Introduction
Meiosis is the special type of recombinative and reductive cell division occurring only in the generation of the gametes or germ cells (oocyte and spermatozoa).
For recombination, meiosis requires that homologous chromosomes are properly paired and aligned by the induction of DNA double-strand breaks by the enzyme SPO11 during the prophase of the first meiotic division.
Meiotic cell division also reduces (halves) the chromosomal content. The overall process of germ cell development is called "gametogenesis" and includes not only meiosis but also the cellular morphological changes, that occur differently in male and female gametes.
|
|
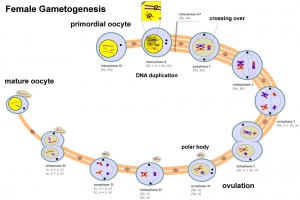
| Female gametogenesis
|
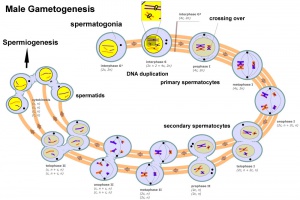
Male gametogenesis
|
Some Recent Findings
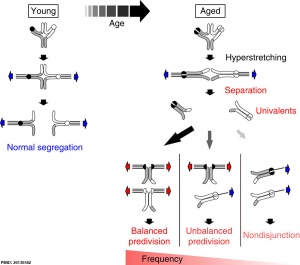
Bivalent separation into univalents precedes age-related meiosis I errors in oocytes
[1]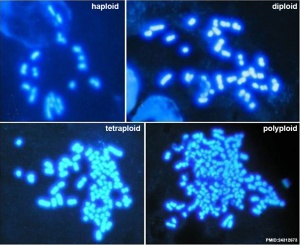
Karyotype of parthenogenetic blastocysts
[2]
- Distinct prophase arrest mechanisms in human male meiosis[3] "To prevent chromosomal aberrations being transmitted to the offspring, strict meiotic checkpoints are in place to remove aberrant spermatocytes. However, in about 1% of males these checkpoints cause complete meiotic arrest leading to azoospermia and subsequent infertility. Here, we unravel two clearly distinct meiotic arrest mechanisms that occur during prophase of human male meiosis. Type I arrested spermatocytes display severe asynapsis of the homologous chromosomes, disturbed XY-body formation and increased expression of the Y chromosome-encoded gene c and seem to activate a DNA damage pathway leading to induction of p63, possibly causing spermatocyte apoptosis. Type II arrested spermatocytes display normal chromosome synapsis, normal XY-body morphology and meiotic crossover formation but have a lowered expression of several cell cycle regulating genes and fail to silence the X chromosome-encoded gene ZFX."
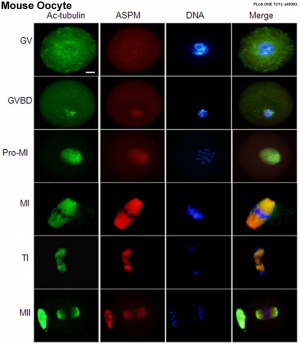
- Review - Actin cytoskeleton dynamics in mammalian oocyte meiosis[4] "During mitosis, cells undergo symmetrical cell division, while oocyte meiotic maturation undergoes two consecutive, asymmetric divisions that generate a totipotent haploid oocyte and two small polar bodies not involved in DNA replication. This specialized division allows most maternal components be maintained in the oocytes for early embryo development. Nuclear positioning, germinal vesicle breakdown, spindle migration, spindle rotation, chromosome segregation, and polar body extrusion are the most critical cellular processes during oocyte meiosis I and II, and a growing number of studies primarily using the mouse oocyte model revealed that actin filaments were critical for these processes, especially for spindle migration. Several important molecules have been reported to be involved in these processes. One family of molecules are the small GTPases, such as Rho GTPases, Ran GTPases, and Rab GTPases and another are the actin nucleators, such as the formin family and the Arp2/3 complex. The present review summarizes recent progress made regarding the roles of actin filaments in the asymmetric oocyte division." oocyte | polar body
- Inefficient Crossover Maturation Underlies Elevated Aneuploidy in Human Female Meiosis[5] "Meiosis is the cellular program that underlies gamete formation. For this program, crossovers between homologous chromosomes play an essential mechanical role to ensure regular segregation. We present a detailed study of crossover formation in human male and female meiosis, enabled by modeling analysis. Results suggest that recombination in the two sexes proceeds analogously and efficiently through most stages. However, specifically in female (but not male), ∼25% of the intermediates that should mature into crossover products actually fail to do so. Further, this "female-specific crossover maturation inefficiency" is inferred to make major contributions to the high level of chromosome mis-segregation and resultant aneuploidy that uniquely afflicts human female oocytes (e.g., giving Down syndrome). Additionally, crossover levels on different chromosomes in the same nucleus tend to co-vary, an effect attributable to global per-nucleus modulation of chromatin loop size. Maturation inefficiency could potentially reflect an evolutionary advantage of increased aneuploidy for human females."
- Altered Crossover Distribution and Frequency in Spermatocytes of Infertile Men with Azoospermia[6] "Our study aims to characterize the crossover distribution in infertile men with non-obstructive (NOA) and obstructive azoospermia (OA) along chromosomes 13, 18 and 21. Eight of the 16 NOA men and five of the 21 OA men in our study displayed reduced crossover frequency compared to control fertile men. Seven NOA men and nine OA men showed altered crossover distributions on at least one of the chromosome arms studied compared to controls. We found that although both NOA and OA men displayed altered crossover distributions, NOA men may be at a higher risk of suffering both altered crossover frequencies and distributions compared to OA men."
- Review - The evolution of meiotic sex and its alternatives[7] "Meiosis is an ancestral, highly conserved process in eukaryotic life cycles, and for all eukaryotes the shared component of sexual reproduction. The benefits and functions of meiosis, however, are still under discussion, especially considering the costs of meiotic sex."
|
| More recent papers
|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
- This search now requires a manual link as the original PubMed extension has been disabled.
- The displayed list of references do not reflect any editorial selection of material based on content or relevance.
- References also appear on this list based upon the date of the actual page viewing.
References listed on the rest of the content page and the associated discussion page (listed under the publication year sub-headings) do include some editorial selection based upon both relevance and availability.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References
Search term: Meiosis | Meiosis 1 | Meiosis 2 | Meiosis crossover | Meiotic spindle | Cohesion | chiasmata
|
|
| Older papers
|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
- From Meiosis to Mitosis: The Astonishing Flexibility of Cell Division Mechanisms in Early Mammalian Development[8] "The execution of female meiosis and the establishment of the zygote is arguably the most critical stage of mammalian development. The egg can be arrested in the prophase of meiosis I for decades, and when it is activated, the spindle is assembled de novo. This spindle must function with the highest of fidelity and yet its assembly is unusually achieved in the absence of conventional centrosomes and with minimal influence of chromatin. Moreover, its dramatic asymmetric positioning is achieved through remarkable properties of the actin cytoskeleton to ensure elimination of the polar bodies. The second meiotic arrest marks a uniquely prolonged metaphase eventually interrupted by egg activation at fertilization to complete meiosis and mark a period of preparation of the male and female pronuclear genomes not only for their entry into the mitotic cleavage divisions but also for the imminent prospect of their zygotic expression."
- Silencing of X-Linked MicroRNAs by Meiotic Sex Chromosome Inactivation [9] "During the pachytene stage of meiosis in male mammals, the X and Y chromosomes are transcriptionally silenced by Meiotic Sex Chromosome Inactivation (MSCI). MSCI is conserved in therian mammals and is essential for normal male fertility. ...Here we use RNA FISH to examine X-miRNA expression in the male germ line. We find that, like protein-coding X-genes, X-miRNAs are expressed prior to prophase I and are thereafter silenced during pachynema. X-miRNA silencing does not occur in mouse models with defective MSCI. Furthermore, X-miRNAs are expressed at pachynema when present as autosomally integrated transgenes. Thus, we conclude that silencing of X-miRNAs during pachynema in wild type males is MSCI-dependent. Importantly, misexpression of X-miRNAs during pachynema causes spermatogenic defects. We propose that MSCI represents a chromosomal mechanism by which X-miRNAs, and other potential X-encoded repressors, can be silenced, thereby regulating genes with critical late spermatogenic functions." spermatozoa
- Bivalent separation into univalents precedes age-related meiosis I errors in oocytes[1] "The frequency of chromosome segregation errors during meiosis I (MI) in oocytes increases with age. The two-hit model suggests that errors are caused by the combination of a first hit that creates susceptible crossover configurations and a second hit comprising an age-related reduction in chromosome cohesion. This model predicts an age-related increase in univalents, but direct evidence of this phenomenon as a major cause of segregation errors has been lacking. Here, we provide the first live analysis of single chromosomes undergoing segregation errors during MI in the oocytes of naturally aged mice. Chromosome tracking reveals that 80% of the errors are preceded by bivalent separation into univalents. The set of the univalents is biased towards balanced and unbalanced predivision of sister chromatids during MI. Moreover, we find univalents predisposed to predivision in human oocytes. This study defines premature bivalent separation into univalents as the primary defect responsible for age-related aneuploidy."
- Premature dyad separation in meiosis II is the major segregation error with maternal age in mouse oocytes[10] "Changes consistent with chromosome cohesion deterioration were found with age, including increased interkinetochore distance and loss of the centromeric protector of cohesion SGO2 in metaphase II arrested (metII) eggs, as well as a rise in the number of weakly attached bivalents in meiosis I (MI) and lagging chromosomes at anaphase I. However, there were no MI errors in congression or biorientation. Instead, premature separation of dyads in meiosis II was the major segregation defect in aged eggs and these were associated with very low levels of SGO2. These data show that although considerable cohesion loss occurs during MI, its consequences are observed during meiosis II, when centromeric cohesion is needed to maintain dyad integrity."
- Chromosomes in the Porcine First Polar Body Possess Competence of Second Meiotic Division within Enucleated MII Stage Oocytes[2] "These results demonstrate that chromosomes in PB1 can participate in normal pre-implantation embryonic development when injected into enucleated MII stage oocytes, and that tetraploid PA blastocysts are produced (although at a low proportion) when PB1 chromosomes are injected into intact MII stage oocytes."
- Bora regulates meiotic spindle assembly and cell cycle during mouse oocyte meiosis[11] "Bora is the binding partner of Aurora A, which is required for its activation and phosphorylation of Polo like kinase 1 (Plk1), and is involved in the spindle assembly and progress of the cell cycle during mitosis. In this study, we examined the expression, localization, and function of Bora during mouse oocyte meiosis. The expression level of Bora was increased during oocyte meiotic maturation, with an elevated level at metaphase. Immunofluorescence analysis showed that Bora was concentrated as a dot shortly after germinal vesicle breakdown (GVBD), associating first with the surrounding chromosomes and then with the spindle throughout the oocyte meiotic maturation. Further experiments confirmed that Bora co-localized with α-tubulin at prometaphase/metaphase, but dissociated from α-tubulin at anaphase/telophase."
|
|
Movies
|
|
A mouse oocyte undergoing meiosis spindle migration followed by first polar body extrusion and MII spindle positioning.[12]
- blue - Hoechst staining of chromosomes.
- green - UtrCH-GFP was used to label cortical changes during spindle migration.
The video shows that cytoplasmic streaming continues to the MII arrest stage to maintain the oocyte set of chromosomes/MII spindle in place close to the cortex. Frames are 11 min apart, and video length is 840 min. Bar, 20 µm.
|
|
|
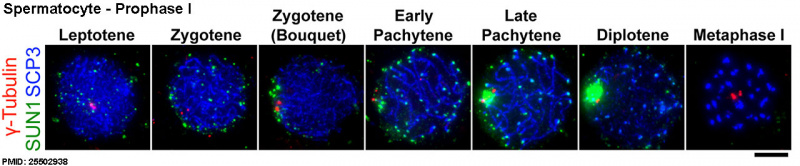
A mouse spermatocyte undergoing meiosis prophase I. The chromosomal telomeres and synaptonemal complexes have been labelled to visualise chromosomal movement within this single nucleus.[13]
|
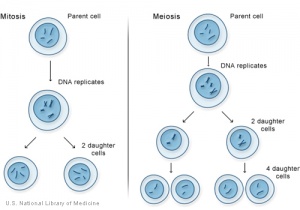
Comparison of Meiosis/Mitosis
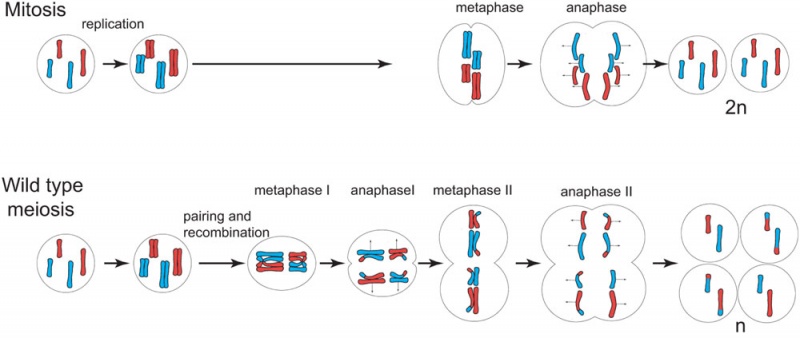
- After DNA replication 2 nuclear (and cell) divisions required to produce haploid gametes
- Each diploid cell in meiosis produces 4 haploid cells (sperm) 1 haploid cell (egg)
- Each diploid cell mitosis produces 2 diploid cells
Meiosis Germ cell division (haploid)
- Reductive division
- Generates haploid gametes (egg, sperm)
- Each genetically distinct from parent
- Genetic recombination (prophase 1)
- Exchanges portions of chromosomes maternal/paternal homologous pairs
- Independent assortment of paternal chromosomes (meiosis 1)
Homologous chromosomes pairing unique to meiosis
- Each chromosome duplicated and exists as attached sister chromatids before pairing occurs
- Genetic Recombination shown by chromosomes part red and part black
- chromosome pairing in meiosis involves crossing-over between homologous chromosomes
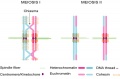
Meiosis I and II
- Meiosis I separates the pairs of homologous chromosomes, reduces the cell from diploid to haploid.
- Meiosis II separates each chromosome into two chromatids (chromosome behavior in meiosis II is like that of mitosis).
Links: Figure 14.32. Comparison of meiosis and mitosis
Prophase I
prophase I
- The homologous chromosomes pair and exchange DNA to form recombinant chromosomes.
- Note - in oocyte development, from birth until puberty oocytes are in "prophase I arrest" at diplotene stage. This is important for sustaining the ovarian oocyte pool and lutenizing hormone (LH) induces resumption of meiosis I.
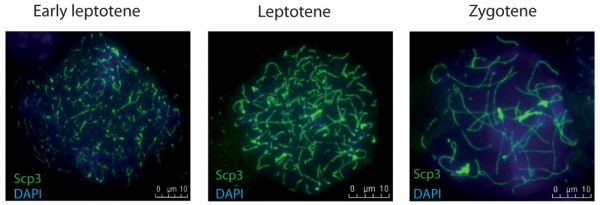
|
| Mouse early meiotic prophase I stages[14]
|
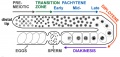
Prophase I is further divided into five stages (phases):
Leptotene
- leptotene phase, leptonema; Greek, leptotene = "thin threads"
- the duplicated paired chromosome homologs condense.
Zygotene
- zygotene phase, zygonema, Greek, zygotene = "paired threads"
- homologous chromosomes become closely associated (synapsis) to form pairs of chromosomes consisting of four chromatids (tetrads).
- the synaptonemal complex begins to form between the two sets of sister chromatids in each bivalent (the duplicated chromosome paired with its homologous duplicated chromosome).
Pachytene
- pachytene phase, pachynema; Greek, pachytene = "thick threads"
- crossing over between pairs of homologous chromosomes (meiotic recombination or synapsis) to form chiasmata (form between two nonsister chromatids at points where they have crossed over)
- synaptonemal complex is complete and can be stable for some time.
- Autosomal non-sister chromatids of homologous chromosomes can now extensively exchange segments in regions of homology.
- Only small regions of non-paired sex chromosomes interact
- Mutations that compromise meiotic recombination in male spermatocytes result in arrest and apoptosis at this stage.
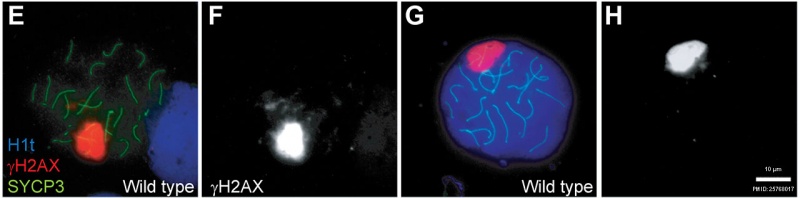
Mouse meiosis pachytene[15]
Diplotene
- diplotene phase, diplonema; Greek, diplonema = "two threads"
- homologous chromosomes begin to separate but remain attached by chiasmata.
- synaptonemal complex degrades and the chromosomes separate from one another a small amount giving this appearance.
- It is possible that some chromosome uncoiling may also occur allowing some gene transcription.
- In the developing human ovary, oocytes remain at the diplotene stage from fetal life through postnatal childhood, until puberty when the lutenizing hormone (LH) surges stimulate the resumption of meiosis.
Diakinesis
- diakinesis phase; Greek, diakinesis = "moving through"
- homologous chromosomes continue to separate, and chiasmata move to the ends of the chromosomes.
- prophase I ends and chromosomes now recondense, transcription stops and the transition to metaphase occurs.
Prometaphase I
- Spindle apparatus formed, and chromosomes attached to spindle fibres by kinetochores.
Metaphase I
- Homologous pairs of chromosomes (bivalents) arranged as a double row along the metaphase plate. The arrangement of the paired chromosomes with respect to the poles of the spindle apparatus is random along the metaphase plate. (This is a source of genetic variation through random assortment, as the paternal and maternal chromosomes in a homologous pair are similar but not identical. The number of possible arrangements is 2n, where n is the number of chromosomes in a haploid set. Human beings have 23 different chromosomes, so the number of possible combinations is 223, which is over 8 million.)
Anaphase I
- The homologous chromosomes in each bivalent are separated and move to the opposite poles of the cell.
Telophase I
- The chromosomes become diffuse and the nuclear membrane reforms.
Cytokinesis I
- Cellular cytoplasmic division to form two new cells, followed by Meiosis II.
- Note - in oocyte meiosis, the extrusion of the first polar body (1 PB) indicates completion of the first meiotic division.
Prophase II
- Chromosomes begin to condense, nuclear membrane breaks down and spindle forms.
Metaphase II
- Spindle fibres attach to chromosomes, chromosomes align in cell centre.
Anaphase II
- Chromosomes separate and move to the opposite poles of the cell.
Telophase II
- Chromosomes reach spindle pole ends and the nuclear membrane reforms.
Cytokinesis
Cellular cytoplasmic division to form new cells.
Meiosis Sex Differences
Female (oogenesis)
- Meiosis initiated once in a finite population of cells
- 1 gamete produced / meiosis
- Completion of meiosis delayed for months or years
- Meiosis arrested at 1st meiotic prophase and reinitiated in a smaller population of cells
- Differentiation of gamete occurs while diploid in first meiotic prophase
- All chromosomes exhibit equivalent transcription and recombination during meiotic prophase
Male (spermatogenesis)
- Meiosis initiated continuously in a mitotically dividing stem cell population
- 4 gametes produced / meiosis
- Meiosis completed in days or weeks
- Meiosis and differentiation proceed continuously without cell cycle arrest
- Differentiation of gamete occurs while haploid after meiosis ends
Sex chromosomes excluded from recombination and transcription during first meiotic prophase
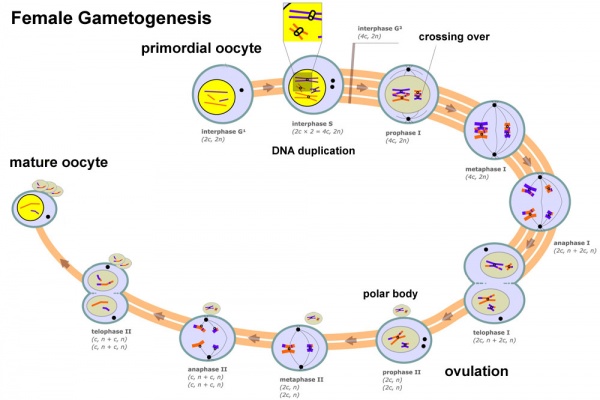
Female Gametogenesis
In females, the total number of eggs ever to be produced are present in the newborn female.
- All eggs are arrested at an early stage of the first meiotic division as a primary oocyte (primordial follicle). Following purberty, during each menstrual cycle, pituitary gonadotrophin stimulates completion of meiosis 1 the day before ovulation.
- In meiosis 1, a diploid cell becomes 2 haploid (23 chromosomes) daughter cells, each chromosome has two chromatids. One cell becomes the secondary oocyte the other cell forms the first polar body.
- The secondary oocyte then commences meiosis 2 which arrests at metaphase and will not continue without fertilization.
- At fertilization meiosis 2 completes, forming a second polar body. Note that the first polar body may also undergo this process forming a third polar body.
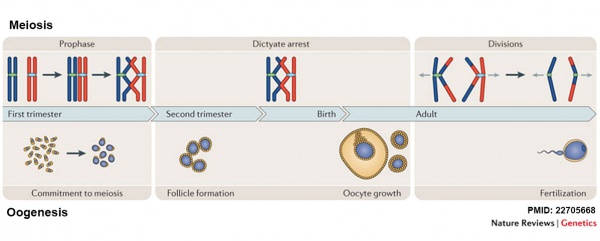
Meiosis and Oogenesis[17]
|
Meiosis - divided into 3 temporally distinct phases.
- Prophase - after DNA replication, homologous chromosomes (shown in red and blue) undergo pairing, synapsis and recombination, and arrest at the diplotene (dictyate) stage.
- Dictyate arrest - oocytes remain in meiotic arrest until the female reaches maturity and the oocyte has completed an extensive period of growth following follicle formation.
- Divisions - luteinizing hormone (LH) surge that triggers ovulation also causes resumption and completion of the first meiotic division in the periovulatory oocyte. The ovulated egg is arrested at second meiotic metaphase, and anaphase onset and completion of meiosis II only occur if the egg is fertilized.
Oogenesis - complex involving 4 distinct phases.
- Commitment to meiosis and meiotic initiation - occurs at GA 8–10 weeks in humans.
- Follicle formation - occurs during the second trimester in humans.
- Oocyte growth - occurs in the sexually mature female under the control of paracrine and endocrine signals. Oocyte growth is thought to take approximately 85 days in humans and typically culminates in the ovulation of a single egg.
- Fertilization - of the ovulated egg results in the completion of the second meiotic division.
|
| Oocyte Meiotic Spindles
|
- Mammalian oocytes have no centrosomes, but still form spindles using many microtubule-organizing centres lacking centrioles. (cells dividing by mitosis have 2 centrosomes to help form the mitotic spindle) PMID 17693257
- Meiotic spindle is a microtubule structure but its relocation in the oocyte cytoplasm involves microfilaments, filamentous-actin structures nucleated by Formin-2 (Fmn2). PMID 19062278
|
Polar Body
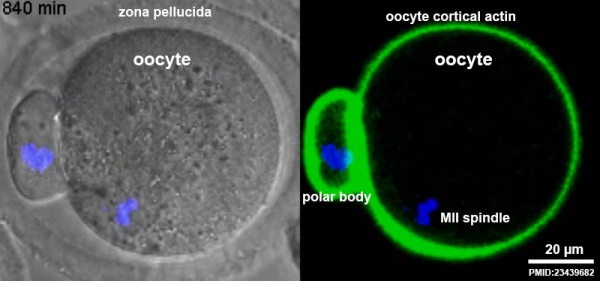
First Polar Body[12]
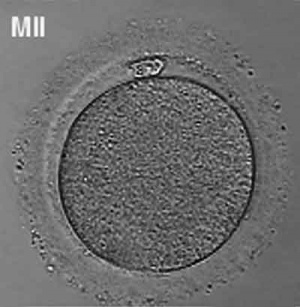
Human oocyte at metaphase II showing polar body at 12 o'clock position.
The breakdown of the germinal vesicle indicates a resumption of meiosis and the extrusion of the first polar body (1 PB) indicates completion of the first meiotic division in human oocytes. The polar body is a small cytoplasmic exclusion body formed to enclose the excess DNA formed during the oocyte (egg) meiosis and following sperm fertilization. There are 2-3 polar bodies derived from the oocyte present in the zygote, the number is dependent upon whether polar body 1 (the first polar body formed during meiosis 1) divides during meiosis 2. This exclusion body contains the excess DNA from the reductive division (the second and third polar bodies are formed from meiosis 2 at fertilization). These polar bodies do not contribute to the future genetic complement of the zygote, embryo or fetus.
Recent research in some species suggest that the space formed by the peripheral polar body (between the oocyte and the zona pellucia) can influence the site of spermatozoa fertilization.
Assisted reproductive techniques involving intracytoplasmic sperm injection (ICSI) have looked at the "quality" of the polar body and found that the morphology is related to mature oocyte viability and has the potential to predict oocyte fertilization rates and pregnancy achievement.[18][19]
- Polar Body Electron Micrographs
First Polar Body cortical granules
First Polar Body chromosomes
First Polar Body microtubules
Second Polar Body chromatin
Second Polar Body cytoplasmic bridge
- Links: Category:Polar Body
Female Abnormalities
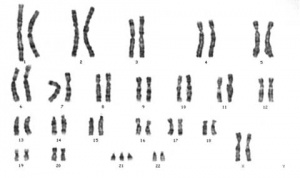
Trisomy 21 female karyotype
Meiotic non-disjunction resulting in aneuploidy, most are embryonic lethal and not seen. The potential for genetic abnormalities increase with maternal age.
- Autosomal chromosome aneuploidy
- Sex chromosome aneuploidy
- Monosomy X - Turner's Syndrome
- trisomy X - Triple-X syndrome
- 47 XXY - Klinefelter's Syndrome
Male Gametogenesis
In males, sperm continues to be generated throughout life from a stem cell population in the testis. Spermatozoa maturation involves two processes meiosis and spermiogenesis
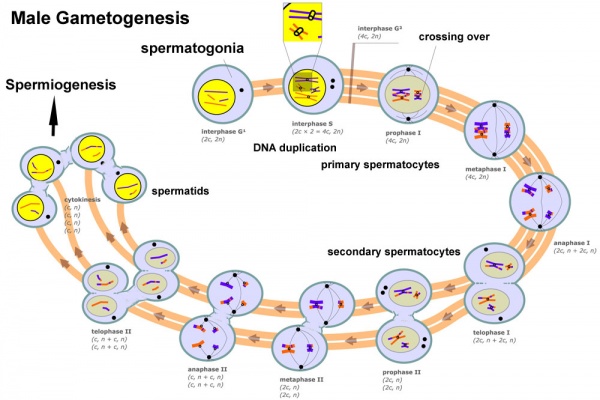
The above figure compares meiosis to the female (the polar bodies have been removed and labelling updated).
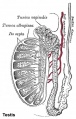
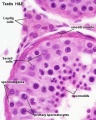
Adult Seminiferous tubule showing spermatozoa developmental stages
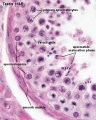
Seminiferous tubule cross-section and supporting cells
Human Spermatozoa Development
- Spermatogenesis process of spermatagonia mature into spermatazoa (sperm).
- Continuously throughout life occurs in the seminiferous tubules in the male gonad- testis (plural testes).
- At puberty spermatagonia activate and proliferate (mitosis).
- about 48 days from entering meiosis until morphologically mature spermatozoa
- about 64 days to complete spermatogenesis, depending reproduction time of spermatogonia
- follicle stimulating hormone (FSH) - stimulates the spermatogenic epithelium
- luteinizing-hormone (LH) - stimulates testosterone production by Leydig cells
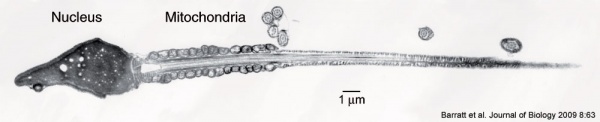
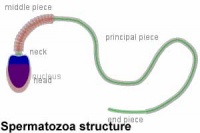
|
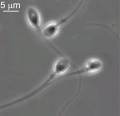
Mature human spermatozoa
- 60 µm long, actively motile
- divided into 3 main regions (head, neck and tail)
- head - (flattened, 5 µm long by 3 µm wide) the nucleus and acrosome. Posterior part of nuclear membrane forms the basal plate.
- neck - (1 µm) attached to basal plate, transverse oriented centriole, contains nine segmented columns of fibrous material, continue as outer dense fibres in tail.
- tail - 3 parts a middle piece, principal piece and end piece
- middle piece - (5 µm long) axonema and dense fibres surrounded by mitochondria
- principal piece - (45 µm long) fibrous sheath interconnected by regularly spaced circumferential hoops
- end piece - (5 µm long) axonema surrounded by small amount of cytoplasm and plasma membrane
|
- Links: Spermatozoa Development
Puberty
- In humans at puberty, hormonal and morphological changes occur within the gonad and other systems (secondary sex characteristics).
- Within the testis the immature Sertoli cells cease to proliferate and differentiate.
- Spermatogonium proliferate and spermatogenesis begins.
- It takes about 70 days for cells to mature from the diploid spermatogonium to a primary spermatocyte.
- This maturation occurs in waves along the seminiferous tubules.
- Links: Puberty
Ejeculate
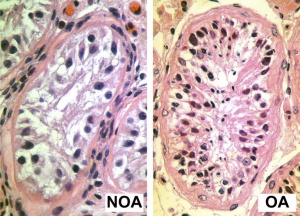
Azoospermia - Non-obstructive azoospermia (NOA) and Obstructive azoospermia (OA)
- release of spermatozoa and accessory gland secretions from the male genital tract (3.5 ml)
- 200-600 million sperm, by volume less than 10 % spermatozoa
- Accessory Gland secretions - 60 % seminal vesicle, 30 % prostate and 10 % bulbourethral
Male Abnormalities
- Oligospermia - (Low Sperm Count) less than 20 million sperm after 72 hour abstinence from sex
- Azoospermia - (Absent Sperm) blockage of duct network
- Immotile Cilia Syndrome - lack of sperm motility
Meiosis in Other Species
- Sea urchin - oocytes complete meiosis before being shed.
- Starfish - oocytes only complete meiosis upon hormonal stimulation.
Abnormalities
Meiotic Nondisjunction
- Occurs when homologues fail to separate during meiotic division I or II
- For example trisomy 21 (Down Syndrome) caused by an extra copy of chromosome 21
- Links: trisomy 21 | Nondisjunction
Chromosomal Translocations
- Philadelphia chromosome
- Chronic myelogenous leukemia
- Piece of Chr9 exchanged with Chr22 Generates truncated abl
Overstimulates cell production
References
- ↑ 1.0 1.1 Sakakibara Y, Hashimoto S, Nakaoka Y, Kouznetsova A, Höög C & Kitajima TS. (2015). Bivalent separation into univalents precedes age-related meiosis I errors in oocytes. Nat Commun , 6, 7550. PMID: 26130582 DOI.
- ↑ 2.0 2.1 Lin T, Diao YF, Kang JW, Lee JE, Kim DK & Jin DI. (2013). Chromosomes in the porcine first polar body possess competence of second meiotic division within enucleated MII stage oocytes. PLoS ONE , 8, e82766. PMID: 24312673 DOI.
- ↑ Jan SZ, Jongejan A, Korver CM, van Daalen SKM, van Pelt AMM, Repping S & Hamer G. (2018). Distinct prophase arrest mechanisms in human male meiosis. Development , 145, . PMID: 29540502 DOI.
- ↑ Duan X & Sun SC. (2018). Actin cytoskeleton dynamics in mammalian oocyte meiosis. Biol. Reprod. , , . PMID: 30010726 DOI.
- ↑ Wang S, Hassold T, Hunt P, White MA, Zickler D, Kleckner N & Zhang L. (2017). Inefficient Crossover Maturation Underlies Elevated Aneuploidy in Human Female Meiosis. Cell , 168, 977-989.e17. PMID: 28262352 DOI.
- ↑ Ren H, Ferguson K, Kirkpatrick G, Vinning T, Chow V & Ma S. (2016). Altered Crossover Distribution and Frequency in Spermatocytes of Infertile Men with Azoospermia. PLoS ONE , 11, e0156817. PMID: 27273078 DOI.
- ↑ Mirzaghaderi G & Hörandl E. (2016). The evolution of meiotic sex and its alternatives. Proc. Biol. Sci. , 283, . PMID: 27605505 DOI.
- ↑ Bury L, Coelho PA & Glover DM. (2016). From Meiosis to Mitosis: The Astonishing Flexibility of Cell Division Mechanisms in Early Mammalian Development. Curr. Top. Dev. Biol. , 120, 125-71. PMID: 27475851 DOI.
- ↑ Royo H, Seitz H, ElInati E, Peters AH, Stadler MB & Turner JM. (2015). Silencing of X-Linked MicroRNAs by Meiotic Sex Chromosome Inactivation. PLoS Genet. , 11, e1005461. PMID: 26509798 DOI.
- ↑ Yun Y, Lane SI & Jones KT. (2014). Premature dyad separation in meiosis II is the major segregation error with maternal age in mouse oocytes. Development , 141, 199-208. PMID: 24346700 DOI.
- ↑ Zhai R, Yuan YF, Zhao Y, Liu XM, Zhen YH, Yang FF, Wang L, Huang CZ, Cao J & Huo LJ. (2013). Bora regulates meiotic spindle assembly and cell cycle during mouse oocyte meiosis. Mol. Reprod. Dev. , 80, 474-87. PMID: 23610072 DOI.
- ↑ 12.0 12.1 Yi K, Rubinstein B, Unruh JR, Guo F, Slaughter BD & Li R. (2013). Sequential actin-based pushing forces drive meiosis I chromosome migration and symmetry breaking in oocytes. J. Cell Biol. , 200, 567-76. PMID: 23439682 DOI.
- ↑ Shibuya H, Morimoto A & Watanabe Y. (2014). The dissection of meiotic chromosome movement in mice using an in vivo electroporation technique. PLoS Genet. , 10, e1004821. PMID: 25502938 DOI.
- ↑ Boku T, Nakane Y, Komada H, Nakagawa M, Hirozane N, Hioki K & Yamamoto M. (1991). [Prognostic factors of "pm" gastric cancer]. Nihon Geka Gakkai Zasshi , 92, 17-23. PMID: 2014022
- ↑ Pacheco S, Marcet-Ortega M, Lange J, Jasin M, Keeney S & Roig I. (2015). The ATM signaling cascade promotes recombination-dependent pachytene arrest in mouse spermatocytes. PLoS Genet. , 11, e1005017. PMID: 25768017 DOI.
- ↑ Xu XL, Ma W, Zhu YB, Wang C, Wang BY, An N, An L, Liu Y, Wu ZH & Tian JH. (2012). The microtubule-associated protein ASPM regulates spindle assembly and meiotic progression in mouse oocytes. PLoS ONE , 7, e49303. PMID: 23152892 DOI.
- ↑ Nagaoka SI, Hassold TJ & Hunt PA. (2012). Human aneuploidy: mechanisms and new insights into an age-old problem. Nat. Rev. Genet. , 13, 493-504. PMID: 22705668 DOI.
- ↑ Ebner T, Yaman C, Moser M, Sommergruber M, Feichtinger O & Tews G. (2000). Prognostic value of first polar body morphology on fertilization rate and embryo quality in intracytoplasmic sperm injection. Hum. Reprod. , 15, 427-30. PMID: 10655316
- ↑ Younis JS, Radin O, Izhaki I & Ben-Ami M. (2009). Does first polar body morphology predict oocyte performance during ICSI treatment?. J. Assist. Reprod. Genet. , 26, 561-7. PMID: 19960239 DOI.
Textbooks
Podcasts
Reviews
Greaney J, Wei Z & Homer H. (2017). Regulation of chromosome segregation in oocytes and the cellular basis for female meiotic errors. Hum. Reprod. Update , , . PMID: 29244163 DOI.
Brar GA & Amon A. (2008). Emerging roles for centromeres in meiosis I chromosome segregation. Nat. Rev. Genet. , 9, 899-910. PMID: 18981989 DOI.
Articles
Zelazowski MJ, Sandoval M, Paniker L, Hamilton HM, Han J, Gribbell MA, Kang R & Cole F. (2017). Age-Dependent Alterations in Meiotic Recombination Cause Chromosome Segregation Errors in Spermatocytes. Cell , 171, 601-614.e13. PMID: 28942922 DOI.
Ren H, Ferguson K, Kirkpatrick G, Vinning T, Chow V & Ma S. (2016). Altered Crossover Distribution and Frequency in Spermatocytes of Infertile Men with Azoospermia. PLoS ONE , 11, e0156817. PMID: 27273078 DOI.
Wang S, Kleckner N & Zhang L. (2017). Crossover maturation inefficiency and aneuploidy in human female meiosis. Cell Cycle , 16, 1017-1019. PMID: 28471715 DOI.
Search Pubmed
Search Pubmed: meiosis
NCBI - Policies and Guidelines | PubMed | Help:Reference Tutorial
Additional Images
Meiotic prophase I stages
Chromosome connections in meiosis
Kinetochore attachment to the spindle and chromosome cohesion in mitosis and meiosis
Human testis NANOG expression
Adult worm hermaphrodite gonad arm
Mouse oocyte microtubule-associated protein
Terms
| Cell Division Terms (expand to view)
|
meiosis | mitosis
- anaphase - (Greek, ana = up, again) Mitosis term referring to the fourth stage, where the paired chromatids now separate and migrate to spindle poles. This is followed by telophase.
- anaphase A - Mitosis term referring to the part of anaphase during which the chromosomes move.
- anaphase B - Mitosis term referring to the part of anaphase during which the poles of the mitotic spindle move apart.
- aneuploidy - (aneuploid) term used to describe an abnormal number of chromosomes mainly (90%) due to chromosome malsegregation mechanisms in maternal meiosis I.
- aster - (Latin, aster = star) star-like object visible in most dividing eukaryotic cells contains the microtubule organizing center.
- astral microtubule - spindle apparatus microtubule (MT) originating from the centrosome which does not connect to a kinetochore. These microtubules only exist during mitosis, the other spindle types are polar and kinetochore microtubules.
- autosomal inheritance - term used in hereditary diseases which means that the disease is due to a DNA error in one of the 22 chromosome pairs that are not sex chromosomes. Both boys and girls can then inherit this error. If the error is in a sex chromosome, the inheritance is said to be sex-linked.
- bivalent - (tetrad) a pair of homologous chromosomes physically held together by at least one DNA crossover.
- bouquet stage - meiosis term for when in prophase transition to the zygotene stage, the chromosome telomeres attachment to the inner nuclear envelope and form a cluster. This occurs before the onset of homologous pairing and synapsis. The name comes from the chromosomes resembling a "bouquet of flowers".
- diploid - (Greek, di = double + ploion = vessel) having two sets of chromosomes (2n), this is the normal euploidy state for all human cells, other than gametes that are haploid (n, a single set of chromosomes).
- diplotene stage- (diplotene phase, diplonema; Greek, diplonema = "two threads") meiotic stage seen during prophase I, the chromosomes separate from one another a small amount giving this appearance. In the developing human ovary, oocytes remain at the diplotene stage from fetal life through postnatal childhood, until puberty when the lutenizing hormone (LH) surges stimulate the resumption of meiosis. Prophase I, is divided into 5 stages (leptotene, zygotene, pachytene, diplotene, diakinesis) based upon changes associated with the synaptonemal complex structure that forms between two pairs of homologous chromosomes.
- euploidy - the normal genome chromosomal set (n, 2n, 3n) or complement for a species, in humans this is diploid (2n). The other classes of numerical chromosomal abnormalities include aneuploidy, polyploidy and mixoploidy.
- FUCCI - Acronym for Fluorescence Ubiquitination Cell Cycle Indicator a molecular tool for identifying the stage in the cell cycle. In G0/G1 cells express a red fluorescent protein and S/G2/M cells express a green fluorescent protein. (More? Tooth Development Movie)
- haploid - (Greek, haploos = single) Having a single set of chromosomes (n) as in mature germ/sex cells (oocyte, spermatozoa) following reductive cell division by meiosis. Normally cells are diploid, containing 2 sets of chromosomes. Ploidy refers to the number of sets of chromosomes in the nucleus of a cell.
- heteroplasmy - presence of more than one type of organellar genome. In humans this can refer to variations in the mitochondrial DNA (mtDNA). (More? PMID 26281784)
- homologous chromosomes - meiosis term for the two matching (maternal and one paternal) chromosomes that align during meiosis I.
- homologous recombination - meiosis term when DNA of homologous chromosomes is covalently exchanged to produce chromosomes with new allele combinations, and also links homologous chromosomes with each other to form a bivalent
- human genome - DNA within the 23 nucleus chromosome pairs and the cytoplasmic mitochondrial DNA.
- kinetochore - the protein structure formed on chromatids where the spindle kinetochore microtubules attach during cell division.
- kinetochore microtubule - spindle apparatus microtubule (MT) that attaches to the chromosome kinetochore by its plus end, the other spindle types are astral and polar microtubules.
- kinesin - a microtubule (MT) motor protein that exists in many isoforms and most move towards the MT positive end. Different isoforms have different functions within the spindle apparatus. PMID 20109570
- meiosis - reductive cell division required to produce germ cells (oocyte, spermatozoa) and for sexual reproduction. Note that only spermatozoa complete meiosis before fertilisation. Chromosome number is reduced from diploid to haploid, during this process maternal and paternal genetic material are exchanged. All other non-germ cells in the body divide by mitosis. (More? Meiosis | Spermatozoa Development | Oocyte Development | Week 1)
- meiosis I - (MI) the first part of meiosis resulting in separation of homologous chromosomes, in humans producing two haploid cells (N chromosomes, 23), a reductional division.
- meiosis II - (MII) the second part of meiosis. In male human spermatogenesis, producing of four haploid cells (23 chromosomes, 1N) from the two haploid cells (23 chromosomes, 1N), each of the chromosomes consisting of two sister chromatids produced in meiosis I. In female human oogenesis, only a single haploid cell (23 chromosomes, 1N) is produced. Meiosis II: Prophase II - Metaphase II - Anaphase II - Telophase II.
- meiotic silencing of unsynapsed chromatin - (MSUC) an aneuploidy protective mechanism for subsequent generations, during meiosis where chromosomes are silenced that fail to pair with their homologous partners.
- merotelic kinetochore - cell division abnormality in chromosomal attachment that occurs when a single kinetochore is attached to microtubules arising from both spindle poles. Normal chromosomal attachment in early mitosis, is by only one of the two sister kinetochores attached to spindle microtubules (monotelic attachment) later sister kinetochores attach to microtubules arising from opposite spindle poles (amphitelic attachment).
- metaphase - mitosis term referring to the third stage where mitotic spindle kinetochore microtubules align chromosomes in one midpoint plane. Metaphase ends when sister kinetochores separate. Originally based on light microscopy of living cells and electron microscopy of fixed and stained cells. A light microscope analysis called a "metaphase spread" was originally used to detect chromosomal abnormalities in cells. Mitosis Phases: prophase - prometaphase - metaphase - anaphase - telophase
- metaphase spread - In mitosis using light microscope analysis originally used to detect chromosomal abnormalities in cells, as chromosomes are only visible during cell division.
- microfilament - (MF) cytoskeleton filament normally required for cytoplasmic intracellular transport, motility and cell shape. Named by the actin monomers assembling into the smallest in cross-section of the three filament systems (microtubules and intermediate filaments). This system is disassembled and reassembled as the contractile ring for cytokinesis (cytoplasm division) following cell division mitosis and meiosis.
- microtubule - (MT) cytoskeleton filament normally required for cytoplasmic intracellular transport and motility. Named by the tubulin monomers assembling into "tubes", and are the largest in cross-section of the three filament systems (microfilaments and intermediate filaments). This system is disassembled and reassembled as the spindle apparatus during cell division.
- mitochondrial DNA - (mtDNA) multiple copies of a small circular DNA molecule located within the mitochondria matrix. In humans 16,568 bp in length containing 37 genes, originally inherited only from the oocyte (maternal inheritance).
- mitosis - (M phase) The normal division of all cells, except germ cells, where chromosome number is maintained (diploid). In germ cell division (oocyte, spermatozoa) meiosis is a modified form of this division resulting in reduction in genetic content (haploid). Mitosis, division of the nucleus, is followed by cytokinesis the division of the cell cytoplasm and the cytoplasmic contents. cytokinesis overlaps with telophase.
- p - chromosome short arm (possibly French, petit) and used along with chromosome and band number to indicate genes located on this arm of the chromosome. The chromosome long arm is identified as q (possibly French, tall) chosen as next letter in alphabet after p. These chromosomal arms are only seen when the chromosome is folded for cell division.
- polar microtubule - spindle apparatus microtubule (MT) that can arise from either pole and overlap at the spindle midzone. This interdigitating structure consisting of antiparallel microtubules is responsible for pushing the poles of the spindle apart. The other spindle types are astral and kinetochore microtubules.
- prometaphase - (Greek, pro = before) mitosis term referring to the second stage, when the nuclear envelope breaks down into vesicles. Microtubules then extend from the centrosomes at the spindle poles (ends) and reach the chromosomes. This is followed by metaphase.
- pronuclear fusion - (Greek, pro = before) the process of the fusion of the two haploid nuclear structures (pronuclei) contributed from the spermatazoa and oocyte to form the first diploid nucleus cell. Can also be called "fusion of pronuclei".
- pronucleus - (Greek, pro = before; plural, pronuclei) the two haploid nuclei or nuclear structures containing the genetic material from the spermatozoa and the oocyte. These two haploid nuclei will fuse together to form the first diploid nucleus cell, the zygote. Therefore the nuclear structures that exist "before the nucleus", the plural term is pronuclei.
- prophase - (Greek, pro = before) - mitosis term referring to the first stage, when the diffusely stained chromatin resolves into discrete chromosomes, each consisting of two chromatids joined together at the centromere.
- prophase I - meiosis term refers to the first phase of meiosis I, which together with meiosis II results in the reductive cell division only occurring gametes. Prophase can be further divided into a number of stages: leptotene zygotene, pachytene, diplotene, diakinesis.
- q - chromosome long arm (possibly French, tall), the next letter in alphabet after p, and used along with chromosome and band number to indicate genes located on this arm of the chromosome. The chromosome short arm is identified as p (possibly French, petit). These chromosomal arms are only seen when the chromosome is folded for cell division.
- S phase - during interphase of cell cycle where DNA is duplicated prior to second growth period (G2 phase) that is followed by mitosis (M phase).
- synapsis - (syndesis) meiosis term for the pairing of two homologous chromosomes that occurs during prophase I.
- synaptonemal complex - meiosis term for a protein structure essential for synapsis of homologous chromosomes. (proteins SCP3 and SCP1).
- telomere - region found at each end of the chromosome and involved in cellular ageing and the capacity for division. The regions consist of repeated sequences protecting the ends of chromosomes and harbour DNA repair proteins. In the absence of the enzyme telomerase, these regions shorten during each cell division and becoming critically short, cell senescence occurs.
- telophase - mitosis term referring to the fifth stage, where the vesicles of the nuclear envelope reform around the daughter cells, the nucleoli reappear and the chromosomes unfold to allow gene expression to begin. This phase overlaps with cytokinesis, the division of the cell cytoplasm.
- telomerase - the enzyme that maintains the chromosome end length, the telomeres, involved in cellular ageing and the capacity for division. Absence of telomerase activity leads to the chromosome ends shorten during each cell division, becoming critically short and cell senescence then occurs.
- tetrad - (bivalent) a pair of homologous chromosomes physically held together by at least one DNA crossover.
|
|
|
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, April 17) Embryology Cell Division - Meiosis. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Cell_Division_-_Meiosis
- What Links Here?
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G