Primordial Germ Cell Development
| Embryology - 26 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
Early in development at the time of gastrulation a small group of cells are "put aside" to later form oocytes and spermatozoa, these cells described as the primordial germ cells (PGCs). The cells migrate initially through the primitive streak into the posterior endoderm that forms the hindgut,[2] and from there later into the genital ridge that will be the site of the developing gonad. The maintenance of pluripotency within this cell population may arise through epigenetic modifications that suppress somatic differentiation programs. These cells differentiate at different times in male testis and female ovary development. Recent molecular studies suggest that final determination occurs after PGCs colonize the developing gonad.[3]
In the male, this population of cells when transformed is also thought to give rise to testicular germ cell tumours.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Primordial Germ Cells | Female Primordial Germ Cells | Male Primordial Germ Cells | testicular germ cell tumour |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Textbooks
- Human Embryology (2nd ed.) Larson Chapter 10 p261-306
- The Developing Human: Clinically Oriented Embryology (6th ed.) Moore and Persaud Chapter 13 p303-346
- Before We Are Born (5th ed.) Moore and Persaud Chapter 14 p289-326
- Essentials of Human Embryology, Larson Chapter 10 p173-205
- Human Embryology, Fitzgerald and Fitzgerald Chapter 21-22 p134-152
- Developmental Biology (6th ed.) Gilbert Chapter 14 Intermediate Mesoderm
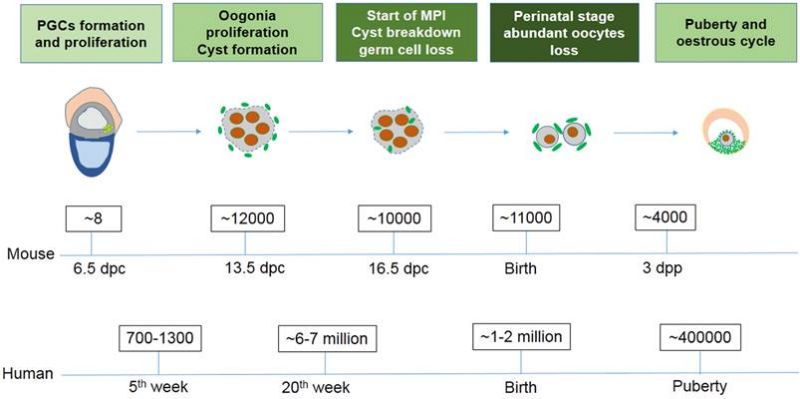
Timeline
Ovary
Mouse and Human Ovarian PGC Timeline Comparison[16]
Testis
Electron microscopy has showed apoptosis features in about 50% of mouse cells[17] and a recent study has shown apoptosis molecular markers expressed in human.[18]
Primordial Germ Cell Migration
Primordial Germ Cells (PGCs) are thought to be the first population of cells to migrate through the primitive streak in early gastrulation.
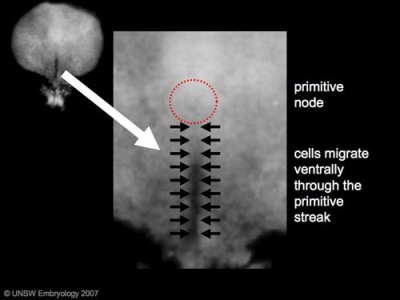
Human Embryonic Disc (Stage 7 week 3, GA week 5)
Week 3 - GA Week 5
- Human embryonic disc showing the primitive streak region where gastrulation occurs, generation the trilaminar embryo.
- Arrows indicate direction of cell migration through the streak.
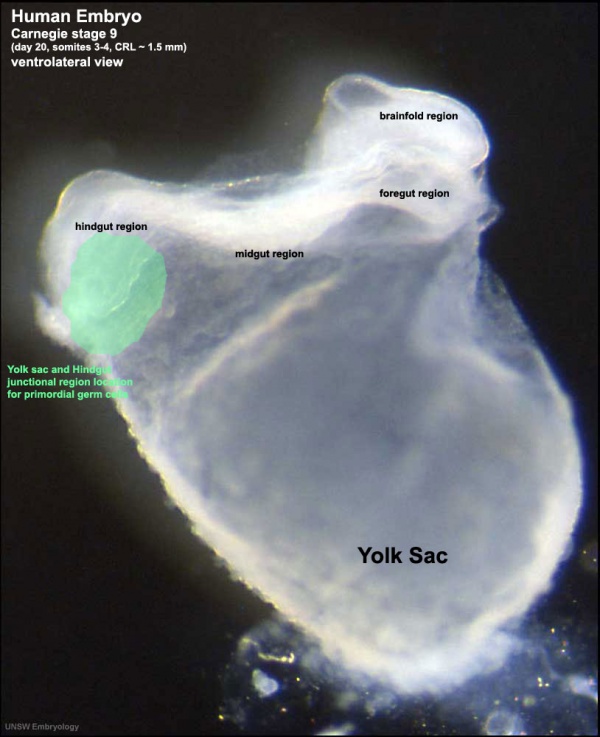
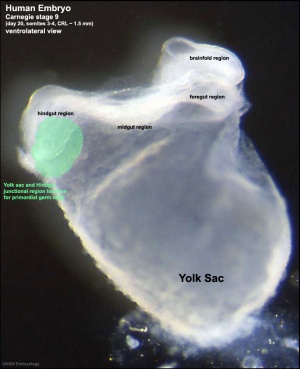
This population of cells then lie at the hindgut and yolk sac junctional region and later migrate into the germinal ridge in early embryonic development.
Human Embryo (Stage 9 GA week 5) primordial germ cell region (green)
- Sacrococcygeal teratomas - Remnant primitive streak cells (most common solid tumor in newborn infants)
- Germline teratoma - (Germinoma) abnormally differentiated/located PGCs fail to die.
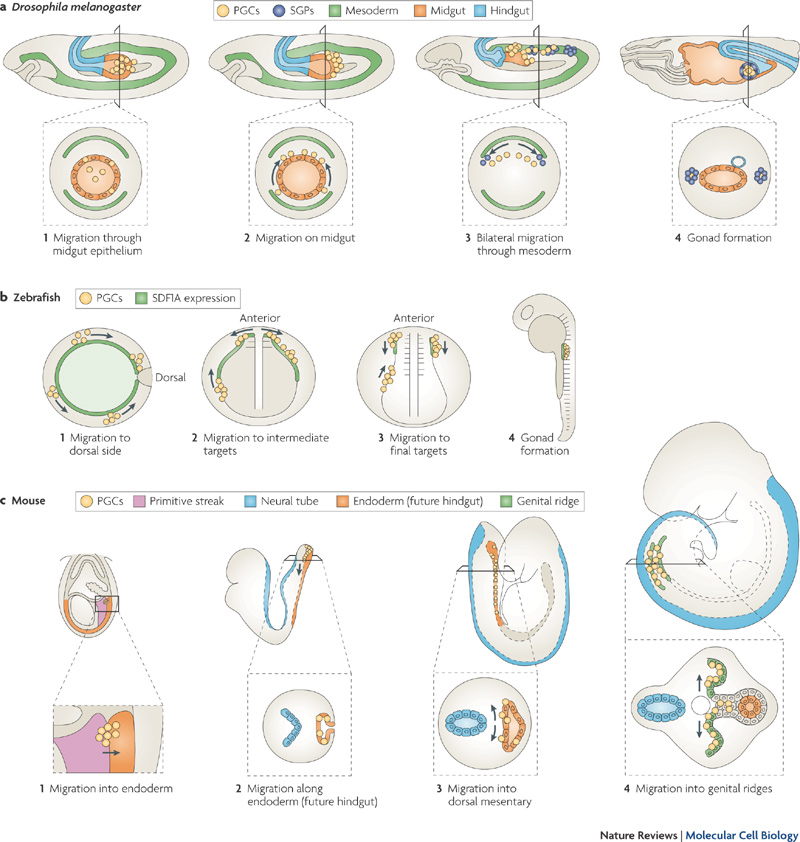
Species Comparison of Migration
Stages of primordial germ cell migration[19]
Mouse Migration Movies
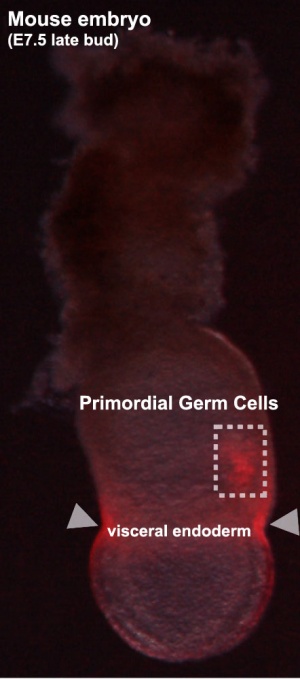
Labeled mouse primordial germ cells (E7.5)[20] |
Labeled mouse primordial germ cells (E10.5) See Mouse Migration Movies]] The movies below show labeled primordial germ cells (green) migrating within the mouse embryo between the periods of E9.0 to E10.5 into the genital ridge region that will later form the gonad. |
Mouse Template:ME11.0 to Template:ME12.0
- Female - genital ridge granulosa cells
- Male - Sertoli cells and Leydig cells
- E11.5 - PGCs are bipotential
| Mouse Primordial Germ Cell Migration | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
| |||||||||
- Links: Mouse Development
Fetal Ovary Meiosis
Fetal ovary meiosis[21]
Ectopic PGCs
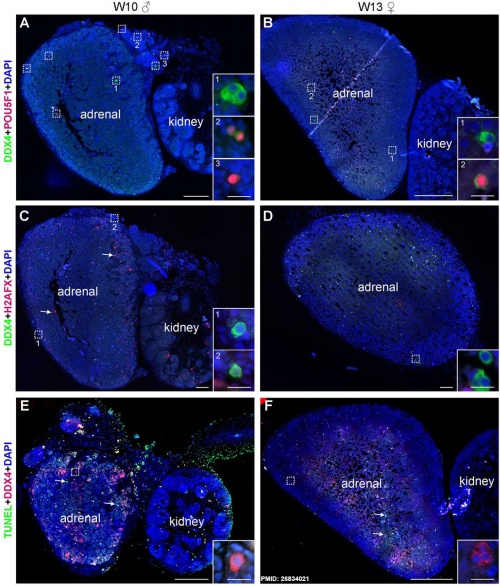
|
Ectopic PGC's in human adrenal gland in first trimester male week 8 (GA week 10) and female week 11 (GA week 13).[21]
|
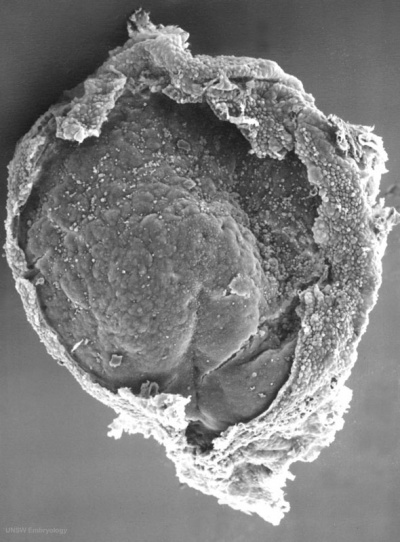
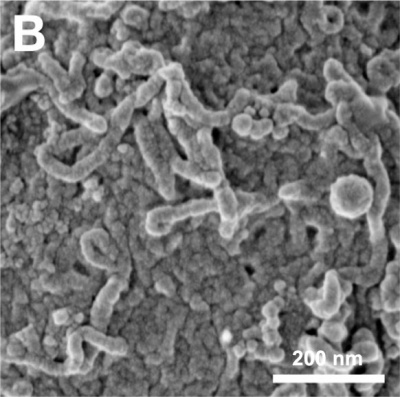
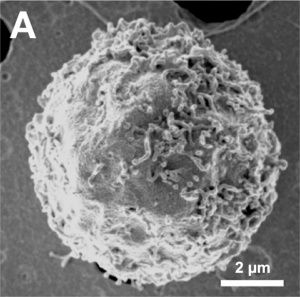
Cell Structure
The images below are scanning electron micrographs of the surface of a chicken primordial germ cell that has been grown in culture.[1]
The first image shows the whole cell and the second image shows detail of the cell surface showing extensions.
DNA Methylation
| Mouse primordial germ cell DNA methylation[22]
Demethylation
|
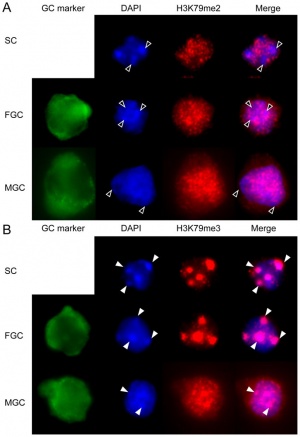
|
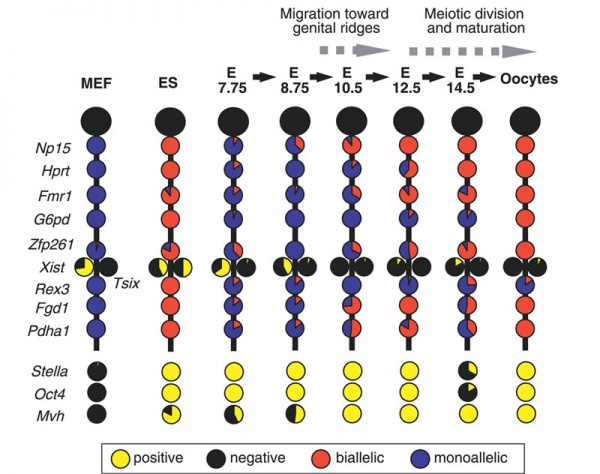
X-linked Gene Expression
Mouse- X-linked gene expression during primordial germ cell development.[23]
Each circle graph indicates the ratio of cells that are positive (yellow) and negative (black) for each gene, and biallelically (red) and monoallelically (blue) expressed in cells positive for each gene.
- Links: X Inactivation | Mouse Development
Molecular
- Prdm1 and Prdm14 - PR domain proteins expressed in mouse (E6.25), suppresses somatic differentiation.
- Sall4 - zinc finger protein, inactivation of this transcription factor in mouse can reduce PGC number.[12]
A study has recently identified 11 genes that are specifically expressed in male and female fetal germ cells, both in vivo and in vitro, but are not expressed in embryonic stem cells.[24]
PGC Markers: alkaline phosphatase-positive, Oct4 (POU5F1), Fragilis (IFITM1)[25], Stella (DPPA3), Dazl, and Vasa (DDX4).
- Steel factor - (KITLG) a ligand for the KIT tyrosine kinase receptor.
- DAZL
- dead end - coding an RNA binding protein mainly expressed in the germ cells of vertebrates.
- Blimp1 - B-Lymphocyte induced maturation protein-1 (PRDM1)
- Prmt5 - protein arginine methyltransferase-5
- Nanog - knockdown induces apoptotic cell death in mouse migrating primordial germ cells.[26]
- AID - Activation-Induced cytidine Deaminase enzyme required for demethylation (removal of CpG methylation). Within the genome, DNA methylation is associated with epigenetic mechanisms and occurs at cytosine residues that are followed by guanines.[27]
Stem Cells
A recent study in chicken has shown that only two key factors are required to convert stem cells into haploid spermatids:[28]
- retinoic acid (RA)
- stem cell factor (SCF)
Abnormalities
Teratomas
Common group of fetal tumors occuring along the body midline, anywhere from the coccyx to the pineal gland, reflecting the developmental PGC migration pathway (for review see[29]).
- Histologically classified as either mature or immature.
- Immature elements consisting principally of primitive neuroglial tissue and neuroepithelial rosettes and have have a generally favorable prognosis.
- Sacrococcygeal teratomas - most common site (70%–80% of all teratomas).
- classified into four types based on the amount of mass present externally versus internally.
Testicular germ cell tumours (seminoma)
References
- ↑ 1.0 1.1 Choi JW, Kim S, Kim TM, Kim YM, Seo HW, Park TS, Jeong JW, Song G & Han JY. (2010). Basic fibroblast growth factor activates MEK/ERK cell signaling pathway and stimulates the proliferation of chicken primordial germ cells. PLoS ONE , 5, e12968. PMID: 20886037 DOI.
- ↑ Fujimoto T, Miyayama Y & Fuyuta M. (1977). The origin, migration and fine morphology of human primordial germ cells. Anat. Rec. , 188, 315-30. PMID: 900520 DOI.
- ↑ 3.0 3.1 Nicholls PK, Schorle H, Naqvi S, Hu YC, Fan Y, Carmell MA, Dobrinski I, Watson AL, Carlson DF, Fahrenkrug SC & Page DC. (2019). Mammalian germ cells are determined after PGC colonization of the nascent gonad. Proc. Natl. Acad. Sci. U.S.A. , 116, 25677-25687. PMID: 31754036 DOI.
- ↑ Hansen CL & Pelegri F. (2021). Primordial Germ Cell Specification in Vertebrate Embryos: Phylogenetic Distribution and Conserved Molecular Features of Preformation and Induction. Front Cell Dev Biol , 9, 730332. PMID: 34604230 DOI.
- ↑ Barnett AA, Nakamura T & Extavour CG. (2019). Hox genes limit germ cell formation in the short germ insect Gryllus bimaculatus. Proc. Natl. Acad. Sci. U.S.A. , 116, 16430-16435. PMID: 31346080 DOI.
- ↑ Kobayashi T & Surani MA. (2018). On the origin of the human germline. Development , 145, . PMID: 30037844 DOI.
- ↑ Yang S, Ding S, He S, He L, Gao K, Peng S & Shuai C. (2019). Differentiation of primordial germ cells from premature ovarian insufficiency-derived induced pluripotent stem cells. Stem Cell Res Ther , 10, 156. PMID: 31151408 DOI.
- ↑ Gallinat A. (1995). [Ambulatory endoscopic surgery--ovary and leiomyoma]. Arch. Gynecol. Obstet. , 257, 17-21. PMID: 8579394
- ↑ de Souza AF, Pieri NCG, Roballo KCS, Bressan FF, Casals JB, Ambrósio CE, Perecin F & Martins DS. (2018). Dynamics of male canine germ cell development. PLoS ONE , 13, e0193026. PMID: 29489867 DOI.
- ↑ Fernández-Pérez D, Brieño-Enríquez MA, Isoler-Alcaraz J, Larriba E & Del Mazo J. (2018). MicroRNA dynamics at the onset of primordial germ and somatic cell sex differentiation during mouse embryonic gonad development. RNA , 24, 287-303. PMID: 29187591 DOI.
- ↑ Wang JQ & Cao WG. (2016). Key Signaling Events for Committing Mouse Pluripotent Stem Cells to the Germline Fate. Biol. Reprod. , 94, 24. PMID: 26674564 DOI.
- ↑ 12.0 12.1 Yamaguchi YL, Tanaka SS, Kumagai M, Fujimoto Y, Terabayashi T, Matsui Y & Nishinakamura R. (2015). Sall4 is essential for mouse primordial germ cell specification by suppressing somatic cell program genes. Stem Cells , 33, 289-300. PMID: 25263278 DOI.
- ↑ Tanaka SS, Yamaguchi YL, Steiner KA, Nakano T, Nishinakamura R, Kwan KM, Behringer RR & Tam PP. (2010). Loss of Lhx1 activity impacts on the localization of primordial germ cells in the mouse. Dev. Dyn. , 239, 2851-9. PMID: 20845430 DOI.
- ↑ 14.0 14.1 Haston KM, Tung JY & Reijo Pera RA. (2009). Dazl functions in maintenance of pluripotency and genetic and epigenetic programs of differentiation in mouse primordial germ cells in vivo and in vitro. PLoS ONE , 4, e5654. PMID: 19468308 DOI.
- ↑ Gu Y, Runyan C, Shoemaker A, Surani A & Wylie C. (2009). Steel factor controls primordial germ cell survival and motility from the time of their specification in the allantois, and provides a continuous niche throughout their migration. Development , 136, 1295-303. PMID: 19279135 DOI.
- ↑ Sun YC, Sun XF, Dyce PW, Shen W & Chen H. (2017). The role of germ cell loss during primordial follicle assembly: a review of current advances. Int. J. Biol. Sci. , 13, 449-457. PMID: 28529453 DOI.
- ↑ Wang RA, Nakane PK & Koji T. (1998). Autonomous cell death of mouse male germ cells during fetal and postnatal period. Biol. Reprod. , 58, 1250-6. PMID: 9603260 DOI.
- ↑ Sakashita A, Kawabata Y, Jincho Y, Tajima S, Kumamoto S, Kobayashi H, Matsui Y & Kono T. (2015). Sex Specification and Heterogeneity of Primordial Germ Cells in Mice. PLoS ONE , 10, e0144836. PMID: 26700643 DOI.
- ↑ Richardson BE & Lehmann R. (2010). Mechanisms guiding primordial germ cell migration: strategies from different organisms. Nat. Rev. Mol. Cell Biol. , 11, 37-49. PMID: 20027186 DOI.
- ↑ Cao L, Shitara H, Sugimoto M, Hayashi J, Abe K & Yonekawa H. (2009). New evidence confirms that the mitochondrial bottleneck is generated without reduction of mitochondrial DNA content in early primordial germ cells of mice. PLoS Genet. , 5, e1000756. PMID: 19997484 DOI.
- ↑ 21.0 21.1 Heeren AM, He N, de Souza AF, Goercharn-Ramlal A, van Iperen L, Roost MS, Gomes Fernandes MM, van der Westerlaken LA & Chuva de Sousa Lopes SM. (2016). On the development of extragonadal and gonadal human germ cells. Biol Open , 5, 185-94. PMID: 26834021 DOI.
- ↑ Abe M, Tsai SY, Jin SG, Pfeifer GP & Szabó PE. (2011). Sex-specific dynamics of global chromatin changes in fetal mouse germ cells. PLoS ONE , 6, e23848. PMID: 21886830 DOI.
- ↑ Sugimoto M & Abe K. (2007). X chromosome reactivation initiates in nascent primordial germ cells in mice. PLoS Genet. , 3, e116. PMID: 17676999 DOI.
- ↑ Sabour D, Araúzo-Bravo MJ, Hübner K, Ko K, Greber B, Gentile L, Stehling M & Schöler HR. (2011). Identification of genes specific to mouse primordial germ cells through dynamic global gene expression. Hum. Mol. Genet. , 20, 115-25. PMID: 20940145 DOI.
- ↑ Lange UC, Saitou M, Western PS, Barton SC & Surani MA. (2003). The fragilis interferon-inducible gene family of transmembrane proteins is associated with germ cell specification in mice. BMC Dev. Biol. , 3, 1. PMID: 12659663
- ↑ Yamaguchi S, Kurimoto K, Yabuta Y, Sasaki H, Nakatsuji N, Saitou M & Tada T. (2009). Conditional knockdown of Nanog induces apoptotic cell death in mouse migrating primordial germ cells. Development , 136, 4011-20. PMID: 19906868 DOI.
- ↑ Sanz LA, Kota SK & Feil R. (2010). Genome-wide DNA demethylation in mammals. Genome Biol. , 11, 110. PMID: 20236475 DOI.
- ↑ Ji M, Tang S, Pei W, Ning M, Ma Y, Li X & Guan W. (2018). Generation of haploid spermatids from chicken embryonal primordial germ cells. Int. J. Mol. Med. , , . PMID: 29620249 DOI.
- ↑ Woodward PJ, Sohaey R, Kennedy A & Koeller KK. (2005). From the archives of the AFIP: a comprehensive review of fetal tumors with pathologic correlation. Radiographics , 25, 215-42. PMID: 15653597 DOI.
Reviews
Yildirim E, Aksoy S, Onel T & Yaba A. (2020). Gonadal development and sex determination in mouse. Reprod Biol , , . PMID: 32147393 DOI.
Grimaldi C & Raz E. (2020). Germ cell migration-Evolutionary issues and current understanding. Semin. Cell Dev. Biol. , 100, 152-159. PMID: 31864795 DOI.
Günesdogan U & Surani MA. (2016). Developmental Competence for Primordial Germ Cell Fate. Curr. Top. Dev. Biol. , 117, 471-96. PMID: 26969996 DOI.
Saitou M & Yamaji M. (2010). Germ cell specification in mice: signaling, transcription regulation, and epigenetic consequences. Reproduction , 139, 931-42. PMID: 20371640 DOI.
Richardson BE & Lehmann R. (2010). Mechanisms guiding primordial germ cell migration: strategies from different organisms. Nat. Rev. Mol. Cell Biol. , 11, 37-49. PMID: 20027186 DOI.
Matsui Y. (2010). The molecular mechanisms regulating germ cell development and potential. J. Androl. , 31, 61-5. PMID: 19875497 DOI.
De Felici M, Farini D & Dolci S. (2009). In or out stemness: comparing growth factor signalling in mouse embryonic stem cells and primordial germ cells. Curr Stem Cell Res Ther , 4, 87-97. PMID: 19442193
Hayashi K, de Sousa Lopes SM & Surani MA. (2007). Germ cell specification in mice. Science , 316, 394-6. PMID: 17446386 DOI.
Matsui Y & Okamura D. (2005). Mechanisms of germ-cell specification in mouse embryos. Bioessays , 27, 136-43. PMID: 15666347 DOI.
Watson CM & Tam PP. (2001). Cell lineage determination in the mouse. Cell Struct. Funct. , 26, 123-9. PMID: 11565804
De Felici M. (2000). Regulation of primordial germ cell development in the mouse. Int. J. Dev. Biol. , 44, 575-80. PMID: 11061420
Articles
Cantú AV & Laird DJ. (2017). A pilgrim's progress: Seeking meaning in primordial germ cell migration. Stem Cell Res , 24, 181-187. PMID: 28754603 DOI.
Hu YC, Nicholls PK, Soh YQ, Daniele JR, Junker JP, van Oudenaarden A & Page DC. (2015). Licensing of primordial germ cells for gametogenesis depends on genital ridge signaling. PLoS Genet. , 11, e1005019. PMID: 25739037 DOI.
West JA, Viswanathan SR, Yabuuchi A, Cunniff K, Takeuchi A, Park IH, Sero JE, Zhu H, Perez-Atayde A, Frazier AL, Surani MA & Daley GQ. (2009). A role for Lin28 in primordial germ-cell development and germ-cell malignancy. Nature , 460, 909-13. PMID: 19578360 DOI.
Durcova-Hills G, Tang F, Doody G, Tooze R & Surani MA. (2008). Reprogramming primordial germ cells into pluripotent stem cells. PLoS ONE , 3, e3531. PMID: 18953407 DOI.
Fujimoto T, Miyayama Y & Fuyuta M. (1977). The origin, migration and fine morphology of human primordial germ cells. Anat. Rec. , 188, 315-30. PMID: 900520 DOI.
Search PubMed
Search Pubmed: Primordial Germ Cell Migration | Primordial Germ Cell | Testicular germ cell tumours
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- Whitehead Institute for Biomedical Research - Page Lab
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 26) Embryology Primordial Germ Cell Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Primordial_Germ_Cell_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G