2017 Group Project 2
| 2017 Student Projects | |||
|---|---|---|---|
|
Kidney
Mark Hill (talk) 16:02, 14 September 2017 (AEST) OK Feedback
- I can see a list of sub-headings but have no feeling for the developmental process.
- There should be lots of images related to development including animal models.
- Where are all the component cells and their developmental origin.
- Molecular regulation of development.
- What are the current directions of research.
Introduction
The kidneys are two bean-shaped organs that play an important role in the renal system (also known as the urinary system). In the womb, the placenta is responsible for maintaining balance of water, nutrients, ion levels, etc. However, after birth, this responsibility is given to the kidneys. As such, they are vital for the everyday functions of the human body as they are the unit responsible for the filtration of blood and subsequent reabsorption of water and other nutrients according to what the body needs. The by-product of this process is urine, and it is excreted from the kidneys into the ureters which then deliver the urine to the urinary bladder.
Filtration is carried out in what is called the "nephron". In humans, the number of nephrons in each kidney ranges from 200,000 to 2.5 million. However, a typical kidney usually has approximately 900,000 to 1 million nephrons. Nephrons are the functional units of kidneys and are independent of each other. https://www.ncbi.nlm.nih.gov/pubmed/21604189
Kidney development in the embryo is known as nephrogenesis and has three stages: pronephros, mesonephros and metanephros. This process typically begins in week 4 of gestation and ends in week 36. About 1 in 500 babies are born with a kidney problem due to developmental abnormalities. https://www.kidney.org/atoz/content/detectkid Z5017644 (talk) 16:39, 31 August 2017 (AEST)
Developmentally, they arise from the intermediate mesoderm. Z5017644 (talk) 22:20, 15 September 2017 (AEST)
Structure of Kidney
A mature kidney is comprised of a cortex, medulla and papilla regions. Nephrons differentiate into many different cell types which are morphologically different. Z5178407 (talk) 19:59, 1 October 2017 (AEDT)
Timeline of Kidney Embryology
Kidney development (nephrogenesis) in humans begins in week 4 and commonly ends in week 36 of gestation. This is a brief timeline of the developmental processes.
| WEEK | DEVELOPMENT |
|---|---|
| 4 (early) | Pronephros begins (first stage of nephrogenesis) |
| 4 (late) | Mesonephros begins (second stage of nephrogenesis) |
| 5 | Metanephros begins (third last stage of nephrogenesis) |
| 6 | Ascension of kidneys from pelvis area begins |
| 8 | Functional kidney is formed. First nephrons are formed, a process that continues until week 36 |
| 9 | Kidneys complete their ascension and now sit just below the adrenal glands |
| 10 | Kidneys now ready to perform filtration |
| 36 | Nephrogenesis completed. No more nephrons formed from this point on. |
| Postnatal | Kidney and its structures continue to mature. |
Z5017644 (talk) 03:36, 16 September 2017 (AEST)
Kidney development
Development of the kidney is called nephrogenesis and it arises from the intermediate mesoderm in the metanephric blastema.. Here are the three main stages of nephrogenesis which begins in week 4 of gestation and ends in week 36. It must be noted that whilst nephrogenesis does not continue beyond week 36, maturation of the kidney and its functional units does continue. Z5017644 (talk) 16:35, 31 August 2017 (AEST)
Nephrogenesis
Nephrogenesis involves two transitory stages, pronephros and mesonephros, which end in a final stage (metanephros) giving us the final product of a functional kidney. It is not to say that kidney is ready and functional once metanephros is reached, but rather, it has reached the stage where its structures are set and can continue to mature into a proper functional kidney that can sustain life.
Z5017644 (talk) 00:49, 16 September 2017 (AEST)
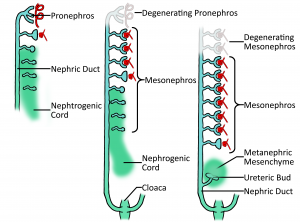
1. Pronephros
The earliest nephric stage in humans (week 4), arising from the intermediate mesoderm near the pharyngeal arches and extend from the 4th to the 14th somites and consists of 6-10 pairs of tubules. These spill into a pair (a 'pair' because there are two kidneys) of primary ducts (nephric or mesonephric duct) that are formed at the same level and go on to extend caudally. The pronephros is a transient structure that disappears completely by the 4th week of human embryonic life, its degradation can be seen in the diagram. (z5015446) Z5017644 (talk) 01:30, 16 September 2017 (AEST)
2. Mesonephros
This stage sees the continuation of the nephric duct caudally, with mesonephric tubules arising laterally from it. Together, these structures are known as the mesonephros and whilst still a transient structure, it has important excretory functions during early embryonic life (4—8 weeks). Gradually the top two thirds of the mesonephros go onto form the genitals, however the last third continues to form the functional kidney. This part of the mesonephros that goes on to form the kidney is known as the nephrogenic chord which is essentially mesenchymal tissue.
Towards the end of week 4 of development, the nephrogenic duct starts to move away from the nephric duct as seen in the diagram, whilst the nephric duct continues to grow caudally.
(z5015446) Z5017644 (talk) 01:30, 16 September 2017 (AEST)
3. Metanephros
At the fifth week of development, a lateral projection called the ureteric bud develops from the nephric duct whilst the nephrogenic chord has now detached itself from the nephric duct to form the mentanephric blastema, which has been described as a cloud of mesenchymal cells[1]. After receiving relevant signalling, the metanephric blastema and ureteric bud interact by way of the blastema 'clouding around' the bud. This cloud takes the traditional kidney-bean shape and just gets bigger as time goes on.
Inside the metanephric blastema, the ureteric bud bifurcates to form the calyces, pelvis, ureter and collecting tubules of the kidney, where as blastema's mesenchymal cells go on to form numerous vesicles which develop into glomeruli and Bowman's capsules. Their interaction allows for the creation of the nephron. However, as general overview, the ureteric bud differentiates to create the renal tubule section of the kidney (reabsorption compartment of kidney), while the metanephric blastema's mesenchymal cells differentiate to create the renal corpuscle section (filtration compartment).
The permanent and functional kidney is now ready at week 8, although nephrons are still being made until approximately week 36 as they are incredibly important for life and therefore, there needs to be a lot of them. (z5015446) Z5017644 (talk) 02:39, 16 September 2017 (AEST)
[1]http://www.sciencedirect.com/science/article/pii/S1534580710002078
Nephron development
Both the mesonephric blastema and ureteric bud contribute to the formation of the nephron and its two main units, the glomerulus and Bowman's capsule.
http://www.sciencedirect.com/science/article/pii/S1084952114002390#bib0025 Z5017644 (talk) 02:41, 16 September 2017 (AEST)
Ascension
During metanephros, the ureteric bud forms the ureter. The ureter of each kidney descends from the kidney complex and connects to the urinary bladder. At week 6, the ureters ascend upwards as the torso of the foetus continues to extend. At week 9, they assume their permanent and proper anatomical position under the adrenal glands.
Genes Expressed
http://www.sciencedirect.com/science/article/pii/S0925477397006679?via%3Dihub https://www.ncbi.nlm.nih.gov/pubmed/11937757 Z5017644 (talk) 04:20, 16 September 2017 (AEST)
Blood Supply
Z5017644 (talk) 04:01, 16 September 2017 (AEST)
Developmental abnormalities
As mentioned in the Introduction, every 1 in 500 newborns suffer from congenital abnormalities of the kidney and urinary tract (CAKUT). Studies have shown that certain CAKUT increases the risks of developing hypertension and cardiovascular diseases at adulthood.
An increase in anti-α smooth muscle actin (α-SMA),vimentin, and fibronectin expression in renal tissue (Balbi et al., 2008; Lamouille et al., 2014) as well as a decrease of the proximal tubule cubulin receptor (Lima et al., 2010) have been associated with disruptions in renal development. The proximal tubule receptor is important as it characterises the epithelial-mesenchymal transition (EMT) process, which is a physiological process that occurs during early embryogenesis, tissue repair, and pathology (Kalluri and Weinberg, 2009).
Vimentin and α-SMA are only expressed before the differentiation or transdifferentiation processes in epithelial cells. During this process, cells can proliferate, migrate and produce extracellular matrix. Thus, these proteins can be utilised as a marker of cell indifferentiation.
Although calcitriol is widely known for its important role in the homeostasis of calcium homeostasis and bone metabolism, recent studies have shown that it is also involved in he homeostasis of other cellular processes. These processes include the control of of autoimmunity, inflammatory process as well as blood pressure. Furthermore, calcitriol participates in the regulation of cell proliferation and differentiation processes, and the regulation of the renin gene.
Kidney developmental abnormalities are diverse and they correspond to defects at different stages of kidney development. Renal vascular anomalies are defects involving renal arteries and renal veins while fusion anomalies results in conditions such as fused pelvic kidney, crossed fused renal ectopia and horseshoe kidney.
Renal agenesis (absence of one or both kidneys) Multiple ureters (more than one ureter draining a kidney) Hypoplastic kidneys (underdevelopment of the kidneys) Dysplastic kidneys (abnormal development of kidneys which arises from tubules failing to branch out completely) Wilms tumours (kidney cancer) Patterning defects
Z5178407 (talk) 15:14, 14 September 2017 (AEST)
Z5178407 (talk) 15:14, 14 September 2017 (AEST)
Current Research
Can kidney disease be associated with nephron number?
Nephron development ceases around week 36 of gestation at the end of nephrogenesis, the body can not create new nephrons beyond that point. Due to the wide range of possible nephron numbers (250,000 - 2.5 million), many investigations have arisen to determine whether a lower nephron count predisposes a person to kidney disease later on in life.
https://www.ncbi.nlm.nih.gov/pubmed/16014104
https://www.ncbi.nlm.nih.gov/pubmed/16774009
https://www.ncbi.nlm.nih.gov/pubmed/21604189
https://www.ncbi.nlm.nih.gov/pubmed/19615565
https://www.ncbi.nlm.nih.gov/pubmed/28818273 Z5017644 (talk) 16:50, 31 August 2017 (AEST)
The Cellular Basis of Kidney Development Mammalian kidney development has helped elucidate the general concepts of mesenchymal-epithelial interactions, inductive signaling, epithelial cell polarization, and branching morphogenesis. Through the use of genetically engineered mouse models, the manipulation of Xenopus and chick embryos, and the identification of human renal disease genes, the molecular bases for many of the early events in the developing kidney are becoming increasingly clear. Early patterning of the kidney region depends on interactions between Pax/Eya/Six genes, with essential roles for lim1 and Odd1. Ureteric bud outgrowth and branching morphogenesis are controlled by the Ret/Gdnf pathway, which is subject to positive and negative regulation by a variety of factors. A clear role for Wnt proteins in induction of the kidney mesenchyme is now well established and complements the classic literature nicely. Patterning along the proximal distal axis as the nephron develops is now being investigated and must involve aspects of Notch signaling. The development of a glomerulus requires interactions between epithelial cells and infiltrating endothelial cells to generate a unique basement membrane. The integrity of the glomerular filter depends in large part on the proteins of the nephrin complex, localized to the slit diaphragm. Despite the kidney's architectural complexity, with the advent of genomics and expression arrays, it is becoming one of the best-characterized organ systems in developmental biology.
WT-1 is Required For Early Kidney Development In humans, germline mutations of the WT-1 tumor suppressor gene are associated with both Wilms' tumors and urogenital malformations. To develop a model system for the molecular analysis of urogenital development, we introduced a mutation into the murine WT-1 tumor suppressor gene by gene targeting in embryonic stem cells. The mutation resulted in embryonic lethality in homozygotes, and examination of mutant embryos revealed a failure of kidney and gonad development. Specifically, at day 11 of gestation, the cells of the metanephric blastema underwent apoptosis, the ureteric bud failed to grow out from the Wolffian duct, and the inductive events that lead to formation of the metanephric kidney did not occur. In addition, the mutation caused abnormal development of the mesothelium, heart, and lungs. Our results establish a crucial role for WT-1 in early urogenital development.
User:Z5015446
Questions for the future
General info on the renal system
Z5017644 (talk) 16:59, 31 August 2017 (AEST)
References
Balbi APC, Marin ECS, Francescato HDC, Costa RS, Coimbra TM. MAPK and angiotensin II receptor in kidney of newborn rats from losartan-treated dams. Pediatr Nephrol. 2008;23:1433–1444. doi: 10.1007/s00467-008-0830-1. [PubMed]
Kalluri R, Weinberg RA. The basics of epithelial-mesenchymal transition. J Clin Invest. 2009;6:1420–1428. doi: 10.1172/JCI39104. [PubMed]
Lamouille S, Jian XJ, Derynck R. Molecular mechanisms of epithelial–mesenchymal transition. Nat Rev Mol Cell Biol. 2014;3:178–196. doi: 10.1038/nrm3758. [PubMed]
Lima WR, et al. ZONAB promotes proliferation and represses differentiation of proximal tubule epithelial cells. J Am Soc Nephrol. 2010;3:478–88. doi: 10.1681/ASN.2009070698. [PubMed]
Z5015446 (talk) 16:40, 31 August 2017 (AEST) Z5015446 (talk) 16:52, 31 August 2017 (AEST)