Neural Crest Development
Introduction
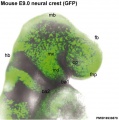
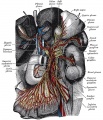
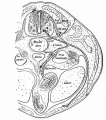
The neural crest are bilaterally paired strips of cells arising in the ectoderm at the margins of the neural tube. These cells migrate to many different locations and differentiate into many cell types within the embryo. This means that many different systems (neural, skin, teeth, head, face, heart, adrenal glands, gastrointestinal tract) will also have a contribution fron the neural crest cells.
In the body region, neural crest cells also contribute the peripheral nervous system (both neurons and glia) consisting of sensory ganglia (dorsal root ganglia), sympathetic and parasympathetic ganglia and neural plexuses within specific tissues/organs.
In the head region, neural crest cells migrate into the pharyngeal arches (as shown in movie below) forming ectomesenchyme contributing tissues which in the body region are typically derived from mesoderm (cartilage, bone, and connective tissue).General neural development is also covered in Neural Notes.
| Neural Crest Links: neural crest | Lecture - Early Neural | Lecture - Neural Crest Development | Lecture Movie | Schwann cell | adrenal | melanocyte | peripheral nervous system | enteric nervous system | cornea | cranial nerve neural crest | head | skull | cardiac neural crest | Nicole Le Douarin | Neural Crest Movies | neural crest abnormalities | Category:Neural Crest | |||
|
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Neural Crest Embryology <pubmed limit=5>Neural Crest Embryology</pubmed> |
Neural Crest Migration
| <mediaplayer width='410' height='340' image="http://php.med.unsw.edu.au/embryology/images/7/7d/Chicken-neural-crest-migration-01.jpg">File:Chicken-neural crest migration 01.mp4</mediaplayer> | 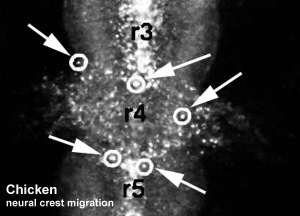
Chicken embryo sequence shows the migration of DiI-labeled neural crest cells towards the branchial arches as the embryo. White rings indicate migration of individual cells. Each image represents 10 confocal sections separated by 10 microns. |
Movie Source: Original Neural Crest movies kindly provided by Paul Kulesa.[6]
Related Movies: Migration 01 | Migration 02 | Migration 03 | Migration 04 | Migration 05 | Migration 06 | Migration 07
Textbooks
Objectives
- Understand the structures derived from ectoderm.
- Understand the formation of neural folds.
- Identify the initial location of neural crest cells in the trilaminar embryo.
- Identify pathways of neural crest migration throughout the embryo.
- To know the major tissues to which neural crest cells contribute.
- To know how abnormalities in development that result from abnormal neural crest cell migration.
- Understand how neural crest cells contribute to the pharyngeal arches and the head structures they form.
Neural Crest Derivatives
A key feature of neural crest is the migration into other embryonic tissues to form specific neural and non-neural populations and structures.
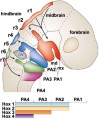
Cranial neural crest
- migration - dorsolaterally and into pharyngeal arches
- craniofacial mesenchyme - cartilage, bone, cranial neurons, glia, and connective tissues of the face
- pharyngeal arches and pouches - thymic cells, tooth odontoblasts, middle ear bones (ossicles) and jaw (mandible)
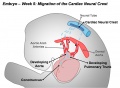
Cardiac neural crest
- migration - located between the cranial and trunk neural crests, overlapping the anterior portion of the vagal neural crest.
- pharyngeal arches - (3,4,6) melanocytes, neurons, cartilage, and connective tissue
- heart outflow tract - aortic arch/pulmonary artery septum, large arteries wall musculoconnective tissue
Trunk neural crest
- migration - two major pathways over somites (dorsolaterally) and between somite and neural tube (ventrolaterally)
- dorsolateral - skin melanocytes
- ventrolaterally - dorsal root ganglia, sympathetic ganglia, adrenal medulla, aortic nerve clusters
Vagal and sacral neural crest
- migration - ventrally into surrounding splanchnic mesenchyme of gastrointestinal tract
- splanchnic mesenchyme - parasympathetic (enteric) ganglia of the gut
Development Overview
The following cranial and trunk data is based upon 185 serially sectioned staged (Carnegie) human embryos.[7]
Cranial Neural Crest
- stage 9 - an indication of mesencephalic neural crest
- stage 10 - trigeminal, facial, and postotic components
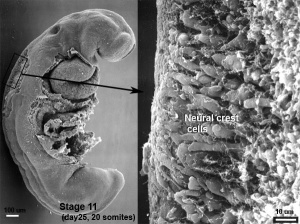
- stage 11 - crest-free zones are soon observable in rhombomere 1, 3, and 5
- stage 12 - rhombomeres 6 and 7 neural crest migrate to pharyngeal arch 3 and then rostrad to the truncus arteriosus
- stage 13 - nasal crest and the terminalis-vomeronasal complex are last of the cranial crest to appear
stages 9-14 - otic vesicle primordium descends
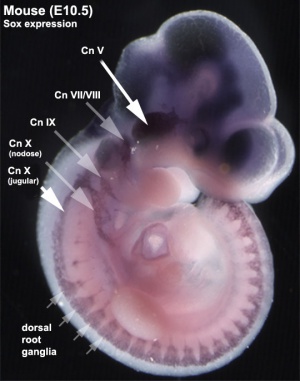
Vagal Neural Crest
Recent research suggests that the vagal neural crest cells are a transitional population that has evolved between the head and the trunk, taking separate pathways to the both the heart and to the gut.[8][9]
Trunk Neural Crest
Spinal ganglia increase in number over time and are in phase with the somites, though not their centre. There are 3 migratory pathways: ventrolateral between dermatomyotome and sclerotome, ventromedial between neural tube and sclerotomes, and lateral between surface ectoderm and dermatomyotome.
- stage 13 - about 19 present
- stage 14 - about 33 present
- stage 15-23 - 30–35 ganglia
Neck and Shoulder
A mouse study using individually labelled cells of postotic neural crest followed the development of the shoulder girdle (clavicle and scapula) that connects the upper limb to the axial skeleton.[10]
- Clavicle is a neural crest-mesodermal structure, posterior dermal clavicle mesoderm.
- Cryptic cell boundaries traverse apparently homogeneous skeleton of the neck and shoulders.
- Bones and muscles code of connectivity that mesenchymal stem cells of both neural crest and mesodermal origin obey
- Neural crest anchors the head onto the anterior lining of the shoulder girdle
- Hox-gene-controlled mesoderm links trunk muscles to the posterior neck and shoulder skeleton.
- Skeleton identified as neural crest-derived is affected in human Klippel-Feil syndrome, Sprengel's deformity and Arnold-Chiari I/II malformation.
Skin Melanocytes
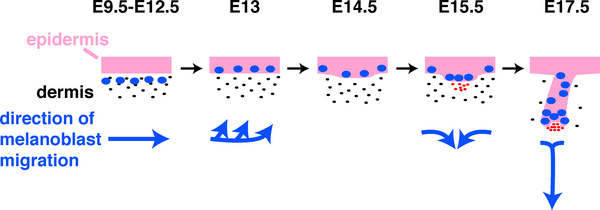
|
|
| Mouse melanocyte migration[11] | Movie Mouse Skin - Melanoblast Migration E14.5[12] |
References
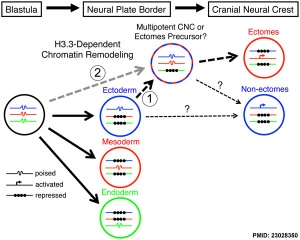
- ↑ 1.0 1.1 <pubmed>23028350</pubmed>| PLoS Genet.
- ↑ <pubmed>21552538</pubmed>
- ↑ <pubmed>20478300</pubmed>
- ↑ <pubmed>20399765</pubmed>
- ↑ <pubmed>20360764</pubmed>
- ↑ <pubmed>10683170</pubmed>
- ↑ <pubmed>17848161</pubmed>| PMC2375817 | J Anat.
- ↑ <pubmed>20962585</pubmed>
- ↑ Bryan R. Kuo, Carol A. Erickson Vagal neural crest cell migratory behavior: A transition between the cranial and trunk crest. Volume 240, Issue 9, pages 2084–2100, September 2011 Dev Dynamics
- ↑ <pubmed>16034409</pubmed>| PMC1352163| Nature
- ↑ <pubmed>16277556</pubmed>| PLoS Biol.
- ↑ <pubmed>20067551</pubmed>| PMC2859249
Reviews
<pubmed>21309066</pubmed> <pubmed>21309065</pubmed> <pubmed>21295685</pubmed> <pubmed>21452438</pubmed>
Articles
Search PubMed
Search April 2010 "Neural Crest Development" - All (4354) Review (843) Free Full Text (1621)
Search Pubmed: Neural Crest Development
Additional Images
Terms
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 26) Embryology Neural Crest Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Neural_Crest_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G