Book - Contributions to Embryology Carnegie Institution No.52
| Embryology - 27 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Ingalls NW. A human embryo at the beginning of segmentation, with special reference to the vascular system. (1920) Contrib. Embryol., Carnegie Inst. Wash. Publ. 274, 11: 61-90.
| Online Editor |
|---|
| This historic 1920 paper by Ingalls (1880-1949) describes a Carnegie Collection Embryo No. 1878 Carnegie stage 9 embryo in Week 3.
|
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
A Human Embryo at the Beginning of Segmentation, with Special Reference to the Vascular System
By N. William Ingalls (With five plates and one text-figure)
Published by the Carnegie Institution of Washington Washington, 1920
- Embryo at Segmentation: Figure A | Plate 1 | Plate 2 | Plate 3 | Plate 4 | Plate 5 | Carnegie stage 9 | Carnegie Embryo 1878
Reference
Ingalls NW. A human embryo at the beginning of segmentation, with special reference to the vascular system. (1920) Contrib. Embryol., Carnegie Inst. Wash. Publ. 274, 11: 61-90.
Cite this page: Hill, M.A. (2024, April 27) Embryology Book - Contributions to Embryology Carnegie Institution No.52. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_Contributions_to_Embryology_Carnegie_Institution_No.52
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G
Introduction
The embryo which forms the subject of this communication is No. 1878 of the Carnegie Embryological Collection. Data relative to its age are rather indefinite, but it is apparent that the menstrual age is in excess of the real or fertilization age. Further evidence of this is found in the fact that the tissues of the ovum (and this applies also to the endometrium) had begun to suffer slightly, with the result that the fixation and staining reactions are not all that could be desired. These changes are not, however, of such a nature or degree as to raise doubts about the specimen being normal. They have been further complicated by what has been in some respects an unfavorable plane of section, so that at times considerable difficulty has been encountered in determining certain of the finer morphological details presented by this particular stage. This account, therefore, will confine itself to a brief description of the essential features of the embryonic body and of the structures immediately adjoining. We shall not enter upon any discussion of the findings in the yolk-sac or chorion, or of the finer histological details of the embryo itself. Evidences of mitotic activity are encountered, but they are not especially frequent.
Concerning the gross specimen we may quote the laboratory notes as follows :
- "Main mass of specimen composed of decidua, clots and scrapings from curettage. In addition to this there is an isolated flattened ovum measuring 7.5 x 10.5 x 12 mm. The specimen is stained pink, like the scrapings obtained from curettage. The bare surface of the chorionic vesicle, which comprises about one-third of area of ovum, is folded somewhat. The villi seem to be well-developed, but are absent also on a narrow strip in the side opposite the bare area. The bare areas look rather thick-walled and the villi rather clubbed.
- "Upon opening the ovum from the bare area it felt rather firm. The chorion was thick and most of the interior was filled with a web-like magma, small portions of which were not unlike absorbent cotton. It was quite easy, however, to remove all the magma without injury to the embryo. The latter looked somewhat mottled, especially in the region of the yolk-sac, which lay somewhat at right angles to the amnion. At. the point of union of the two a somewhat elongated, opaque disc could be seen quite distinctly."
External Form, Embryo and Adnexa
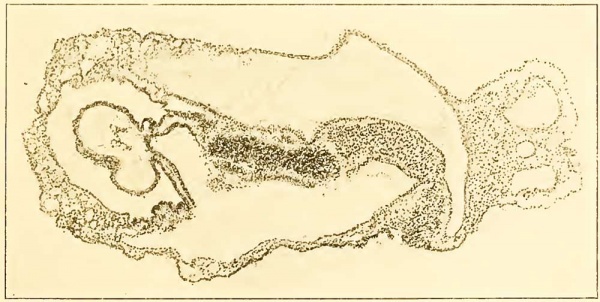
The form of the embryonic body (figs. 1, 2, 3, and 4) is in general quite similar to a larger and older specimen, No. 391, 2 mm. (Dandy, 1910), and a much more advanced one, No. 1201, also 2 mm., both of the Carnegie Collection. In all three, as in so many other embryos of about this stage, there is a well-marked dorsal concavity. In No. 1878 this flexure is very abrupt, even more marked than in No. 1201 (Evans and Bartelmez, 1917), and is most acute about, or slightly in advance of, the fine between the future brain and cord. Posterior to the angle thus formed is a short portion of the dorsum of the embryo which is very nearly straight, while beyond this is a gentle rounded elevation completing the embryonic body. Seen in profile, No. 1878 is strikingly like the older but slightly shorter embryo (H 98), 1.27 mm., recently described by Wilson (1914, plate III). The Kromer-Pfannenstiel embryo, while only a little longer (1.8 mm.) is much more advanced. The model shows the body of this embryo to be practically straight (Kromer, 1903; Keibel and Elze, 1908). Indeed, in the model there seems to be hardly room enough for the heart and pericardial cavity, as they have been described and figured in section, between the brain and the yolk-sac.
There can be no doubt that the early development of the heart, and especially of the myoepicardial mantle and pericardial coelom, plays a very important role in the lifting up of the head. This general effect on the anterior end of the embryo could be accentuated only by the precocious development of the forebrain. Further evidence of such an elevating process may be seen in the relation of the amniotic reflection anteriorly, where, immediately below the stomodeum it is lifted up as a saddle-like ridge by the large pericardial coelom beneath. In addition, one may conclude that a large yolk-sac would have a similar tendency to raise the heart and with it, of course, the entire anterior end of the embryo. This might help explain the condition in the Kromer embryo, where the yolk-sac is not very large. While doubtless subject to not a little individual and relative variation, the yolk-sac, throughout most of its history, especially later and possibly also very early, is rather small as compared with the embryo and body-stalk. For a very brief period, however, it undergoes a rapid increase in dimensions and the relative size of the embryo and sac is, for a short time, reversed. In the Kromer specimen the relation of the two is being changed for the second and last time. Certainly the embryonic body is weaker and more pliable about its middle than anywhere else, and all things considered it is difficult to see how it could possibly be bent in the opposite direct ion. A dorsal flexure, normal or otherwise, is what one might expect at this time and, until more is definitely known as to the possible factors involved, some degree at least of such a flexion will have to be looked upon as normal. On the whole, it would be better to speak of an elevation or erection of the head, as also suggested by Wilson, and so put the emphasis where it undoubtedly belongs. There is no reason why a curve of this character may not later be wiped out entirely, as happens to so many others in the embryo, of which we will mention only the dorsal curvature of later stages, which is subject to very considerable individual variation. The deep, sharp kinks shown by the His embryos Lg. and Sch., 2.15 and 2.2 mm. respectively, may very well lie abnormal, and, except their direction, they apparently have little if anything in common with the so-called flexure which we have just discussed. In the same category would fall the embryo Delaf. of Eternod (1909), noted as "2.1 mm. (?) enselle," admittedly an old alcohol specimen. These kinks involve the body at a definitely more caudal point. They occur at a later stage in development, at a time when the diminishing yolk-sac not only offers less support to the embryo, but would be in a favorable position, on account of its narrowing attachments, to actually exert some traction upon the delicate body of the embryo. It is quite conceivable that the earlier normal curve might grade insensibly into the later abnormal one. Wilson's specimen was, we imagine, very near the parting of the ways, where it would have either gradually straightened out or have suffered, through some influence, a much sharper degree of flexion.
No attempt will be made to institute a formal and detailed comparison of No. 1878 with other embryos, nor to determine its developmental status as regards them. Loose comparisons between embryos lose much of their apparent meaning on account of the variability in rate and manner of development. To such variation we have recently drawn attention (1918) and fortunate or extreme instances of it may yet throw considerable light on early human ontogeny.
The greatest length of embryo No. 1878, measured on a straight line from the anterior (later ventral) surface of the head just above the stomodeum to the cloacal membrane, is, as determined on the model, 1.38 mm. Of this, rather more than onehalf will later be taken up into the head, the heart being now frankly in the head region, while the rest of the future body is represented by the remaining posterior part of the embryo, of which a small portion is occupied by the primitive streak.
The elevation of the posterior end of the body is not due solely to the sinking in of the region in front of it, but to a considerable extent to the developmental changes which will very shortly entirely remodel this part of the embryo. The anterior slope appears as a part of the general dorsal concavity, the posterior runs down to the extreme end of the body close to the body-stalk, while its lateral surfaces fall off quite rapidly to the attachment of the amnion. Anteriorly and less deeply over its summit it is grooved by the neural folds, which extend about onethird the way down the posterior slope, of which a little less than the caudal half is occupied by the primitive streak. The last-mentioned region, in part at least, corresponds to the steep declivity so well marked in the Glaevecke embryo of v. Spee and the 1.3 mm. stage of Eternod. This portion, due to its active growth, will not only contribute in length to the embryonic body but will also bring about the so-called folding-off of its caudal extremity. The results of this process are the formation of the hind-gut, the allantois then appearing as an appendage of the gut instead of the yolk-sac, and finally the shifting to the ventral surface of the originally dorsally placed cloacal membrane. Around the posterior margin of the tail-bud thus formed, the primitive streak may persist for some time. These changes, due to an active outgrowth rather than to any passive folding, are just beginning in No. 391 and are well under way in Kromer's embryo.
Concerning the body-stalk and amnion a few words here will suffice. The former is very large and massive, in fact much more bulky than the entire embryonic body, and is roughly spindle-shaped with its smaller end attached to the chorion quite obliquely. Its shape and size are due to the enormous vessels which it contains, especially about its middle third, where it bulges prominently into the amniotic cavity. On account of its relations to the body-stalk, the amnion shows two deep, lateral recesses on either side of the stalk. From the left recess there passes out into the stalk a long, irregular, hollow outgrowth which we may identify as an amniotic duct (fig. 3). Its length, including the funnel-shaped origin from the general amniotic cavity, is about 0.6 mm. The duct crosses the body-stalk almost at right angles, close to the surface of the latter, and ends very near the distal extremity of the allantois and close to the chorionic surface of the body-stalk a short distance below the attachment of the stalk to the chorion. The lumen is very irregular, at times wanting, and is lined by cells which vary from low cuboidal to frankly cylindrical. Irregular thickenings or solid masses of cells also occur in the walls, some of which seem to have almost lost their connection with the duct. The whole structure is obviously undergoing retrogressive changes. Near the summit of the amnion there occurs a second small, slightly branched, but quite irregular diverticulum (not shown in the illustrations), which penetrates for some little distance into the substance of the body-stalk in the region of its attachment to the chorion. Tins slender, tubular structure is much smaller than the amniotic duct mentioned above; it is also less sharply demarcated from the surrounding mesenchyme, while its cells are rather more vacuolated.
Embryo and Ectoderm
The external configuration of the embryonic body is determined in large measure by the stage of development of the nervous system, which is here in the form of a groove, widely open throughout its entire extent. One may say that about onehalf of' this groove represents the future brain, and that at about the junction of this with the cord, or even a little in advance of this point, is where the flexure of the embryo is most marked. The primitive forebrain is especially prominent, being represented by two conspicuous crescentic folds, highest and broadest behind and approaching each other anteriorly, where they are separated by a shallow furrow. Behind, these foldsare convex, separated by a deep groove, and the brain-wall here is not especially thick. In front their median slopes are distinctly concave, passing gently into the central depression of the neural groove. A similar, even more marked concavity is found in this region in embryo No. 1201. It is at this point, in front, that the walls of the neural groove are thickest, indicating the position of the later optic pits. The median groove (the bottom of the neural groove) is continued over the anterior surface of the head, spreading out and terminating in the buccopharyngeal" membrane just above the attachment of the somatopleure. This portion of the brain, which we have identified as prosencephalon, is widest about its center, overhangs posteriorly the sides of the head ventral to it, and is placed at approximately right angles to the remaining caudal part of the brain.
Just behind the angle thus formed the brain is slightly narrower, but it soon increases again in diameter to form a spindle-shaped enlargement, the posterior limits of which are indistinct, This latter enlargement, which is not especially conspicuous, is the rhombencephalon, while the faint constricted portion in front of it we take to be the mesencephalon, although a part of what we have called forebrain, just at the angle, may later give rise to more or less of the mid-brain. In other'words, the cephalic flexure is very well marked at this early stage. A better term would be mid-brain flexure, since it is only the nervous system which is bent, and this over the anterior, blind end of the fore-gut. A straightening out of the body of the embryo will render this flexure even more typical and conspicuous. The appearance of the fore-brain is here quite different from the condition as usually figured, where the neural folds are represented as being strongly convex. The remainder of the nervous system, represented by the cord, is a deep, open furrow in the back of the embryo. Caudally, there is a rather sudden decrease in the thickness of the ectoderm forming the folds ; the groove also becomes very faint and shallow, but can be traced as far back as the primitive node. About the middle of the dorsal concavity, in the region which may be taken as upper cervical or lower medulla, the interval between the crests of the neural folds is less than elsewhere, and here the closure will first begin.
In the surface ectoderm on either side, about opposite the middle of the rhombencephalon, are two not very sharply delimited, moderate thickenings, which are the otic plates. They are in the main quite symmetrical, somewhat elongated, and measure about 0.08 mm in diameter. The basal surface of the ectoderm here is cleaner and sharper than in the adjacent body-wall and hence more like the neural folds. These thickened areas are somewhat more extensive than shown in figures 1 and 2, but all of the material may not be found later in the otic vesicles, some being utilized in the separation off and covering in of the same. As indicative of a ganglion crest we may note that toward the anterior limit of the rhombencephalon there appear two fairly distinct and symmetrical collections of cells, of rather short extent, capping the neural folds on either side — the primordia of the ganglia of the fifth cranial nerves (figs. 1 and 2).
At the very posterior end of the embryo, occupying the lower half of the caudal slope of this part of the body, is the primitive streak. From the primitive node to the cloacal membrane it measures 0.13 mm. in length and is marked by a broad, shallow groove. At the primitive node there is a rather diffuse, loose connection between the three germ layers; the ectoderm is here distinctly thickened and faint indications of the remains of the dorsal opening of an archenteric canal can be made out. The latter seems to penetrate about half way to the entoderm and may therefore be in relation with the posterior end of the chorda, but nothing definite as regards this can be made out. Ventral and lateral to the primitive streak there is an abundance of rather loose mesenchyme.
The anterior (later ventral) surface of the head is regularly convex, with the median groove and beginning stomodeum as already noted. On either side, where this surface passes into the lateral surfaces of the head, are two quite distinct vertical grooves, the one on the left being rather deeper, shorter, and nearer the median line. Internally there is, on the left side, a shallow, poorly defined extension of somewhat thickened entoderm toward the ectodermic groove. The lateral surface of the head region is marked on the left side by two distinct, vertical, parallel furrows (fig. 1). The anterior one is short and is simply a fold involving the entire thickness of the body-wall, projecting internally into the pericardial coelom. The other groove is much longer and if continued upward would cut the neural tube in the region of the mid-brain flexure. Corresponding with this posterior groove there is a similar one, on the right side, best marked low down, where its ectoderm lies close to the pharyngeal entoderm.
Entoderm and Derivatives
The gut-tract (fig. 3) is represented at this stage by a well-defined fore-gut and what is combined mid-gut and hind-gut, There is as yet no real hind-gut, since the cloacal membrane is in the roof, while just behind this the allantois comes off from a rather deep, laterally compressed, funnel-shaped outpocketing of the yolk-sac, the roof of which is formed by the entoderm in the region of the cloacal membrane and primitive streak.
The fore-gut, about 0.5 mm. in length, makes a very decided angle with the future mid-gut, due to the erection of the whole anterior end of the embryonic body. It is at this time quite large and capacious, determining, together wtih the neural folds and heart, the configuration of this part of the embryo. Dorsally, in the mid-line, chordal region, it is in close relation throughout with the thin floor plate of the neural groove. In general, the dorso-ventral diameter is distinctly less than the transverse; its anterior half is definitely enlarged, especially in its transverse measurements, the future oral and pharyngeal regions. The shape of the cavity of the fore-gut varies considerably in its anterior and posterior portions. In the latter, from just behind the first pocket and thyroid to its communication with the yolk-sac, it may best be described as triangular or even T-shaped on section. Internally the dorsal wall is concave in the median line and then passes out to the roof in the rounded lateral angles. Externally, in the roof of these lateral recesses, on either side of the median ridge, are two rather faint, broad furrows which may be termed aortic and which are lost anteriorly on the roof of the pharynx. The two ventro-lateral walls appear as if pushed in toward the center, with the result that there is present a deep median trough in the floor and two lateral somewhat shallower grooves on either side, but close to the roof of the gut.
Externally, the fore-gut shows a prominent ventral keel corresponding to the internal median groove and two laterally projecting ridges which fade out behind on the yolk-sac. On either side of this keel the entodermal walls are concave, the dorsal wall is in general convex, the opposite holding for the interior. This ventral keel is in intimate relation with the developing heart, projecting through as it were between the two layers of the wide mesocardium until it lies quite close to the endothelial tube of the heart, The two dorsally placed, lateral grooves or recesses in the gut-wall lie behind the attachment of the mesocardium and behind the dorsal wall of the pericardial coelom, projecting toward, but at some distance from, the beginning pleuro-peritoneal passages. Traced forward, the ventro-lateral walls of the gut open out as the space demanded by the developing heart gradually decreases. The dorso-lateral grooves likewise fade out as the result of these changes in the lateral walls. Just where these grooves merge into the enlarged anterior part of the fore-gut we may locate the earliest trace of the first entodermic pocket. On the right side there appears a small furrow, which extends for a short distance ventrally and very slightly posteriorly across the lateral wall, where it comes into relation with a second groove, which, if continued, would cut the median line just in front of the thyroid (fig. 4).
Externally in the ectoderm there is a well-marked linear depression of considerably greater extent than the small furrow just noted, but having the same direction. Here over an extent of about 0.04 mm. the ectoderm and entoderm are quite close together, no mesenchymal cells intervening, but the two layers are not in actual contact. Just in front of this groove lies one of the anterior, finger-like prolongations of the pericardial coelom, while still farther forward are found the beginnings of the ventral aorta and first arch. However, on this (the right) side there are other indistinct furrows in both the ectoderm and entoderm, but they sustain no relation to each other and we would look upon the condition described above as an early indication of the first cleft. On the left side this small ventral furrow is not as definite, the gut here being more evenly expanded. Externally, as previously noted, there is a groove in the ectoderm very similar to that on the right, but everywhere between it and the entoderm there is mesenchyme present. At no point is there any well-defined variation in thickness in either ectoderm or entoderm corresponding with these grooves. The right side is slightly in advance of the left and the same appears to be the case in the two other embryos we have mentioned, No. 391 and No. 1201.
The buccopharyngeal membrane is well defined, measuring about 0.05 mm. from side to side and slightly more from above downwards. The ectoderm and entoderm over this area are in intimate contact; the former shows here a definite increase in thickness over the neighboring ectoderm, while the entoderm is not locally changed, but is distinctly thicker over the floor and side-walls of the pharynx than elsewhere. Externally the membrane lies immediately above the reflections of the somatopleure from the body to form the anterior limit of the pericardial cavity. It occupies the floor and side-walls of a shallow depression, which is continued by a definite furrow over the anterior end of the head into the open neural tube. The membrane seems to be rather widely separated from the first cleft, i. e., the latter appears quite far back on the side of the head, but later development will approximate the two considerably.
In the midventral fine, just anterior to the point where the deep ventral trough of the fore-gut begins to widen out, is the primordium of the thyroid. It is a short, well-circumscribed outgrowth from the floor of the pharynx, indicated internally by a very short but well-defined lumen. In the model this outgrowth is located slightly to the right and directed also quite definitely in the same direction. The relation to the first internal pocket has been already noted. The thyroid is here in close relation with that part of the cardiac plexus which will later give rise to the ventral aorta and beginning of the first arch, in practically the same location in which we have described it for a much older stage (1907, p. 548). In No. 391 there seems to be an early thyroid in the same position as regards the heart, but this can not be determined with certainty on account of defects in the sections.
It is unfortunate that no definite statements can be made concerning the chorda, particularly as regards its anterior and posterior extremities. In addition to the unfavorable plane of the sections through the posterior two-thirds of the specimen there is (particularly in front of this) considerable loss of tissue involving the chordal region and the floor of the neural groove. When these regions are figured the defective tissues have been drawn in, but only for the purpose of completing the illustration. In the very roof of the pharynx, where the plane of sections is obliquely tangential, and so again most unfavorable, there are certain appearances which might be interpreted as indicating the presence of chordal tissue extending almost as far forward as the buccopharyngeal membrane. Elsewhere in the embryo there is a defect, probably more than a simple tear in the sections, involving the floor of neural groove, and also, but to a much greater extent, the roof of the gut-tract. Along this line, both within the gut cavity and externally in the bottom of the neural tube, is a considerable amount of cell detritus, probably largely chordal in origin. This disintegration of the chorda may be due to certain peculiar properties of the tissue. It might further be construed as evidence of a weak connection with the entoderm. Regarding the posterior end of the chorda, nothing whatever can be made out beyond the fusion of ectoderm and entoderm at the primitive node. In general the condition of the chorda is probably much the same as in No. 391, namely, a chordal plate, nowhere entirely free from the entoderm and blending posteriorly with the primitive node.
The allantois is especially well developed in this case, long and regular throughout its course. Its origin from the yolk-sac has already been noted. It enters the body-stalk, lying near what may be termed the caudal border of the stalk and surrounded on the other sides by large anastomosing blood-channels. The lumen is lined by low columnar cells which become more flattened distally, while the caliber gradually increases toward its free, somewhat flattened extremity, which lies very near the tip of the amniotic duct. The length of the allantois is 1.25 mm. and it is everywhere at a considerable distance from the amnion, but its relation to the exocoelom is quite different. Along what we have called the caudal surface of the body-stalk are two prominent longitudinal ridges due to the underlying arterise umbilicales. Underneath the deep groove thus formed, and situated between the vessels, runs the allantois. Here there is a prolonged area of contact between the mesothelium of the body-stalk and the epithelium of the allantois, about 0.3 mm. in extent or about one-fourth the entire length of the duct (fig. 3). The breadth of contact gradually increases as the distal, slightly dilated portion of the allantois is reached, where several epithelial cells are involved. Nowhere is there any interruption or loss of independence in either the epithelium or mesothelium, neither are there any apparent changes in the two cell-layers. What significance, if any, may attach to this we can not say; at all events it is not of regular occurrence. It may possibly be reminiscent of a larger vascular and functionating allantois, free in the exocoelom.
Immediately dorsal to the origin of the allantois from the yolk-sac, i.e., in the roof of the gut, or yolk-sac, and very close to the caudal limit of the amnion, is the cloacal membrane." It is in a way hardly a membrane, being rather a solid, cylindrical mass of cells, uniting the ectoderm and entoderm, much as in Wilson's embryo H 3 (1914).
As regards the yolk-sac, even approximate dimensions can not be given, on account of the extensive folding which has occurred here. It seems rather larger in the models than in the photographs and is absolutely and relatively larger than in No. 391, or in the Kromer specimen; in the latter the yolk-sac is quite small. Figure 3 shows a short, well-marked diverticulum of the yolk-sac very near the embryo. Its lumen is lined by high columnar cells, the whole knob-like structure projecting into a mass of vascular mesenchyme. If it were shifted forward a fraction of a millimeter into the septum transversum and nearer to the omphalomesenteric veins we would have, not one of the so-called glands or crypts of the yolk-sac, but instead, the very beginning of hepatic development. As it is, one is reminded of the hepatic function ascribed to the epithelial tubules and crypts of the yolk-sac by Graf Spee (1896) and others. All things considered, we think that the umbilical vesicle, so-called, may subserve functions other than those of a purely haematopoietic nature (cf. Eternod, 1909, 1913; Jordon, 1910). Very near the diverticulum just mentioned is a smaller, solid outgrowth of the entoderm into the adjacent mesoderm.
Mesoderm and Coelom, Segmentation
The mesoderm of the body of the embryo is in the form of two narrow, compact bands, lying between the walls of the neural groove, the surface ectoderm and the entoderm of the yolk-sac. Toward the neural folds it is best defined, its other limits being much less sharp. Laterally it becomes looser and is continued into the mesot helium covering the yolk-sac and amnion, while caudad it passes into the diffuse mesenchyme of the primitive-streak region, where it is continued across the median line. Cephalad these paired mesodermic masses break up to form the scattered mesenchyme of the head and are also very clearly in connection with the thickened pericardial mesothelium as well (text-fig. A). The mesoderm of the body (the two compact bands as above noted) is in the first stages of segmentation. Its medial border is rather uneven, but exhibits certain very definite, fairly symmetrical, deep indentations which will later separate off, partially at least, the somites of this region.
Near the mesial margin of the mesodermic mass are formed the beginnings of the so-called myoccels, very small or indicated only by the arrangement of the surrounding cells. Of these minute closed cavities there are three on the left side and two on the right, while between them are the indentations just mentioned. There is no indication here of a lateral extension of the myoccels, either to help form, or set up a secondary connection with, the intraembryonic ceelom, as is described by Keibel and Elze (1906) in the case of the second somite in an embryo of 1.38 mm. The embryonic ceelom is, except for the pericardial cavity and the small myoccels just noted, practically non-existent as yet, but a transitory or inconstant connection between it and the cavities of the somites may be established later. We have here, therefore, evidence of three segments on one side and two on the other; of these the middle or second segment on the left is the most distinct. The anterior limits of the first segment on both sides and of the third on the left and the second on the right are very uncertain (figs. 1 and 2).
The segments thus far differentiated lie just in advance of the center of the dorsal concavity and a short distance farther forward the unsegmented mesoderm becomes continuous with the walls of the pericardial cavity. As regards the nervous system, their location would be about the junction of brain and cord and we may look upon them as occipital somites. In the cervical somites which will form later, one may expect a better differentiation and sharper demarcation both between the individual somites and between these and the lateral mesoderm. Aside from the pericardial cavity (described later) and the myoccels, practically the only evidence of the formation of the intraembryonic coelom is found in the presence of a number of small, tubular prolongations of the exocoelom into the lateral mesoderm. They present very much the same appearance as shown by Dandy (1910), but are rather smaller. A few other unconnected spaces occur in the lateral mesoderm, forerunners of the embryonic body-cavity.
Vascular System
By far the most interesting and important results of the study of this embryo are those which pertain to early phases of vascular development. For the sake of convenience in treatment we may recognize four regions, viz., the chorion, bodystalk, yolk-sac, and embryo, which are also the various regions in which independent blood-vessel formation has occurred, or where this process is still more or less active. As will be seen later, these vascular areas are not only almost entirely independent of each other, but the primitive vessels within these various regions show certain differentiating characters.
Chorion
Blood-vessels are present in large numbers throughout that portion of the vesicle wall which forms a part of the series with the embryo (fig. 3) . The remainder of the chorion is also doubtless vascularized, but probably to a somewhat less extent. In the villi vessels are abundant as slender anastomosing channels or cords, continuous with the deeper vessels of the chorionic wall, presenting the picture of a loose, wide-meshed plexus. While for the most part forming a continuous network, there are certainly many detached strands, although, in view of the beginning abnormal cytological changes, which are more marked here than anywhere else in the specimen, this condition need not be looked upon as representing the usual method of development. The general swelling of the stroma in many of the villi may be in some measure responsible for the presence of what appear to be solid cords of cells rather than open vessels, although the latter do occur, often quite small but not infrequently of considerable size, particularly near the bases of the larger villi.
In the chorion proper, vascular channels are much more numerous and well defined, while the stroma is in a practically normal condition. The vascular picture here is in marked contrast with that in the villi, or indeed with that of all other parts of the ovum. There exists a rich plexus of vessels occupying the entire thickness of vesicle wall. They have in general an arrangement parallel with the surface and vary considerably in caliber. In contradistinction to the vessels in the villi those in the chorion are widely open, even though many of them are very small. That a few of them might be solid can not be excluded, but the appearance in all cases is rather an extremely fine lumen or simply an approximation of the walls. A second and quite conspicuous distinction in these vessels is their relatively thick walls, due to the increase in number and greater condensation of the cytoplasmic processes immediately around them, so that the vessels, aside from their lumina, are quite prominent features in a section through the vesicle wall. Near the attachment of the embryo these vessels are larger and rather more numerous and have already established a slender connection with the vascular channels in the bodystalk. Formed elements are found, with certainty, nowhere in the chorionic vessels except in the immediate neighborhood of the body-stalk. They are present here only in very small numbers, having made their way upward from the larger vessels farther down in the stalk. Hofbauer cells are occasionally present in the villi and abundant in the chorion, particularly near the attachment of the embryo.
Body-Stalk
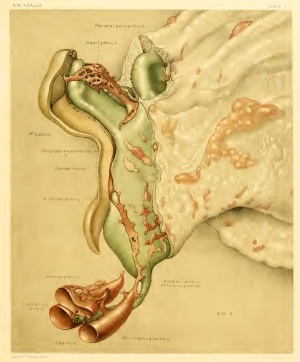
It is in the body-stalk that vascular channels show their greatest development at this stage, where, as has been previously mentioned, they are largely responsible for the size and shape of this structure (figs. 3 and 4).
About the level of the origin of the allantois, we may place the lower or embryonic extremity of the two umbilical arteries; that is, the limit of those portions of the arteries which are formed earlier than the part upon the yolk-sac, and in loco within the body-stalk. The vessel on the left extends to a slightly lower level than that on the right and is in adcUtion distinctly larger, although in both cases the arteries end quite abruptly instead of running out gradually as one or more smaller channels. For a very short distance these arteries are represented by spaces in the mesenchyme, devoid of a definite endothelial coat, but containing each a wellmarked blood island connected with the surrounding stroma by numerous cytoplasmic processes. A recognizable limit between a definite vessel and the apparently neutral mesenchyme is entirely absent, although the transition is to some extent bridged by the presence of blood islands. Followed distally, the left artery rapidly acquires a definite coat, although its lumen is yet almost entirely filled by a large, rather compact blood island attached to the walls by numerous cytoplasmic strands (fig. 9). Blood-islands have been noted in the body-stalk on many occasions, as by Debeyre (1912), Grosser (1913), and Bremer (1914). In our embryo they resemble the blood-islands of the yolk-sac much more than those of the body-stalk described by the investigators just mentioned, at least as far as we can make out from their descriptions or illustrations.
The right artery is not only much smaller at first, but on the side toward the embryo its endothelial wall is deficient for a short distance, where its lumen becomes continuous with an extensive, irregular, but not definitely lined space, by means of which an open connection is established between the artery and the posterior part of the vitelline plexus (fig. 10). This channel can be traced forward and inward through the wall of the yolk-sac and splanchnopleure until, as a much smaller but more definitely lined vessel, it becomes continuous with the small offshoot from the right aorta, the first vitelline (umbilical) artery. On the left side we can make out no connection between the umbilical artery and the vitelline plexus, although the latter shows much the same condition as on the right side except for the communication with the artery. There may well be some exchange of fluid through the loose t issue on the left side, but the more open anastomosis on the right may help explain the smaller size of the artery on this side, since a free flow to or from the yolk-sac is possible.
Traced away from the embryo, the blood-islands in the umbilical arteries gradually become smaller and looser, their connections with the vessel-walls grow less conspicuous, while the individual elements become rounded and more distinct and also fewer in number until both vessels are entirely empty. These early blood cells, where they are not especially numerous, are attached to the anterior wall of the vessel, i.e., on the side toward the embryo. In this location the wall is often less sharply defined than where there are no blood-cells forming, although such cells may be connected with a perfectly definite vessel wall.
Both arteries present, low down, a few very short branches which are usually much less distinctly lined than the main vessels. A little farther from the embryo a number of ventral branches are found which anastomose and increase in size until there is formed a plexus of large, irregular vessels entirely filling the bodystalk between the amnion in front and the umbilical arteries and allantois behind. Distal to the giving off of these branches, the arteries are somewhat reduced and also more nearly equal in size. They become, however, progressively larger, especially the left, but give off very few branches until, near the free end of the allantois, they rapidly lose their identity by extensive anastomoses with the venous plexus in front. Small, scattered masses of blood-cells are found in both arteries, being more numerous in the left. At the very distal extremity of the left artery, in the channels which connect it with the ventral plexus, there is again blood-island formation. The right artery, although it has many connections with the plexus, can be followed farther toward the chorion than the left.
Ventral to the umbilical arteries and the allantois which courses between them lies the dense, irregular vascular plexus referred to above. Beginning low down in the body-stalk, where it is connected with the arteries, it increases rapidly in size and complexity and at a higher level has incorporated in it the two arteries. Beyond this point of union the plexus dwindles very markedly, so that the distal portion of the body-stalk is occupied by but a few small vessels, through which a narrow connection is set up with the vessels in the chorion. The walls of the plexus are not always as sharply defined as in the arteries, being also distinctly thinner and much less conspicuous. Small anastomosing channels, offshoots from the plexus, are especially in evidence immediately around the allantois. Blood-cells are present to about the same extent as in the arteries. No direct connections exist between the plexus (the future umbilical veins) and either the yolk-sac or the beginning umbilical veins which form in the body of the embryo. This latter communication doubtless forms at a slightly more advanced stage. It is at the chorionic end of the body-stalk that the greatest transformation would be required to bring about separate arterial and venous channels, instead of the single set of vessels which occupies this location at present.
The stroma of the body-stalk varies somewhat in different localities. It is densest and most cellular close to the embryo'; at a little distance its cellular content becomes much less marked, while the cytoplasmic reticulum is very conspicuous. Along the posterior border, near the allantois and the umbilical arteries, this tissue is always much less rich in nuclei than around the plexus in front. Often the wide space between the arteries, allantois, and the investing mesothelium is bridged by a very delicate network of cytodesmata almost devoid of nuclei. At the smaller, chorionic end of the stalk, where the venous plexus is running out, the stroma gradually loses its delicate reticular appearance, becomes denser, and assumes a more fibrous aspect much like the mesoderm of the chorionic wall. The vessels in this part of the stalk, which establish the slender and circuitous connection between the plexus and the chorionic vessels, are quite small and thick-walled, and so in both respects more like those of the chorion than those of the body-stalk. They may be looked upon, therefore, as ingrowths from the chorion to meet the independently formed vessels of the stalk. Over the distal part of the body-stalk the mesothelial investment is gradually lost, and in fact close to its attachment to the chorion the limits of the stalk fade almost imperceptibly into the adjacent coagulum in the exocoelom. From the mesothelium there are found short, tubular or funnelshaped ingrowths, but these are few in number and have no relation to the underlying vessels.
Yolk-Sac
On account of the extreme distortion of the yolk-sac, due to folding and partial collapse, it is not always possible to locate exactly many of the vascular rudiments. Blood-islands are most numerous in the region of the fundus and are also scattered sparsely over the remainder of the vesicle, being rather more frequent laterally and posteriorly, while a few are found quite close to the embryo. Vessels in various stages of development are most conspicuous in these same areas, but no special attempt has been made to trace them or to determine their relations more definitely. On what appears to be the posterior surface of the sac, there are a considerable number of large, well-defined channels which are not even remotely connected by open vessels with those close to the embryo. As the embryo is approached, all the vascular features become less frequent, more scattered, and more difficult to recognize, especially lateral to the anterior part of the embryo. Any connection between the vessels or heart of the embryo and those of the adjacent yolk-sac anteriorly by way of the vitelline (omphalomesenteric) veins is at best very attenuated and indirect; a continuous, open endothelium-lined path is not present; for, whatever interchange of fluids may be going on between the embryo and yolk-sac in this region, there are no corresponding morphological features to be recognized.
Posteriorly, however, nearer the attachment of the body-stalk, conditions prevail which may be described as a vitelline plexus (fig. 4), much better developed here than farther forward. This plexus is made up of short, open endothelial channels, solid cords, scattered angiocysts, and occasional small blood-islands, with the better-developed vessels exhibiting a tendency to assume a course at right angles to the axis of the embryo. It is impossible to determine the extent to which these various vascular primordia may be united at this stage to form a more or less continuous network. Conditions here are more like the "rete periintestinale" of Felix (1910) than those shown by Bremer (1912) in the rabbit. The posterior and mesial limit of the vitelline plexus on the right side is formed by a large, fairly definite channel, for the most part quite unconnected with the general plexus in front of and lateral to it. Near its origin on the yolk-sac it exhibits a small, patent, but not yet definitely walled communication with the umbilical artery in the body-stalk. Traced inward and forward in the splanchnopleure it becomes progressively larger, its proximal portion being a distinct endothelium-lined tube. It possesses here a few short sprouts and then, considerably reduced in size, turns inward to join what seems to be the posterior end of the dorsal aorta (fig. 4) . Just where it passes inward to meet the aorta, a small branch runs forward which is apparently destined to establish a second, more anterior connection between the aorta and vitelline plexus. Both the aortic and the vitelline ends of this latter vessel are in evidence, but the intervening portion is rather problematical, at best a slender cellular connection. We have here a nascent vitelline artery, the second of the series of roots of the future umbilical artery. On the left side no indication can be discovered of any connection between the aorta and the plexus. There is, however, a well-marked blood-channel already laid down on the periphery of the plexus, but still lacking its connections with the aorta in front and the body-stalk behind. At the aortic end a communication may be in process of formation by means of single cells, but the line of section is much less favorable for determining this than on the right side.
As just noted, the anterior part of the vitelline plexus is much less developed than the posterior. A connection between the former, the radicles of the vitelline or omphalomesenteric veins, and the proximal portion of the vein as it lies in the septum transversum, is either absent or so small as to escape detection. If there is an interruption it occurs just at the lateral border of the embryonic body. Substantially the same conditions are present on both sides. If not already present there will be established here very soon a second connection between the intraembryonic and extraembryonic vessels, in this case by way of the venous end of the heart. Apparently the posterior (aortic) connection antedates the anterior or omphalomesenteric union.
Heart and Embryonic Vessels
In considering the cardiac plexus (figs. 4, 7, and 8) we may begin with the omphalomesenteric roots, single on either side, beginning in the septum transversum near the lateral border of the embryo. Passing inward and forward through the septum, beneath the dorsal recesses, they turn cephalad under cover of the myoepicardial mantle and unite with each other to form a single median mass. Of the two vessels, without regard to their structure, whether hollow or otherwise, the left is rather larger than the right. Small where they may be called the omphalomesenteric veins, and again where they unite in the median line, they are in the interval of a plexiform nature, slightly more open on the right, and dip down on both sides as far as the lowest limit of the myoepicardial mantle. Two small cross anastomoses are present between these caudal halves of the heart plexus. About its middle and for a short extent the plexus is essentially a single median mass, fairly uniform and irregularly stellate or crescentic in section. Cephalad it broadens out rapidly, becomes more complicated and very markedly plexiform, while its constituent elements again assume a roughly symmetrical, bilateral arrangement. At the same time it begins to extend around onto the lateral surface of the pharynx. At the level of the thyroid, high up under the mantle, the cardiac mass breaks up into two roughly symmetrical portions (the right more markedly plexiform) which pass cephalad and dorsad over the ventre (-lateral surfaces of the pharynx and rapidly run out on either side on the roof of the fore-gut near its lateral margins. These structures represent the first two aortic arches and are perfectly definite endothelium-lined channels. They cross the lateral wall of the pharynx some distance in front of what we have called the first entodermic pocket, about midway between this and the buccopharyngeal membrane. At their plexiform origins both arches are sharply limited endothelial channels lying close to the pharyngeal entoderm. From the anterior convexity of the course described by these vessels there is given off a short sprout, the beginning of the internal carotid. Beyond the origin of the carotid the dorsal aortse can be traced for only a very short distance, when they fade out in the aortic grooves on the roof of the fore-gut in the region dorsal to the first pocket. There are no indications of a second arch unless one chooses to put such an interpretation upon certain irregularities found along the lateral border of the plexus in its anterior third and at the base of the first arch.
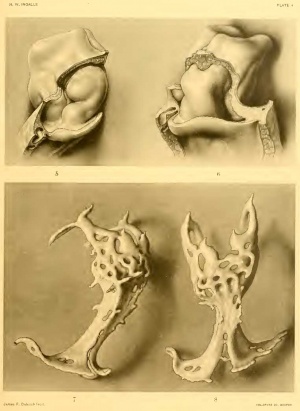
Concerning the structure of the heart plexus (text-figure A and fig. 11) a few words are necessary, since the reconstructions give no adequate expression of its essential characters. Only the lateral extremities of the omphalomesenteric veins and the anterior, bifurcated end of the plexus, where the first arches take origin, are genuine endothelium-lined vessels. Between these points the heart is represented by a very irregular, cellular mass, roughly crescentic in section, its concavity directed backward toward the fore-gut, The illustrations do not bring out fully the great irregularity in the surface of this mass, since it has not been possible to reproduce the numberless cell processes with which it is thickly studded. The entire cardiac plexus is extensively vacuolated, the cavities being for the most part separate. Toward the arterial end of the heart these spaces become more frequent, larger, and more often confluent, There is in this stage a freer pathway for diffusion currents outside the plexus, in the wide space underneath the mantle, than there is within the plexus itself, only the two extreme ends of which are pervious. A certain similarity obtains between conditions represented here and those shown by Parker (1915, plate I, fig. 5) in a 15-16 somite embryo of Perameles nasula, but only as regards the general form of the heart, since the vascular development at this stage in Perameles is comparatively very far advanced. In her text-figure 14 the heart in cross-section is quite like that of our embryo, except that it presents two separate endothelial tubes instead of a single angiocystic mass. One may conclude that in man the two halves of the heart fuse at a very early period in their development, if there is actually a stage in which they are two such independent structures. It is conceivable that in the heart before us we might later have found two quite independent endothelial tubes, but the probability seems very remote. In man fusion antedates the complete canalization of the heart, while in most forms the reverse is the case (Parker, 1. c; Bremer, 1912; Schulte, 1916; Wang, 1918).
As regards the actual processes which give rise to the lumen of the heart, the same principles obtain in man as have been described in the cat by Schulte (1. c). ( ompared with endothelium formation elsewhere, there is in the heart a relatively large amount of mesenchyme and very little endothelium. We can find nothing indicative of the origin of this mesenchyme or of its being increased in amount except by its own proliferation. It is everywhere at a considerable distance from the myoepicardial mantle, but its most caudal extensions lie very close to the entoderm. The entire heart has obviously arisen in loco and not by the invasion of vasculogenic elements from without. It is not possible to say to what extent the heart, as shown in figures 7 and 8, is to be derived from a single paired or even unpaired primordium by a simple process of growth; there has doubtless been not a little accretion of angioblastic material, and this probably more extensive at the cephalic end than elsewhere. The continued formation of angiocysts, their extension and coalescence, will soon transform the impervious mass as it now exists into an open passage between the omphalomesenteric veins and the first aortic arches. By this time we might expect to find a free communication between the venous end of the heart and the anterior part of the vitelline plexus and also a dorsal aorta continuous and patent throughout its extent.
Posterior to the first pocket the dorsal aorta? are in process of formation, as evidenced by the presence of a number of small vesicles, or even less definitely limited spaces, in the rather dense mesenchyme close to the roof of the fore-gut. About opposite the intestinal portal the right aorta gradually becomes more evident and can be traced backward, lying close to the entoderm, as far as or possibly a little beyond the point where it has its lateral connection with the posterior portion of the vitelline plexus, and thence with the artery of the body-stalk (fig. 4). Toward its posterior end the aorta is distinctly dilated and there is here a possible second, more anterior vitelline artery in process of formation. Anterior to this dilation is a small, much constricted, but probably pervious segment of the vessel. From the intestinal portal to the vitelline artery the aorta is for the most part a definitely lined channel; the constriction just noted may possibly be solid, and also near the origin of the vitelline artery its walls are, in a few places, almost deficient. Whether its posterior end is open or really closed off is very difficult to determine. Beyond the latter point a few scattered but conspicuous cells are found, quite different from the other cells near them, especially as regards their larger, longer, and more deeply staining processes. They are to be looked upon as material for the further growth and extension of the aorta or its branches. Situated in the line of the aorta and laterally toward the yolk-sac, these cells show the orientation described for similar cells in the cat by Schulte (1914), forecasting, as it were, the long series of vitelline arteries which is to follow. There are in addition, also at the posterior end of the aorta, indications of a very few short, mesially directed branches, not always closed off from the wide intercellular spaces adjoining, which represent the first dorsal rami of the aorta. The exact location of the vitelline artery can not be determined; continuing the segments backward would place it about the region of the sixth, the future third cervical. Felix places the anterior connection with the aorta at the seventh segment, in the stage of 5-6 somites, while in a 2.6 mm. embryo with 13-14 somites this connection has shifted back to the tenth segment on the right side and beyond the twelfth on the left.
On the left side certain minor differences are to be observed. In the region of the somites the aorta is larger than on the right. This is followed by a short stretch where the vessel is possibly interrupted but represented, at least in part, by a solid chain of large cells. Its posterior end is enlarged like that on the right, but, instead of being open, is occupied by a small blood island (fig. 12). The conditions here must be essentially the same as those described by Miller and McWhorter in the chick (1914), where the posterior third of the aorta on the operated side is represented by an elongated, cord-like blood island, whose cells "are identical with the cells in the blood islands of the normal area opaca " (I.e., p. 209) . In this particular case, as well as in our own, the diffusion, not to mention any circulation, must be extremely tardy in this solid vessel. With the establishment of even very slender connections, such as are present on the opposite side in both embryos, and the ensuing rise in the rate of fluid interchange, we may expect a certain change in the appearance of the blood islands such as are found in the umbilical veins of our own embryo, or their gradual disappearance as well. The only formed elements which we have found in the embryonic vessels, aside from the blood island just noted, are a few cells in the caudal portion of the right aorta, both conditions recalling the cell clusters described in this vessel at much later stages in other animals (Emmel, 1916; Jordan, 1917). As regards branches on the left side, very little can be made out. The same angioblastic cells are present as on the right side, but they are rather less conspicuous. If there is a vitelline artery already laid down here it is very indefinite and located at the caudal end of the aorta in the region occupied by the blood island. Both aortae lie very close to the entoderm; the wide space intervening between these vessels and the mesoderm is bridged by great numbers of cell processes of varying size and shape and is also comparatively free from nuclei. This space is rather wider than that between the mesoderm and ectoderm. The surface of the mesodermic mass toward the entoderm is much more irregular and there are more isolated mesenchymal elements here than underneath the ectoderm. From the mesoderm there are undoubtedly many cells migrating out, where they may very easily come into intimate relation with the aorta or assist in forming some of its first branches (Schulte, 1914).
It is not only in the splanchnopleure that vasculogenesis is in progress, but also, although to a much less extent, in the somatopleure as well. There is to be found on the somatopleure on the left side, very near its reflection to form the amnion, a very definitely lined, thin-walled vessel — the left umbilical vein (figs. 2 and 6). It is of considerable antero-posterior extent and in its cephalic portion is a very sharply defined channel, but with extremely thin walls. At this end it runs out as a solid spindle-shaped sprout in a region almost devoid of cells. Its posterior extremity is gradually lost in the loose mesenchyme under the ectoderm. The vein presents two or three mesial branches, more distinct in front, through which a connection will be set up with the earlier formed omphalomesenteric root of the heart, the vitello-umbilical vein. Conditions on the right side are too uncertain to warrant description. On both sides, however, there occur, farther back, scattered vasofactive cells and small angiocysts in the somatopleure, or even in the amnion close by, marking out the course of the umbilical vein backward toward the body-stalk. At present, however, the embryonic part of the umbilical vein is entirely isolated and independent. A few short branches come off the venous plexus of the bodystalk at its lower ends; these are more conspicuous on the right side and may aid later in completing the afferent umbilical channel.
Myoepicardial Mantle and Pericardial Coelom
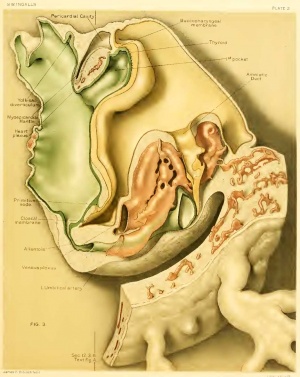
The pericardial cavity and myoepicardial mantle, the latter with its contained plexiform heart already described, are of particular interest at this stage (text-figure A, and figs. 5, 6, and 11). In transverse section the cavity throughout is crescenticin outline and corresponds, in its antero-posterior extent, quite closely with that of the fore-gut. The floor of the cavity is concave, narrow, and roughened internally by a number of small, radiating ridges in the thickened mesothelium. The cephalic limits of the cavity project upward as two narrow slits on either side of the fore-gut (or mouth) lateral to the buccopharyngeal membrane. Similar diverticula of the pericardial cavity have been noted in a 4.9 mm. embryo (Ingalls, 1907, 1. c, p. 553). The recess on the right side is somewhat more extensive, terminating in three fingerlike prolongations. As the dorsal wall of the cavity we may describe the region about the two extremities or horns of the crescent which the cavity presents on section. Cephalad this is nothing more than the bottom of a narrow cleft between the splanchnic mesoderm (myoepicardial mantle) and the somatopleure. Farther back this cleft gradually widens out, especially on the right side, due in part to the narrowing of the mesocardium — a sort of undermining.
This increase in width continues to a point about opposite the slight constriction which can be seen in the myoepicardial mantle. Just below, i. e., posterior to this, there is a sudden increase in the transverse dimensions of the cavity, due to the presence at this level of two deep, well-marked, dorsally or dorso-caudally directed, roughly funnel-shaped diverticula which penetrate for some distance into the mass of mesoderm which farther back is beginning to break up into segments. These diverticula take origin from the widest part of the pericardial coelom and moreover close to its lateral wall on either side. Mesially they are in relation with, or even extend dorsally beyond, the dorso-lateral grooves of the fore-gut. Of the outpocketings just noted (recessus parietales dorsales) the right is better defined and more extensive in all its measurements, and is possibly situated a trifle more cephalad than the left. As far as can be determined they are yet blind, having effected no connection with the coelom elsewhere, although we can not deny the possibility of such a connection on the left side. From the deepest point of the recess on the left side there extends backward for a short distance into the paraxial mesoderm (that is, into a region definitely lateral to that in which more caudally the myoccels are to be found) a very slender cleft which possibly, at its posterior end, is in relation with some of the coelomic spaces already noted (p. 56) as ingrowths, in part at least, from the extra-embryonic coelom. A second less notable cleft appears at a level slightly more caudad. On the right side essentially the same picture is presented as described above, except that the smaller, more caudal extension is missing. As may be seen, the dorsal recesses are much nearer the floor than the roof of the pericardial cavity; later this relation is reversed. However, it may be noted in passing that the upper part of the pericardial cavity is in relation with vessels which will later come to lie without this cavity, viz., the aortic arches. Under the inner, mesial wall of these recesses lie the dorso-lateral grooves of the foregut as previously noted; the future pulmonary anlage is more ventral and possibly also farther cephalad. Anteriorly they are roofed in by the somatopleure near its reflection to form the amnion. Their floor below is splanchnopleure, which mesially and ventrally forms a sort of sling, constituting the floor of the pericardial cavity, the whole being the septum transversum, around the dorsal border of which, at the anterior intestinal portal, the fore-gut and mid-gut communicate.
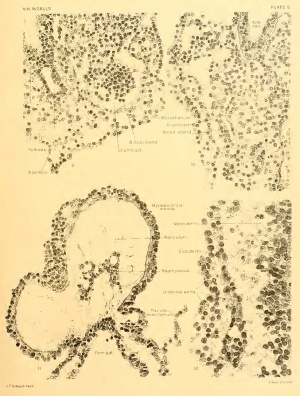
Figure A. Section 12-3-6, X90. The line of this section is shown in figure 3 on plate 2. Transversely the plane of section is somewhat oblique, so that structures on the left of the median line (above in the illustration) are cut at a higher level than those on the right. The section cuts just behind the primitive node, through the anterior intestinal portal and approximately, though obliquely, through the middle of the heart. On the right in the figure appears the body-stalk with the two umbilical veins and allantois between them. Three cavities are seen: above in the illustration is the amniotic cavity; below is the yolk-sac, communicating anteriorly with the foregut; farther forward, in the median line, is the pericardial coelom with its contained heart. The central, densely cellular mass is a tangential section of the left neural fold. Between the entoderm of the foregut and the superficial ectoderm, the mesoderm can be traced forward to the point where it splits to become continuous with the visceral and parietal layers of the pericardial cavity. (Cf. also text and fig. 11, plate 5.)
Into the pericardial cavity, and indeed determining its shape although by no means filling it, projects the myoepicardial mantle. Its ventral and lateral surfaces are free; above and below it is reflected back as the parietal mesothelium, while dorsally this same reflection constitutes the posterior mesocardium. The two layers of the mesocardium are, however, widely separated, for between them lies the ventral keel of the fore-gut, even beyond what may be considered as the ventral limits of the mesocardium, while farther cephalad is the bulging floor of the pharynx. In the region of the dorsal recesses the two layers of mesocardium are most closely approximated but still separated by the fore-gut. It is this undermining which will soon free the heart except at its two ends, the communication thus established between the two sides persisting as the transverse sinus of the pericardium.
The myoepicardial mantle or, to be brief but less exact, the heart, is practically straight and of almost uniform width, but slightly broader below; its ventral, tree surface is distinctly convex antero-posteriorly, as well as from side to side. This ventral surface is rather more strongly convex below than above, and again along its left margin, and passes insensibly into the lateral surfaces. Of these the left is the simpler, presenting caudally and dorsally a slight bulging due to the undermining just dorsal to it, The right side is throughout more strongly convex and is traversed about its middle by a deep transverse furrow below which the heart is distinctly thinner dorso-ventrally. The ventral end of this furrow is continuous with a much shallower, broader groove which passes caudad and slightly mesad over the ventral surface of the heart, The deep furrow on the right, together with a much more diffuse but roughly corresponding depression on the left, brings about a slight constriction of the heart about or a little below the middle. This constriction corresponds approximately to the anterior part of the narrowest portion of the cardiac plexus. The relief of the right side of the heart is in marked contrast with that on the left, in that the former shows a well-marked bulging above the transverse groove, while below this groove is a small, but very distinct, shoulderlike prominence. On the left side is a slight prominence anteriorly which is separated by a gentle concavity from a more pronounced elevation at the caudal end of the heart. It must be remembered that what we have just described as the heartnamely, the myoepicardial mantle— particularly the very anterior end of it, has its form determined far more by the pharynx beneath it than by the vascular primordia wit Inn. To what extent the form of the mantle and its external relief are influenced at this stage by the structures beneath is a question, but it would seem that the development of the mantle is in advance of the endothelial elements within, and so, in a measure, independent of them. Further, there are found here not only the future heart, but also the ventral aorta and the beginning of the first arches.
In a structure relatively so short and really comprising at one end less and at the other end more than the future heart, one can not expect to find much indication of the later differentiation of the tubular heart, The moderate constriction in the mantle described above really does not involve the ventral surface of the heart, If we identify the deep incisure on the right side as the beginning bulbo-ventricular groove, then! but only as regards the mantle, the atrial and descending portions of the ventricular loop are conspicuously shorter than the remainder from which will arise the ascending limb of the loop and the bulbus. This prominent shoulder on the right is very similar to that shown in cat embryos (Schulte, 1906), except that here, due to the late fusion of the mantles, the same prominence with the bulbo-ventricular groove above is formed on both sides, in each half of the myoepicardial mantle. In the heart of Xo. 391 (2 mm.) as shown by Mall (1912), the groove is farther from the attached, venous end of the heart and is, moreover, in its proper position on the left side. Were the relations of the early endothelial tube within more in accord with the form of the mantle one would feel less compunction in hanging on this little projecture on the right side of the heart speculations a propos of an incipient dextrocardia or situs inversus, without, however, being able to offer much more to substantiate such a claim, or anything by way of explanation.
Careful examination of the myocardial wall fails to reveal anything which might be construed as distortion or post-mortem change of any kind. In the region of the groove there is a slight thickening of the mantle and its free, coelomic surface is distinctly more irregular than elsewhere from the heaping up of its cells. At the dorsal end of the bulbo-ventricular groove is a deep triangular depression continued forward and backward along the line of the mesocardium. On the left side the undermining of the mantle is well marked only in its caudal half, the deepest point being distinctly below, caudad to the center of the mantle. The establishment of a connection here across the median line, the future transverse sinus of the pericardium, awaits only the withdrawal of the ventral keel of the fore-gut. This pushing inward of the mesocardium at a definitely more caudal point on the left side (on the right this process is more extensive but also more anterior), together with the deep groove on the right side and the fact that the ventral, transverse convexity of the mantle is sharpest toward the left side of this surface, leads one to the conclusion that there is developing in this case a reversed ventricular loop — that is, a loop directed toward the left instead of toward the right. Another bit of evidence which might be adduced in this connection is the fact that the right lateral border of the cardiac plexus is more concave than the left, while the median fused portion of the plexus shows a very definite keel, directed ventrally and to the left. This last-mentioned character is most noticeable on section, as if the endothelial structures were reaching out toward the left, anticipating the formation of a ventricular loop whose apex will be directed toward that side.
As regards the relations of the myoepicardial mantle and its inclosed plexus, the evidence here also points to a beginning reversed ventricular loop. The shallow sagittal groove already noted on the ventral surface of the mantle in its caudal third is situated slightly to the right of the open space between the omphalomesenteric roots; in other words, it overlies the left border of the expanded portion of the right venous root. This sagittal groove is located to the right of the median line. Traced upward, it turns outward, toward the right, to become continuous with the ventral end of the bulbo-ventricular groove. Tin's shallow, sagittal furrow will probably later be completely effaced, since we can hardly see that it might represent the atrio-ventricular constriction. Whether or not it is an indication of a possible earlier and greater independence of the two halves of the mantle, which appears doubtful, must remain for the present an open question. At the point where the grooves become confluent they are situated to the right of the plexus, so that the shoulder on the right is quite out of relation with the plexus, containing only the fine fibrillar network to be described later. On the whole, then, the endothelial component of the heart is shifted a little toward the left within the mantle, besides showing a slight but definite bowing toward the same side. While obviously the plexus must follow the bending of the tubular mantle, made possible by the early breaking through of the mesocardium, there is nothing at present to prevent the plexus from undergoing considerable changes in position within, and independent of, the mantle. All things considered, one can not well avoid the conclusion that we have before us about the earliest recognizable stage of dextrocardia, infrequent enough in the adult, to which might have been added later a more or less complete situs inversus viscerum.
The apparently low, caudal location of the bulboventricular groove is to be explained by conditions under the upper part of the mantle. There is found here, yet in relation with the pericardial cavity, the anterior, most obviously plexiform segment of the so-called heart plexus, which represents, however, the ventral aortae, first arches, and very possibly other aortic arches (Bremer, 1912), as well as the distal extremity of the bulbus. This portion of the mantle is also much thinner than that farther back, where it will form the wall of the ventricular loop, and the protoplasmic network is distinctly less delicate and regular.
As regards the future subdivisions of the endothelial portion of the heart at this stage, little more than their obvious spatial sequence can be recognized. In its caudal, expanded third is represented the sinus venosus, the wide transverse extent of which is already an accomplished fact. There remains only the complete fusion of its two lateral components, already inaugurated by two small connecting channels. The anterior third of the endothelial heart is frankly a plexus, the distal portion of which is made up of bilaterally arranged, open endothelium-lined vessels. From this segment must be derived the termination of the bulbus, the ventral aortae, and the anterior aortic arches. The remaining unpaired middle third of the heart plexus contains, therefore, the bulk of the future organ, aside from the mantle. Any definite limits can not be given, but in the formation of the ventricular loop which will arise from its rapid growth and elongation, it seems probable that the caudal third of the plexus will be encroached upon more and drawn into the ventricular loop, as atrium, to a greater extent than the anterior third will be incorporated into the bulbus.
Between the myoepicardial mantle, and the endocardial primordium within, there exists a wide space winch is more extensive and characteristic in the ventral and lateral regions than in the region dorsal to the heart plexus (text-figure A and fig. 11). This interval is bridged over by innumerable fine protoplasmic fibers which have their origin, or attachment, on the mantle externally and the surface of the plexus internally. They were described long ago by His, and more recently by Mall (1912), and there can be no doubt but that they represent an essential and perfectly normal constituent of the embryonic heart, It may be granted, of course, that their appearance and arrangement may be altered in a variety of ways by reagents or post-mortem changes. The extent and importance of these intercellular and interepithelial connections of early development (the eytodesmata) may be seen from the detailed accounts of Szily (1904, 1908) and Held (1909). Taken en masse, as they are found throughout the embryo, they make up the mesostroma of Studnicka (1911, 1912). Aside from their obvious office of retaining the early cell masses and layers in proper order and arrangement (Szily), they would also afford adequate means for the necessary diffusion of fluids at this stage, while later they may play an important role in connection with developing nerve paths (Held). The embryonic heart offers an example of the special development of the eytodesmata just mentioned. In the particular case in question their arrangement is essentially radial, extending from the future endothelial tube within to the under surface of the mantle externally, and also, but to a slightly less extent, to the ventral wall of the fore-gut behind. As a rule, the course described by the fibrils is rather wavy and irregular, often simulating a loose, much drawn out reticulum, and they show a distinct tendency to clump together, especially internally, where they spring from the various prominences and ridges of the plexus. That they exert any traction upon the nascent endothelium, or later upon the heart tube, drawing them out at divers points as has been suggested, is altogether possible, but appears to us unlikely. Often between the fibrils there occur regular, open spaces, possibly to some extent of the nature of artefacts, and where there are cells in close relation with them they may simulate angiocysts. Identical conditions are figured by Szily in the chick heart (1. c, 1908, figs. 5 and 8). A large space of this kind is found quite constantly in the dorsal concavity of the plexus, between it and the fore-gut. The great development of such a protoplasmic, intercellular network between the myocardial and endocardial layers of the heart wall is doubtless to be looked upon as a preparatory feature, facilitating or initiating the later invasion of the space thus occupied by the ventricular myocardium on the one hand and, on the other, by the endothelium at various points in the formation of the endocardial cushions and other connective tissue of endothelial origin. Toward the anterior limits of the myoendocardial space the characteristic arrangement of the fibrils is gradually obscured; they become more irregular, less numerous, and the whole picture is that of a coarser, less orderly framework, until it finally becomes indistinguishable from the ordinary mesostroma of the neighboring somatopleure. The arrangement of the reticulum in the heart under consideration is quite different from that shown by Szily (1. c, 1904) in the heart of chick embryos, where the network is looser and the fibrils are arranged without any special regularity, no predominently radial disposition being in evidence. Similar differences are obvious in the section through the heart of a 15-somite human embryo as given by Tandler (1912). Here, although it is of a much later stage, the network, as in the chick, has about as indefinite an arrangement as could be desired. The nearest approach to the conditions present in our case are those shown by the former writer, but taken from the bulbus cordis of a 32-hour chick embryo (1. c, 1908, figs. 5 and 6).
The inner basal surface of the mantle, to which are attached the fibrils, is remarkably smooth and even, more so in fact than the same surface on any of the other cell-layers of the embryo except the medullary folds and otic plates. Closer examination of this surface, however, reveals a fine cremation of the edge on section, representing the points of union between the fibrils and the mesothelial cells of which they constitute a part (cf. Szily, 1. c, 1908, fig. 6). Anteriorly, where the fibrillar network loses its delicate, uniform characters, the under surface of the mantle suffers in a similar way, becoming very irregular and uneven. The variations in the nature of the mesostroma in different regions of the embryo are essentially quantitative in character. All conceivable gradations are represented, as may be seen in the examples furnished by Szily. Free cells or small cell-masses in the reticulum are practically restricted to the anterior third of the plexus, and here again they are more in evidence around the caudal limits of the plexus, where considerable remodeling will take place, than farther forward where definite bloodvessels represent the beginning of the first arches.
The free, coelomic surface of the mantle is relatively rough and uneven ; the cells are frequently clumped together in small (at times hollow) masses projecting into the pericardial cavity. The mantle is thickest behind and gradually thins out to the line of reflection as the parietal layer at the top of the pericardial cavity. Details of its structure can not be made out; in its anterior portion there is but a single layer of cells; elsewhere and particularly low down there may be more than one. Nowhere, however, is there any break in the clear-cut basal surface of the mantle or any indication on the part of its cells of an invasion of the underlying reticulum. The parietal layer of the pericardial mesothelium is a single layer throughout. Thinnest ventrally, it becomes thicker over the lateral walls, while dorsally and where it forms the mesocardium its cells are cuboidal or low columnar in type. In the dorsal recesses, where they are expanded to join the general pericardial cavity, their walls exhibit considerable irregularity in the form of deep, narrow clefts or tiny out-pocketings lined by low columnar cells.
A consideration of the vascular features presented by this particular example of early human development confirms one in the idea that the ability of the mesoderm to give rise to vascular endothelium, at least at certain stages and in man, is practically coextensive with the limits of this category of embryonic cells, indicating a certain catholicity in tliis respect, As regards free blood-cells, the case is apparently somewhat different. There are, as previously noted, four separate regions in which blood-vessels arise; their connections are secondary, being established at various times and in a number of chfferent places. These regions are the chorion, body-stalk, yolk-sac, and embryo. To these might be added the amnion, unless one draws the line between this membrane and the somatopleure so as to leave all the vasofactive material, such as the angiocysts of the umbilical line, well within the embryonic body. The sites of blood-cell formation are most widely distributed in the yolk-sac, while in the body of the embryo they seem to be very sharply restricted (aorta). (Closer phylogenetic relation of the dorsal aorta with the vitelline plexus.) In the chorion there are, at present, no indications of the production of formed blood elements.
In the body-stalk and chorion the process of vessel formation, by accretion and accessions from without, may already have been superseded by the direct growth and extension of preexisting vessels. In the yolk-sac and embryo both processes are still going on hand in hand. There remains, however, much to be done in the way of linking up, not only the intraembryonic vessels with each other and with the heart, but also in establishing relations between the intrasomatic and extrasomatic channels by way of the heart, aortae, and umbilical veins. There is an unmistakable precocity in the vascular development in man, a certain element of haste almost, so that after a given stage has been reached the ground plan of the embryonic circulatory system is sketched out with all possible expedition. Obviously, the most promising method of procedure would be to begin construction at every suitable point. The paramount importance of an adequate circulatory mechanism, as well as its extreme susceptibility, is too well known to call for comment. It is likewise unnecessary to rehearse the evidence that the vessels of the embryonic body are largely formed in loco instead of by any invasion from without (Halm, 1909; Miller and McWhorter, 1914; Schulte, 1914; Reagan, 1915; Sabin, 1917).
In the particular case in question three main groups of embryonic vessels may be recognized — the heart, aortae, and umbilical veins, each group completely isolated, only the aorta having a connection with the extraembryonic vessels and this with certainty only on one side. The aorta and umbilical vein are both still in process of formation at a number of separate points. To what extent this might also have been true for the heart and first arches we can not say, but doubtless the principles involved were essentially the same. Conditions here are such that the many connections still required to complete the vascular circuit may be made within a very short period of time. It would be useless to speculate concerning priority or sequence, since there must be considerable variation; but in any case the union of the intraembryonic and extraembryonic portions of the umbilical vein would appear to be a relatively late event.
The vascular system, as we find it in this embryo, is really of little more importance to its possessor at this particular moment than are the future respiratory and excretory systems, of which there are as yet not even the faintest traces. The special organs for these last-named functions will appear in their own good time. In the meantime, however, these and other functions will be discharged in other ways, but even now, as later, only upon the basis of an adequate circulatory mechanism. At first the modest needs of the embryo, as regards a circulating or diffusible medium, will be met by the relatively great surface which its component parts present to the surrounding fluid, as in the yolk-sac and coelom. There can be no doubt that important nutritive material finds its way to the young embryo through the coelom, or secondarily through the yolk-sac. Without saying that the fluid within the umbilical vesicle differs materially from that outside, one may still conceive that its lining entoderm may play some small, though evanescent role, and that its mesoderm and blood-channels have something more to do than the formation of early blood-cells and vitelline vessels.
In addition to the extensive absorptive or excretive surfaces offered by the early embryo, we have a second intermediary mechanism for adequate diffusion in the form of the widely distributed mesostroma. The obstacles which this lastnamed tissue offers to the countless tiny currents resulting from cell activity must be almost nil. It is in large measure in these Lilliputian forces, as well as in the various and varying chemical changes which are constantly going on, that one must seek the explanation for the earliest vasculogenic conditions. Cell growth and migrations, cell form and orientation, vacuolation and the formations of cysts and definite channels are all, in part, the local reaction to a present stimulus. Endothelium comes into existence in response to certain influences, and in the face of new conditions it may again revert or give rise to the indifferent tissue from which it sprang. Even after the establishment of a complete, closed circulation, the wide intercellular, often avascular spaces of the embryo must play an important role in the exchange of various fluids. Gradually, however, this feature becomes less conspicuous, but in the last analysis the adult conditions are exactly the same, where the individual cells live, move, and have their being in the same primitive way, in the tissue fluids which bathe them.
From the very first there has been what we may call a physiological circulation, howbeit very primitive, but nevertheless quite sufficient. The necessary interchanges and transference of various substances are soon facilitated and accelerated by the gradual appearance of freer pathways and more sharply limited channels in definite localities. These early vessels, with their thin or at times deficient walls, must confer a material benefit upon the adjacent regions, even before the heart is capable of strengthening or directing the feeble currents within them. Soon, of course, the early, primitive arrangements must give way before the powerful pulsations of a functionating heart, which remains henceforth a sine qua non of further development.
That the vascular system, as we find it here, can not be injected goes without saying. In the first place there is a most conspicuous lack of continuity, and even where present the vessels do not always present a definite wall separating their lumina from the wide intercellular spaces without. Extravasations under any conditions of intravascular pressure would obviously be a foregone conclusion. One speaks of extravasation because the starting-point is the lumen of the vessel; but here, in many cases, the vessels might be injected from the loose mesostroma around them. The differences between vessel walls and deficiencies in the same are morphological rather than physiological, distinctions of degree rather than of quality, for in both instances there is an interchange of material in both directions.
We would take occasion at this time to express our thanks to Dr. George L. Streeter for so generously placing the facilities of the Carnegie Laboratory at our disposal. Appreciation is also due to Mr. 0. 0. Heard for invaluable aid rendered in preparing the models from which the accompanying illustrations were made; and to Mr. James F. Didusch for the excellent maimer in which these illustrations were carried out.
Bibliography
Bremer, J. L., 1912. The development of the aorta and the aortic arches in rabbits. Am. Jour. Anat., vol. 13. ■ , 1914. The earliest blood-vessel in man. Ibid., vol. 16.
Dandy, W. E., 1910. A human embryo with seven pairs of somites measuring about 2 mm. in length. Am. Jour. Anat., vol. 10.
Debeyre, A., 1912. Description d'un embryon humain de mm. 9. Jour, de l'Anat. et de la Physiol., t. 48.
Emmel, V. E., 1916. The cell clusters in the dorsal aorta of mammalian embryos. Am. Jour. Anat., vol. 19.
Eternod, A. C. F., 1899. Premiers stades de la circulation sanguine dans l'oeuf et l'embryon humains. Anat. Anz., Bd. 15. , 1909. L'oeuf humain implantation et gestation, trophoderme et placenta. Geneve. , 1913. Les premiers stades de developpement de l'oeuf humain. 27th International Congress of Medicine, London, 1913. Section I. Anatomy and Embryology.
Evans, H. M., and (I. W. Bartelmez, 1917. A human embryo of seven to eight somites. Anat. Record, vol. 11, p. 355-356.
Felix, W., 1910. Zur Entwickelungegeschichte der Rumpfarterien des menschlichen Embryo. Morph. Jahrb., Bd. 41.
Grosser, O., 1913. Ein menschlicher Embryo mit Chordakanal. Anat. Hefte, Bd. 47.
Hahn, Herman, 1909. Experimentelle Studien uber die Kntstchung des Blutes und der ersten Gefiissen beim Htihnchen. I Theil. Intraembryonale Gefasse. Arch. f. Entwicklungsmechanik. Bd. 27.
Held, H., 1909. Die Entwicklung des Nervengewebes bei den Wirbeltieren. Leipzig.
His, W.. lssO-5. Anatomic menschhcher Embryonen. Lepzig.
Ingalls, N. W., 1907. Beschreibung eines menschlichen Embryos von 4.9 mm. Arch. f. mik. Anat., Bd. 70.
1918. A human embryo before the appearance of the myotomes. Contributions to Embryology, vol. 7, Carnegie Inst. Wash. Pub. No. 227.
Jordan, H. E., 1910. A microscopic study of the umbilical vesicle of a 13 mm. human embryo, with special reference to the entodermal tubules and the blood islands. Anat. Anz., Bd. 37.
1917. Aortic clusters in vertebrate embryos. Anat. Record, vol. 11, p. 372.
Keibel, F., and C. Elze, 1908. Normentafeln zur Entwickelungsgeschichte des Menschen. Jena.
Kromer, 1903. Wachsmodell eines jungen menschlichen Embryo. Verh. d. Ges. f. Gynakologie.
Mall FP. On the development of the human heart. (1912) Amer. J Anat. 13: 249-298.
Miller, A. M., and J. E. McWhorter, 1914. Experiments on the development of blood-vessels in the area pellucida and embryonic body of the chick. Anat. Record, vol. 8.
Parker, Katherine M., 1915. The early development of the heart and anterior vessels in Marsupials, with special reference to Parameles. Proc. Zool. Soe. London. Part III.
Reagen, F. P., 1915. Vascularization phenomena in fragments of embryonic bodies completely isolated from yolk-sac blastoderm. Anat. Record, vol. 9.
Sabin, F. R., 1917. Origin and development of the primitive vessels of the chick and of the pig. Contributions to Embryology, vol. 6, Carnegie Inst. Wash. Pub. No. 226.
Schulte, H. von W., 1914. Early stages in vasculogenesis in the cat (Felis domestica), with special reference to the mesenchymal origin of endothelium. Memoirs Wistar Institute, No. 3. , 1916. The fusion of the cardiac anlages and the formation of the cardiac loop in the cat (Felis domestica). Am. Jour. Anat., vol. 20.
Spee, Graf, 1896. Zur Demonstration ilber die Entwicklung der Drusen des menschlichen Dottersacks. Anat. Anz., Bd. 12.
SnnNicKA, F. K., 1911. Das Mesenchym und das Mesostroma der Froschlarven und deren Produkte. Anat. Anz., Bd. 40. , 1912. Die Plasmodesmen und die Cytodesmen. Ibid.
v. Szily, A., 1904. Zur Glaskorperfrage. Anat. Anz., Bd. 24. , 1908. Ueber das Entstehen eines fibrillaren
Stutzgewebes in Embryo und dessen Verhaltnis zur Glaskorperfrage. Anat. Hefte, Bd. 35.
Tandler J. The Development of the Heart. (1912) Sect. II, chapt. 18, vol. 2, in Keibel F. and Mall FP. Manual of Human Embryology II. (1912) J. B. Lippincott Company, Philadelphia., pp. 534-570.
Wang, C. C, 1918. The earliest stages of development of the blood-vessels and of the heart in ferret embryos. Jour. Anat. and Physiol., vol. 52.
Wilson JT. Observations upon young human embryos. (1914) J Anat Physiol., 48(3): 315-51 PMID 17233002 PMC1288949
Descriptions of Plates
- Embryo at Segmentation: Figure A | Plate 1 | Plate 2 | Plate 3 | Plate 4 | Plate 5 | Carnegie stage 9 | Carnegie Embryo 1878
Reference
Ingalls NW. A human embryo at the beginning of segmentation, with special reference to the vascular system. (1920) Contrib. Embryol., Carnegie Inst. Wash. Publ. 274, 11: 61-90.
Cite this page: Hill, M.A. (2024, April 27) Embryology Book - Contributions to Embryology Carnegie Institution No.52. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_Contributions_to_Embryology_Carnegie_Institution_No.52
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G
Plate 1
| Fig. 1. Left lateral view of model of embryonic body viewed in the plane of section.
X 100. Yolk-sac, amnion, and body-stalk have been cut away. Fig. 2. Dorsal view of the same model. Both views show open neural groove. Otic plates and V ganglia appear as darker, rounded areas, the former larger and farther lateral, in the region of the rhombencephalon. Outlines of the underlying somites and in figure 2 the left umbilical vein. At the posterior end of the body the broad, dark line indicates the primitive-streak region with the primitive node at its anterior end and the cloacal membrane at the posterior end. Projecting from the cut surface of the body-stalk are the two umbilical arteries, with their branches to form the ventral venous plexus (cf . figs. 3 and 4) ; between the arteries is the allantois. In figure 1, first ectodermic pocket in front of left otic plate, opposite midbrain flexure. |
Plate 2
| Fig. 3. Chorion, body-stalk, and embryo, the last on sagittal section, view as in figure 1. X 90.
Ectoderm is shown in yellow, entoderm in green, vessels in red. Chorion shows numerous small vessels and the slender connection with the large venous plexus of body-stalk. Lower anastomosis between umbilical artery and ventral venous plexus not shown. Distal portion of allantois in contact with mesothelium of body-stalk. Primitive streak between primitive node and cloacal membrane. Heart plexus suspended between myoepicardial mantle and fore-gut (cf. figs. 7 and 8 on plate 4). Line of section 12-3-6, text-figure a, is indicated. |
Plate 3
| Fig. 4. Vascular system as seen from the right side. X 100. Color scheme as in figure 3.
The ectoderm has been cut away close to the border of the medullary folds. The mesoderm of the embryo and adjacent yolksac has been removed to show the vessels embedded in it. In the vitelline plexus and that part of it which represents the yolk-sac portion of the vitelline (umbilical) artery, the endothelial vessels are represented in darker color than the less definitely lined channels. The same is true for the anterior portion of the dorsal aorta and the isolated vesicles in the roof of the fore-gut. Scattered blood-islands and other vascular formations are seen through the mesoderm of the yolk-sac. Anterior to the branch of the aorta (marked vitelline artery) is a second, more doubtful connection between the aorta and the vitelline plexus. |
Plate 4
| Fig. 5. Right ventrolateral view of heart, myoepicardial mantle, and pericardial cavity. X 100.
|
Plate 5
(Further details concerning the figures on this plate will be found in the text.)
| Fig. 9. Section 12-1-2. X 350. Portion of body-stalk low down, showing blood-islands in left umbilical artery. Toward the yolk-sac, above in the illustration, the lumen of the artery communicates freely with the spaces in the mesenchyme.
|
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 27) Embryology Book - Contributions to Embryology Carnegie Institution No.52. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_Contributions_to_Embryology_Carnegie_Institution_No.52
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G