2017 Group Project 4
| 2017 Student Projects | |||
|---|---|---|---|
|
Eye Development
Introduction
The eye is a complex sensory structure which allows a variety of species to intake and process visual information from the world around us. Most structures forming the human eye are developed between the embryonic weeks 3 through 10, deriving primarily from ectoderm, neural crest cells and mesenchyme.
This project page will start with a description of the functional anatomy of the adult eye, continue with a description and timeline of the embryonic formation and signaling pathways of important structures of the human eye and animal models and finish with an examination of congenital abnormalities, current research and pending questions for the near future.
We hope that this page provides you with an enjoyable, comprehensive introduction to the embryology of the eye (we have aimed to keep it a concise 'reachable' page!)
Anatomy of the Adult Eye
To discuss the key anatomical components of the eye, we will break down the eye in parts[1]:
Supporting Structures (Orbit, Extraocular Muscles, Lacrimal Gland, Eyelid)
The orbit consists of a framework of bones and connective tissue which provide structural support and protection to the sensitive human eye. 7 bones contribute to the orbit of the eye: frontal, lacrimal, sphenoid, zygomatic, ethmoid, maxillary, palatine. Several openings exist in the orbital structure, 2 key fissures are the superior orbital fissure and inferior orbital fissure and the posterior optic canal. These openings allow cranial nerves passageway.
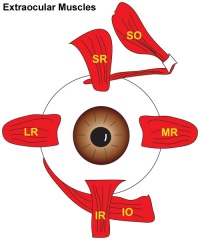
A set of 6 extra-ocular muscles allow for a strong voluntary control of the movement of the eye. These muscles include the superior rectus, inferior rectus, lateral rectus, medial rectus, inferior oblique, superior oblique muscles. These muscles are some of the smallest in the human body and are able to produce fast, controlled motion to focus on an object of interest.
The eyelid is the anterior covering of the eye, consisting of a thin fold of skin. It is controlled by the levitator palpebrae superioris muscle. Supero-lateral to the eyelid is the lacrimal gland, which secretes a film of tear, an important component to keeping the anterior surface of the eye moist. The eyelid assists in this process by opening and closing, or blinking, which spreads the tear film across the surface of the eye. Debris caught in the tear film is excreted through the lacrimal duct which is located on the infero-medial surface of the eyelid.
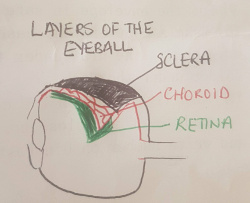
Layers of the Eyeball (Sclera, Choroid, Retina)
The superficial layer of the eyeball is the sclera, a thick fibrous layer which thickens as it extends posteriorly. The sclera is visible as the white part of our eye. Its function is to provide structural stability to the eye and serves as attachment site for extra-ocular muscle insertions. Deep to the sclera is the choroid, a vascular network which provides nourishment to the layers of the eyeball and plays a large role in the growth of the eye. Deep to the choroid is the retina, a network of nerve fibres which transmit visual information. The retina contains 2 types of photoreceptors which convert visible light into nerve impulses (rods and cones). Rods work best in low light environments while cones work best in high light enthronements and detect colour and sharp detail.[2]: The retina does not extend to the anterior part of the eyeball, unlike the sclera and choroid layers.
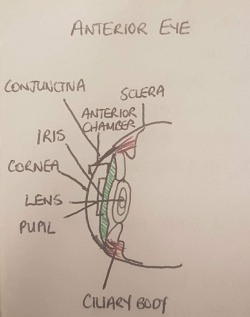
Anterior structure (Lens, Cornea, Pupil, Iris, Ciliary Body, Conjunctiva, Anterior Chamber)
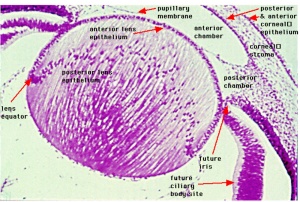
The most anterior component of the eyeball is the cornea, which is a multilayered avascular transparent structure. Its major function is to refract incoming light to assist image formation on the retina (contributes about 75% of total refraction). Continuous with the cornea superiorly and inferiorly is the conjunctiva. Deep to the cornea is the iris, the pigmented ring of the eye. The iris consists of 2 muscles which act to enhance and diminish the size of the pupil (the aperture surrounded by the iris) allowing light to enter the eye. Deep to the pupil and iris is the lens, which is a transparent structure which contributes the other 25% of the eye's refractive power. The lens changes shape through accommodation (focusing) controlled by the ciliary body muscles, changing the position of images to form them on the retina. The ciliary body is the anterior extension of the choroid. Between the anterior surface of the lens and posterior surface of the cornea lies the anterior chamber. This chamber is filled with aqueous humour supplied by the ciliary body, which provides nutrients to ocular structures and maintains intra-ocular pressure.
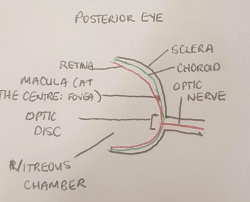
Posterior structure (Optic Nerve, Optic Disc, Macula, Fovea, Posterior Chamber)
Found in the posterior region of the retina is the macula, a central spot in which a high concentration of cone photoreceptors are found. The direct centre of the macula is called the fovea which contains solely cone photoreceptors and has a high workload and hence a strong supporting vascular structure[3]:. Found in the most posterior part of the eye is the optic nerve (which is coupled with the central retinal artery and central retinal vein), responsible for transporting nerve impulses to the cerebral cortex for processing. The opening of the optic nerve is called the optic disc, which contains no photoreceptors and is the blind spot of the eye. Between the posterior surface of the lens, the retina and the optic disc is the posterior chamber. The posterior chamber contains the vitreous humour, a jelly like substance with a high viscosity which maintains intra-ocular pressure.
Embryonic Contributions
The development of the eye is a complex process. It involves the formation of the retina and the lens, which we together call the eyeball. It also involves the formation of auxiliary eye structures which are the eyelid, lacrimal gland, and cornea. The retina is formed from the neuroectoderm, the lens from the surface ectoderm and the auxiliary tissues are formed from the head surface ectoderm, neural crest cells and the head mesoderm [4].
| Embryonic contributions | Eye component |
|---|---|
| Neuroectoderm of the forebrain | Retina, Posterior layers of the iris, The optic nerve |
| Surface ectoderm of the head | The lens of the eye, The corneal epithelium |
| Mesoderm between the neuroectoderm and the surface ectoderm | The fibrous and vascular coats of the eye |
| Neural crest cells | Choroid, Sclera, Corneal endothelium |
Timeline of embryonic development
| WEEK | DEVELOPMENT |
|---|---|
| 3-4 | Eye fields, Optic vesicles |
| 5-6 | Optic Cup, Lens Vesicle, Choroid Fissure, Hyaloid Artery |
| 7-8 | Cornea, Anterior Chamber, Pupillary Membrane, Lens, Retina |
| 8-10 | Eyelids |
| 9-15 | Iris, Ciliary Body |
Carnegie Stages
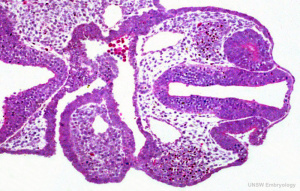
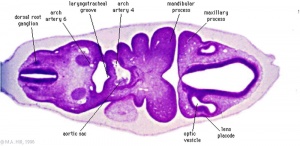
Carnegie Stages are a system of 23 stages used to describe developmental events of the vertebrate embryo. At stage 10 we see early signs related to eye development. The table below collates and summarises some of the work of Anthony A. Person [5].
| Stage | Events |
| Stage 10 (22 days) | The optic primordium has developed. Two small grooves develop on each side of the developing forebrain in the neural folds - these are the optic grooves. The optic placode has begun to develop, which is seen as a small thickening of the surface ectoderm lateral to the hindbrain. |
| Stage 11 (24 days) | Optic vesicle begins to form from the optic groove. |
| Stage 12 (26 days) | The optic vesicles have extended from the forebrain to the surface ectoderm. The optic vesicle now lies close to the surface ectoderm. |
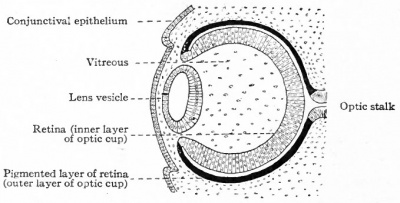
| Stage 13 (28 days) | The optic vesicle interacts with the surface ectoderm and will induce this ectoderm to form the lens placode, which is the precursor of the lens. Optic evagination and differentiation makes it possible to identify some parts of retina, including the future pigmented layer of retina and optic stalk. |
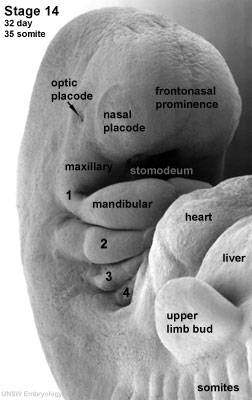
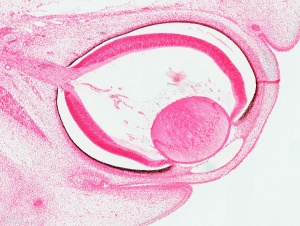
| Stage 14 (32 days) | The lens placode is indented by the lens pit and is cup-shaped. |
| Stage 15 (33 days) | The lens pit will close and form a circle that separates from the surface ectoderm and becomes the lens vesicle. The lens vesicle and optic cup lie close to the surface ectoderm, which creates a slight elevation in the region of the eye. |
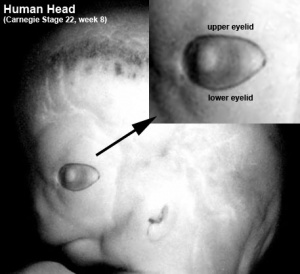
| Stage 16 (37 days ) | The lens body has grown and now has a D-shaped lens cavity. The first indication of the development of the eyelids is visible as eyelid grooves above and below the eye. |
| Stage 17 - 19 | Retinal pigment is visible and the retinal fissure is almost closed. The eyes are still laterally placed but begin migration to a more anterior position. The lower eyelid fold develops first, followed by the upper eyelid fold. |
| Stage 18 (44 days) | Mesenchyme invades the region between the lens epithelium and the surface ectoderm. The eyes have shifted to a more anterior position. The grooves above and below the eyes are deeper but have not joined yet. |
| Stage 19 - 22 | The eyelid folds develop into the eyelids and the upper and the lower eyelids now meet at the outer canthus (the outer corner of the eye). |
| Stage 20 (51 days) | The lens cavity is lost. The upper and lower lids meet laterally and medially. The eyelids now partly cover the eye. The lens suture begins to form. |
| Stage 23 (57 days) | End of embryonic period. The face begins to look human. Eyelids closure is complete. |
Development of the eye components
Introduction
<html5media width=“480" height="360">https://www.youtube.com/embed/ghHDFWlfpoQ</html5media>
The eye starts to develop at 22 days. The optic grooves (sulci) appears in the neural folds at the cranial end of the embryo. When the neural fold fuse to form the forebrain, the optic grooves will form optic vesicles. The optic vesicles are continuous cavities from the cavity of the forebrain and project from the wall of the forebrain into the mesenchyme. The optic vesicle extends from the diencephalon and will come in contact with the surface ectoderm of the head. This induces the formation of a lens placode [6].
The surface ectoderm near the optic vesicles will thicken and form the lens placodes. The lens placodes will sink into the surface ectoderm and form lens pits. The edges of the lens pits will travel towards each other and fuse to form round lens vesicles, which will later lose connection with the surface ectoderm. The optic vesicles keep developing, eventually forming double-walled optic cups which are connected to the brain by the optic stalk. The two layers of the optic cup will differentiate in different directions. The cells of the outer layer will produce melanin pigment and later become the pigmented retina. The cells of the inner layer of the optic cup will proliferate fast and develop glia, ganglion cells, interneurons and light-sensitive photoreceptor neurons. These cells are in the neural retina. The ganglion cells of the retina are neurons that send signals to the brain. The axons of the ganglion cells of the neural retina will grow in the wall of the optic stalk. The cavity in the optic nerve will start disappearing, and instead, the axons of the ganglion cells will form the optic nerve. The optic stalk is now the optic nerve [7]
The optic cups will fold inwards around the lens while the lens vesicles have grown inwards losing their connection with the surface ectoderm and placing them in the cavities of the optic cups. The retinal fissures (linear grooves) will develop and cover the ventral surface of the optic cups and down to the optic stalk. The retinal fissures contain vascular mesenchyme and hyaloid blood vessels will develop here. The hyaloid artery supplies the structures in the eye with blood and the hyaloid vein will return the blood from these structures [7].
Formation of the optic vesicle
It is a specific area of the neural ectoderm that will become the optic vesicle - this happens because of a group of transcription factors - Six3, Pax6, and Rx1. These transcription factors are expressed in the most anterior tip of the neural plate. This area will split into bilateral regions and form the optic vesicles. The Pax6 protein has shown to be especially important for the development of the lens and retina. This protein is important for photoreceptive cells in all phyla. Pax6 is also present in the murine forebrain, hindbrain, and nasal placodes, but the eyes are most sensitive its absence [7].
The sonic hedgehog gene is important for the separation of the single eye field into two fields. If this gene is inhibited, the eye field will not split which will result in cyclopia, a single eye in the center of the face [7].
Retina
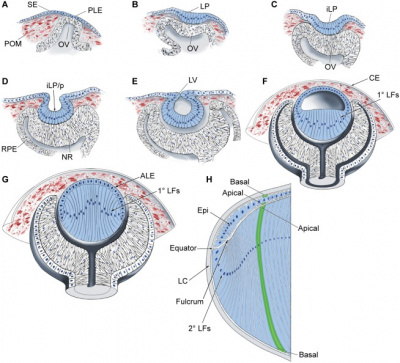
The retina is the essential component of the eye with the primary function of photoreception [8]. Formation of the retina begins with the specification of retinal cells in the anterior neuroectoderm. The first morphological sign of eye development is the formation of two lateral grooves in the anterior neuroectoderm, collectively referred to as optic sulci. The cells of the optic sulci will evaginate and form the optic vesicle. The distal portion of the optic vesicle will form the retina and the proximal will form the optic stalk [9]. The walls of the optic cup will develop into the two layers of the retina. The thin, outer layer of the optic cup will become the pigment layer of the retina and the thick, inner layer of the optic cup will differentiate into the neural retina. Forkhead transcription factors regulate the proliferation and differentiation of the retinal precursor cells. During the embryonic and early foetal periods, the two layers of the retina are separated by an intra-retinal space, derived from the cavity of the optic cup. Eventually, the two layers of the retina fuse and the intra-retinal space disappears [6].
When the lens is developing the inner layer of the optic cup starts to proliferate and forms a thick neuroepithelium which will later differentiate into the neural retina. The neural retina contains photoreceptors and the cell bodies of neurons and is the light-sensitive region of the retina. The axons of the ganglion cells in the neural retina grow in the wall of the optic stalk and will form the optic nerve [6].
Lens
Human lens induction occurs at around 28 days and is completed around day 56. The surface ectoderm will thicken near the optic vesicle and create the lens placode and later form the lens vesicle [10]. Lens cells come from ectoderm and differentiate into either lens fibres or the lens epithelium. The anterior monolayer of epithelial cells of the lens will create the lens epithelium, which makes the sheet of cuboidal epithelium covering the anterior surface of the lens. The posterior lens vesicle cells will produce the linear primary fibre cells aligned parallel to the optic axis. These fibres will create the lens mass and form the embryonic lens nucleus. The lens epithelial cells will keep proliferating and produce new cells which generate secondary lens fibre cells. These rows of cells will form the outer shells and keep the lens growing throughout life. This makes the eye lens unique - it will have an addition of new cells inside the surrounding capsule all the time [11].
Ciliary Body
The ciliary body is a muscular and secretory tissue and is located directly behind the lens. The ciliary body forms part of the anterior segment of the eye and is an important regulator of eye physiology and the vision. The ciliary body produces the aqueous humour which fills the anterior chamber, nourishing the lens and cornea while also maintaining intra-ocular pressure. The ciliary body also synthesises collagenIX and tenasin-C [12].
The ciliary body extends from the iris root (anteriorly) to the ora serrata (posteriorly). It consist of ciliary muscles and ciliary processes. Each ciliary process (fold) is covered by a double-layered secretory epithelium; the outer pigmented and the inner un-pigmented (closes to the lens) layers. The epithelial layers of the ciliary body come from the retina of the optic cup. The inner non-pigmented ciliary epithelium is connected with the neural retina and the outer pigmented ciliary epithelium which is connected with the retinal pigmented epithelium. The epithelium of the iris is the further anterior extension. The epithelial layers are associated with a stroma containing the ciliary muscle. The ciliary muscle is complex, but can be divided into three portions; anterior, posterior and internal [13].
In the chick eye development, we see the mesenchyme grow together on the margin of the optic cup and will form the stroma of the ciliary body and the iris, which is located more anteriorly. It is unknown how if the lens has a role in inducing the secretory ciliary body epithelium and the muscular iris epithelium [14].
Iris
The iris is the circular, pigmented muscular portion of the eye with a central aperture that is the pupil. The main function of this round muscle is to manipulate pupil diameter as an adjustment to various conditions, regulating the amount of light entering the eye. This is influenced by dilator pupillae (radial) and sphincter pupillae (constricting) muscles. Iris pigmentation is dependent on the abundance and distribution of certain types of melanin in the posterior epithelium of the iris (eumelanin and pheomelanin).
The iris develops at the end of the third month of development as a thin layer derived from the anterior rim of the optic cup [15]. It is formed by the pigment-containing internal and external layers of the optic cup and by a layer of vascularised connective tissue which also contains the pupillary muscles. The internal layers of the optic cup form the non-pigmented iris parts whereas the external layers of the optic cup develop into the pigmented epithelial structures. [16]. Iris stroma develops as a result of neural crest cell migration – within this stroma, the dilator and constrictor muscles develop from the neuroectoderm of the optic cup. In the anterior of the eye, the optic epithelium is non-neural and matures as ciliary body and iris epithelia [16].
Cornea
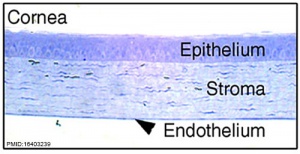
The cornea is the transparent and avascular component located at the anterior-most section of the eye. It is composed of five layers stemming from the 3 main layers of epithelium, stroma and endothelium:
- Corneal epithelium (most anteriorly) – consisting of multiple layers of cells which rest on a basement membrane.
- Bowman’s membrane – a clear, acellular homogenous layer. This layer is a modified portion of the stroma and has no regenerative capacity as a response to damage [17].
- Corneal stroma – largest portion of the retina consisting of collagenous tissue.
- Descemet’s membrane – a homogenous, elastic true basement layer with a regenerative capacity.
- Corneal endothelium – a single layer of cuboidal cells responsible for the maintenance of the dehydrative nature of corneal tissue. Cite error: The opening
<ref>tag is malformed or has a bad name.
Corneal development involves interactions between ectoderm-derived tissues. More specifically, the corneal epithelium arises from interactions between the cranial ectoderm and optic vesicles derived from the neural ectoderm [18]. The neural tube and the adjacent ectoderm give rise to the neural crest cells which are multipotent with a migratory capacity. These cells contribute to the formation of corneal epithelium and the stromal cells. Innervation of the cornea stroma and epithelium is derived from both the neural crest and the ectodermal placode Cite error: The opening <ref> tag is malformed or has a bad name. The endothelium is comprised of differentiated neural crest cells which contribute to the later formation of Descemet’s membrane [19].
Aqueous Chambers
There are two aqueous chambers of the eye: anterior and poster. The anterior chamber is filled with a plasma-like fluid called aqueous humour, produced by the ciliary body and trabecular meshwork in 3 main stages: diffusion, ultrafiltration and active secretion [20]. The main function of this fluid is to maintain the intra-ocular pressure as well as provide structural integrity to the round shape of the eyeball. This structure also has a role in the immune response and provides nutrition to the avascular tissue (e.g., the cornea and lens). [21].
The anterior chamber is defined as the developed space posterior to the cornea (between the cornea and the iris) and develops at the stage of differentiation of the mesodermal portion of the iris [16]. The space defining the posterior chamber develops as the mesenchyme between the lens and the peripheral iris.
Choroid and Sclera
The choroid and sclera surround the eye and are located adjacent to each other (the choroid lines the inner portion of the sclera). The sclera is the outer, fibrous white layer of the eye which functions as a supportive wall of the eye ball and is continuous with the cornea Cite error: The opening <ref> tag is malformed or has a bad name . The heavy vasculature of the choroid provides a major supply of oxygen and nourishment to the retina [22].
They are derived from interactions between the neural crest and mesoderm-derived mesenchyme. The sclera forms as a result of condensation of the mesenchyme outside the optic cup [23].
The choroid arises from the mesenchyme surrounding the optic vesicle and the cranial neural crest cells. The first vasculature structures appear in the choroid during week 15 in the form of arterioles and venules; veins and arteries become distinguishable by week 22 [24].
Extraocular muscles
The extraocular muscles are responsible for eye movement within the eye orbit – a critical aspect of sight and communication. 6 main muscles contribute to eye movement. These include: superior rectus(SR), inferior rectus(IR), lateral rectus (LR), medial rectus (MR), superior oblique (SO) and inferior oblique (IO).
The development of these muscles is dependent on interactions between the eye, its muscles and the neural crest. [25].
Somites supplied by cranial nerve III forms 5 of the 6 extraocular muscles which form an interlocking tendinous ring made of fibrous connective tissue. The 5 muscles include the lateral, medial, superior and inferior rectus, and the superior oblique muscle. This common tendinous ring is known as the annulus of Zinn and surrounds the entry of the optic nerve. The remaining sixth muscle is the inferior oblique muscle and is separate from the aforementioned tendinous ring due to its distinct point of origin. Cite error: The opening <ref> tag is malformed or has a bad name.
Eyelids
The eyelids are upper and lower folds of skin functioning as a barrier protecting the anterior surface of the eye from direct local injury. Eyelids also regulate light exposure, distribute precorneal tear film over the corneal surface when blinking and are involved in functions involving tear flow [26]. No sign of eyelid fold development is evident at week 5 however at this stage the lens pit begins to invaginate from the surface ectoderm [27]. Week 6 initiates the timeline of eyelid development as small depressions emerge in the surface ectoderm both above and below the developing eye – these rapidly deepen to form the folds of the eyelids [28]. The lower eyelid fold develops first and, by the end of week 6, the lower fold is more distinct than the upper eyelid fold [29]. Week 7 marks the point at which both the upper and lower folds are distinctly developed and during this stage, the upper lid assumes its more dominant role [27]. The eyelid is not fully closed - there is a gap between the folds and the cornea is slightly visible – they eventually undergo fusion in week 8 [27].
Lacrimal Glands
The lacrimal glands are exocrine glands located in the upper lateral region of each eye orbit which secrete the aqueous potion of the tear film. This film is continuously secreted and functions to clean and protect the external surface of the eye, keeping the environment constantly moist and preventing possible injury from dust and other particles [30]. Lacrimal gland morphogenesis is divided into three stages.
- The first is the presumptive glandular stage. This stage involves the thickening of the conjunctival superior fornix epithelium and the condensing of the surrounding mesenchyme [31].
- The second is the bud stage. This stage is considered the initial sign of glandular formation due to the development of nodular structures and lumina within epithelia buds [31].
- The third is the glandular maturity stage (week 9-16). In this stage, the lacrimal gland begins to morphologically mirror the adult gland [31].
Signalling pathways and transcription factors involved throughout eye development
Eye field development
Many signalling pathways and transcription factors are involved in the development of the eye field (which forms in addition to the telencephalon and diencephalon as part of division of the anterior neural plate). Eye progenitor cells are guided into the eye field region by fibroblast growth factor, bone morphogenetic protein and Wnt-signalling pathways [32]. Additionally, interactions between canonical and non-canonical Wnt-signalling have been shown to maintain the eye field border [33].
The cells of the eye field express eye field transcription factors, some of which include Rx1/Rax, Pax6, Lhx2, Six3 and Otx2. Six3, Pax6, Otx2 and Rx1 specify retinal lineage and regulate progenitor cell movement to guide them to their correct locations [32]. Six3 also partly mediates Wnt-signalling repression, allowing for the development of the optic vesicles from the eye field [34]. Lhx2 maintains optic lineage in eye progenitor cells and suppresses the potential for differentiation towards different fates [35].
Eye field division and progression to optic cup
TGF-β, FGF and Shh family factors secreted by the axial mesoderm underlying the eye field initiate its splitting into two bilateral, symmetric eye primordia [34]. These factors also regulate the expression of transcription factors Pax2, Pax6, Vax1 and Vax2, thereby establishing the boundaries and patterning the proxiodistal and ventronasal axes of the optic vesicles [32].
Despite the close relationship between cellular proliferation and morphogenesis, optic vesicle evagination does not appear to be mainly driven by proliferation [34]. Alternatively, it has been shown that retinoic acid has critical importance in optic vesicle evagination to form the optic cup [32]. Rx3 activity has also been observed to have involvement in optic vesicle evagination [34].
From optic cup to developed eye
Lens placode maturation and lens development is dependent on Pax6 expression and regulation. During placode formation bone morphogenetic protein and fibroblast growth factor signalling regulate Pax6. Within the presumptive lens ectoderm, Pax6 is regulated by Six3 and Meis. However, within the presumptive epithelium Pax6 is a regulating factor, regulating the expression of Sox2 and eventually Six3 once the lens placode is formed [32].
Meanwhile, in the primordial retina retinal progenitor cells become retinal ganglion cells through the expression of the basic helix-loop-helix transcription factor ath5 (which is regulated by Hh and Fgf signalling. The amacrine, horizontal and bipolar cells of the retina are generated by a variety of transcription factors, some of which include Pax6, Six3 and Foxn4 [32]. Crx and Otx2 positive cells progress towards photoreceptor fates and are further developed into rods and cones based on specific transcription factor expression (TRβ2 and RxRγ expression is required for cones, where as Nrl, Nr2e3 and Ascl1 expression is required for rods) [36]. The transcription factors required for the development of the retinal pigment epithelium include Mitf, Otx1/Otx2 and Pax6. TGF-β, FGF, BMP and Hh signalling from the surrounding mesenchyme have also been shown to help induce retinal pigment epithelium fate in progenitor cells [32].
Within the choroid, the expression and regulation of many different factors and signalling pathways is required for the development of proper arterial-venous connections, allowing for the functional circulation of nutrients and oxygen throughout the eye. VEGF, bFGF, PDGF and PEDF (from the retinal pigment epithelium) drive the development and maturation of choroidal blood vessels. Vascular patterning is regulated by the interactions between VEGF and Delta-like 4 factors. Additionally, Notch signalling has been shown to have importance in the formation of capillary beds [32].
Eye formation in the absence of the retina
The gene Rx is a key component in the formation of retinal structures. Studies have shown that mice lacking Rx function do not form optic sulci or optic vesicles and do not have retina-specific gene expression. There has been made studies in human, medaka, zebrafish and Xenopus which suggest that Rx genes are required for the formation of the vertebrate retina. It is also shown that in Rx-deficient mouse embryos the mature lens does not develop. This indicates that retinal cells are necessary for lens formation, and if the mouse is lacking Rx gene, then it will not display any retina-specific gene expression [4].
It is thought that signaling from the optic vesicle is important to activate the lens-specific gene and the formation of the lens placode. If there are no retinal cells generated, then the morphogenesis of the optic cup does not take place and the lens will not form. The study also showed that elimination of β-catenin expression in the head surface ectoderm in Rx-deficient embryos would develop a lens-like structure even though there were no optic vesicle/cup. Elimination of β-catenin lead to upregulation of Pax6, which is a gene that has a key role in lens formation. This indicates that the optic vesicle/cup might not be required for lens formation. The lens developed smaller than in wild type embryos, which means that the optic cup/vesicle might have an effect on a determination of the size of the lens. It could be that the developing optic vesicle/cup shields the developing lens placode from signals for other tissues that could disturb the lens formation [4].
It is not fully understood if the formation of the auxiliary eye structures is dependent on the development of the eyeball and its components, the retina and the lens. A study made my Eric C. Swindell and al. showed that auxiliary eye structures form even in the absence of retinal morphogenesis and retina-specific gene expression. This indicates that there are two separate developmental processes involved in the formation of the eye and its associated structures. In the absence of the retina and the lens, we still see specific gene expression in the surface ectoderm initiating the formation of auxiliary eye structures like eyelids and lacrimal glands [4].
Current research
Iris epithelium cells as a potential source for regenerative medicine for retinal pigment epithelium degeneration
The retinal pigment epithelium (RPE) is a monolayer of neural-crest-derived cells and is located between the photoreceptors and the choroid. There are many disorders that involve dysfunctional RPE and leads to retinal degenerative diseases. There is no treatment for these diseases, therefore, one of the most promising future therapy for RPE related disorders is to replace the cells of the dysfunctional RPE [37]..
Studies have focused on the development and use of induced pluripotent stem cells (iPSC) for cell replacement therapy. It is possible to differentiate iPSC in vitro towards RPE cells and use them for experimental transplantation studies in animal models. But an alternative strategy for a retinal replacement is based on transdifferentiation (direct conversion) and involves transforming an adult somatic cell into another adult somatic cell. Use of overexpression of cell-lineage specific genes can convert one cell into another cell type and skip the pluripotent state. Iris epithelium (IE) cells might be able to be transdifferentiated into the RPE since both RPE and IE are neuro-epithelia and has a common embryological origin - they both derive from neuroectoderm of the developing optic cup [37].
A study made by Anna Bennis et al. [37]. has shown that the canonical pathways for the most highly expressed genes of the IE and the RPE were very similar. Wnt signaling pathways has shown to be active in the IE but not in the RPE. The Wnt signaling pathway consists of a group of signal transduction pathways that has an influence on cell fate determination and cell proliferation during embryonic development. Activation of Wnt SP makes it possible to reprogram somatic cells (also retinal neurons) into iPSC and is important for the differentiation of pluripotent stem cells to RPE cells. The high expression of Wnt SP genes in the iE suggests that the IE has a multipotent character during life and therefore could be transformed into RPE cells used for RPE replacement [37].
Developing Retinal Ganglion Cells from Pluripotent Cells
A 2016 paper [38] addressed the difficulty of culturing retinal ganglion cells (RGC) from pluripotent cells (both embryonic and enducded) due to a lack of known specific markers to communicate the cell to develop into a RGC. This study found a method of developing RGC through a stepwise approach involving retinal progentior cells. The RGC cells created showed no functional difference that normal RGC cells. Applications of this discovery have included helping to treat patients with congenital glaucoma abnormalities
UNSW Optometry and Vision Science Current Projects
Additionally, UNSW has a number of current research projects currently being undertaken [39]
A few interesting topics include:
- Biochemical changes in the tear film in relationship to the corneal structural changes in diabetes so as to be able to predict nerve damage earlier
- The use of video games and virtual reality (visual electrophysiology) to help treat some congenital anomalies such as lazy eye
- How retinal circuitry operates with regards to neurotransmitter release, receptor location and receptor function, allowing the development of a retinal roadmap which can be applied to retinal diseases
Animal Models
Zebrafish
The zebrafish species is used to investigate ocular development, function and disease as it possesses the similar morphology to a range of other vertebrates (including humans). The advantages of zebrafish models include: they are able to quickly reach their sexual maturity, their behaviour can be observed quite early on because of their rapid development and most importantly, their embryos are translucent. Zebrafish are also most active during the day and as a result, their retinas contain a large number of diverse cone subtypes in additions to rods. [40]
Through studying zebrafish, it was found that that a mutation in chokh/rx3 gene resulted in the absence of eyes during the earliest stages of development, due to the failure in developing retinal progenitor cells. [41]. Additionally, the optical vesicle was inverted and the neuronal differentiation was blocked. The study also found that the DNA-binding homeodomain and the entire C-terminal portion led to a nonsense mutation that resulted in complete loss of function in the eye. It was concluded that the gene chokh/rx3 is important for the developing optic primordia. [40]
Mouse
One of the many studies carried out on mice investigated the effects of certain transcriptional and growth factors to ocular development. The absence of Krüppel-like transcription factor (Klf4) in the cornea of a knockout mice resulted in increased corneal epithelial fragility, stromal oedema and defective lens after eight weeks. This suggests that Klf4 transcription factor affects the structural integrity of corneal epithelium, maintenance of stromal hydration levels and the development of conjunctival goblet cells, deeming it to be a gene of interest to treat ocular surface conditions such as dry eyes, Meesmann's dystrophy, and Steven's-Johnson syndrome. [42]
Chick
Chick embryos have been used to investigate retina cell proliferation and regeneration, where an interactive relationship between the Sonic hedgehog (Shh) pathway and the fibroblast growth factor-2 (FGF2) (expressed in the ciliary body or the ciliary marginal zone) was observed. It was found that the activation of one pathway activates the other and likewise, inhibiting one of the factors that inhibit the other pathway. The experiment also demonstrated that FGF and Shh are required for cell survival after retina removal [43]. The significance of discovering this relationship between these pathways in the chick embryo would provide more understanding of retinal regeneration that could lead to restoration of vision from retinal lesions or retinal degeneration.
Congenital Abnormalities
Although quite rare, abnormalities can occur during the embryonic development that resulting in detrimental effects to the features of the eyes, their function as well as the continuing normal development after birth. These abnormalities can occur unilaterally or bilaterally. It is also common for most reported cases to possess multiple eye abnormalities. Thus, the estimated prevalence statistics listed in the table can overlap.
| Congenital Anomalies | Description | Epidemiology |
|---|---|---|
| Optic Nerve Hypoplasia | A condition where the optic nerves are underdeveloped. | Prevalence: Reported to be the most common abnormality [44]
|
| Coloboma | A condition where parts of the eye is missing and failed to develop normally. | Prevalence: Ocular coloboma was estimated to be 8.0 per 100 000 cases [45] |
| Anopthalmia | The absence of the eyes. | Prevalence: Anophthalmia-Microphthalmia Syndrome was estimated to be 5.3 per 100 000 cases [45]
Cause: SOX2 gene mutation [46] |
| Microphtalmia | Abnormal reduction in size of the eyeballs. | |
| Aniridia | Complete or partial absence of the iris. | Prevalence: Estimated to be 1.5 per 100 000 cases [45] |
| Ptosis ('Lazy Eye') | Structural abnormalities in the eyelid, can be congenital - specifically the failure of innervation of the oculomotor nerve for levator palpebrae superioris [47] - fixable through surgery | Prevalence: a relatively well known abnormality |
| Keratoglobus | Structural abnormalities in the formation of the cornea - unknown cause - fixable through surgery[48] | Prevalence: understood to be a rare condition[48] |
| Aphakia | The absense of a lens in the eye, causing a loss of accommodation and hence far-sightedness. People with aphakia have relatively small pupils which are unable to dilate as much [49] | Prevalence: a rare congenital abnormality |
Future questions
Will it be possible to have eye transplants in the future?
Lots of research has been done in making a whole-eye transplantation successful in order to finally cure blindness. Blindness is a worldwide problem and is currently untreatable due to the fact that retinal ganglion cells and the optic nerves do not regenerate. This can result from diseases including macular degeneration and end of stage glaucoma. The first successful eye transplantation was led by Stone and Cole in 1943[50] investigating 104 samples of the salamander species, Amblystoma Punctatum, and one of its eyes were either implanted back into the same animal or it was completely transplanted to a new host. It was reported that circulation in the iris was re-established, retina was able to regenerate, a new optic nerve was connected to the brain through the optic chiasma and ocular movements were regained. Vision was reestablished by the second month. In a more recent study, in 2015, Li et al.[51] investigated the feasibility of eye transplantation by observing the surviving 15 rat models out of the original 22. They were able to confirm the integrity and functional return of vision after whole-eye surgeries with the aid of advance techniques that are available today. The Ear and Eye Foundation of Pittsburgh[52] is an organization that is currently running a project with lead researchers to discover a way for the optic nerves to regenerate and to cure blindness by 2020.
Will the bionic eye pass clinical trials in the next few years?
The bionic eye is an external visual device that has been in the making for the last several years. It hopes to bring back full vision through the aid of a pair of glasses with a camera attached to it, which then transmits high-frequency radio signals to the microchip implanted in the users' eye. These signals are converted into electrical impulses by the electrodes implanted in the chip, which then stimulates the retinal cells and passes through the optic nerve. Electrical impulses travels to the vision processing centres in the brain and these are then interpreted as an image. At the moment, users must learn to understand light flashes and visual patterns and convert them into images they can interpret. Current prototypes require the users to have a previously working eyes, which means their optic nerves must still be relatively healthy and their visual cortex must be developed. Patients who have suffered from retinitis pigmentosa and age-related macular degeneration would benefit this technology. Bionic Vision Australia[53] is one of the leading organisations that are aiming to treat blindness and are set to perform clinical trials on two of the devices of the bionic eye implants soon. Their objectives are to develop strategies that can improve the affected visual pathway to the brain by stimulating the remaining working retinal cells and also develop safe surgical procedures.
Glossary
| Term | Definition |
| Age-related macular degeneration | An eye disease with its onset usually after age 60 that can progressively destroy the macula, the central portion of the retina, impairing central vision. |
| Aqueous humour | A transparent fluid similar to plasma with low protein concentrations secreted by the ciliary epithelium. |
| Choroid | The pigmented vascular layer of the eye located between the retina and sclera. |
| Choroid fissure | A ventral groove formed by the invagination of the optic cup located at the bottom of the optic vesicle through which blood vessels enter the eye. |
| Ciliary body | The connection between the iris and choroid consisting of the ciliary muscle, ciliary processes and the ciliary ring. Alters the shape of the lens and ciliary epithelium in order to focus on an image. |
| Cornea | The transparent anterior part of the external coat of the eye covering the iris and pupil and continuous with the sclera. |
| Ectoderm | The outermost primary germ cell layer in the early embryo. |
| Endoderm | The innermost primary germ cell layer in the early embryo. |
| Ectodermal placode | A neurogenic placode consisting of an area of thickened epithelium in the embryonic head ectoderm layer that gives rise to neurons and other structures of the sensory system. |
| Extraocular muscles | Muscles controlling eye movement. |
| Fovea | A small depression in the retina where visual acuity is the highest. |
| Hyaloid vasculature | The temporary circulatory system in fetal eyes which spontaneously degenerate when the retinal blood vessels begin to develop. |
| Intraocular pressure | Fluid pressure inside the eye created through continued renewal of fluids. |
| Iris | The coloured muscular ring surrounding the pupil which manipulates the diameter and size of the pupil and controls the amount of light entering the eye. |
| Lens | The transparent biconvex structure posterior to the iris with the main function of focusing light onto the retina in order to form clear images of objects at various distances. |
| Lens pit | A pit-like depression in the fetal head where the lens develops. |
| Lens placode | The placode developing in the ectoderm overlying the optic vesicle, eventually becoming the lens of the eye. |
| Lens vesicle | The ectodermal invagination in the embryo formed opposite the optic cup and gives rise to the lens. |
| Lumina | the central cavity of a tubular structure. |
| Macula | An oval-shaped pigmented area where there is the largest density of cone cells and is involved in producing the sharpness of central vision. |
| Mesenchyme | Loosely organised embryonic connective tissue originating from the mesoderm. |
| Neural crest | A temporary group of embryonic ectodermal cells. |
| Neural retina | Refers to three layers of neural cells within the retina (photoreceptor cells, bipolar cells and ganglion cells). |
| Neural tube | A hollow tubular structure as a result of neural plate folding which later develops into the brain and spinal cord. |
| Neuroepithelium | Epithelium consisting of specialised cells which detect sensory triggers for reception of external stimuli. |
| Optic cup | A structure of the diencephalon formed after optic vesicle folding. This structure gives rise to the retina. |
| Optic groove | Two small grooves on each side of the developing forebrain in the neural folds. Also chiasmatic groove. |
| Optic nerve | Cranial nerve II which transmits impulses to the brain from visual information detected by the photoreceptive retina. |
| Optic stalk | Pair of slender embryonic structures that give rise to the optic nerve. |
| Optic vesicle | An evagination of each lateral all of the embryonic vertebrate forebrain from which the nervous structures of the eye develop. |
| Pupil | The central opening in the iris, allowing and regulating the entry of light into the eye. |
| Retina | The sensory membrane that forms the inner lining of the eye containing rods and cones with the main function of photoreception. Nerve impulses in response to visual information are transmitted to the brain via the optic nerve. |
| Retinitis Pigmentosa | A chronic hereditary eye disease characterized by breakdown and loss of cells in the retina leading to progressive visual loss. |
| Sclera | The dense, white, fibrous membrane that, along with the cornea, forms the external protective covering of the eye |
| Tear film | A layer of tears which spreads over the eye, ultimately maintaining the health of the ocular surface by making the external surface smooth and clear.
|
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
References
- ↑ Chen, J., 2011. Handbook of Visual Display Technology. Springer Berlin Heidelberg. Chapter 2.1
- ↑ Curcio CA, Hendrickson AE. Organization and development of the primate photoreceptor mosaic. Prog Ret Ret. 1991;10:89–120.
- ↑ <pubmed>6462623</pubmed>
- ↑ 4.0 4.1 4.2 4.3 <pubmed>PMC3104407</pubmed>
- ↑ <pubmed>7364662</pubmed>
- ↑ 6.0 6.1 6.2 Schoenwolf, G.C., Bleyl, S.B., Brauer, P.R., Francis-West, P.H. & Philippa H. (2015). Larsen's human embryology (5th ed.). New York; Edinburgh: Churchill Livingstone.
- ↑ 7.0 7.1 7.2 7.3 Gilbert SF. Developmental Biology. 6th edition. Sunderland (MA): Sinauer Associates; 2000. Available from: https://www.ncbi.nlm.nih.gov/books/NBK9983/
- ↑ <pubmed>7084144</pubmed>
- ↑ <pubmed>18675797 </pubmed>
- ↑ <pubmed>20171212 </pubmed>
- ↑ <pubmed>25406393</pubmed>
- ↑ <pubmed>17275804</pubmed>
- ↑ <pubmed>12127103</pubmed>
- ↑ <pubmed>17275804</pubmed>
- ↑ <pubmed>22219630</pubmed>
- ↑ 16.0 16.1 16.2 <pubmed>18168498</pubmed>
- ↑ <pubmed>7084144</pubmed>
- ↑ <pubmed>26310148</pubmed>
- ↑ <pubmed>6511224</pubmed>
- ↑ <pubmed>6763801</pubmed>
- ↑ <pubmed>21293732</pubmed>
- ↑ <pubmed>20044062</pubmed>
- ↑ <pubmed>1628748</pubmed>
- ↑ <pubmed>2199235</pubmed>
- ↑ <pubmed>21482859</pubmed>
- ↑ <pubmed>1993591</pubmed>
- ↑ 27.0 27.1 27.2 <pubmed>27124372</pubmed>
- ↑ <pubmed>7364662</pubmed>
- ↑ <pubmed>21416630</pubmed>
- ↑ <pubmed>17001201</pubmed>
- ↑ 31.0 31.1 31.2 <pubmed>14635806</pubmed>
- ↑ 32.0 32.1 32.2 32.3 32.4 32.5 32.6 32.7 <pubmed>25476579</pubmed>
- ↑ <pubmed>16413771</pubmed>
- ↑ 34.0 34.1 34.2 34.3 <pubmed>23684892 </pubmed>
- ↑ <pubmed>23595746</pubmed>
- ↑ <pubmed>20648062</pubmed>
- ↑ 37.0 37.1 37.2 37.3 <pubmed>PMC5565104</pubmed>
- ↑ Ohlemacher, S. K., Sridhar, A., Xiao, Y., Hochstetler, A. E., Sarfarazi, M., Cummins, T. R. and Meyer, J. S. (2016), Stepwise Differentiation of Retinal Ganglion Cells from Human Pluripotent Stem Cells Enables Analysis of Glaucomatous Neurodegeneration. Stem Cells, 34: 1553–1562. doi:10.1002/stem.2356
- ↑ https://www.optometry.unsw.edu.au/research/current-research
- ↑ 40.0 40.1 Glass, A. S. & Dahm, R. (2003). The Zebrafish as a Model Organism for Eye Development. Opthalmic Research, 36(1), 4-24. doi: 10.1159/000076105
- ↑ Loosli, F., Staub, W., Finger-Baier, K. C., Ober, E. A., Verkade, H., Wittbrodt, J. & Baier, H. (2003). Loss of eyes in zebrafish caused by mutation of chokh/rx3. EMBO Reports, 4(9), 894-899. doi: 10.1038/sj.embor.embor919
- ↑ Swamynathan, S. K., Katz, J. P., Kaestner, K. H., Ashery-Padan, R., Crawford, M. A. & Piatigorsky, J. (2007). Conditional deletion of the mouse Klf4 gene results in corneal epithelial fragility, stromal edema, and loss of conjunctival goblet cells. Molecular Cell Biology, 27(1), 182-94. doi: 10.1128/MCB.00846-06
- ↑ Spence, J. R., Aycinena, J. & Del Rio-Tsonis, K. (2007). Fibroblast growth factor–hedgehog interdependence during retina regeneration. Developmental Dynamics, 236(5), 1161-1174. doi: 10.1002/dvdy.21115
- ↑ Ryabets-Lienhard, A., Stewart, C., Borchert, M., & Geffner, M. E. (2016). The Optic Nerve Hypoplasia Spectrum - Review of the Literature and Clinical Guidelines. Advances in Pediatrics, 63, 127-146. doi: 10.1016/j.yapd.2016.04.009
- ↑ 45.0 45.1 45.2 Orphanet Report Series – Rare Diseases Collection. (2017). Prevalence of rare diseases: Bibliographic data (no. 2). Paris. France: Author. Retrieved from: [[1]]
- ↑ Mauri, L., Franzoni, A., Scarcello, M., Sala, S., Garavelli, L. Modugno, A., ... Penco, S. (2015). SOX2, OTX2 and PAX6 analysis in subjects with anophthalmia and microphthalmia. European Journal of Medical Genetics, 58, 66-70. doi: 10.1016/j.ejmg.2014.12.005
- ↑ <pubmed>8759493</pubmed>
- ↑ 48.0 48.1 <pubmed>23807384</pubmed>
- ↑ <pubmed>28913511</pubmed>
- ↑ Stone, L. S. & Cole, C. H. (1943). Grafted Eyes of Young and Old Adult Salamanders (Amblystoma Punctatum) Showing Return of Vision. Yale Journal of Biology and Medicine, 15(5), 735-754.2. PMCID: PMC2601300
- ↑ Li, Y., Komatsu, C., Wang, B., Miller, M., Wang, H., van der Merwe, Y., Ho, L., Kostereva, N., Zhang, W., Xiao, B., Davidson, E., Solari, M., Steketee, M. B., Guo, S., Kagemann, L., Wollstein, G., Schuman, J., Chan, K., Gorantla, V. S., Washington, K. M. (2015). Abstract 112: Evaluation of Viability, Structural Integrity and Functional Outcome after Whole Eye Transplantation. Plastic & Reconstructive Surgery, 135(5), 82. "PMC2601300"
- ↑ Ear and Eye Foundation of Pittsburgh: https://eyeandear.org/our-research/ophthalmology/the-whole-eye-transplant-project
- ↑ Bionic Vision Australia: http://bionicvision.org.au/about/research_plan