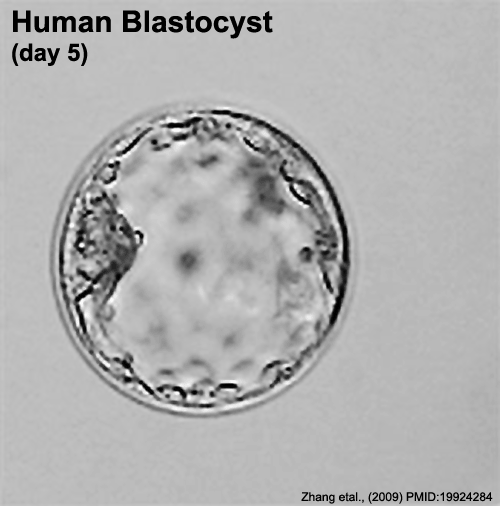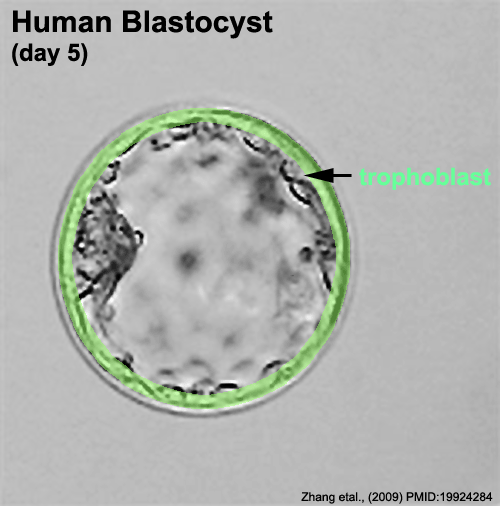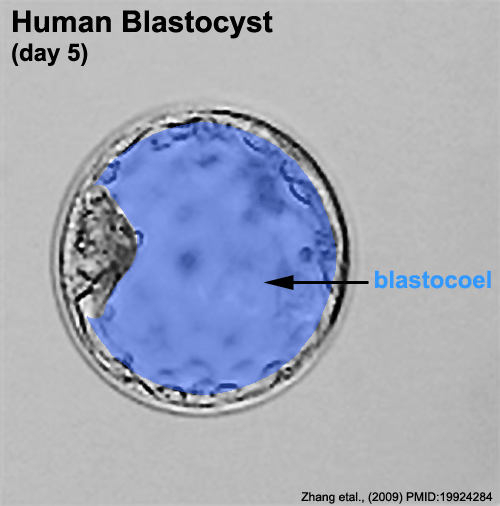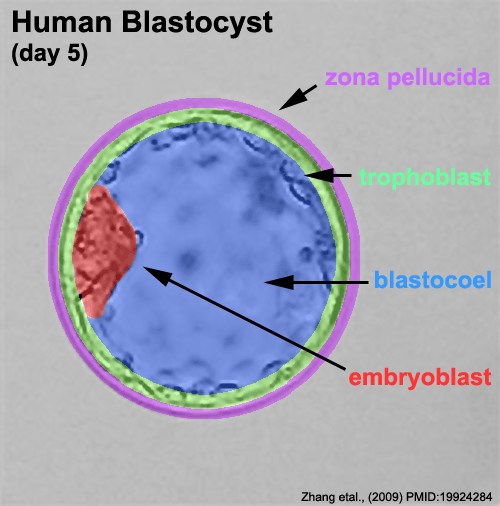
Zona pellucida
| Embryology - 27 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
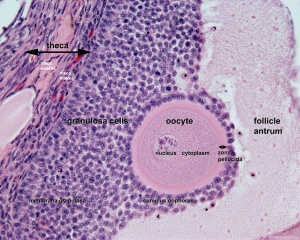
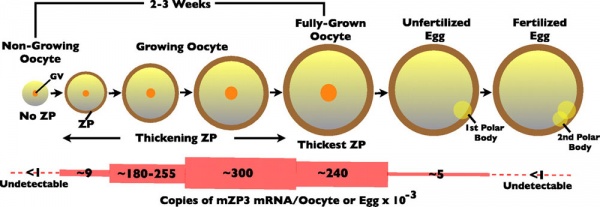
(Latin, zona pellucida = transparent zone) The zona pellucida (ZP) is a specialized extracellular matrix surrounding the developing oocyte (egg, ovum) within each follicle within the ovary. This thick matrix is thought to be formed by secretions from the oocyte and the follicle granulosa cells and in human oocytes consists of four types of zona pellucida glycoproteins ZP1, ZP2, ZP3 and ZP4 which have different roles in fertilization. Note that mice also have 4 ZP genes, but multiple stop and missense codons in ZP4 means that only three are produced. Polymers of ZP2 and ZP3 organized into extended filaments that are cross-linked by ZP1 homodimers.
Note that depending upon species and type of study, the zona pellucida can also be called the: oolemma, egg coat or vitelline membrane.
The zona pellucid has many different roles including in oocyte development, protection during growth and transport, fertilization, spermatozoa binding, preventing polyspermy, blastocyst development, and preventing premature implantation (ectopic pregnancy).
|
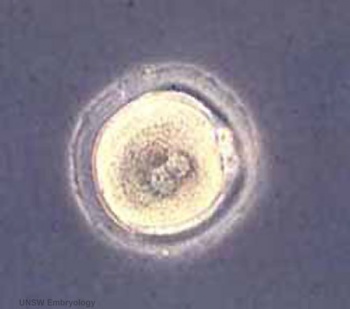
|
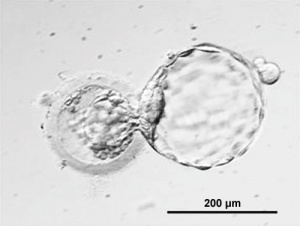
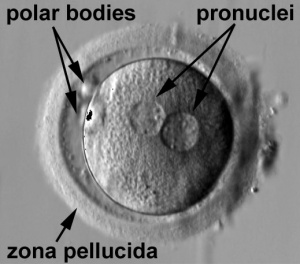
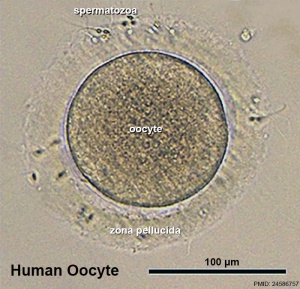
| Human oocyte contained inside zona pellucida.[1] | Early zygote inside zona pellucida |
In human development, during the first week of development following fertilization the zona pellucida remains surrounding the blastocyst from which it "hatches" to commence implantation.
Some Recent Findings

|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Zona pellucida Development | Zona pellucida |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
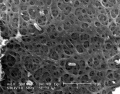
Three-dimensional Structure
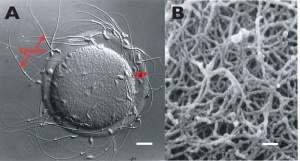
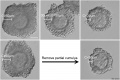
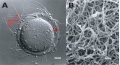
Electron microscopic studies have shown a variety of appearances from a porous, net-like structure to a nearly smooth and compact structure.
An scanning electron microscopic review[10] of both human and mouse zona pellucida structures at ovulation show:
- spongy ZP appearance well correlates with mature oocytes.
- a delicate meshwork of thin interconnected filaments
- a regular alternating pattern of wide and tight meshes.
- wide meshes correspond to "pores" of the "spongy" ZP.
- tight meshes correspond to the compact parts of the ZP surrounding the pores.
Human Zona Pellucida
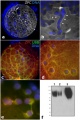
An immunochemistry study[11] of human ZP proteins expression during folliculogenesis within the ovary primordial follicle identified the presence of ZP1 and ZP3 in most follicles (93% and 95%, respectively), with ZP2 only in 32% of these follicles. The ZP proteins were also detected in the cytoplasm of cuboidal granulosa cells. This localization suggested to the authors that these proteins had been present since oogenesis.
Mouse Zona Pellucida
Mouse zona pellucida development.[9]
In the mouse ovarian oocytes, and not granulosa cells, are the only source for mouse ZP glycoproteins.<[12]
Zona Pellucida Glycoprotein Features

- A "ZP domain", which is a signature domain comprised of approximately 260 amino acid (aa) residues.
- An N-terminal hydrophobic signal peptide sequence.
- A potential N- and O-linked glycosylation sites.
- A C-terminal hydrophobic transmembrane-like domain (TMD)
- A potential consensus proprotein convertase (furin) cleavage site (CFCS) upstream of transmembrane-like domain (TMD).
Zona Pellucida glycoprotein 1
(ZP1) A 638 amino acid glycoprotein located in the zona pellucida, synthesized by and surrounding the oocyte.
- Links: OMIM - ZP1
Zona Pellucida glycoprotein 2
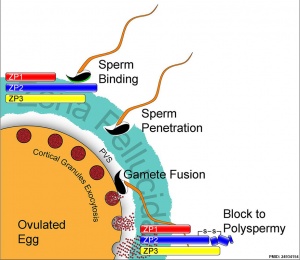
(ZP2) A 745 amino acid glycoprotein located in the zona pellucida, synthesized by and surrounding the oocyte. The protein acts as a secondary sperm receptor that binds sperm only after the induction of the sperm acrosome reaction. Before fertilization ZP2 binds spermatozoa. After fertilization ZP2 is proteolytically cleaved as an initial block to polyspermy.
- Links: OMIM - ZP2
Zona Pellucida glycoprotein 3
Zona Pellucida Glycoprotein 3 (ZP3) A 424 amino acid glycoprotein located in the zona pellucida, synthesized by and surrounding the oocyte required for initial zona matrix formation and during fertilization for species-specific sperm binding. Now thought to exist in 2 isoforms ZP3A and ZP3B (a second polymorphic allele).
A study using the mouse oocyte,[14] has shown that ZP3 is also required for germinal vesicle breakdown in oocyte meiosis. ZP3 was initially expressed in the nucleus during prophase, and then translocates to the zona pellucida. Several potential ZP3-interacting proteins for the germinal vesicle effect were identified in this study: Protein tyrosine phosphatase, receptor type K (Ptprk), Aryl hydrocarbon receptor-interacting protein-like 1 (Aipl1), and Diaphanous related formin 2 (Diaph2).
- Links: OMIM - ZP3
Zona Pellucida glycoprotein 4
(ZP4) A 540 amino acid glycoprotein located in the zona pellucida, synthesized by and surrounding the oocyte required for initial zona matrix formation and along with ZP3 during fertilization for inducing the acrosome reaction and inhibited the binding of spermatozoa to zona pellucida in a time- and dose-dependent reaction. Associated with several mammalian species (human, rat, hamster and rabbit) but not all mammalian species.
A rabbit ZP4 knockout study showed an important ZP structural role for this protein.[15] Earlier ovulation, fertilization and development to blastocyst were normal, though embryos covered by a ZP4-devoided zona, had a thinner, more permeable, and disorganized and fenestrated structure.
- Links: OMIM - ZP4
Zona Pellucida Binding Protein
(ZPBP) Not a zona pellucida protein, but a spermatozoa protein found located on the acrosome surface and one of several proteins that participate in secondary binding between acrosome-reacted sperm and the zona pellucida.
- Links: OMIM - ZPBP
Zona Pellucida Birefringence
(ZPB) Optical property of the zona pellucida using polarization imaging when viewed microscopically. This property has been used to qualitatively predict the developmental potential of a in vitro matured metaphase-II (MII) oocytes.
High birefringence, compared to low, has been associated with oocytes contributing to conception cycles when compared with those of nonconception cycles and higher implantation, pregnancy, and live birth rates from transferred oocytes.[16][7] This work follows on from the earlier discovery of an increased birefringence in the meiotic spindle an early indicator of oocyte activation[17]
Oocyte zona birefringence intensity is associated with embryonic implantation potential in ICSI cycles[16]
- "Using zona birefringence as the only selection criterion, two fertilized oocytes, preferably derived from high birefringence (HZB) oocytes, were selected for further culture and transfer. The required criteria were met by 135 ICSI cycles (124 patients; 34.9 ± 4.1 years of age). Embryos for transfer were used in 20 cycles derived from HZB/HZB oocytes, in 50 cycles from HZB/LZB oocytes and in 65 from LZB/LZB oocytes. The corresponding implantation (P < 0.025), pregnancy (P < 0.005) and live birth (P < 0.025) rates were significantly different between HZB/HZB and HZB/LZB versus LZB/LZB group. Embryo development was superior in embryos derived from HZB oocytes."
Zona pellucida birefringence in in vivo and in vitro matured oocytes[7]
The percentage of high birefringence oocytes was higher in immature than in mature oocytes (40.1 vs. 23.6%). Among immature oocytes, an increased percentage of HB in prophase-I stage oocytes compared to metaphase I stage oocytes was also observed (50.7 vs. 25.0%). However, the percentage of HB oocytes did not change when comparing oocytes before and after in vitro maturation for both prophase I and metaphase I oocytes. No influence of ZPB was observed on the spontaneous in vitro maturation potential. Exclusively for metaphase II retrieved oocytes, a positive influence of ZPB on fertilization (odds ratio [OR], 1.78; 95% confidence interval [CI], 1.27-2.49) and embryo quality (OR, 2.28; 95% CI, 1.04-4.99) was noted.
Birefringence - Defined as the double refraction of light in a transparent, molecularly ordered material, which is manifested by the existence of orientation-dependent differences in refractive index.
Abnormalities
Empty Follicle Syndrome
Empty Follicle Syndrome (EFS) is clinically defined as the failure to aspirate oocytes from mature ovarian follicles during in vitro fertilization. At least one genetic cause has been identified, a missense mutation in zona pellucida protein 3 (ZP3).[18]
Zona Pellucida Antibodies
The zona pellucida has been shown to contain tissue-specific and species-specific antigens that can generate antibodies that inhibit sperm attachment. Anti-zona pellucida antibodies have been shown to occur in human serum though the cause of their generation is largely unknown.[19] It has been suggested that this may be a contributing factor to infertility[20] and premature ovarian failure.[21]
Note the generation of zona pellucida antibodies from specific antigens has also been used in controlling animal fertility.[22] This has also been suggested as a possible target in generating a contraceptive vaccine for controlling human fertility.[22]
Terms
- acrosome reaction - The chemical change within the spermatozoa following binding to the zona pellucida, that leads to the release of acrosomal enzymatic contents. These enzymes degrade the zona pellucida and allow a spermatozoa to penetrate an oocyte.
- granulosa cell - A specific cell type that proliferates in association with the oocyte within the developing follicles of the ovary. These cells form the follicle stratum granulosa and are also given specific names based upon their position within the follicle. In the antral follicle, membrana granulosa sits on the follicular basal lamina and lines the antrum as a stratified epithelium. The cumulus oophorus is a column of granulosa cells that attaches the oocyte to the follicle wall. The corona radiata are the granulosa cells that directly surround the oocyte, and are released along with it at ovulation. Following ovulation the corona radiata provide physical protection to the oocyte and granulosa cells within the ovulating follicle contribute to corpus luteum.
References
- ↑ 1.0 1.1 Shi W, Xu B, Wu LM, Jin RT, Luan HB, Luo LH, Zhu Q, Johansson L, Liu YS & Tong XH. (2014). Oocytes with a dark zona pellucida demonstrate lower fertilization, implantation and clinical pregnancy rates in IVF/ICSI cycles. PLoS ONE , 9, e89409. PMID: 24586757 DOI.
- ↑ Zhou HX, Ma YZ, Liu YL, Chen Y, Zhou CJ, Wu SN, Shen JP & Liang CG. (2014). Assessment of mouse germinal vesicle stage oocyte quality by evaluating the cumulus layer, zona pellucida, and perivitelline space. PLoS ONE , 9, e105812. PMID: 25144310 DOI.
- ↑ Dai C, Chen Y, Hu L, Du J, Gong F, Dai J, Zhang S, Wang M, Chen J, Guo J, Zheng W, Lu C, Wu Y, Lu G & Lin G. (2019). ZP1 mutations are associated with empty follicle syndrome: evidence for the existence of an intact oocyte and a zona pellucida in follicles up to the early antral stage. A case report. Hum. Reprod. , 34, 2201-2207. PMID: 31734689 DOI.
- ↑ Bokhove M & Jovine L. (2018). Structure of Zona Pellucida Module Proteins. Curr. Top. Dev. Biol. , 130, 413-442. PMID: 29853186 DOI.
- ↑ Kellam L, Pastorelli LM, Bastida AM, Senkbeil A, Montgomery S, Fishel S & Campbell A. (2017). Perivitelline threads in cleavage-stage human embryos: observations using time-lapse imaging. Reprod. Biomed. Online , 35, 646-656. PMID: 29074360 DOI.
- ↑ 6.0 6.1 Izquierdo-Rico MJ, Gimeno L, Jiménez-Cervantes C, Ballesta J & Avilés M. (2011). Biosynthesis of hamster zona pellucida is restricted to the oocyte. Theriogenology , 75, 463-72. PMID: 21074836 DOI.
- ↑ 7.0 7.1 7.2 de Almeida Ferreira Braga DP, de Cássia Savio Figueira R, Queiroz P, Madaschi C, Iaconelli A & Borges E. (2010). Zona pellucida birefringence in in vivo and in vitro matured oocytes. Fertil. Steril. , 94, 2050-3. PMID: 20079896 DOI.
- ↑ Ganguly A, Bukovsky A, Sharma RK, Bansal P, Bhandari B & Gupta SK. (2010). In humans, zona pellucida glycoprotein-1 binds to spermatozoa and induces acrosomal exocytosis. Hum. Reprod. , 25, 1643-56. PMID: 20504872 DOI.
- ↑ 9.0 9.1 Wassarman PM. (2008). Zona pellucida glycoproteins. J. Biol. Chem. , 283, 24285-9. PMID: 18539589 DOI.
- ↑ Familiari G, Relucenti M, Heyn R, Micara G & Correr S. (2006). Three-dimensional structure of the zona pellucida at ovulation. Microsc. Res. Tech. , 69, 415-26. PMID: 16703610 DOI.
- ↑ Gook DA, Edgar DH, Borg J & Martic M. (2008). Detection of zona pellucida proteins during human folliculogenesis. Hum. Reprod. , 23, 394-402. PMID: 18033806 DOI.
- ↑ El-Mestrah M, Castle PE, Borossa G & Kan FW. (2002). Subcellular distribution of ZP1, ZP2, and ZP3 glycoproteins during folliculogenesis and demonstration of their topographical disposition within the zona matrix of mouse ovarian oocytes. Biol. Reprod. , 66, 866-76. PMID: 11906903
- ↑ Avella MA, Baibakov B & Dean J. (2014). A single domain of the ZP2 zona pellucida protein mediates gamete recognition in mice and humans. J. Cell Biol. , 205, 801-9. PMID: 24934154 DOI.
- ↑ Gao LL, Zhou CX, Zhang XL, Liu P, Jin Z, Zhu GY, Ma Y, Li J, Yang ZX & Zhang D. (2017). ZP3 is Required for Germinal Vesicle Breakdown in Mouse Oocyte Meiosis. Sci Rep , 7, 41272. PMID: 28145526 DOI.
- ↑ Lamas-Toranzo I, Fonseca Balvís N, Querejeta-Fernández A, Izquierdo-Rico MJ, González-Brusi L, Lorenzo PL, García-Rebollar P, Avilés M & Bermejo-Álvarez P. (2019). ZP4 confers structural properties to the zona pellucida essential for embryo development. Elife , 8, . PMID: 31635692 DOI.
- ↑ 16.0 16.1 Montag M, Schimming T, Köster M, Zhou C, Dorn C, Rösing B, van der Ven H & Ven der Ven K. (2008). Oocyte zona birefringence intensity is associated with embryonic implantation potential in ICSI cycles. Reprod. Biomed. Online , 16, 239-44. PMID: 18284880
- ↑ Liu L, Trimarchi JR, Oldenbourg R & Keefe DL. (2000). Increased birefringence in the meiotic spindle provides a new marker for the onset of activation in living oocytes. Biol. Reprod. , 63, 251-8. PMID: 10859266
- ↑ Chen T, Bian Y, Liu X, Zhao S, Wu K, Yan L, Li M, Yang Z, Liu H, Zhao H & Chen ZJ. (2017). A Recurrent Missense Mutation in ZP3 Causes Empty Follicle Syndrome and Female Infertility. Am. J. Hum. Genet. , 101, 459-465. PMID: 28886344 DOI.
- ↑ Caudle MR & Shivers CA. (1989). Current status of anti-zona pellucida antibodies. Am. J. Reprod. Immunol. , 21, 57-60. PMID: 2697213
- ↑ Koyama K, Hasegawa A, Mochida N & Calongos G. (2005). Follicular dysfunction induced by autoimmunity to zona pellucida. Reprod Biol , 5, 269-78. PMID: 16372044
- ↑ Takamizawa S, Shibahara H, Shibayama T & Suzuki M. (2007). Detection of antizona pellucida antibodies in the sera from premature ovarian failure patients by a highly specific test. Fertil. Steril. , 88, 925-32. PMID: 17349637 DOI.
- ↑ 22.0 22.1 Naz RK, Gupta SK, Gupta JC, Vyas HK & Talwar AG. (2005). Recent advances in contraceptive vaccine development: a mini-review. Hum. Reprod. , 20, 3271-83. PMID: 16113040 DOI.
Reviews
Gupta SK. (2018). The Human Egg's Zona Pellucida. Curr. Top. Dev. Biol. , 130, 379-411. PMID: 29853184 DOI.
Nishio S, Okumura H & Matsuda T. (2018). Egg-Coat and Zona Pellucida Proteins of Chicken as a Typical Species of Aves. Curr. Top. Dev. Biol. , 130, 307-329. PMID: 29853181 DOI.
Hasegawa A, Tanaka H & Shibahara H. (2014). Infertility and Immunocontraception based on zona pellucida. Reprod. Med. Biol. , 13, 1-9. PMID: 29699147 DOI.
Gupta SK. (2015). Role of zona pellucida glycoproteins during fertilization in humans. J. Reprod. Immunol. , 108, 90-7. PMID: 25445843 DOI.
Wassarman PM. (2008). Zona pellucida glycoproteins. J. Biol. Chem. , 283, 24285-9. PMID: 18539589 DOI.
Articles
Hirohashi N & Yanagimachi R. (2018). Sperm acrosome reaction: Its site and role in fertilization. Biol. Reprod. , , . PMID: 29462288 DOI.
Liu W, Bai D, Chen J & Gao S. (2017). Additive-effect pattern of both ZP2 and ZP3 in human and mouse. Hum. Genet. , 136, 1493-1495. PMID: 29094202 DOI.
Liu W, Li K, Bai D, Yin J, Tang Y, Chi F, Zhang L, Wang Y, Pan J, Liang S, Guo Y, Ruan J, Kou X, Zhao Y, Wang H, Chen J, Teng X & Gao S. (2017). Dosage effects of ZP2 and ZP3 heterozygous mutations cause human infertility. Hum. Genet. , 136, 975-985. PMID: 28646452 DOI.
Chen T, Bian Y, Liu X, Zhao S, Wu K, Yan L, Li M, Yang Z, Liu H, Zhao H & Chen ZJ. (2017). A Recurrent Missense Mutation in ZP3 Causes Empty Follicle Syndrome and Female Infertility. Am. J. Hum. Genet. , 101, 459-465. PMID: 28886344 DOI.
Bookshelf
Search NCBI Bookshelf zona pellucida | acrosome reaction
Search Pubmed
July 2010 "zona pellucida" All (4801) Review (582) Free Full Text (1408)
Search Pubmed: zona pellucida | acrosome reaction | Zona pellucida protein 1 | Zona pellucida protein 2 | Zona pellucida protein 3 | Zona pellucida protein 4
Search Images: zona pellucida
Additional Images
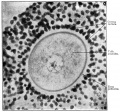
Mouse oocyte and zona pellucida - light and TEM
Mouse oocyte and zona pellucida TEM
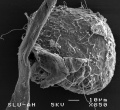
Cat oocyte zona pellucida SEM
Cat oocyte zona pellucida SEM
Cat oocyte zona pellucida spermatozoa bound SEM
Hamster oocyte zona pellucida SEM
Pig ZPC deposition in oocyte-cumulus complexes
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 27) Embryology Zona pellucida. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Zona_pellucida
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G