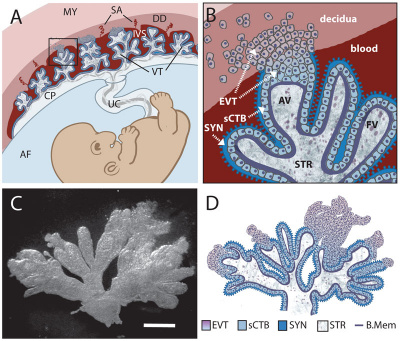
Placenta - Villi Development
| Embryology - 21 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
This page introduces an overview of aspects of the basic fetal subunit of the placenta, the placental villi development. In early placentation, each villi proceeds through a similar initial program of development. In later placentation, villi morphologically differentiate into a limited range of villi functional changes reflecting their specialization. The major initial contribution is from the trophoblast shell that surrounds the conceptus and later by the development of extraembryonic mesoderm and blood vessel differentiation.
There are three main types of trophoblast cells that differentiate:
- villous cytotrophoblasts
- extravillous cytotrophoblasts
- syncytiotrophoblasts that form by fusion of villous cytotrophoblasts
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Placental Villi | Hofbauer cells |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Cytotrophoblast Layer
There is a new interpretation of the changes that are occuring in the cytotrophoblast (CTB) layer during early to full-term human placenta development. Traditionally the interpretation was that the cytotrophoblast layer thinned and became discontinuous towards term. The thinning is thought due to the epithelium surface expanding at a faster rate than its volume. Two recent studies suggest that while the cytotrophoblast layer does indeed thin, it does not become discontinuous.
Syncytiotrophoblast Layer
The syncytiotrophoblast (STB) layer forms the epithelial covering of the entire villous tree. These cells are multinucleated, terminally-differentiated syncytium formed by the fusion of the underlying progenitor cytotrophoblast (CTB) cells. The process is described as "syncytialization" and is mediated by syncytin-1, an envelope protein of a human endogenous retrovirus W (HERV-W). The differentiation is regulated by chorionic gonadotropin (hCG) and the fusion of cytotrophoblast cells is ongoing during placental development.
Cellular parts derived from the syncytiotrophoblasts (apoptotic nuclei and microparticulate debris) can be shed into the maternal blood in which they are bathed. The apototic process appears to be part of the fusion mechanism between cytotrophoblast and the overlying multinucleate syncytiotrophoblast layer.
Studies have suggested that these cells are transcriptionally inactive. A recent study using a number of different detection techniques now suggests that at least some of the cells nuclei may still be transcriptionally inactive.
Mesenchymal Villi
Mesenchymal villi generate all other villous types:
- immature intermediate villi
- stem villi
- mature intermediate villi
- terminal villi
Mesenchymal villi continuously form out of the trophoblastic sprouts throughout pregnancy and have been considered the basis for growth and differentiation of the villous trees.
Villi Stages
Early chorionic (placental) villi development is defined by the contribution number and type of layers. In early embryonic placental development, villi proceed through each of these stages, while later fetal development consists mainly of branching of existing villi.
Primary Villi
Week 2 - (GA week 4) first stage of chorionic villi development, trophoblastic shell cells (syncitiotrophoblasts and cytotrophoblasts) form finger-like extensions into maternal decidua.

Secondary Villi
Week 3 - (GA week 5) second stage of chorionic villi development, extraembryonic mesoderm grows into villi, covers entire surface of chorionic sac.
Basal region will form chorionic plate.
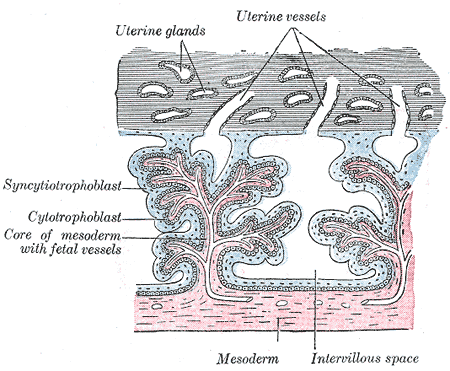
Tertiary Villi
Week 4 - (GA week 6) third stage of chorionic villi development, mesenchyme differentiates into blood vessels and cells, forms arteriocapillary network, fuse with placental vessels, developing in connecting stalk.
Villi Types
Later villi development has additional morphological and functional descriptions.
- stem villi - or anchoring villi, cytotrophoblast cells attached to maternal tissue.
- branched villi - grow from sides of stem villi, region of main exchange, surrounded by maternal blood in intervillous spaces.
- terminal villi - not active outgrowths caused by proliferation of the trophoblast. Passive protrusions induced by capillary coiling due to growth of the fetal capillaries within the mature intermediate villi (third trimester).
- chorionic plate - region of membrane at the base of the villi through which placental arteries and vein passes.
Hofbauer Cells

The placental macrophages known as Hofbauer cells[4] can be seen located within the villi and may have different origins depending on gestational age.[5]
- early pregnancy - may differentiate from villous mesenchymal stem cells of the stroma or monocyte progenitor cells from the hypoblast-derived yolk sac
- later pregnancy - may originate from fetal hematopoietic stem cells. For example, fetal monocytes that were recruited to the placenta.
Hofbauer Cell features:
- highly vacuolated cells
- located the core stroma of placental villi and cord
- macrophages with micropinocytotic activity and phagocytosis ability
- possible paracrine role for early stages of placental vasculogenesis
A figure showing the changes in placental villi between early (first trimester) and late (third trimester) placental development.
The Hofbauer cells can also be a site of placental transmitted pathogen infection and act as a reservoir for fetal infection.
Villi Trimester Development
Trimester 1 and 2
In the first two trimesters they are the forerunners of the immature intermediate villi, whereas in the last trimester the mesenchymal villi are transformed into mature intermediate villi. Immature intermediate villi formed during the first two trimesters are developmental steps towards the stem villi.
Trimester 3
Mature intermediate villi develop during the last trimester, produce numerous terminal villi. Terminal villi are not active outgrowths caused by proliferation of the trophoblast, but rather passive protrusions induced by capillary coiling due to excessive longitudinal growth of the fetal capillaries within the mature intermediate villi. The arrangement of the capillary bed in the terminal villi can vary from simple U-like loops to a richly branched network due to capillary elongation and sprouting.
Some text modified from[6], see also[7]
Placental Villi Blood Vessels
Some of the following data is from a histological study of human placental villi.[8]
- macrophage-like cells first cells to differentiate at day 21 (post-conception) from mesenchymal precursors.
- haemangioblastic cell cords (angiogenic cell cords, ACC) at day 21 (post-conception) also from mesenchymal cells, are the precursors of the capillary endothelium and haematopoietic stem cells
- pericytes form later and are a third cell population derived from the mesenchymal cells
- first main vascular patterns grow towards the longitudinal axis of the developing villi
- capillary basal lamina cannot be detected earlier than in the third trimester
- third trimester - fetal villous angiogenesis occurs by proliferation of the existing endothelium and pericytes rather than through haemangioblastic cells.
Human Villi Timeline
| Fertilization Age
(weeks) |
Gestational Age
(weeks) |
Vessel Lumen Diameter
(range in microns, µm) |
Features |
| 3 to 4 | 5 and 6 | 10 - 15 |
|
| 5 to 6 | 7 and 8 | 10 - 26 |
|
| 7 to 8 | 9 and 10 | 60 - 75 two central vessels
26 - 34 capillary network |
|
| 9 to 10 | 11 and 12 | 70 - 90 two central vessels
26 - 34 capillary network |
|
| Vill development data based upon immunochemistry confocal laser scanning microscope (CLSM) study[9] with clinical gestational age (GA) from last menstrual period (LMP) and has been corrected for post-conception (fertilization) age, approximately 14 days later.
CD31 - (PECAM-1, Platelet Endothelial Cell Adhesion Molecule) is a cluster of differentiation molecule found on endothelial and other blood cells. | |||
Extravillous Trophoblast Outgrowth
During the first trimester of human pregnancy, extravillous trophoblasts (EVT) from placental villi invade the decidua temporarily occluding the spiral arteries. This occusion prevents maternal blood flow and creates a low-oxygen environment, that may play a role in the regulation of extravillous trophoblast outgrowth.
A study has shown that the early placenta ( under 11 weeks of gestation) responds to oxygen concentration, whereas villi from older placentae (11 or 12 weeks) show no differential response.[10]
References
- ↑ Swiderska-Chadaj Z, Markiewicz T, Koktysz R & Cierniak S. (2018). Image processing methods for the structural detection and gradation of placental villi. Comput. Biol. Med. , 100, 259-269. PMID: 28797713 DOI.
- ↑ Leavey K, Benton SJ, Grynspan D, Bainbridge SA, Morgen EK & Cox BJ. (2017). Gene markers of normal villous maturation and their expression in placentas with maturational pathology. Placenta , 58, 52-59. PMID: 28962696 DOI.
- ↑ Lorenzi T, Turi A, Lorenzi M, Paolinelli F, Mancioli F, La Sala L, Morroni M, Ciarmela P, Mantovani A, Tranquilli AL, Castellucci M & Marzioni D. (2012). Placental expression of CD100, CD72 and CD45 is dysregulated in human miscarriage. PLoS ONE , 7, e35232. PMID: 22606231 DOI.
- ↑ Wynn RM. (1967). Derivation and ultrastructure of the so-called Hofbauer cell. Am. J. Obstet. Gynecol. , 97, 235-48. PMID: 6017029
- ↑ Reyes L, Wolfe B & Golos T. (2017). Hofbauer Cells: Placental Macrophages of Fetal Origin. Results Probl Cell Differ , 62, 45-60. PMID: 28455705 DOI.
- ↑ Castellucci M, Scheper M, Scheffen I, Celona A & Kaufmann P. (1990). The development of the human placental villous tree. Anat. Embryol. , 181, 117-28. PMID: 2327595
- ↑ Castellucci M, Kosanke G, Verdenelli F, Huppertz B & Kaufmann P. (2000). Villous sprouting: fundamental mechanisms of human placental development. Hum. Reprod. Update , 6, 485-94. PMID: 11045879
- ↑ Demir R, Kaufmann P, Castellucci M, Erbengi T & Kotowski A. (1989). Fetal vasculogenesis and angiogenesis in human placental villi. Acta Anat (Basel) , 136, 190-203. PMID: 2481376
- ↑ Lisman BA, van den Hoff MJ, Boer K, Bleker OP, van Groningen K & Exalto N. (2007). The architecture of first trimester chorionic villous vascularization: a confocal laser scanning microscopical study. Hum. Reprod. , 22, 2254-60. PMID: 17545656 DOI.
- ↑ James JL, Stone PR & Chamley LW. (2006). The effects of oxygen concentration and gestational age on extravillous trophoblast outgrowth in a human first trimester villous explant model. Hum. Reprod. , 21, 2699-705. PMID: 16807282 DOI.
Reviews
Articles
Search PubMed
Search April 2010
- Placenta Development - All (10032) Review (1896) Free Full Text (2435)
- Placental Development - All (9103) Review (1762) Free Full Text (2219)
- Placental Membranes - All (10083) Review (748) Free Full Text (1728)
Search Pubmed: Placenta Villi Development | Placenta Development
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 21) Embryology Placenta - Villi Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Placenta_-_Villi_Development
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G