ANAT2341 Lab 7: Difference between revisions
m (→References) |
|||
| Line 38: | Line 38: | ||
===References=== | ===References=== | ||
<pubmed>20182616</pubmed> | |||
<pubmed>21147753</pubmed> | |||
<pubmed>25343988</pubmed> | |||
<pubmed>21620133</pubmed> | |||
<pubmed>22484060</pubmed> | |||
Revision as of 11:43, 18 July 2017
1. QUIZ
2. Guest Lecturer - Hongjun Shi
Introduction
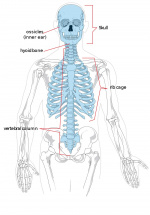
The defining feature of vertebrates is the vertebral column which is composed of a series of structurally similar bone units – vertebrae along the body axis. The segmental pattern of the vertebral column is established during early embryo development when the somites are rhythmically produced from the paraxial mesoderm (Bailey and Dale, 2001). Somitogenesis requires the interaction of two components, referred to as the clock and wavefront (Aulehla and Pourquie, 2010; Pourquie, 2011). The wavefront (determination front) determines the future somite boundary and is created by opposing gradients of FGF and Wnt signaling (caudal-rostral) and retinoic acid signaling (rostral-caudal) in the presomitic mesoderm (PSM). At the same time, PSM cells express a number of genes in the FGF, Wnt and Notch signaling pathway in an oscillatory pattern (the clock), and bands of expression appear to move in a caudal-to-rostral direction. When the periodic signal reaches the determination front, a somite can form. Disruption of somitogenesis by genetic mutations results in vertebral defects. For example, in humans and mice, homozygous mutation of the DLL3, LFNG, MESP2, HES7 and RIPPLY2 (Pourquie, 2011) (McInerney-Leo et al., 2015) which are either components or downstream targets of Notch signalling leads to extensive vertebral malformation.
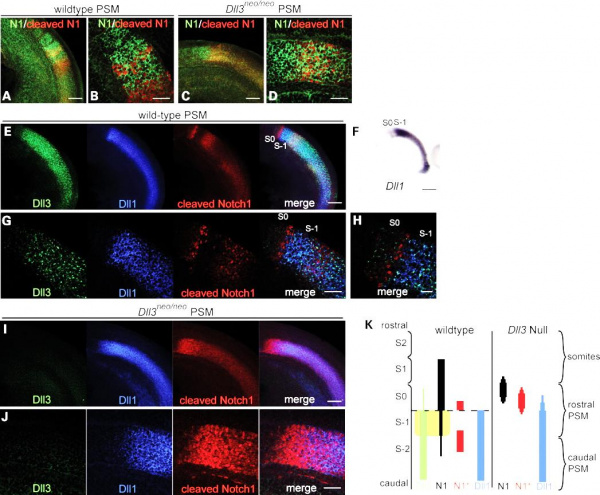
Figure 1: Deletion of Dll3 gene casues failure of Notch1 signalling to restrict to a thin stripe of cells. In stead a broad rostral domain of Notch1 signalling is observed (Chapman et al., 2011).
In addition to genetic mutations, environmental insults during embryo development may also interfere with somitogenesis. For example, hypoxia during pregnancy can inhibit FGF signaling in the PSM and cause segmentation defects in mice. Heterozygous mutation in the Notch signaling genes increase the susceptibility to segmentation defects when the mice are exposed to mild hypoxia (Sparrow et al., 2012).
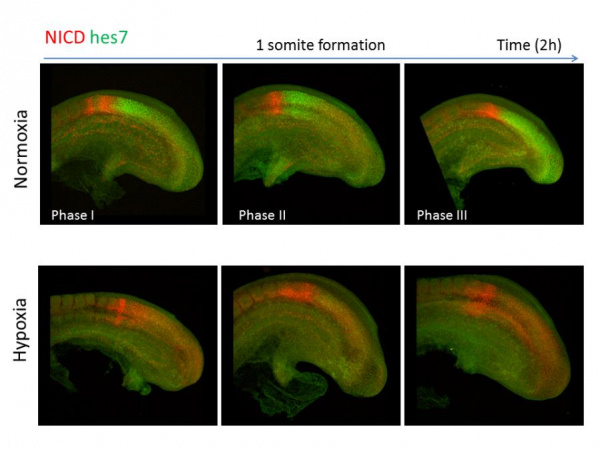
Figure 2: Oscillatory pattern of Hes7 expression and Notch signaling in PSM under normal conditions. Under the hypoxic condition, Hes7 protein expression is lost and a broader expression domain of Notch signaling is observed in PSM.
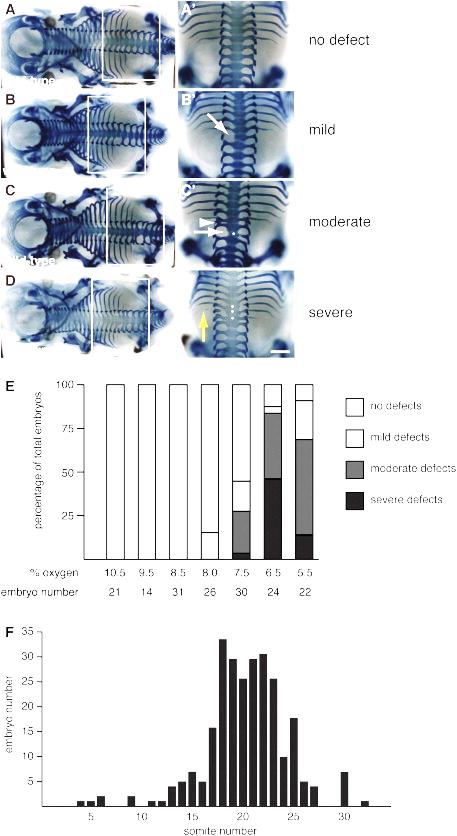
Figure 3: Vertebral defects induced by hypoxia (Sparrow et al., 2012).
References
<pubmed>20182616</pubmed> <pubmed>21147753</pubmed> <pubmed>25343988</pubmed> <pubmed>21620133</pubmed> <pubmed>22484060</pubmed>
Links: Axial Skeleton Development | Somitogenesis Molecular Movie