseits diirfte aber der erste ventral Seitenbronchus der linken Seiten dem der rechten plus dem eparteriellen Bronchus entsprechen. Der erste linke ventralbronchus zeigt namlich einen nach vorwarts strebenden Ast, der in seiner Gestalt nicht allein Aenlichkeit aufweist mit dem eparteriellen Bronchus bei verschiedenen Thieren; sondern er ist auch geradezu in einem eparteriellen Gebiet gelegen, wenn man von einem ahnlichen, aber doch etwas veranderten Gesiclitspunkte aus, als es Aeby gethan, zwischen dem eparteriellen und hyparteriellen Bronchialgebiet unterscheidet.” Aeby looked upon this apical branch of the 1st lateral on the left side as a simple side branch, which extends up into the apex of the lung having a certain outward similarity to Lateral 1, which might, he pointedly remarks, lead to erroneous assumptions. This branch was named by His, the Bronchus ascendens, an element, which substitutes in the left lung for the unpaired eparterial bronchus in the right, a view in which he is supported by Robinson. N arath and Willach, on the other hand, as stated above, look upon it as the equivalent of the eparterial bronchus, a homology which is affirmed by Minot, Huntington, Merkel, and Blisnianskaja, but d’Hardiviller, and Nicholas and Dimitrova accept the conclusions of Aeby, His, and Robinson. That is to say, d’Hardiviller accepts them in so far as they regard the left apical bronchus of Narath, a true side branch of the 2d lateral trunk and not the equivalent of the eparterial bronchus on the right side.
In following, step by step, the appearance of the secondary divisions of Lateral 2, in the pig, we find on the right side the dorsal fork is turned downwards and outwards owing to the presence of Lateral 1 above it, in consequence of which, it becomes the large dorsoinferior branch of L. 2. On the right side, however, this unobstructed branch extends upwards toward the apex of the lung and substitutes, as Aeby and His pointed out, for the suppression of left L. 1. It is, however, a true side branch of Lateral 2 and is not to be regarded as the homologue of right Lateral 1, which in the vast majority of cases is unpaired.
Lateral Bronchi
Kolliker, who worked on the rabbit, agrees with the observations of Remak on the chick in finding the first branches of the stem bronchus growing lateralwards and dorsalwards. He did not, however, give the lateral group a special name. Aeby, whose observations were made upon full—grown material, designated them ventral bronchi in contradistinction to the dorsal group, both of which arise in the hyparterial region from independent origins, while in the eparterial region the dorsoventral bronchus uninfluenced by the pulmonary artery has a common origin from a point on the stem bronchus or trachea midway between the origin of the dorsal and ventral bronchi in the hyparterial group. Al-though His would have preferred the term lateral bronchi, he follows the description of Aeby, while Robinson is really the first to take his term lateral bronchi from the topography of the embryonic lung. Zumstein and Nicholas and Dimitrova have accepted Robinson’s terminology, while Willach, Narath, Merkel, and Bremer have followed Aeby.
Although he believes the selection an unhappy one, Narath, like His, uses the term “ ventral ” simply because it has received general acceptance in the literature and because the bronchi run to the ventral part of the lung. All of the lateral group receive a topographical nomenclature from Ewart, while d’Hardiviller calls them “external bronchi,” and Blisnianskaja “ the ventrolateral” group. Curiously enough, these are the only branches of the entire bronchial tree which all authors unanimously agree, despite the different terminology, are wholly independent derivatives of the stem bronchus.
Owing to the topography of the origin of this series of bronchi from the lateral wall of the stem, the author has followed Robinson, Zunistein, and Nicolas and Dimitrova in their nomenclature instead of Aeby and His. This is quite logical for, as His has pointed out, all of the ventral characteristics of this group are secondary to their later growth ventralwards in the space between the diaphragm and chest wall. The spiral line formed by joining the origins of the lateral bronchi on the stem represents the extent of ventral growth of these bronchi, as the upper elements reach farther ventralwards than the lower and consequently the torsion of the stem is greatest above and gradually diminishes as the lower elements are reached. These occupy practically the lateral plane of their origin. finally, the presence of a real set of ventral bronchi in many species renders the change in the nomenclature urgent.
Dorsal Bronchi
With the exception of Ewart, d’Hardiviller, and Blisnianskaja, all authors designate this group the dorsal bronchi. d’Hardiviller calls them posterior bronchi, while the latter classifies them as a dorsolateral group. There is also a general agreement that they are independent derivations of the stem bronchus, although Narath, without absolutely pledging himself to this view, is inclined to look upon them as a group primarily derived from the lateral series. He reaches this conviction partly because he regards the “ Eparterial ” bronchus as the first dorsal bronchus and a definite dorsal, branch of Lateral 1 and partly because they bear a certain similarity to branches of the lateral group. In consequence of the shifting of his Dorsal 1 up on to the trachea or stem bronchus, Narath regards Aeby’s D. 1, D. 2, D. 3, etc., as D. 2, D. 3, and D. 4, respectively. In looking upon the dorsal group as derivatives of the lateral bronchi, Narath has the support of Blisnianskaja, who argues if the “eparterial” is a dorsolateral bronchus, it is reasonable to suppose the remainder of the series are similarly derived. Neither of these authors, however, have followed the wandering step-by-step either of the eparterial or the dorsal branches on to the stem bronchus. They are, on the contrary, independent derivatives of the stem and, like the lateral series, are to be considered as a group of principal bronchi.
Phylogenetically they are one of the most sharply differentiated groups of the stem. We have designated the dorsal series, D. 2, D. 3, D. 4, etc., to keep their numerals in harmony with that of the larger lateral bronchi, although it is clear, of course, that our D. 2 is the first element of the dorsal series.
Ventral Bronchi
Because of their extreme variability, Aeby looked upon this group as accessory bronchi, which had their origin in the lateral series and subsequently wandered to take up a position on the stem bronchus. Among this group he classifies the Bronchus cardiacus. These conclusions were obtained from the study of adult specimens, so Aeby brings no definite proof of their wandering. His does not mention them, while Willach, also without evidence, seems to accept Aeby’s view. They are, according to Robinson, a definite group of independent bronchi, which he terms ventral. N arath accepts the older view of Aeby, but like that author, his conclusions, with the exception of the infracardiac bronchus, are drawn from comparative study of corrosions of the adult lungs. Moreover, even in the case of the Bronchus cardiacus, Narath acknowledges embryology brings no direct proof of a wandering in the sense of Aeby.
d’Hardiviller clings to the expression accessory, although he regards this group, which he terms anterior bronchi as independent derivations of the stem bronchus. In the latter View he is supported by Nicholas and Dimitrova who, like Robinson, term them ventral branches of the stem.
The results obtained from the pig indicate that the ventral bronchi are independent derivatives of the stem and do not form first on the lateral series and then secondarily become transplanted on to the main bronchus.
Ventral 2, Bronchus Cardiacus
This bronchus Aeby looked upon as the most important of the ventroaccessory group. Derived primarily from the second lateral bronchus, it takes its place upon the stem bronchus between it and L. 3. In many species it supplies a separate lobe, the Lobus infracardiacus instead of being included in the Lobus inferior. In his investigations on the human lung, His, from its size, the position of its origin, and its precocious development looks upon the Bronchus cardiacus as an independent element which appears out of the regular schematic order, a view with which Willach agrees. Robinson, while accepting the ontogenetic interpretation of His, believes with Aeby in its phylogenetic derivation from the Lateral 2. In holding that it may arise either from the second lateral or the stem bronchus, Zumstein takes a combined view, that is to say, in some instances it is an accessory bronchus and in others it is an independent structure. Narath is a most decided supporter of Aeby’s doctrine, both from an embryological and a comparative point of view, but thinks L. 3 and L. 4, as well as the second lateral bronchus may give rise to this trunk, a View in which he is supported by Merkel and Blisnianskaja. d’Hardiviller and Nicholas and Dimitrova, however, look upon it as one of the principal branches of the stem bronchus. In the pig, the independence of this element is shown with great clearness where it forms the largest element of the ventral group of bronchi. Its hyperdevelopment apparently results from the increase in the respiratory surface by the utilization of the space between the heart and liver rnedialwards to the two stem bronchi for lung tissue. It is unpaired, like Lateral 1 and with that element destroys the symmetry of the tree.
Medial Bronchi
Aeby’s idea in classifying this group as dorsoaccessory, that is to say, branches originating on the dorsal bronchi and wandering on to the stem bronchus was practically the same as in the case of his ventroaccessory group, namely, their inconstancy and the existence, in a series of adult lungs, of bronchi, which looked like transition stages between the origin of a medial element on a dorsal trunk and its final position on the stem bronchus. Willach, without definite observations, supported this view, while Robinson, who calls them dorsointernal bronchi and believes them accessory, in the sense of Aeby, describes their origin by means of a splitting of the division between the two buds of a dorsal bronchus down to the main bronchus leaving the inner one of the buds with an independent origin on the stem. Zumstein speaks of them as medial and independent in which he has the support of Nicholas and Dimitrova and d"Hardiviller, although the latter designates the group as an internal series. Merkel accepts the older doctrine of Aeby.
Narath, also, believes from both embryological grounds and from comparative anatomy that these bronchi can be traced as branches of the dorsal group. A criticism of his view has already been given. In the pig, they are irregular, but independent products of the stem. As they never occur more than a short distance above L. 4, we find the reason lies in the presence of the oesophagus, which prevents the development of medial bronchi above that level.
The main results of the preceding paper may be expressed in the following:
Résuimé.
1. The anlage of the lungs in the pig is unpaired and asymmetrical.
It arises from the ventral part of the head gut behind the Sinus venosus, as a ventral outgrowth, preceded by a lateral flattening of the foregut below the gill pouches and the appearance of longitudinal furrows, which divide the fore gut into two parts, a ventral respiratory portion and a dorsal digestive segment. From the lower part of the anlage the lungs arise, from the upper the trachea. If there is a serial phylogenetic association between the pulmonary anlage and the gill pouches, as some authors maintain, the connection is lost in the pig, for the lungs originate well below the gill area and distinctly ventralwards to the series of bronchial pouches. From the caudal extremity of the pulmonary anlage, arise two lateral outgrowths, giving rise to the stem bronchi. These, like the anlage itself, are asymmetrical, the right growing lateralwards and caudalwards, while the left extends almost directly horizontal. Then the respiratory and digestive portions begin to separate, a process, which begins from the caudal end of the anlage and extends upwards along the line formed by the two longitudinal furrows, freeing the respiratory apparatus from the oesophagus. In its subsequent growth, the pulmonary anlage enlarges, the tips of the stem bronchi dilate, and begin to bend dorsalwards around the oesophagus. This results in the formation of the primitive lung sacs. At this time, the production of the bronchi begins. They are readily divided into four series from the topography of their origin, namely, lateral, dorsal, ventral, and medial.
2. The first lateral bronchus, the so-called “eparterial bronchus,” is, in the pig, unpaired and arises as a later'al outgrowth from the right side of the trachea, just above the roots of the two stem bronchi. It is distinctly lateral in origin and bears a serial relationship to the remainder of the lateral bronchi. Its position in mammals varies, sometimes it is on the stem bronchus, but it is often situated on the trachea.
This difierence can usually be explained by the point of origin of the two stem bronchi with reference to the pair designated as Lateral 2.
If the stems originate low down, then Lateral 1 is thrown on to the trachea, while if their origin is higher up, the first lateral arises from the stem bronchus. Apparently Lateral 1 is characteristic of mammals and, according to Aeby, of birds. A bronchus corresponding to it has not been found either in reptilia or amphibia. In almost all mammals it is an unpaired element. No satisfactory proof has even been brought to show a bilateral development of Lateral 1 with a subsequent degeneration of the left bronchus, notwithstanding the fact that this process has been described in two species. At no time in the life history of the pig is there a Lateral 1 formed on the left side. There is furthermore no embryological evidence to show a relationship between Lateral 1 and the dorsal series of bronchi. These characteristics are secondary and result from the antagonistic effects of the growth of Lateral 1 and Lateral 2. The latter is forced somewhat ventralwards, while the former is pressed dorsalwards, until its lower branches lie above the dorsal series of bronchi.
3. The remainder of the lateral series originate in succession from the lateral side of the stem bronchus as lateral outgrowths or hernialike expansions of the Wall of the stem bronchus near the terminal bud.
These elements in their growth outwards finally reach the chest wall.
Here they are compelled to grow in the space between the ribs and the liver and consequently follow the curvature of the chest wall which ultimately gives them, more or less, the appearance of ventral bronchi, a fact which led Aeby, who studied only the finished tree, to call them the ventral series.
4. The dorsal series of bronchi, originating like the lateral group as outgrowths from the stem bronchus, are usually paired. They alternate with the paired lateral bronchi and are independent productions of the _stem. They do not either ontogenetically or phylogenetically originate from the lateral bronchi. For convenience, the first pair are called Dorsal 2, to keep the designation harmonious with the larger series of lateral bronchi.
5. The ventral bronchi originate as outgrowths from the ventral surface of the stem. They, like the other series, are independent productions of the main bronchus. They are not originally formed on the lateral bronchi and subsequently transferred to the stem bronchus. Consequently, they are chief bronchi and not accessory in the sense of Aeby.
In the pig and in the great majority of mammals, left Ventral 2 is suppressed. With the absence of left Lateral 1, it destroys the absolute symmetry of the mammalian lung. The cause for the remarkable hyperdevelopment of the Ventral 2 on the right side in most mammals is undoubtedly due to the effort to increase the respiratory area by filling the space that intervenes between the heart and diaphragm with the Lobus infracardiacus. The remainder of the ventral series are usually paired in the pig and like the dorsal series ordinarily alternate with the larger lateral bronchi. As a rule their roots are placed on the ventral surface of the stem midway between the adjacent lateral elements and opposite the corresponding dorsal bronchi. The first ventral element is designated Ventral 2 on account of its topographical relationship to Lateral 2.
6. The medial bronchi are, like the other series, produced by medial outgrowths from the stem. They are not formed on the dorsal bronchi and then transferred to the stem. They rarely occur higher than the level of Lateral 4 and are extremely irregular in their arrangement.
7. Noteworthy are the great variations found in the production of the various bronchi. The lateral series are by far the most constant elements of the tree. Still, it is not uncommon to find either an extra element formed or else to see one of the usual elements suppressed. As the common number of lateral elements is six on the right side and five on the left, the extremes may vary between five and seven on the right and four and six on the left. In the case of the dorsal series, the variation is even more marked than in the lateral, thus, one element may be suppressed, leaving the dorsal area between two adjacent lateral bronchi naked or, else, an extra element may be formed, giving two dorsal elements in a single interspace. The ventral series is still more variable than the dorsal, so much so, in fact, as to make it uncommon even in the pig where these elements are unusually well developed, to find a series complete, of course, with the exception of left Ventral 2, which is always suppressed. It is not uncommon to find several elements of this series absent at once. Like the dorsal bronchi, they may also be reduplicated in a single interspace. The medial bronchi are the most variable of the four types. They may not be present at all, they may be present only on one side, or they may be reduplicated in a single interspace, but, in the pig, they never occur higher on the stem than the level of the fourth lateral bronchus. The reason for this fact lies in the presence of the oesophagus above this point, which allows no space for the development of medial elements from this portion of the stem bronchus.
8. The following formula would represent the complete series of principal bronchi in the lung of the pig: Tnscnm.
Lateral 1.
Right Stem Bronchus. Left Stem Bronchus.
Lateral 2. Lateral 2.
Dorsal 2. Dorsal 2.
Ventral 2. ’ Lateral 3. Lateral 3.
Dorsal 3. Dorsal 3.
Ventral 3. Ventral 3.
Lateral 4. Lateral 4.
Dorsal 4. Dorsal 4.
Ventral 4. Ventral 4.
Medial 4. Medial 4.
Lateral 5. Lateral 5.
Dorsal 5. Dorsal 5.
Ventral 5. Ventral 5.
Media] 5. Medial 5.
Lateral 6. Lateral 6.
It is extremely rare to find a tree as complete as the one expressed in this formula. A number of bronchi may be missing or else some may be reduplicated.
9. The whole series of bronchi show a most remarkable adaptation to the space in which they have to grow. This is true of both the chief bronchi as well as their smaller subdivisions. When, for example, a bronchus is suppressed, an adjacent branch will grow into the area usually supplied by the missing element, substituting for its loss. It is in this way that we obtain the large series of pictures which suggest a wandering of the secondary branches from the lateral and dorsal elements on to the stem bronchus. After a careful study of this point, it may be definitely stated that bronchi never wander. They remain firmly fixed on the stem or side branches where they originate. Not uncommonly their direction may be altered, however, by changes in the space in which they develop.
This response on the part of the growing bronchi to their space relationships is also shown in the course or direction of the principal elements as Well as their secondary branches. We have, therefore, Lateral 1 produced and growing into the area between the upper part of the heart and chest wall. Owing to the larger space just beside the vertebral column and the antagonism between it and Lateral 2, the lower branches of Lateral 1 are forced dorsalwards until it resembles superficially a dorsal bronchus. The second lateral bronchi develop in the region between the chest wall, heart, and liver. The area in which the remainder of stem has to grow has in cross-section practically the shape of an isosceles triangle. The stem, occupying a point about the middle of the base, sends three sets of branches, namely, dorsal, lateral, and ventral, directed into the angles of the triangle where they would have the most freedom to develop. Between the roots of the two stem bronchi runs the oesophagus, leaving no place for the development of median branches in this region. At the level of Lateral 4, however, below the oesophagus more room occurs and, consequently, we observe in this region the formation of medial bronchi. Undoubtedly the difference in the branching of the stem in the Lobus inferior of the human lung when compared with the pig may be sought in its altered topography owing to the erect posture which changes principally the position of the liver.
This adaptation on the part of the lungs to their environment is to be expected for they are relatively late accessions to the animal economy and are of no known use to the organism during the period of gestation.
Accordingly as the heart and liver are both phylogenetically older than the lungs and also are of known functional value during foetal life, it is natural that the latter should adapt themselves to the early needs of older organs.
10. The growth of the main series of bronchi is monopodial in character, that is to say, they are produced without a definite division of the end bud. New elements are not always produced from the end bud, but may be formed from the stem some distance from its terminus.
The process is successive, that is to say, the elements are produced one after another from above downwards, recapitulating the method of growth shown in simpler animals like the reptiles, for example. When a new element is about to be produced, one notes an increase in the number of karyokinetic figures in the epithelium in the region of the new branch.
The basement membrane becomes less distinct and the connective-tissue nuclei in the surrounding mesoderm are more closely packed together.
In this region a slight bulging of the epithelium is then noted, which increases until a small elevation is raised upon the surface of the stem.
This increases in size, yielding a rounded projection, which gradually emancipates itself and gives rise to a new bronchus. The process is essentially the same whether it occurs in the neighborhood of the terminal bud or higher up on the stem. In general, we may say, the lateral and medial elements are produced nearer the terminal end of the main bronchus, while the dorsal and ventral elements are formed somewhat higher up, often where the stem has regained its cylindrical form.
Subsequent division of the branches may occur either by monopody or dichotomy. Often monopodial production of buds persists for one or two generations on the main bronchi, then the method becomes dichotomous, either equal or unequal in nature depending somewhat on the space in which the bronchi have to divide. In the case of equal division of the bud, however, one fork grows on to become the stem while the other remains as the side branch. The ifirst division of the main bronchi may, it is well to note, be dichotomous as in the case of Lateral 1 and Lateral 2. Thus in its growth, the mammalian lung recapitulates the history of the simpler lungs of lower animals.
11. The pulmonary arteries in the pig arise from the pulmonary arches as Bremer has described. At first, they run parallel, then bend towards each other, sending out anastamoses, which yield finally a common trunk with two origins above and two arteries below. Later the upper part of the right artery degenerates and with it the right pulmonary arch. At 5 mm. before the pulmonary arteries may be followed as far as the anlage of the lungs, the pulmonary vein may be seen as a slight ingrowth from the undivided portion of the Sinus venosus, passing through the Mesocardium posterior towards the pulmonary anlage. It forms almost in the medial plane. With this establishment of the venous outlet ventralwards to the anlage, the arteries, as the growth of the organ proceeds, are naturally developed from the capillary plexus on the dorsal side of the primitive bronchi. This fixes the arteries with reference to the stem bronchi before any of the side branches are produced. As the pulmonary anlage projects some distance ventralwards from the head gut, Lateral 1, the “eparterial” bronchus, develops above the artery, while Lateral 2 and the remainder of the principal branches originate below. Thus, the two regions of the tree have a difierent topography with reference to the pulmonary artery, but this vessel has no fundamental influence on the structure of the two parts, nor does it differentiate the tree into two regions of different morphological significance as Aeby has maintained.
The entire primitive tree is surrounded by a capillary plexus. As the bronchi grow, and produce new branches, arteries are developed from this plexus on the dorsal side of the tree as the artery lies dorsalwards and lateralwards to the stem. From this position, arteries to the lateral bronchi run out above and behind them. The branches to the dorsal bronchi pass dorsalwards along the lateral aspect of these elements. To the ventral series, arteries pass around the lateral aspect of the stem bronchus beneath the root of the corresponding lateral bronchus to gain the outer aspect of the ventral bronchus along which they run. The medial bronchi receive their supply from branches that originate from the main artery and pass around the dorsal aspect of the stem to run on the dorsal surface of the medial bronchi. As the right pulmonary artery runs ventralwards to Lateral 1 the artery to that bronchus develops on its ventral surface.
In the younger stages, both the aortic arch and the Ductus arteriosus lie well above the level of Lateral 1:. As the embryo increases in age, there is a gradual descent of the heart and with it, the great vessels.
At 15 cm. one observes the Ductus arteriosus at the level of Lateral 1; at 22 cm. the aortic arch reaches this point, while at birth both vessels lie below the bronchus.
12. The pulmonary vein develops in pigs about 5 mm. long as an ingrowth from the undivided portion of the Sinus venosus at the level of the pulmonary anlage. As the stem bronchi increase in size, right and left pulmonary veins develop from the capillary plexus which surround them. These, naturally, form on the ventral surface, with the bronchi between them and the arteries. Similarly, as the various principal bronchi are produced from the stem bronchus, veins are formed from the capillary plexus. The veins from the lateral bronchi lie below and ventralwards to the bronchi, those from the dorsal elements run along the medial aspect of the air passages to empty into pulmonary veins lying ventralwards to the stems. The veins from the ventral bronchi extend along the medial aspect of the bronchus and terminate directly into the pulmonary veins; those from the medial bronchi extend along their ventral surface to empty in the larger veins accompanying the stems. The vein from Lateral 1 runs along the ventral aspect of the bronchus somewhat ventralwards to the corresponding artery. This forms the single exception to the general alternation of artery, bronchus, and vein. As the embryo increases in age, the Vena pulmonalis, which originates near the midline, is gradually pushed to the left by the increasing asymmetry of the heart, until it finally comes to lie over the area of the stem bronchus where a left Ventral 2 would have developed if such a bronchus were present. The hyperdevelopment of the Bronchus infracardiacus associated with the development of the Vena cava inferior to the right of that bronchus aids in pushing the Vena pulmonalis to the left.
13. The asymmetry of the mammalian lung is associated with the asymmetrical development of the heart and its great vessels. In the descent of the aortic arch and the Ductus arteriosus during embryonic life from a point above the origin of Lateral 1 to a point below, we have an explanation for the suppression of this element on the left side, for if this bronchus were formed, both aorta and the Botallian duct would be caught upon it and their descent prevented. Likewise the Vena pulmonalis appears in the midline and is carried to the left until it finally rests on the portion of the stem where a left Ventral 2 should develop.
The usual suppression of these two elements, therefore, must be looked upon as a phylogenetic provision to allow for the descent of the great vessels on the one hand and the shifting of the Vena pulmonalis on the other. It is noteworthy that in those animals where these bronchi are formed on both sides, they are so situated as to offer no resistance to either of these features of the development of the great vessels.
14. The mesodermic portion of the lungs is derived from the general mesoderm about the head gut. As the bronchi appear, this is pushed out into the primitive coelom to form two irregular swellings, marking the anlagen of the two wings of the lungs. With the appearance of Lateral 1, on the right side, and Lateral 2, on each stem bronchus, swellings are observed on the two simple lungs just over these bronchi, giving rise to the simplest form of the Lobus superior, Lobus medius, on the right side, and the Lobus superior on the left. The remainder of the mesoderm about the stem bronchus forms the anlage of the Lobus inferior on each side. With the formation of Ventral 2, the Bronchus infracardiacus, a swelling from -the mesoderm forms over it which is the anlage of the Lobus infracardiacus. These swellings are first surrounded by shallow grooves, which with the rapid growth of the bronchi beneath, rapidly develop into deep fissures separating the various lobes from each other. With the further growth of these bronchi and the appearance of the series of bronchi on the stem, projections and fissures are formed over and between them and in the mesoderm. These are equivalent in all respects except in age and size, to the earlier fissures and swellings, but, under ordinary circumstances, never give rise to distinct lobes. This is due to the more rapid growth of the first bronchi, to the gradual increasing density of the mesoderm, and, lastly, to the environment of the several lobes of the lung. The right Lobus superior, containing Lateral 1 does not belong to the dorsal region of the lung as some authors hold, but to the lateral. The characters which make it appear as a dorsal segment are secondary and not primary. Likewise the portion of the left Lobus superior containing the apical bronchus belongs to the lateral region and not to the dorsal. As in the case of the right Lobus superior, its dorsal characteristics are secondary. This segment is to be compared to the portion of the right Lobus medius which contains the main dorsoinferior bronchus. Moreover, the entire left Lobus superior is the ontogenetic equivalent of the right Lobus medius. The right Lobus superior is an unpaired lobe and has no equivalent in the left lung. The same thing is true of the Lobus infracardiacus.
Lobe formation varies greatly in different species. In the majority of mammals, there are three or four lobes on the right side, arising from Lateral 1, Lateral 2, Ventral 2, and the stem bronchus, while, on the left side, there are ordinarily two formed from Lateral 2, and the stem.
Extremes of variation occur, however, between a lobeless lung in which none of the bronchi subdivide it and a multilobar lung in which most of the principal bronchi have segmented the wing into a series of small lobes. Apparently, the division of the lung into lobes is of no general morphological significance.
15. In the light of recent researches on the reptilian, amphibian, and avian lung, it is possible to take a new viewpoint for the development of the mammalian lung. The lungs of lower animals, we now know, are products of monopodial growth. The simple lungs of reptilia are capable of producing monopodially outgrowths in any direction (Hesser).
These may become specialized in certain species and have a definite topography. As we mount the animal scale, the necessity of an increased respiratory surface finally results in the transformation of the original simple lung into a conducting apparatus, which is represented in the mammalian lung by the stem bronchus and its chief branches. The simple lungs may no longer be compared to the Lobuli respiratorii of the mammalian lung, for the latter represent new elements which with the increased respiratory surface are added peripherally to the simpler lungs as these become transformed into bronchi. With the addition of these new elements, the respiratory function also wanders peripheralwards, so that the portion of the mammalian tree which represents the simpler lungs undergoes a change of physiological function. Its phylogenetic relationship to the simple lungs is shown by the monopodial growth of the mammalian stem bronchus and its principal branches, which recapitulate ontogenetically the growth process of the simple lungs before producing dichotomously the prepheral respiratory structures which are used in mammalian respiration. In certain animals, moreover, the stem bronchus and its branches retain for a period in their li.fe history their respiratory function. In monotremes and marsupialia, the young are transferred to the pouch and compelled to carry on their own respiration when only the stem bronchus and its chief branches are formed. The ordinary respiratory structures used in the adult stage, are produced at a later period. We have, thus, both a physiological and an ontogenetic proof that the simple lungs correspond, in mammals, only to the stem bronchus and its chief branches.
The great majority of mammalian lungs are asymmetrical, the asymmetry consisting in the presence of an unpaired Lateral 1 and an unpaired Ventral 2, both of which occur on the right side. Some mammalian lungs are symmetrical and considerable effort has been made to explain all the asymmetrical lungs on the basis of the minority of symmetrical ones. The asymmetrical lung, however, must be regarded as typical for mammals. The two bronchi responsible for the asymmetry are, so far as we know, characteristic of the mammalian and avian (Aeby) lung as similar bronchi have never been described in the lungs of lower animals. The cause for the asymmetry, apparently lies in the necessity of leaving space for the descent of the heart and great vessels, by the suppression of left Lateral 1, on the one hand, and to allow room for the shifting of the heart which draws the Vena pulmonalis to the left by the suppression of left Ventral 2, on the other. In those lungs where these two elements, which are usually missing, are found, they are appar ently so placed as not to interfere with these features of the development of the heart.
16. In the organogenesis of the lungs, we have the stem and main bronchi consisting of simple tubes lined by a double layer of epithelium, the inner of which is columnar, while the outer is composed of smaller polygonal cells. This simple tube is surrounded by a membrane. propria produced largely by the deposit of fibrils from the exoplasm of the connective—tissue syncytium, composing the mesoblastic portion of the lungs at this early stage. As the bronchi grow, a layer of spindle cells differentiate from the mesoderm, which are transformed into the muscular coat of the bronchi. Later still, a chondrification of the perimuscular syncytium takes place from which the cartilaginous rings of the trachea and the bronchial cartilages are formed. With these changes, the connective-tissue fibrils become grouped into trabeculas about the bronchi and in the submucosa. Later, the mucosa is thrown into a series of longitudinal folds, while from the cuticular border of the inner row of cells, cilia develop. From the bottom of the crypt—li-ke invaginations formed by the longitudinal folds of epithelium, glands begin to grow into the submucosa, which sometimes pass between the developing muscle bundles into the deeper layers of this coat. As this process takes place, there is a differentiation of some of the epithelium into goblet cells, a process, which one also observes in the glands, giving rise to a series of submucous glands with partly serous and partly mucous cells. While these changes occur in the mucosa, the cartilages are also growing, and with them a further differentiation of the framework into distinct fibrous trabeculae takes place. As we follow the bronchi peripheralwards, they become simpler and essentially younger in structure and yet, develop their adult characteristics in precisely the same Way. The epithelium soon becomes single layered and of a columnar type as the periphery is reached. finally it takes on a distinct, flat, cubical form. The Lobuli respiratorii begin to develop in pigs about 19 cm. long by a slight dilatation of the growing ends of the bronchi. These represent the bronchioli. Later Bronchioli respiratorii are then formed, having a_ progressively flattened epithelium, which runs over into Ductuli alveolares.
These are present at the age represented by a pig 22 cm. long. Subsequently, Atria, Sacculi alveolares, and Alveoli pulmonis form in the prenatal. period, all of which have the characteristic flattened respiratory epithelium. And finally, after birth, there is a dilatation of the lobules and a further flattening of the epithelium occurs, and before the pig is half grown, a muscle layer develops about the air passages as far as the Atria, where it stops in sphincter-like bands. One finds at no period in the life history of the pig’s lung, openings or fenestrae which communicate between adjacent respiratory lobules. The latter form independently at the growing ends of the tree and as they approximate each other, the interalveolar framework can always be demonstrated between them Without interruptions suggestive of fenestrae connecting adjacent alveoli.
17. The framework of the lungs develops from a general syncytium forming the mesodermic anlagen of the lung wings. By a gradual differentiation of connective-tissue fibrils from the exoplasmic part of the syncytium, the framework becomes denser and, finally, at 8 cm., a suggestion of lobulation is obtained about the end branches of the growing bronchi. Within these connective-tissue lobules, the framework differentiates as the embryo grows, forming simultaneously basement membranes for the young bronchial buds. At the same time, the interlobular fibers and those below the pleura, unite to form trabeculac. As the Lobuli respiratorii, towards the end of foetal life, begin to impinge on each other, the interalveolar framework and the two adjacent basement membranes are pressed together into a single wall or septum in which the blood-vessels run. These lobules persist until adult life, although they may become compound by the rupture of the interlobular septa and the subsequent confluence of several adjacent lobules. This process ordinarily takes place at the base, leaving the periphery of the compound lobule separated by partial septa.
18. The lymphatics appear at the root of the lung in an embryo 4-5 cm. in length. Accompanying the bronchi and pulmonary vessels, they gradually grow in for some distance until the smaller air passages are reached, when they leave these structures and grow towards the pleura in the interspaces between the smaller bronchi, in what represent the primitive interlobular spaces. In this way they aid in the dilferentiation of the connective-tissue lobules. The reason for this course is not entirely clear, but it may be due to the increasing density of the framework about the bronchi, which forces the later-appearing lymphatics into the interlobular spaces as a locus minoris resistentiae. Upon reaching the pleura, they turn and form a plexus in the subpleural connective tissue. Here and there, they may be seen penetrating the lobules, but cannot be followed for any distance in them. At 23 cm., the first evidence of the submucous plexus is seen in the stern bronchi.
Literature
AEBY.—Der Bronchialbaum der Saiigethiere und des Menschen. Leipzig, 1880.
v. BAER.—Ueber Entwickelungsgeschichte der Thiere. Konigsberg, 1828.
BIcHA'r.—Anatomie descriptive. Paris, 1829.
BLISNIANSKAJA.—Dle Entwickelungsgeschichte der menschlichen Lungen. Diss. Zurich, 1904.
BoNNEr.——Handbuch d. vergl. Histologie u. Physiologie der Haussaugethiere. Bd. 2, Berlin, 1892.
BBEMER.—American Journal of Anatomy. Vol. I, 1902.
American Journal of Anatomy. Vol. III, 1904.
CAn1A'r.—Jour. de l’Anatomie et de la Physiologie. 1877.
COUNCILMAN.—J011I‘I'la.l Boston Med. Soc. Vol. IV, 1901.
EWABT.——The Bronchi and Pulmonary Blood-vessels. London, 1899.
fiscHELIs.—Beltrage zur Kenntniss der Entwicklungsgeschichte der Lunge. Diss. Berlin, 1885.
FLINT.—JOl1!1S Hopkins Hospital Reports. Vol. X, 1900.
Journal of Medical Research. Vol. VII, 1902.
(1) American Journal of Anatomy. Vol. II, 1903.
(2) Archiv. f. Anat. u. Ent. Anat. Abth. 1903.
(1) Anatomischer Anzelger. 1906.
(2) Anatomischer Anzeiger. 1906.
FoL.—Recueil zoologique suisse. T. 1, 1884.
GEGENBAUER.-——GI‘11ndI‘iSS der Vergleichenden Anatomie. Leipzig, 1874.
Vergleichende Anatomie der Wirbelthiere. Bd. 2, 1901.
G6TTE.—Beitrage zur Entwicklungsgeschichte des Darmkanals im Hiinchen. Tiibingen, 1867.
Die Entwicklungsgeschichte der Unke. Leipzig, 1875.
Zotilogischer Jahrbiicher Anat. Abth. 1904.
GREIL.—A1lat. Hefte. Bd. 29, Hft. 3, 1905.
GUIEYssE.—Jour. de 1’Anatomie et de la Physiologie. T. 34, 1898.
n’HAm>1v1LLER.——(1) Comptes Rendus de la Société de Biologie. Vol. III, 1896.
(2) Bibliographie Anatomique. Vol. IV, No. 5; Vol. V, No. 1, 1896-1897.
(1) Bibliographie Anatomique. Vol. V, No. 1, 1897.
(2) Bibliographie Anatomique. Vol. V, No. 6, 1897.
(3) Comptes Rendus de la Société de Biologie. Vol. 4, November 20, December 4, December 11, 1897.
HESSER.—-Anat. Hefte. Bd. 29, Hft. 2, 1905.
HIs.—Untersuchungen iiber die erste Anlage des Wirbelthierleibes. Leipzig, 1868.
Arch. f. Anat. u. Entwick. Anat. Abth. 1887.
HOCHSTETTER.—Zeit. fur wissenschaft. Mikroskopie. Bd. 15, 1898.
HUN'r1NoToN.—Anna1s of the New York Academy of Science. Vol. XI, 1898.
JUSTESEN.—A1'Ch. f. Mik. Anat. 1900.
KAsTscHENKo.—Arch. f. Anat. u. Phys. Anat. Abth. 1887.
K6LL1KER.—Entwicklungsgeschichte des Menschen. Leipzig, 1879.
Kt'I'r'rNER.—Virchow’s Archiv. 1876.
Lmzmo.—-Lehrbuch der Histologie des Menschen und der Thiere. Frankfort A/M, 1857.
LAGUESSE ET n’HAnD1v1LLER.—Comptes Rend. Soc. Biol. T. 5, 1898.
Mall FP. Development of the connective tissues from the connective syncytium. (1902) Amer. J Anat. 1(3): 329-366
MERKEL.-—HaI1db11ch der Anatomie des Menschen (Von Bardeleben). Jena, 1902.
MEYER.—Lehrbuch der Anatomie des Menschen. Leipzig, 1861.
Mu.LER.—Journa1 of Morphology. 1893.
Arch. f. Anat. u. Phys. Anat. Abth. 1900.
Minot CS. Human Embryology. (1897) London: The Macmillan Company.
Mos1m.—Arch. f. Mik. Anat. Bd. 60, 1902.
Mt‘ILLER.—Jena.ische Zeitschrift fur Naturwissenshaft. Bd. 32 (N. F. Bd. 25), 1898.
NARATH.——Verhand1. der Anat. Gesellsch. 1892.
Zoologische Forschungsreisen in Australien u. dem Malayischen Archipel. (Semen), Iena, I896.
Bibliotheca. medica. Abth. A, Hft. 3, 1901.
NICHOLAS AND DIMITROVA.--COII1pteS Rendus de la Societé de Biologie. 1897.
RATHKE.—Verh. d. Kais. Leop. Carol. Akad. d. Nat. Bonn, 1828.
Entwicklungsgeschichte der Natter. Konigsberg, 1839.
REMAK.——Untersuchungen iiber die Entwickelung der Wirbelthiere. Berlin, 1855.
RoBINsoN.—Journal of Anatomy and Physiology. 1889.
RiinINGE1z.—Topographisch-chirurgische Anatomie des Menschen. Stuttgart, 1873.
SAKURAI.—Ana.tomischer Anzeiger. 1904.
SCHMALHAUSEN.—AI1a.t0IniSche1‘ Anzeiger. Bd. 27, 1905.
SCHMIDT.-—Cited by His (87). 1870.
SEEssEL.—Arch. f. Anat. u. Phys. Anat. Abth. 1877.
SELENKA.—Studien uber die Entwickelungsgeschichte der Thiere. Heft 1, das Opossum. Wiesbaden, 1887.
Zeit. f. wiss. Zool. Bd. 16, 1866.
STIEDA.—Zelt. f. wiss. Zool. Suppl. Bd. 1878.
S'ross.~—Untersuchungen iiber die Entwicklung der Verdauungsorgane. Diss. Leipzig, 1892.
THOMA.-—Untersuchungen fiber die Histogenese und Histodynamik des Gef§.sssystems. Stuttgart, 1893.
UsKow.—Arch. f. Mik. Anat. Bd. 22, 1883.
WEBER AND BUv1GNmR.——Bibliogra.phie Anatomique. T. 12, 1903.
WILLAcH.——Beitra.ge zur Entwicklung der Lunge bei Saiigethieren. 0ster wieck-Harz (Vickfeld), 1888.
ZUMsrEIN.—Sitzungsberichte der Gesell. zur Befiird. d. gesammt. Naturwiss. zu Marburg. Marz, 1889.
Sitzungsberichte der Gesell. zur Befiird. d. gesammt. Naturwiss. zu Marburg. Februar, 1891.
Sitzungsberichte der Gesell. zur Befiird. d. gesammt. Naturwiss. zu Marburg. Marz, 1892.
Sitzungsberichte der Gesell. zur Beford. d. gesammt. Naturwiss. zu Marburg. Mai, 1900.
Explanation of the Plates
Plate I
Figs. 1-14. are magnified 50 diameters. Pulmonary arteries red, pulmonary veins blue, bronchi white.
Fig. 1. Reconstruction of a portion of the head gut of a pig’s embryo 3 mm. long. Ventral view.
Fig. 2. Dorsal View of the same reconstruction.
Fig. 3. Reconstruction of a. portion of the head gut of a pig's embryo 5 mm. long. Ventral view.
Fig. 4. Dorsal view of the same reconstruction.
Fig. 5 Reconstruction of the bronchial tree of a pig 6 mm. long.
Fig. 6. Dorsal view of the same reconstruction.
Fig. 7 Reconstruction of the bronchial tree of a pig 7.5 mm. long.
Fig. 8 Dorsal View of the same reconstruction.
Fig. 9. Reconstruction of the bronchial tree of a pig 8.5 mm. long.
Fig. 10. Dorsal View of the same reconstruction.
Fig. 11. Reconstruction of the bronchial tree of a pig 10 mm. long.
Fig. 12. Dorsal view of the same reconstruction.
Fig. 13. Reconstruction of the bronchial tree of a pig 12 mm. long.
Fig. 14. Dorsal view of the same reconstruction.
Plate II
figs. 15-19. are magnified 50 diameters. Pulmonary arteries red, pulmonary veins blue, bronchi white.
fiG. 15. Reconstruction of the bronchial tree of a pig 13.5 mm. long.
fiG. 16. Dorsal View of the same reconstruction.
fiG. 17. Reconstruction of the bronchial tree of a pig 15 mm. long.
fiG. 18. Dorsal view of the same reconstruction.
fiG. 19. Reconstruction of the bronchial tree of a pig 18.5 mm. long.
Plate III
fig. 20. magnified 50 diameters. Pulmonary arteries red, pulmonary veins blue, bronchi white.
fiG. 20. Dorsal view of the same reconstruction.
Plate IV
figs. 21-25.
fiG. 21. Celluloid corrosion of the bronchial tree of a pig’s embryo 5 cm. long. X 2.
fiG. 22. Celluloid corrosion of the bronchial tree of a pig’s embryo 7 cm. long. X 2.
fiG. 23. Wood’s metal corrosion of the bronchial tree of a pig’s embryo 18 cm. long.
In this specimen one lateral bronchus on each side is suppressed, giving five laterals on the right and four on the left, instead of the usual complement of six and five respectively. Ventral 3 on both sides is suppressed. Substituting for these branches are ventral branches of the adjacent lateral bronchi, while on the right side a lateral division from the inferior branch of V. 2 also extends into the region usually supplied by right V. 3.
fiG. 24. Dorsal View of the same preparation.
Dorsal 3 on the left side is suppressed. It is compensated for partly by Dorsal 2 growing lower than usual and partly by branches from Medial 4 on that side. On the right side Dorsal 4 is reduplicated, the upper element growing dorsolateralwards, the lower directly dorsal. Medial 4 and 5 are present on both sides.
fiG. 25. Wood’s metal corrosion of the lung of a suckling pig two days old. Ventral view. X 2.
Ventral 2 is broken off to show the dorsal bronchi. In places where the metal has passed into the smaller bronchi, the dichotomy is well shown.
The branches are schematic in .their arrangement with the exception of Ventral 5 on the left side, which is reduplicated, and an extra irregular lateral branch is interpolated on the right side.
Abbreviations
b = Gill pouch.
a : Head gut.
o = Pulmonary anlage.
h = Ductus hepaticus.
o = CEsophagus.
ad = Arteria. pulmonalis dextra.
T = Trachea.
d = Right stem bronchus.
s = Left stem bronchus.
as = Arteria pulmonalls ainistra.
12 = Vena pulmonalis.
L. 1, L. 2, L. 3, L. 4, L. 5, L. 6, etc.=The lateral series of bronchi.
D. 2, D. 3, D. 4, D. 5, D. 6, etc. =The dorsal series of bronchi.
V. 2, V. 3, V. 4, V. 5, V. 6, etc. = The ventral series of bronchi.
M. 4, M. 5, etc.=The medial series of bronchi.
ap : = Apical branch of left L. 2.
m = Medial branch.
d = Dorsal branch.
1: Lateral branch.
17 = Ventral branch.
8 = Superior branch.
i = Inferior branch.
In the combined abbreviations: di = Dorsoinferior branch.
Lb = Lateroinferior branch.
vs = Ventrosuperior branch, etc.
Reference
Flint JM. The development of the lungs. (1906) Amer. J Anat. 6: 1-137.
Cite this page: Hill, M.A. (2026, April 5) Embryology Paper - The development of the lungs. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_The_development_of_the_lungs
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G