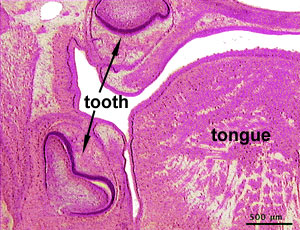
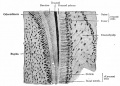
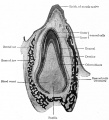
Integumentary System - Tooth Development
| Embryology - 27 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
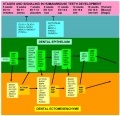
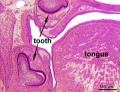
The tooth is an extrordinary integumentary system specialization providing insights into epitheilal/mesenchymal (ectoderm of the first pharyngeal arch and neural crest, ectomesenchymal cells) interactions in development and has a major contribution from the neural crest. (More? Neural Crest Development)
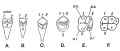
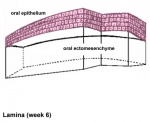
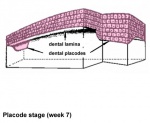
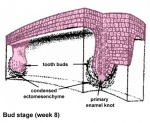
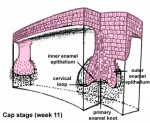
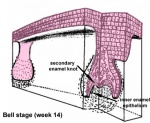
After placed formation, there are four key morphological stages describing the early tooth development: bud, cap, bell, and terminal differentiation.
| lamina | placode | bud | cap | bell |
|---|---|---|---|---|
|
|
|
|
|
Recent molecular animal model studies have shown that epithelial Wnt/β-catenin signaling is sufficient to initiate tooth development through activating Shh, Bmps, Fgfs and Wnts in the dental epithelium.[1] This then initiates the expression of odontogenic genes in the underlying mesenchyme.
| Historic Embryology |
|
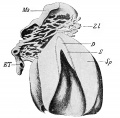
Oscar Hertwig (1849-1922) was a German embryologist and anatomist who first identified in amphibia tooth development, and was subsequently named Hertwig's epithelial root sheath (HERS). In amphibia, this is a permanent structure. In mammals, this is a transient structure, assembled during early tooth root formation and elongation. Later the HERS becomes fenestrated and reduced to the epithelial rests of Malassez (ERM).[2] |
Links: gastrointestinal tract
| Historic Tooth Development |
|---|
| 1902 Tooth Development | 1912 The Teeth | 1921 The Teeth |
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Tooth Embryology | Tooth Development | Odontoblast Development | dentinogenesis | Ameloblast Development | enamelogenesis
|
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Textbooks
- Human Embryology (2nd ed.) Larson Chapter 14 p443-455
- The Developing Human: Clinically Oriented Embryology (6th ed.) Moore and Persaud Chapter 20: P513-529
- Before We Are Born (5th ed.) Moore and Persaud Chapter 21: P481-496
- Essentials of Human Embryology Larson Chapter 14: P303-315
- Human Embryology, Fitzgerald and Fitzgerald
- Color Atlas of Clinical Embryology Moore Persaud and Shiota Chapter 15: p231-236
Movies
| <html5media height="540" width="512">File:Mouse tooth movie 01.mp4</html5media> | This time-lapse movie from a mouse embryo (E 12.5–13.5) cultured for 5 days ex vivo, images were taken at 30-min intervals.[12]
The tooth germ is from a developing molar and the lingual side is on the left.
|
Development Overview
- ectoderm, mesoderm and neural crest ectomesenchyme contribute
- inductive influence of neural crest with overlying ectoderm
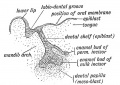
Odontoblast Cells
The odontoblast cells (dentinoblasts) are a population of neural crest-derived mesenchymal cells.
- differentiate under the influence of the enamel epithelium
- form predentin
- calcifies to form dentin (protects the dental pulp)
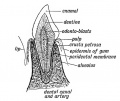
Dentin
- Collagen - collagen type I (about 90%) collagen type III and type V
- Proteoglycans - chondroitin sulphate (biglycan and decorin), heparan sulphate (entactin and perlecan), keratan sulphate, and dermatan sulphate
- Laminins - only at epithelial-mesenchymal junction
- Formation - before tooth eruption (primary dentin) rate of 4–8 μm/day, after tooth eruption (secondary dentin) rate of 0.5 μm/day.
Abnormalities - Dentinogenesis Imperfecta Type I, II and III, Dentin Dysplasia Type I and II,
Osteogenesis Imperfecta
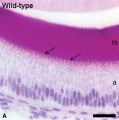
Ameloblast Cells
The ameloblast cells a population of ectoderm-derived oral epithelium cells that produce the tooth enamel.
- Molecular - BMP and FGF
- tooth growth occurs in ossifying jaws
- periodontal ligament holds tooth in bone socket
Epithelial Root Sheath
The Epithelial Root Sheath or Hertwig's epithelial root sheath (HERS) these epithelial cells differentiate into cementoblasts through epithelial mesenchymal transition (EMT) and during the tooth root development also induce odontoblastic differentiation of the dental papilla through epithelial mesenchymal interaction (EMI). Recent research has identified a method to establish HERS cell lines.[13]
Tooth Stages
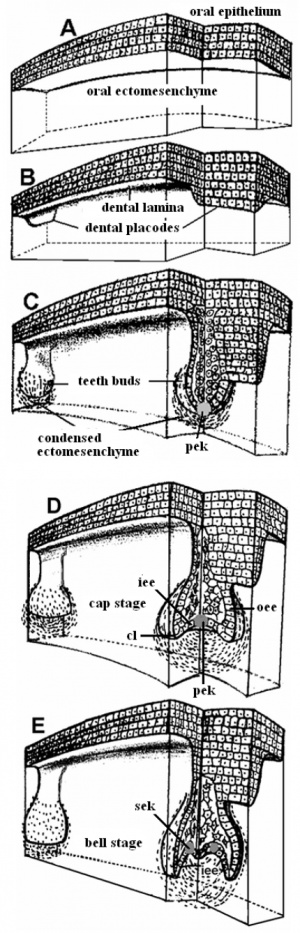
| Stage | Human (weeks) |
Mouse (days) | |
| lamina | 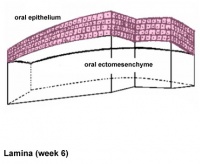
|
Week 6 | E 11 |
| placode | 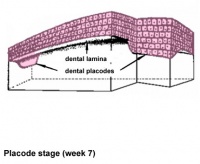
|
Week 7 | E 11.5 |
| bud | 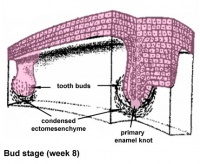
|
Week 8 | E 12.5 |
| cap | 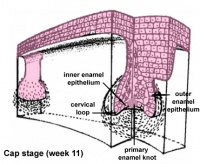
|
Week 11 | E 14.5 |
| bell | 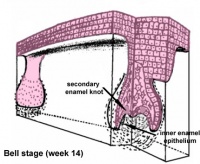
|
Week 14 | E 15.5 |
| Tooth Stages | |||
|---|---|---|---|
| Stage | Human (weeks) |
Mouse (days) | |
| lamina | 
|
Week 6 | E11 |
| placode | 
|
Week 7 | E11.5 |
| bud | 
|
Week 8 | E12.5 |
| cap | 
|
Week 11 | E14.5 |
| bell | 
|
Week 14 | E15.5 |
Image Links: all stages | lamina | placode stage | bud stage | cap stage | bell stage
Human 2 Sets of Teeth
Human Dentition Timeline
| Milk Dentition | |
|---|---|
| Median incisors | 6th to 8th month |
| Lateral incisors | 8th to 12th month |
| First molars | 12th to 16th month |
| Canines 1 | 7th to 20th month |
| Second molars | 20th to 24th month |
| Permanent Dentition | |
| First molars | 7th year |
| Median incisors | 8th year |
| Lateral incisors | 9th year |
| First premolars | 10th year |
| Second premolars | 11th year |
| Canines | 13th to 14th year |
| Second molars | 13th to 14th year |
| Third molars | 17th to 40th year |
| Approximate timings only, vary considerably according to racial, climatic, and nutritive conditions. Data source: Keibel and Mall (1912)[15] The teeth Links: tooth | |
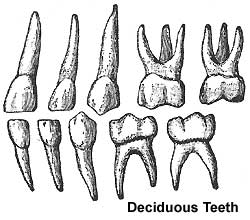
Deciduous Teeth
- 20 deciduous teeth
- Differential rates of growth, shed at different times over 20 year period
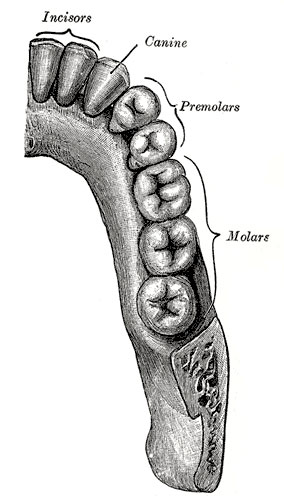
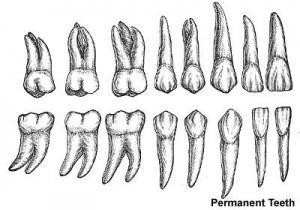
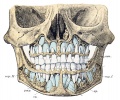
Permanent Teeth
- 32 permanent teeth
- Incisors - sharp cutting edge, adapted for biting the food.
- Canines - are larger and stronger than the incisors. The upper canines have also been called the "eye teeth", while the lower canines "stomach teeth".
- Premolars - or Bicuspid teeth are smaller and shorter than the canines.
- Molars - are the largest teeth adapted for grinding and pounding food.
Epithelial Mesenchymal Interaction
local ectodermal thickening expresses several signaling molecules these in turn signal to the underlying mesenchyme triggering mesenchymal condensation (epithelially expressed Bmp4 induces Msx1 and Lef1 as well as itself in the underlying mesenchyme)
Four epithelial signaling molecules, Bmp2, Shh, Wnt10a, and Wnt10b, in the early inductive cascade, each signal has a distinct molecular action on the jaw mesenchyme.
Mouse (E11 and E12) - all four genes are specifically expressed in the epithelium.
Shh and Wnt10b induce general Hedgehog and Wnt targets, Ptc and Gli for Shh and Lef1 for Wnt10b,
Bmp2 is able to induce tooth-specific expression of Msx1.
(Text above modified from: Hélène R. Dassule and Andrew P. McMahon Developmental Biology, v 202, n 2, October 15, 1998, p215-227)
(More? Epithelial Mesenchymal Interaction)
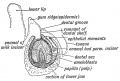
Periodontal Ligament
The tooth is not anchored directly onto its bony socket (alveolar bone) but held in place by the periodontal ligament (PDL), a specialized connective tissue structure that surrounds the tooth root coating of cementum.
The additional roles of the PDL are to also act as; a shock absorber, transmitter of chewing forces (from tooth to bone), sensory information (heat, cold, pressure and pain).
The collagen fiber bundles within the ligament are called "Sharpey’s fibres".
Cementum (from investing layer of the dental follicle) is contiguous layer with the periodontal ligament on one surface and firmly adherent to dentine on the other surface.
Molecular Tooth Development
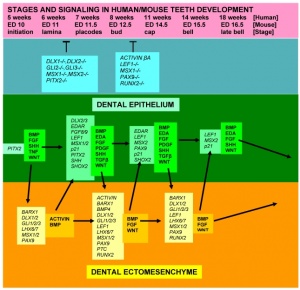
More than 300 genes have been associated with tooth development including: BMP4, FGF8, MSX1, MSX2, PAX9, PITX2, SHOX2, Delta/Notch, Hox-8, Runx2
Most recent review in Developmental Dynamics by Lin D, Huang Y, He F, Gu S, Zhang G, Chen Y, Zhang Y. Expression survey of genes critical for tooth development in the human embryonic tooth germ. Dev Dyn. 2007 Mar 29.
Amelogenin - abundant protein secreted by ameloblasts which is a major component of tooth enamel.
The papers below are from UNSW Embryology (version 3), information requires updating.
Bone Morphogenic Protein (BMP) / Fibroblast Growth Factor (FGF)
Growth factors in the BMP- and FGF-families are expressed in dental epithelium during initiation of tooth development and their effects on the underlying mesenchyme mimic those of the epithelium. They upregulate the expression of many genes, including the homeobox-containing Msx-1 and Msx-2, and stimulate cell proliferation suggesting that they may act as epithelial signals transmitting epithelial-mesenchymal interactions. During subsequent morphogenesis, when the characteristic shapes of individual teeth develop as a result from folding of the dental epithelium, several signal molecules including Sonic hedgehog, Bmps-2, 4, 7 and Fgf-4 are expressed specifically in restricted and transient epithelial cell clusters, called enamel knots.
(Text: Irma Thesleff and Carin Sahlberg Seminars in Cell & Developmental Biology, v 7, n 2, April, 1996, p185-193)
Delta/Notch
The expression pattern of Delta 1 in ameloblasts and odontoblasts is complementary to Notch1, Notch2, and Notch3 expression in adjacent epithelial and mesenchymal cells. Notch1 and Notch2 are upregulated in explants of dental mesenchyme adjacent to implanted cells expressing Delta1, suggesting that feedback regulation by Delta-Notch signaling ensures the spatial segregation of Notch receptors and ligands. TGF1 and BMPs induce Delta1 expression in dental mesenchyme explants at the stage at which Delta1 is upregulated in vivo, but not at earlier stages. In contrast to the Notch family receptors and their ligand Jagged1, expression of Delta1 in the tooth germ is not affected by epithelial-mesenchymal interactions, showing that the Notch receptors and their two ligands Jagged1 and Delta1 are subject to different regulations.
Text: Mitsiadis etal Developmental Biology,v 204, n 2, December 15, 1998, p420-431
BMP4 expression by the interaction of Pax9 with Msx1 at the level of transcription and protein complex determines the fate of the transition from bud to cap stage during tooth development.[16]
Twist1 a basic helix-loop-helix-containing transcription factor expressed in dental mesenchyme during the early stages of tooth development acts through the FGF signaling pathway.[17]
Foxi3 forkhead-box transcription factor inhibits formation of enamel knots and cervical loops and therefore the differentiation of dental epithelium.[18]
Hey1 and Hey2 basic helix-loop-helix type transcription factors are differently expressed during mouse tooth development[19] Hey family (also known as Chf, Herp, Hesr, and Hrt).
- Hey2 transcripts were restricted to the undifferentiated inner enamel epithelium and down-regulated in preameloblasts and ameloblasts.
- Hey1 expressed in pre-ameloblasts and down-regulated in differentiated ameloblasts.
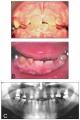
Abnormalities
| ICD-11 face, mouth or teeth | |
|---|---|
|
LA30 Structural developmental anomalies of teeth and periodontal tissues
| |
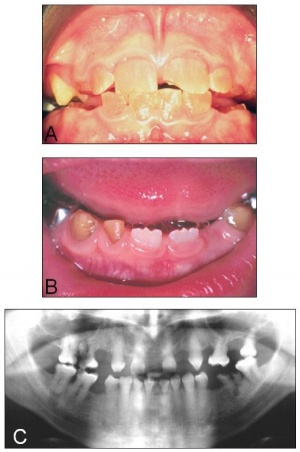
Adontia
A total lack of tooth development.
Dentinogenesis imperfecta
The teeth are translucent and often roughened with severe amber discolouration. Discoloured teeth with an opalescent sheen, dentin does not support enamel (dentin sialophosphoprotein mutation)
Dentine dysplasia
The primary teeth are translucent and amber in colour whereas the erupting secondary central incisors are of normal appearance.
Amelogenesis Imperfecta
Abnormal tooth enamel formation (AMELX, ENAM, KLK4, MMP20).
Dens Evaginatus
Dental anomaly mainly affecting premolars in people of Mongolian origin.
Hypodontia
Lack of development of one or more teeth.
Hypohidrotic Ectodermal Dysplasia
Maldevelopment of one or more ectodermal-derived tissues.
Microdontia
Small teeth.
Hutchinson's teeth
(Hutchinson's incisor, Hutchinson's sign, Hutchinson-Boeck teeth) Historic clinical term for an infant tooth abnormality associated with congenital syphilis. Teeth are smaller, more widely spaced than normal and have notches on the biting surfaces. Named after Jonathan Hutchinson (1828 – 1913) an English surgeon and pathologist, who first described this association.
References
- ↑ 1.0 1.1 Zhou N, Li N, Liu J, Wang Y, Gao J, Wu Y, Chen X, Liu C & Xiao J. (2018). Persistent Wnt/β-catenin signaling in mouse epithelium induces the ectopic Dspp expression in cheek mesenchyme. Organogenesis , , 1-12. PMID: 30570432 DOI.
- ↑ Luan X, Ito Y & Diekwisch TG. (2006). Evolution and development of Hertwig's epithelial root sheath. Dev. Dyn. , 235, 1167-80. PMID: 16450392 DOI.
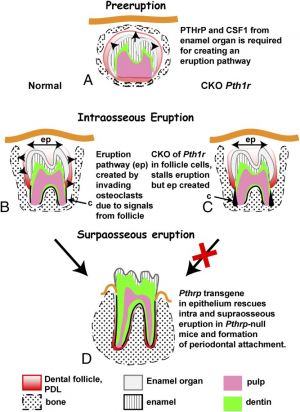
- ↑ Richman JM. (2019). Shedding new light on the mysteries of tooth eruption. Proc. Natl. Acad. Sci. U.S.A. , 116, 353-355. PMID: 30602459 DOI.
- ↑ Zhang S, Li X, Wang S, Yang Y, Guo W, Chen G & Tian W. (2020). Immortalized Hertwig's epithelial root sheath cell line works as model for epithelial-mesenchymal interaction during tooth root formation. J. Cell. Physiol. , 235, 2698-2709. PMID: 31512758 DOI.
- ↑ Zhang J, Liao L, Li Y, Xu Y, Guo W, Tian W & Zou S. (2019). Parathyroid hormone-related peptide (1-34) promotes tooth eruption and inhibits osteogenesis of dental follicle cells during tooth development. J. Cell. Physiol. , 234, 11900-11911. PMID: 30584670 DOI.
- ↑ Takahashi A, Nagata M, Gupta A, Matsushita Y, Yamaguchi T, Mizuhashi K, Maki K, Ruellas AC, Cevidanes LS, Kronenberg HM, Ono N & Ono W. (2019). Autocrine regulation of mesenchymal progenitor cell fates orchestrates tooth eruption. Proc. Natl. Acad. Sci. U.S.A. , 116, 575-580. PMID: 30509999 DOI.
- ↑ Seo H, Amano T, Seki R, Sagai T, Kim J, Cho SW & Shiroishi T. (2018). Upstream Enhancer Elements of Shh Regulate Oral and Dental Patterning. J. Dent. Res. , , 22034518758642. PMID: 29481312 DOI.
- ↑ Bonczek O, Balcar VJ & Šerý O. (2017). PAX9 gene mutations and tooth agenesis: A review. Clin. Genet. , 92, 467-476. PMID: 28155232 DOI.
- ↑ Li J, Chatzeli L, Panousopoulou E, Tucker AS & Green JB. (2016). Epithelial stratification and placode invagination are separable functions in early morphogenesis of the molar tooth. Development , 143, 670-81. PMID: 26755699 DOI.
- ↑ Huang X, Bringas P, Slavkin HC & Chai Y. (2009). Fate of HERS during tooth root development. Dev. Biol. , 334, 22-30. PMID: 19576204 DOI.
- ↑ Lin D, Huang Y, He F, Gu S, Zhang G, Chen Y & Zhang Y. (2007). Expression survey of genes critical for tooth development in the human embryonic tooth germ. Dev. Dyn. , 236, 1307-12. PMID: 17394220 DOI.
- ↑ Morita R, Kihira M, Nakatsu Y, Nomoto Y, Ogawa M, Ohashi K, Mizuno K, Tachikawa T, Ishimoto Y, Morishita Y & Tsuji T. (2016). Coordination of Cellular Dynamics Contributes to Tooth Epithelium Deformations. PLoS ONE , 11, e0161336. PMID: 27588418 DOI.
- ↑ Li X, Zhang S, Zhang Z, Guo W, Chen G & Tian W. (2019). Development of immortalized Hertwig's epithelial root sheath cell lines for cementum and dentin regeneration. Stem Cell Res Ther , 10, 3. PMID: 30606270 DOI.
- ↑ 14.0 14.1 Koussoulakou DS, Margaritis LH & Koussoulakos SL. (2009). A curriculum vitae of teeth: evolution, generation, regeneration. Int. J. Biol. Sci. , 5, 226-43. PMID: 19266065
- ↑ Grosser O. Lewis FT. and McMurrich JP. The Development of the Digestive Tract and of the Organs of Respiration. (1912) chapter 17, vol. 2, in Keibel F. and Mall FP. Manual of Human Embryology II. (1912) J. B. Lippincott Company, Philadelphia.
- ↑ Ogawa T, Kapadia H, Feng JQ, Raghow R, Peters H & D'Souza RN. (2006). Functional consequences of interactions between Pax9 and Msx1 genes in normal and abnormal tooth development. J. Biol. Chem. , 281, 18363-9. PMID: 16651263 DOI.
- ↑ Meng T, Huang Y, Wang S, Zhang H, Dechow PC, Wang X, Qin C, Shi B, D'Souza RN & Lu Y. (2015). Twist1 Is Essential for Tooth Morphogenesis and Odontoblast Differentiation. J. Biol. Chem. , 290, 29593-602. PMID: 26487719 DOI.
- ↑ Jussila M, Aalto AJ, Sanz Navarro M, Shirokova V, Balic A, Kallonen A, Ohyama T, Groves AK, Mikkola ML & Thesleff I. (2015). Suppression of epithelial differentiation by Foxi3 is essential for molar crown patterning. Development , 142, 3954-63. PMID: 26450968 DOI.
- ↑ Kibe K, Nakatomi M, Kataoka S, Toyono T & Seta Y. (2018). Hey1 and Hey2 are differently expressed during mouse tooth development. Gene Expr. Patterns , 27, 99-105. PMID: 29155305 DOI.
- ↑ Barron MJ, McDonnell ST, Mackie I & Dixon MJ. (2008). Hereditary dentine disorders: dentinogenesis imperfecta and dentine dysplasia. Orphanet J Rare Dis , 3, 31. PMID: 19021896 DOI.
Journals
Reviews
Chang B, Svoboda KKH & Liu X. (2019). Cell polarization: From epithelial cells to odontoblasts. Eur. J. Cell Biol. , 98, 1-11. PMID: 30473389 DOI.
Bonczek O, Balcar VJ & Šerý O. (2017). PAX9 gene mutations and tooth agenesis: A review. Clin. Genet. , 92, 467-476. PMID: 28155232 DOI.
Kawashima N & Okiji T. (2016). Odontoblasts: Specialized hard-tissue-forming cells in the dentin-pulp complex. Congenit Anom (Kyoto) , 56, 144-53. PMID: 27131345 DOI.
Peterkova R, Hovorakova M, Peterka M & Lesot H. (2014). Three-dimensional analysis of the early development of the dentition. Aust Dent J , 59 Suppl 1, 55-80. PMID: 24495023 DOI.
Bei M. (2009). Molecular genetics of tooth development. Curr. Opin. Genet. Dev. , 19, 504-10. PMID: 19875280 DOI.
Seppala M, Zoupa M, Onyekwelu O & Cobourne MT. (2006). Tooth development: 1. Generating teeth in the embryo. Dent Update , 33, 582-4, 586-8, 590-1. PMID: 17209531
Thesleff I. (2006). The genetic basis of tooth development and dental defects. Am. J. Med. Genet. A , 140, 2530-5. PMID: 16838332 DOI.
Tompkins K. (2006). Molecular mechanisms of cytodifferentiation in mammalian tooth development. Connect. Tissue Res. , 47, 111-8. PMID: 16753804 DOI.
Cobourne MT & Sharpe PT. (2003). Tooth and jaw: molecular mechanisms of patterning in the first branchial arch. Arch. Oral Biol. , 48, 1-14. PMID: 12615136
Sharpe PT. (2001). Neural crest and tooth morphogenesis. Adv. Dent. Res. , 15, 4-7. PMID: 12640730 DOI.
Articles
Yuan X, Cao X & Yang S. (2019). IFT80 is required for stem cell proliferation, differentiation, and odontoblast polarization during tooth development. Cell Death Dis , 10, 63. PMID: 30683845 DOI.
Lin D, Huang Y, He F, Gu S, Zhang G, Chen Y & Zhang Y. (2007). Expression survey of genes critical for tooth development in the human embryonic tooth germ. Dev. Dyn. , 236, 1307-12. PMID: 17394220 DOI.
Nakatomi M, Morita I, Eto K & Ota MS. (2006). Sonic hedgehog signaling is important in tooth root development. J. Dent. Res. , 85, 427-31. PMID: 16632755 DOI.
Ogawa T, Kapadia H, Feng JQ, Raghow R, Peters H & D'Souza RN. (2006). Functional consequences of interactions between Pax9 and Msx1 genes in normal and abnormal tooth development. J. Biol. Chem. , 281, 18363-9. PMID: 16651263 DOI.
Kettunen P & Thesleff I. (1998). Expression and function of FGFs-4, -8, and -9 suggest functional redundancy and repetitive use as epithelial signals during tooth morphogenesis. Dev. Dyn. , 211, 256-68. PMID: 9520113 <256::AID-AJA7>3.0.CO;2-G DOI.
Search PubMed
Search Pubmed: Tooth Development | odontogenesis | tooth morphogenesis | adontia | amelogenesis imperfecta | dens evaginatus | hypodontia
Additional Images
Category:Tooth | Category:Integumentary
Historic Images
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Template:Ref- Keith1902 Development and Morphology of the Teeth (1902)
Grosser O. Lewis FT. and McMurrich JP. The Development of the Digestive Tract and of the Organs of Respiration. (1912) chapter 17, vol. 2, in Keibel F. and Mall FP. Manual of Human Embryology II. (1912) J. B. Lippincott Company, Philadelphia. The Teeth (1912)
Bailey FR. and Miller AM. Text-Book of Embryology (1921) New York: William Wood and Co. The Teeth (1921)
Terms
| Tooth Terms | ||
|---|---|---|
Tooth Development
| ||
|
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- StemBook - Tooth organogenesis and regeneration
- University of Helsinki Gene Expression in Tooth
- American Dental Association Overview - Tooth
- Columbia University Medical Centre Illustrations: How a Tooth Decays
- Merck Tooth disorders
- Nemours Foundation Teething Tots
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 27) Embryology Integumentary System - Tooth Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Integumentary_System_-_Tooth_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G