Respiratory System - Diaphragm
| Embryology - 21 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
The diaphragm is part of the musculoskeletal system, along with ribs and intercostals, that mechanically support respiration. In humans, the muscles of the diaphragm arise from somite level 3 to 5 (C3 to C5), which also corresponds to the levels of segmental nerves providing innervation of the diaphragm. The diaphragm begins functioning prenatally, and in the third trimester preparatory fetal respiratory movements occur, that are thought to have a number of roles in late respiratory and perhaps neural development.
Failure of complete diaphragm development can lead to a herniation of abdominal components through channels or gaps in the developing diaphragm into the pleural cavity.
The respiratory system does not carry out its physiological function (gas exchange) prenatally and postnatally the lungs continue to grow for another 8+ years. Respiration also is dependent upon the cardiovascular system, pulmonary circulation, developed prenatally activated and altered postnatally.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Diaphragm development | Fetal respiratory movements | Phrenic nerve development | Congenital diaphragmatic hernia |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
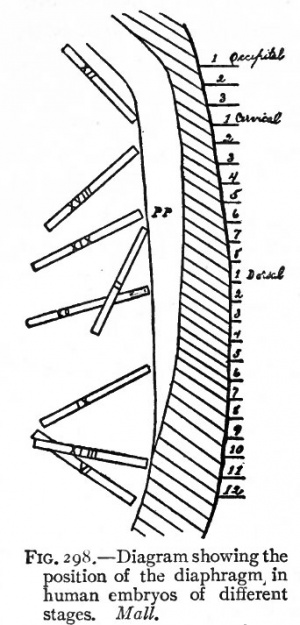
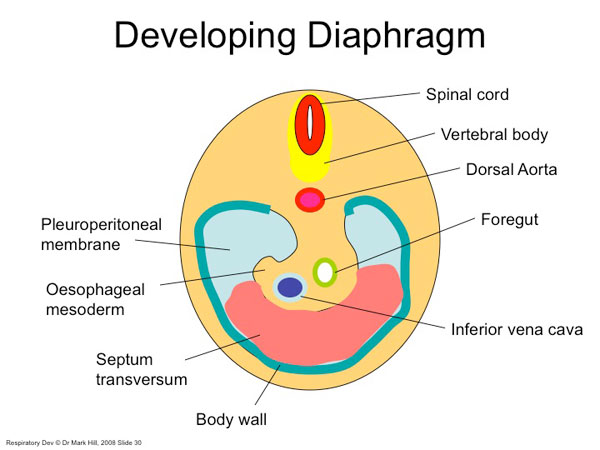
Diaphragm Components
|
Five elements contribute to the diaphragm.
septum transversum - central tendon 3rd to 5th somite - musculature of diaphragm (More? Somitogenesis) ventral pleural sac - connective tissue mesentry of oesophagus - connective tissue around oesophasus and IVC (More? Gastrointestinal Tract Development) pleuroperitoneal membranes - connective tissue around central tendon |
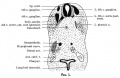
Pleuroperitoneal Fold
| The transient pleuroperitoneal fold (PPF) arise from the posterior body wall, and appears in late week 4 (Carnegie stage 13/14, CRL 6mm) and is present until week 6 (Carnegie stage 17, CRL 14mm).[5] After this time it can no longer be separated from the diaphragm. These pair of folds have a triangular shape and abnormalities in their development is related to congenital diaphragmatic hernia (CDH). | 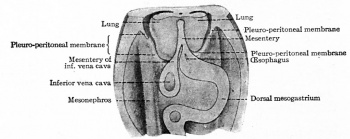
Rat pleuro-peritoneal membranes (historic drawing) |
Diaphragm Innervation
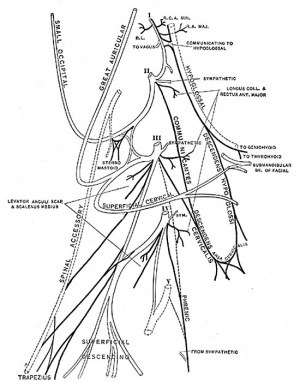 Adult Cervical Plexus (phrenic nerve shown lower right) |
Innervation of the human diaphragm is by the phrenic nerves, arising from the same segmental levels from which the diaphragm skeletal muscles arise, segmental levels C3 to C5.
The paired phrenic nerves are mixed containing motor neurons for the diaphragm and sensory nerves for other abdominal structures (mediastinum, pleura, liver, gall bladder). The two adult phrenic nerves differ in their length, and also in their anatomical relations at the upper part of the thorax. Netrin signaling may be important in early phrenic nerve growth, as knockout mice show incomplete phrenic nerve innervation of the diaphragm. Other respiratory muscles include the intercostals which are innervated by the intercostal nerves arising from segmental levels T1 to T11. |
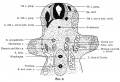
Historic
The images below are descending sections from a study of the 6 mm human embryo (~Carnegies stage 14) phrenic nerve by Amin (1914).[8]
Fetal Respiratory Movements
Fetal respiratory movements (FRM) or Fetal breathing movements (FBM) are thought to be regular muscular contrations occurring in the third trimester, preparing the respiratory muscular system for neonatal function and to also have a role in late lung development.
The majority of FBM research has been carried out in fetal sheep.[9]
Hypoxia, decreased oxygen levels, blocks these movements by inhibition of the brain stem respiratory centres.
Hypercapnia, increased carbon dioxide levels, or acid cerebrospinal fluid perfusion can cause an increase in the regularity and depth of breathing.
Hormones also affect fetal breathing movements: inhibitors of prostaglandin synthetase (indomethacin, meclofenamate or aspirin) induce continuous fetal breathing movements, while prostaglandin E2 arrests fetal breathing.
- The effect of fetal breathing movements on pulmonary blood flow in fetal sheep.[9] "FBM do not increase mean blood flow through the left pulmonary artery; thus, it is unlikely that FBM stimulate lung growth through changes in pulmonary blood flow."
- Maturation of fetal breathing activity.[10]
- The central control of fetal breathing and skeletal muscle movements.[11]
Hypercapnia (Greek, hyper = "above" and kapnos = "smoke") or hypercarbia increased carbon dioxide levels.
Abnormalities
LB00.0 Congenital Diaphragmatic Hernia
| ICD-11 |
|---|
|
LB00 Structural developmental anomalies of diaphragm
|
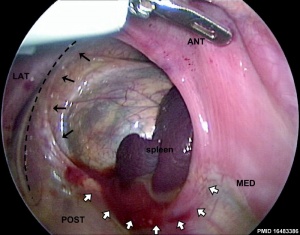
Failure of the pleuroperitoneal foramen (foramen of Bochdalek) to close allows viscera into thorax. Intestine, stomach or spleen can enter the pleural cavity, compressing the lung. This can also be associated with abnormal lung development.[13]
Bochdalek hernia - most common on the posterior left side (85%).

|
Left posterolateral diaphragmatic hernia[14]
|
Australian national rate (1982-1992) 2.1 - 3.8 /10,000 births.[15]
- Prenatal detection and outcome of congenital diaphragmatic hernia: a French registry-based study.[16] "501 cases of CDH were identified from a total of 1,835,022 live births (2.7/10 000 live births). The overall prenatal detection rate was 54%."
- Outcomes of congenital diaphragmatic hernia: a population-based study in Western Australia.[17] "Ninety-two percent of postoperative infants survived beyond 1 year of age, as did 80% of infants who reached the surgical referral center. However, only 52% of live-born infants, 32% of all cases, and 16% of all prenatally diagnosed cases survived. Therefore, the overall mortality rate for this condition remains high, despite increased prenatal detection, transfer to tertiary institutions for delivery, and advances in neonatal care, and is influenced significantly by the rate of prenatal termination. In our study, 33% of all cases of CDH and 49% of prenatally diagnosed fetuses underwent elective termination of pregnancy. This large number of fetal terminations confounds the accurate assessment of the true outcomes of this condition."
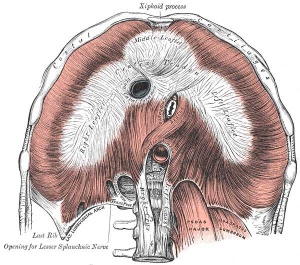
Adult Diaphragm

References
- ↑ Takahashi T, Friedmacher F, Zimmer J & Puri P. (2019). Ephrin-B1, -B2, and -B4 Expression is Decreased in Developing Diaphragms and Lungs of Fetal Rats with Nitrofen-Induced Congenital Diaphragmatic Hernia. Eur J Pediatr Surg , 29, 113-119. PMID: 30469162 DOI.
- ↑ Sefton EM, Gallardo M & Kardon G. (2018). Developmental origin and morphogenesis of the diaphragm, an essential mammalian muscle. Dev. Biol. , , . PMID: 29679560 DOI.
- ↑ Kardon G, Ackerman KG, McCulley DJ, Shen Y, Wynn J, Shang L, Bogenschutz E, Sun X & Chung WK. (2017). Congenital diaphragmatic hernias: from genes to mechanisms to therapies. Dis Model Mech , 10, 955-970. PMID: 28768736 DOI.
- ↑ Coles GL & Ackerman KG. (2013). Kif7 is required for the patterning and differentiation of the diaphragm in a model of syndromic congenital diaphragmatic hernia. Proc. Natl. Acad. Sci. U.S.A. , 110, E1898-905. PMID: 23650387 DOI.
- ↑ Clugston RD, Zhang W & Greer JJ. (2010). Early development of the primordial mammalian diaphragm and cellular mechanisms of nitrofen-induced congenital diaphragmatic hernia. Birth Defects Res. Part A Clin. Mol. Teratol. , 88, 15-24. PMID: 19711422 DOI.
- ↑ Mantilla CB & Sieck GC. (2008). Key aspects of phrenic motoneuron and diaphragm muscle development during the perinatal period. J. Appl. Physiol. , 104, 1818-27. PMID: 18403452 DOI.
- ↑ Burgess RW, Jucius TJ & Ackerman SL. (2006). Motor axon guidance of the mammalian trochlear and phrenic nerves: dependence on the netrin receptor Unc5c and modifier loci. J. Neurosci. , 26, 5756-66. PMID: 16723533 DOI.
- ↑ Amin M. The course of the phrenic nerve in the embryo. (1914) J Anat Physiol. 48(2): 215-8. PMID 17232992
- ↑ 9.0 9.1 Savich RD, Guerra FA, Lee CC & Kitterman JA. (1994). The effect of fetal breathing movements on pulmonary blood flow in fetal sheep. Pediatr. Res. , 35, 484-9. PMID: 8047386
- ↑ Blanco CE. (1994). Maturation of fetal breathing activity. Biol. Neonate , 65, 182-8. PMID: 8038281 DOI.
- ↑ Dawes GS. (1984). The central control of fetal breathing and skeletal muscle movements. J. Physiol. (Lond.) , 346, 1-18. PMID: 6422029
- ↑ Fisher JC & Bodenstein L. (2006). Computer simulation analysis of normal and abnormal development of the mammalian diaphragm. Theor Biol Med Model , 3, 9. PMID: 16483386 DOI.
- ↑ Ameis D, Khoshgoo N & Keijzer R. (2017). Abnormal lung development in congenital diaphragmatic hernia. Semin. Pediatr. Surg. , 26, 123-128. PMID: 28641748 DOI.
- ↑ Tovar JA. (2012). Congenital diaphragmatic hernia. Orphanet J Rare Dis , 7, 1. PMID: 22214468 DOI.
- ↑ P. Lancaster and E. Pedisich Congenital Malformations Australia 1981-1992 ISSN 1321-8352.
- ↑ Gallot D, Boda C, Ughetto S, Perthus I, Robert-Gnansia E, Francannet C, Laurichesse-Delmas H, Jani J, Coste K, Deprest J, Labbe A, Sapin V & Lemery D. (2007). Prenatal detection and outcome of congenital diaphragmatic hernia: a French registry-based study. Ultrasound Obstet Gynecol , 29, 276-83. PMID: 17177265 DOI.
- ↑ Colvin J, Bower C, Dickinson JE & Sokol J. (2005). Outcomes of congenital diaphragmatic hernia: a population-based study in Western Australia. Pediatrics , 116, e356-63. PMID: 16140678 DOI.
Reviews
Koo CW, Johnson TF, Gierada DS, White DB, Blackmon S, Matsumoto JM, Choe J, Allen MS, Levin DL & Kuzo RS. (2018). The breadth of the diaphragm: updates in embryogenesis and role of imaging. Br J Radiol , 91, 20170600. PMID: 29485899 DOI.
Sefton EM, Gallardo M & Kardon G. (2018). Developmental origin and morphogenesis of the diaphragm, an essential mammalian muscle. Dev. Biol. , , . PMID: 29679560 DOI.
Tovar JA. (2012). Congenital diaphragmatic hernia. Orphanet J Rare Dis , 7, 1. PMID: 22214468 DOI.
Pober BR. (2008). Genetic aspects of human congenital diaphragmatic hernia. Clin. Genet. , 74, 1-15. PMID: 18510546 DOI.
Mantilla CB & Sieck GC. (2008). Key aspects of phrenic motoneuron and diaphragm muscle development during the perinatal period. J. Appl. Physiol. , 104, 1818-27. PMID: 18403452 DOI.
Burgess RW, Jucius TJ & Ackerman SL. (2006). Motor axon guidance of the mammalian trochlear and phrenic nerves: dependence on the netrin receptor Unc5c and modifier loci. J. Neurosci. , 26, 5756-66. PMID: 16723533 DOI.
Articles
Paris ND, Coles GL & Ackerman KG. (2015). Wt1 and β-catenin cooperatively regulate diaphragm development in the mouse. Dev. Biol. , 407, 40-56. PMID: 26278035 DOI.
Merrell AJ, Ellis BJ, Fox ZD, Lawson JA, Weiss JA & Kardon G. (2015). Muscle connective tissue controls development of the diaphragm and is a source of congenital diaphragmatic hernias. Nat. Genet. , 47, 496-504. PMID: 25807280 DOI.
Hayashi S, Fukuzawa Y, Rodríguez-Vázquez JF, Cho BH, Verdugo-López S, Murakami G & Nakano T. (2011). Pleuroperitoneal canal closure and the fetal adrenal gland. Anat Rec (Hoboken) , 294, 633-44. PMID: 21370493 DOI.
Search PubMed Now
Search PubMed Now: diaphragm development | phrenic nerve development | fetal respiratory movements | Pubmed - Congenital Diaphragmatic Hernia | OMIM - Congenital Diaphragmatic Hernia
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 21) Embryology Respiratory System - Diaphragm. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Respiratory_System_-_Diaphragm
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G