Endocrine - Thyroid Development
| Embryology - 20 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
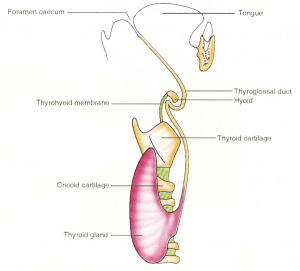
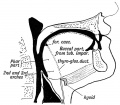
The boundary endoderm in the floor region forms a pocket (marked by the foramen cecum) that separates from the surface and forms the thyroid. Cells originate on the surface of the floor and descend into mesoderm above aortic sac and into the hypopharyngeal eminence as "cords". These cells continue to descend until they reach their final destination in the neck adjacent to the thyroid cartilage.
This pathway forms a temporary duct (thyroglossal duct). There are abnormalities of incomplete or excessive descent of these thyroid precursor cells. The thyroid is one of the earliest endocrine organs to differentiate and has an important hormonal role in embryonic development. The early bundle of cells then forms the thyroid by first dividing to form 2 lobes separated by a narrow connecting isthmus.
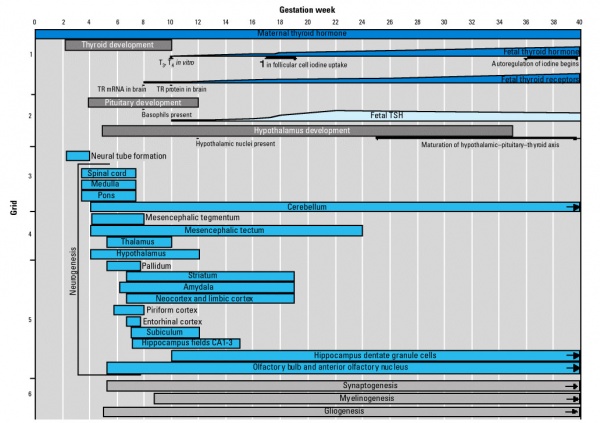
In the first trimester, the developing fetus is initially dependent upon maternal thyroid hormone crossing the placental barrier.[1] Around week 16 (GA week 18) the fetal thyroid becomes active enough to support the fetal requirements for neural development.
- Maternal thyroid hormone - required for early stages of brain development (Maternal Thyroid recent studies show both high and low thyroid hormone impact)
- Fetal functions from week 10 - required for neural development, stimulates metabolism (protein, carbohydrate, lipid), reduced/absence = cretinism (see abnormalities)
- Fetal fully functional 16 - 18 weeks - (GA 18-20 weeks)
- Hormones - (amino acid derivatives) Thyroxine (T4), Triiodothyronine (T3)
Maternal thyroid function also changes in early pregnancy, through pituitary changes, resulting in thyroid stimulating hormone (TSH) levels decreasing during the transition from pre-pregnancy to early pregnancy.[2] This change in TSH was less predictable in women with thyroid antibodies. Generally maternal TSH and free T4 are maintained within the same range as those in nonpregnant women.
Hyperthyroidism in adults is generally treated with antithyroid drugs (ATDs), some of these drugs have been identified as teratogens during pregnancy. (More? Antithyroid Drugs)
Iodine deficiency disorders (IDD) is the single most common cause of preventable mental retardation and brain damage in the world. Iodine (Greek, ioeides = violet) is required for the synthesis of thyroid hormone, a key regulator of neurological development. IDD causes goiters and decreases the production of hormones vital to growth and development. (More? iodine deficiency)
head | iodine deficiency | Antithyroid Drugs
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Thyroid Embryology | http://www.ncbi.nlm.nih.gov/pubmed/?term=thyroglossal+duct thyroglossal duct] |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Reading
- Human Embryology (2nd ed.) Larson
- The Developing Human: Clinically Oriented Embryology (6th ed.) Moore and Persaud Ch10: p230-233, Ch12: p280-282, Ch13: p319-347
- Before We Are Born (5th ed.) Moore and Persaud
- Essentials of Human Embryology Larson
- Human Embryology Fitzgerald and Fitzgerald Ch24: p166-167
| Thyroid | |
|---|---|

|
Endocrinology - An Integrated Approach Stephen Nussey and Saffron Whitehead
St. George's Hospital Medical School, London, UK Oxford: BIOS Scientific Publishers; 2001. ISBN-10: 1-85996-252-1 Copyright © 2001, BIOS Scientific Publishers Limited. Bookshelf |
Chapter 3. The thyroid gland
| |
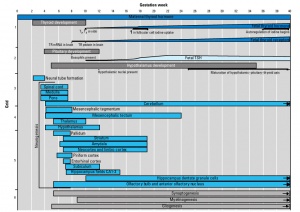
Development Overview
|
|
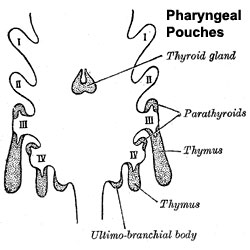
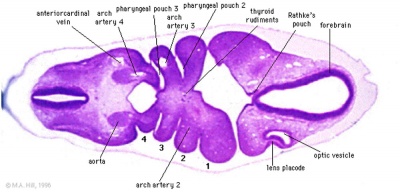
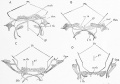
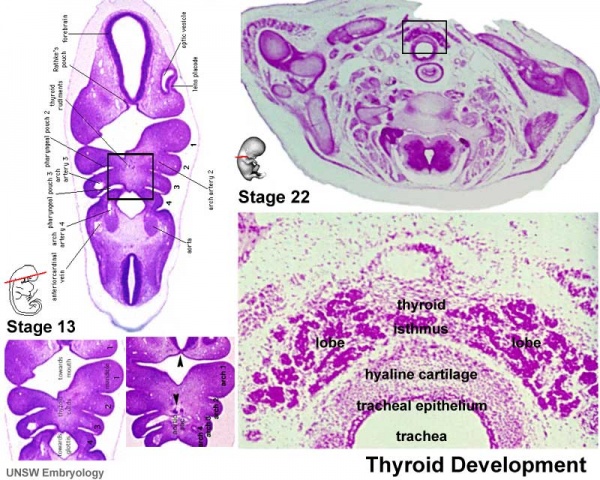
| Foramen Caecum | Stage 13 and Stage 22 thyroid development |
- thyroid median endodermal thickening in the floor of pharynx, outpouch – thyroid diverticulum
- tongue grows, cells descend in neck
- thyroglossal duct - proximal end at the foramen cecum of tongue thyroglossal duct
- thyroid diverticulum - hollow then solid, right and left lobes, central isthmus
Thyroid Timeline
- 24 days - thyroid median endodermal thickening in the floor of pharynx, outpouch – thyroid diverticulum
- Week 11 - colloid appearance in thyroid follicles, iodine and thyroid hormone (TH) synthesis growth factors (insulin-like, epidermal) stimulates follicular growth
Stage 13
- Links: Stage 13 Sections | Carnegie stage 13
Stage 22
- Links: Stage 22 Sections | Carnegie stage 22
Thyroid System and Neural Development
Fetal Thyroid Hormone
- Initial secreted biologically inactivated by modification, late fetal secretion develops brown fat
- Iodine deficiency- during this period, leads to neurological defects (cretinism)
- Birth - TSH levels increase, thyroxine (T3) and T4 levels increase to 24 h, then 5-7 days postnatal decline to normal levels
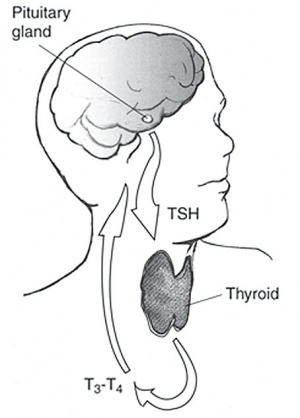
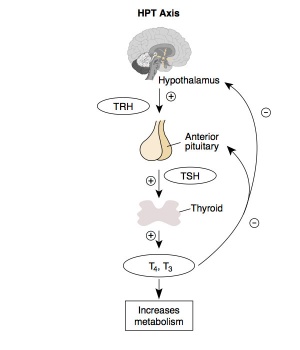
Thyroid Hormone
Thyroid hormone is synthesized in the thyroid gland by the iodination of tyrosines (monoiodotyrosine) and the coupling of iodotyrosines (diiodotyrosine) in the thyroglobulin.
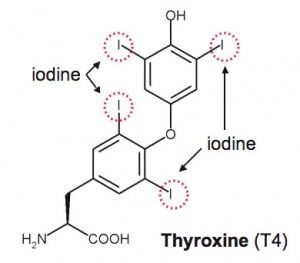
Thyroxine (T4) molecular structure showing iodine positions (red rings). |
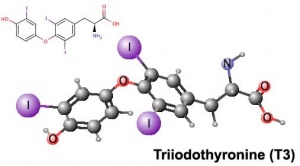
Triiodothyronine (T3) |
| Thyroxine (T4) - (Mr 777) majority of thyroid hormone derived from the thyroid gland. Thyroxine is released from thyroglobulin by proteolysis and secreted into the blood. | Triiodothyronine (T3) - synthesized and secreted by the thyroid gland in much smaller quantities than thyroxine (T4), though T3 is mainly used by tissues. Derived mainly from peripheral monodeiodination of T4 (at the 5' position of the outer ring of the iodothyronine nucleus). |
Deiodinases
| A group of enzymes that can modify the thyroid prohormone secreted by the thyroid gland either activating (deiodinase 2) or deactivating (deiodinase 3) thyroid hormone. Their role is thought to regulate the amount of active hormone available locally in a tissue.
Deiodinase 3
|
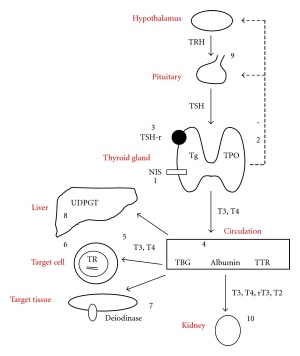
Thyroid hormone homeostasis |
Links: PubChem - T4 | PubChem - T3
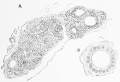
Calcitonin
Calcitonin is synthesized by parafollicular cells (C cells) interspersed in the connective tissue between the follicles and the blood capillary network.[9]
Maternal Thyroid
Maternal thyroid related changes during pregnancy[10]:
- stimulation of maternal thyroid gland by elevated levels of human chorionic gonadotropin (hCG)
- occurs mainly near end of first trimester associated with a transient lowering in serum TSH
- increase in serum thyroxine-binding globulin levels
- small decrease in free hormone concentrations (in iodine-sufficient conditions) significantly amplified in iodine restriction or overt iodine deficiency
- trend toward a slight increase in basal thyrotropin (TSH) values between first trimester and term
- modifications of the peripheral metabolism of maternal thyroid hormones
Maternal thyroid hormone crosses the placenta possibly by 3 transport protein family mechanisms[1]
- thyoorganic anion-transporting polypeptide
- L-type amino acid transporter
- monocarboxylate transporter
| Pregnancy Time | Thyrotropin (TSH) mIU/L | Free Thyroxine (FT4) pmol/L |
|---|---|---|
| 9-12 weeks | 0.18 - 2.99 | 11 - 19.2 |
| second trimester | 0.11 - 3.98 | 10.5 - 18.2 |
| third trimester | 0.48 - 4.71 | 9.0 - 16.1 |
| Total T4 (TT4) reference interval after 19 weeks GA was 77 - 186 nmol/L.
Data[11] - 95th percentile reference | ||
A study has identified an association of maternal thyroid function during early pregnancy with child brain morphology and IQ[12]
- Maternal levels of free T4 during pregnancy correlate with their offspring's IQ, cortex volume and grey matter volume
• Both the upper and lower limits of normal maternal levels of free T4 during pregnancy are associated with reduced child IQ, as well as decreased child cortex and grey matter volume • Maternal levels of TSH during pregnancy are not associated with child IQ or child brain morphology
In the placenta, the inner ring placental deiodinase inactivates most of the maternal T4 to reverse T3 (rT3). During fetal development to term, up to 30% of the fetal thyroid hormones are of maternal origin.
Abnormalities

There are structural anatomical variations and thyroid under and over development, there are a number of abnormalities associated with the persistence of the embryological path of thyroid cell descent, the thyroglossal duct.
In addition there are abnormalities due to low iodine environmental conditions of growth and development. This has subsequent effects upon neural and renal development.
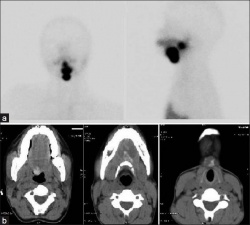
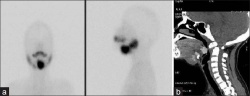
Lingual thyroid gland
The failure of thyroid descent.
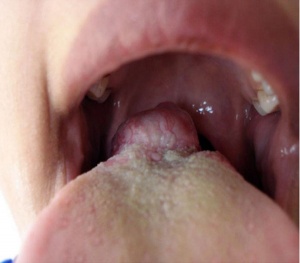
|
|
|
| Ectopic thyroid - lingual[14] | Sublingual, suprahyoid and subhyoid[15] | Sublingual and suprahyoid[15] |
Thyroglossal Cyst
The persistence of the thyroglossal duct.
Thyroglossal fistula
The partial degeneration of the thyroglossal duct.
Abnormal development of the thyroid
Incomplete or excessive descent.
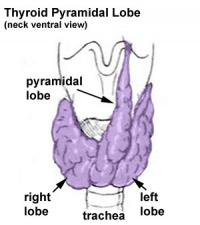
Pyramidal Lobe
|
This additional lobe is formed from the isthmus (50% of people) attached to hyoid bone distal end of thryoglossal duct. |
Agenesis of Isthmus
![Thyroid agenesis of isthmus[16]](/embryology/images/thumb/a/a1/Thyroid-agenesis_of_isthmus.jpg/200px-Thyroid-agenesis_of_isthmus.jpg)
|
An absence of the isthmus with the lateral lobes positioned independently on either side of the trachea. The normal isthmus measures about 1.25 cm transversely as well as vertically and is located anterior to the second and third tracheal cartilages.[16] |
Congenital Hypothyroidism
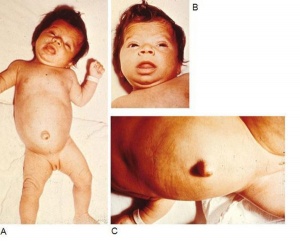
Occurs approximately 1 in 3000 births, associated with neurological abnormalities. This abnormality can occur through either dysgenesis or agenesis of the thyroid gland development or abnormal thyroid hormone production. The condition can be classified as a permanent and transient form.
American Academy of Pediatrics 2006 - Update of newborn screening and therapy for congenital hypothyroidism[18] "Unrecognized congenital hypothyroidism leads to mental retardation. Newborn screening and thyroid therapy started within 2 weeks of age can normalize cognitive development. The primary thyroid-stimulating hormone screening has become standard in many parts of the world. However, newborn thyroid screening is not yet universal in some countries. Initial dosage of 10 to 15 microg/kg levothyroxine is recommended. The goals of thyroid hormone therapy should be to maintain frequent evaluations of total thyroxine or free thyroxine in the upper half of the reference range during the first 3 years of life and to normalize the serum thyroid-stimulating hormone concentration to ensure optimal thyroid hormone dosage and compliance. Improvements in screening and therapy have led to improved developmental outcomes in adults with congenital hypothyroidism who are now in their 20s and 30s. Thyroid hormone regimens used today are more aggressive in targeting early correction of thyroid-stimulating hormone than were those used 20 or even 10 years ago. Thus, newborn infants with congenital hypothyroidism today may have an even better intellectual and neurologic prognosis."
Congenital Hypothyroidism Classification
| Classification | Etiology |
| Primary | Thyroid dysgenesis (developmental anomaly)
Thyroid dyshormonogenesis (impaired hormone production) Resistance to TSH binding or signaling |
| Central | Isolated TSH deficiency
Thyrotropin-releasing hormone deficiency Thyrotropin-releasing hormone resistance Deficiency in pituitary development transcription factors |
| Peripheral | Resistance to thyroid hormone
Abnormalities of thyroid hormone transport |
| Syndromic | Pendred syndrome
Bamforth-Lazarus syndrome Ectodermal dysplasia Hypothyroidism Kocher - Deber - Semilange syndrome Benign chorea - hypothyroidism Choreoathetosis Obesity - colitis |
| Transient | Maternal intake of antithyroid drugs
Transplacental passage of maternal TSH receptor blocking antibodies Maternal and neonatal iodine deficiency or excess Heterozygous mutations of THOX2 or DUOXA2 Congenital hepatic hemangioma/hemangioendothelioma |
| Based on Table 3 from review on congenital hypothyroidism.[17] |
genital - hypothyroidism can also impact upon male genital development, by inhibiting testicular growth, through a delay of Sertoli cell differentiation and proliferation.
Iodine Deficiency
Iodine deficiency disorder (IDD) is the single most common cause of preventable mental retardation and brain damage in the world (More? Abnormal Development - Iodine Deficiency). It is required for synthesis of thyroid hormone, which in turn regulates aspects of neural development.
Worldwide:
1.6 billion people are at risk IDD affects 50 million children 100,000 cretins are born every year It causes goiters and decreases the production of hormones vital to growth and development. Children with IDD can grow up stunted, apathetic, mentally retarded and incapable of normal movement, speech or hearing. IDD in pregnant women cause miscarriage, stillbirth and mentally retarded children.
A teaspoon of iodine is all a person requires in a lifetime, but because iodine cannot be stored for long periods by the body, tiny amounts are needed regularly. In areas of endemic iodine deficiency, where soil and therefore crops and grazing animals do not provide sufficient dietary iodine to the populace, food fortification and supplementation have proven highly successful and sustainable interventions.
Iodized salt programs and iodized oil supplements are the most common tools in the fight against IDD.
(Data: ICCIDD)
Prolonged intake of large amounts (excess) of iodide can increase the incidence of goiter and/or hypothyroidism in humans. African Congo appears to be the only country that appears to have a dietary excess.
- Links: Iodine Deficiency
Maternal Abnormalities
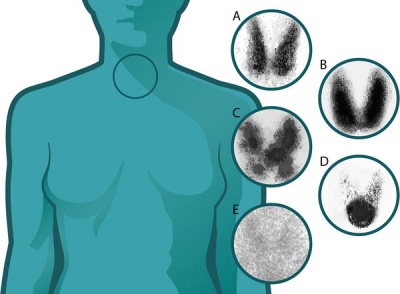
Thyroid uptake scan images of normal and abnormal thyroids (A) Normal
(B) Graves disease: diffuse increased uptake in both thyroid lobes.
(C) Toxic multinodular goiter (TMNG): "hot" and "cold" areas of uneven uptake.
(D) Toxic adenoma: increased uptake in a single nodule with suppression of the surrounding thyroid.
(E) Thyroiditis: decreased or absent uptake.
Thyroid uptake scans image[19] Maternal Graves Disease - "The dose of anti-thyroid drug usually needs to be decreased during pregnancy, and often Graves disease remits completely and the medication can be withdrawn. This is probably due to the overall immunosuppressive effect of pregnancy."
Graves' disease in mothers can cause thyrotoxic fetus - may have increased fetal motility and develop a range of abnormalities including: goitre, tachycardia, heart failure associated hydrops, growth retardation, craniosynostosis and accelerated bone maturation.
Maternal Hashimoto's Thyroiditis (common autoimmune thyroid disease) usually no consequences on fetal thyroid, even if antibodies (anti-TPO and anti-Tg) found in the newborn due to transplacental passage.
maternal hypothyroxinemia
Links: NIH Genes & Disease - Chapter 41 - Endocrine | EPA (USA) - Radiation Technetium
Environmental Thyroid Disruptors

There are several environmental compounds (chemicals) that are suspected of being thyroid disruptors including:
- halogenated phenolic compounds (3,3',5,5'-tetrabromobisphenol A, 3,3',5,5'-tetrachlorobisphenol A, 4-hydroxy-2',3,4',5,6'-pentachlorobiphenyl)
- phenol compounds (pentachlorophenol, 2,4,6-triiodophenol)
They have been demonstrated to induce partial agonistic and/or complex competitive/uncompetitive antagonistic responces in cell culture.
Bisphenol A - monomer used to manufacture polycarbonate plastic, possibly disrupts thyroid hormone function and affects neocortical development (accelerating neuronal differentiation/migration). (More? EHP - Bisphenol A Need for a New Risk Assessment)
Genes
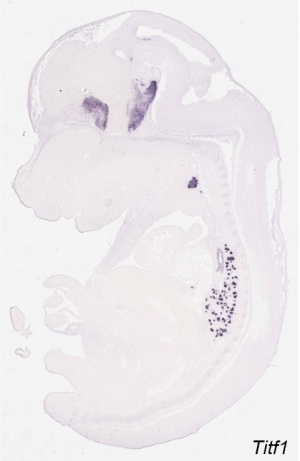
- Thyroid Transcriptor Factors (TTF) - TTF-1, TTF-2, PAX-8
- Fetal hypothyroidism from low levels of these transcription factors- Pit-1, Prop-1, LHX-3
- Thyrotropin-releasing hormone (TRH)
- Thyroid-stimulating hormone (TSH)
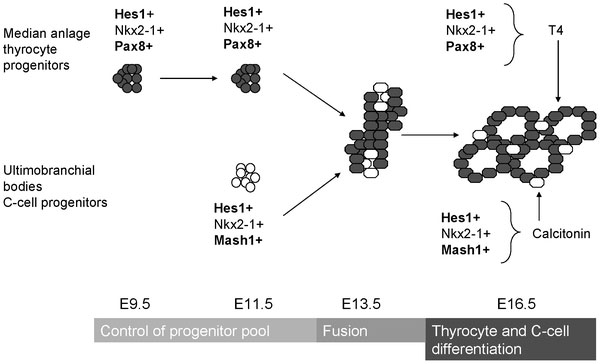
Mouse thyroid Hes1 model[21]
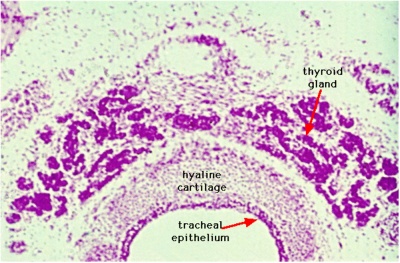
Thyroid Images
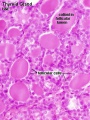
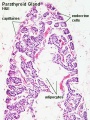
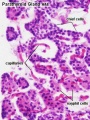
Adult Histology
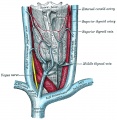
Anatomy
References
- ↑ 1.0 1.1 James SR, Franklyn JA & Kilby MD. (2007). Placental transport of thyroid hormone. Best Pract. Res. Clin. Endocrinol. Metab. , 21, 253-64. PMID: 17574007 DOI.
- ↑ Balthazar U & Steiner AZ. (2012). Periconceptional changes in thyroid function: a longitudinal study. Reprod. Biol. Endocrinol. , 10, 20. PMID: 22436200 DOI.
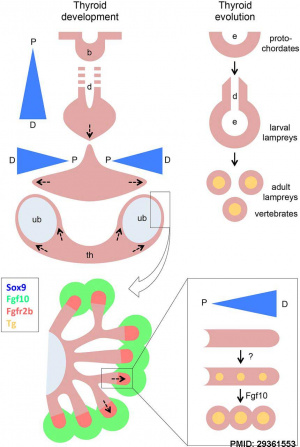
- ↑ 3.0 3.1 Liang S, Johansson E, Barila G, Altschuler DL, Fagman H & Nilsson M. (2018). A branching morphogenesis program governs embryonic growth of the thyroid gland. Development , 145, . PMID: 29361553 DOI.
- ↑ Nilsson M & Fagman H. (2017). Development of the thyroid gland. Development , 144, 2123-2140. PMID: 28634271 DOI.
- ↑ Villacorte M, Delmarcelle AS, Lernoux M, Bouquet M, Lemoine P, Bolsée J, Umans L, de Sousa Lopes SC, Van Der Smissen P, Sasaki T, Bommer G, Henriet P, Refetoff S, Lemaigre FP, Zwijsen A, Courtoy PJ & Pierreux CE. (2016). Thyroid follicle development requires Smad1/5- and endothelial cell-dependent basement membrane assembly. Development , 143, 1958-70. PMID: 27068110 DOI.
- ↑ Takanashi Y, Honkura Y, Rodriguez-Vazquez JF, Murakami G, Kawase T & Katori Y. (2015). Pyramidal lobe of the thyroid gland and the thyroglossal duct remnant: a study using human fetal sections. Ann. Anat. , 197, 29-37. PMID: 25458181 DOI.
- ↑ Lania G, Zhang Z, Huynh T, Caprio C, Moon AM, Vitelli F & Baldini A. (2009). Early thyroid development requires a Tbx1-Fgf8 pathway. Dev. Biol. , 328, 109-17. PMID: 19389367 DOI.
- ↑ Zimmermann MB. (2009). Iodine deficiency in pregnancy and the effects of maternal iodine supplementation on the offspring: a review. Am. J. Clin. Nutr. , 89, 668S-72S. PMID: 19088150 DOI.
- ↑ Hazard JB. (1977). The C cells (parafollicular cells) of the thyroid gland and medullary thyroid carcinoma. A review. Am. J. Pathol. , 88, 213-50. PMID: 18012
- ↑ Glinoer D. (1999). What happens to the normal thyroid during pregnancy?. Thyroid , 9, 631-5. PMID: 10447005 DOI.
- ↑ Donovan LE, Metcalfe A, Chin AC, Yamamoto JM, Virtanen H, Johnson JA & Krause R. (2018). A Practical Approach for the Verification and Determination of Site and Trimester Specific Reference Intervals for Thyroid Function Tests in Pregnancy. Thyroid , , . PMID: 30595114 DOI.
- ↑ Korevaar TI, Muetzel R, Medici M, Chaker L, Jaddoe VW, de Rijke YB, Steegers EA, Visser TJ, White T, Tiemeier H & Peeters RP. (2016). Association of maternal thyroid function during early pregnancy with offspring IQ and brain morphology in childhood: a population-based prospective cohort study. Lancet Diabetes Endocrinol , 4, 35-43. PMID: 26497402 DOI.
- ↑ 13.0 13.1 Hartoft-Nielsen ML, Boas M, Bliddal S, Rasmussen AK, Main K & Feldt-Rasmussen U. (2011). Do Thyroid Disrupting Chemicals Influence Foetal Development during Pregnancy?. J Thyroid Res , 2011, 342189. PMID: 21918727 DOI.
- ↑ Kumar Choudhury B, Kaimal Saikia U, Sarma D, Saikia M, Dutta Choudhury S, Barua S & Dewri S. (2011). Dual ectopic thyroid with normally located thyroid: a case report. J Thyroid Res , 2011, 159703. PMID: 21765986 DOI.
- ↑ 15.0 15.1 Jain A & Pathak S. (2010). Rare developmental abnormalities of thyroid gland, especially multiple ectopia: A review and our experience. Indian J Nucl Med , 25, 143-6. PMID: 21713222 DOI.
- ↑ 16.0 16.1 Dixit D, Shilpa MB, Harsh MP & Ravishankar MV. (2009). Agenesis of isthmus of thyroid gland in adult human cadavers: a case series. Cases J , 2, 6640. PMID: 20181171 DOI.
- ↑ 17.0 17.1 Rastogi MV & LaFranchi SH. (2010). Congenital hypothyroidism. Orphanet J Rare Dis , 5, 17. PMID: 20537182 DOI.
- ↑ Rose SR, Brown RS, Foley T, Kaplowitz PB, Kaye CI, Sundararajan S & Varma SK. (2006). Update of newborn screening and therapy for congenital hypothyroidism. Pediatrics , 117, 2290-303. PMID: 16740880 DOI.
- ↑ Perros P. (2005). Thyrotoxicosis and pregnancy. PLoS Med. , 2, e370. PMID: 16363909 DOI.
- ↑ Diez-Roux G, Banfi S, Sultan M, Geffers L, Anand S, Rozado D, Magen A, Canidio E, Pagani M, Peluso I, Lin-Marq N, Koch M, Bilio M, Cantiello I, Verde R, De Masi C, Bianchi SA, Cicchini J, Perroud E, Mehmeti S, Dagand E, Schrinner S, Nürnberger A, Schmidt K, Metz K, Zwingmann C, Brieske N, Springer C, Hernandez AM, Herzog S, Grabbe F, Sieverding C, Fischer B, Schrader K, Brockmeyer M, Dettmer S, Helbig C, Alunni V, Battaini MA, Mura C, Henrichsen CN, Garcia-Lopez R, Echevarria D, Puelles E, Garcia-Calero E, Kruse S, Uhr M, Kauck C, Feng G, Milyaev N, Ong CK, Kumar L, Lam M, Semple CA, Gyenesei A, Mundlos S, Radelof U, Lehrach H, Sarmientos P, Reymond A, Davidson DR, Dollé P, Antonarakis SE, Yaspo ML, Martinez S, Baldock RA, Eichele G & Ballabio A. (2011). A high-resolution anatomical atlas of the transcriptome in the mouse embryo. PLoS Biol. , 9, e1000582. PMID: 21267068 DOI.
- ↑ Carre A, Rachdi L, Tron E, Richard B, Castanet M, Schlumberger M, Bidart JM, Szinnai G & Polak M. (2011). Hes1 is required for appropriate morphogenesis and differentiation during mouse thyroid gland development. PLoS ONE , 6, e16752. PMID: 21364918 DOI.
Journals
- Thyroid Thyroid The official journal of the American Thyroid Association.
Online Textbooks
Endocrinology: An Integrated Approach Nussey, S.S. and Whitehead, S.A. Oxford, UK: BIOS Scientific Publishers, Ltd; 2001. table of Contents
- The Thyroid Gland
- Surgical anatomy and embryology of the thyroid gland
- Hypothyroidism in infancy and childhood
NIH Genes & Disease Chapter 41 - Endocrine
Developmental Biology (6th ed) Gilbert, Scott F. Sunderland (MA): Sinauer Associates, Inc.; c2000.
- Is the endostyle the precursor of the thyroid gland?
- Environmental thyroid hormone disruptors
- table 13.1. Some derivatives of the neural crest
Molecular Biology of the Cell (4th Edn) Alberts, Bruce; Johnson, Alexander; Lewis, Julian; Raff, Martin; Roberts, Keith; Walter, Peter. New York: Garland Publishing; 2002.
Health Services/Technology Assessment Text (HSTAT) Bethesda (MD): National Library of Medicine (US), 2003 Oct. Thyroid Gland search Results
Reviews
Silva JF, Ocarino NM & Serakides R. (2018). Thyroid hormones and female reproduction. Biol. Reprod. , , . PMID: 29767691 DOI.
Landers K & Richard K. (2017). Traversing barriers - How thyroid hormones pass placental, blood-brain and blood-cerebrospinal fluid barriers. Mol. Cell. Endocrinol. , 458, 22-28. PMID: 28153799 DOI.
Chan SY, Vasilopoulou E & Kilby MD. (2009). The role of the placenta in thyroid hormone delivery to the fetus. Nat Clin Pract Endocrinol Metab , 5, 45-54. PMID: 19079273 DOI.
Postiglione MP, Parlato R, Rodriguez-Mallon A, Rosica A, Mithbaokar P, Maresca M, Marians RC, Davies TF, Zannini MS, De Felice M & Di Lauro R. (2002). Role of the thyroid-stimulating hormone receptor signaling in development and differentiation of the thyroid gland. Proc. Natl. Acad. Sci. U.S.A. , 99, 15462-7. PMID: 12432093 DOI.
Park SM & Chatterjee VK. (2005). Genetics of congenital hypothyroidism. J. Med. Genet. , 42, 379-89. PMID: 15863666 DOI.
De Felice M, Postiglione MP & Di Lauro R. (2004). Minireview: thyrotropin receptor signaling in development and differentiation of the thyroid gland: insights from mouse models and human diseases. Endocrinology , 145, 4062-7. PMID: 15231702 DOI.
Grüters A, Biebermann H & Krude H. (2003). Neonatal thyroid disorders. Horm. Res. , 59 Suppl 1, 24-9. PMID: 12566717 DOI.
Articles
Abu-Khudir R, Paquette J, Lefort A, Libert F, Chanoine JP, Vassart G & Deladoëy J. (2010). Transcriptome, methylome and genomic variations analysis of ectopic thyroid glands. PLoS ONE , 5, e13420. PMID: 20976176 DOI.
Villa-Cuesta E & Modolell J. (2005). Mutual repression between msh and Iro-C is an essential component of the boundary between body wall and wing in Drosophila. Development , 132, 4087-96. PMID: 16093324 DOI.
Iskaros J, Pickard M, Evans I, Sinha A, Hardiman P & Ekins R. (2000). Thyroid hormone receptor gene expression in first trimester human fetal brain. J. Clin. Endocrinol. Metab. , 85, 2620-3. PMID: 10902817 DOI.
Search PubMed
Search Pubmed: thyroid development | Congenital Hypothyroidism
Additional Images
Historic
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Terms
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- International Council for the Control of Iodine Deficiency Disorders
- World Health Organization- Micronutrient deficiencies Eliminating iodine deficiency disorders
- British Thyroid Association The official site of the British Thyroid Assocation, a non-profit making Learned Society of professional clinical specialist doctors and scientists in the United Kingdom who manage patients with thyroid disease and/or are researching into the thyroid and its diseases in humans.
- WHO Nutrition Guidelines
- American Thyroid Association American Thyroid Association - Patient Information
- Thyroid Foundation of America
- Endocrine Society (USA)
- European Thyroid Association
- Latin-American Thyroid Association
- [www.aota.or.kr Asia and Oceana Thyroid Association]
- Society for Endocrinology (UK)
- British Association of Endocrine Surgeons
- [www.aace.com American Association of Clinical Endocrinologists]
- Thyroid Disease Manager USA -- this site includes textbook on the Thyroid. .
- [www.synthroid.com Knoll Pharmaceuticals]- makers of SYNTHROID
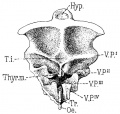
- Gray's Anatomy - The Ductless Glands | The thyroid gland and its relations | Floor of pharynx of human embryo about twenty-six days old. (From model by Peters) | 4b. The Parathyroid Glands
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 20) Embryology Endocrine - Thyroid Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Endocrine_-_Thyroid_Development
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G