2015 Group Project 5
| 2015 Student Projects | ||||
|---|---|---|---|---|
| 2015 Projects: Three Person Embryos | Ovarian Hyper-stimulation Syndrome | Polycystic Ovarian Syndrome | Male Infertility | Oncofertility | Preimplantation Genetic Diagnosis | Students | ||||
| 2015 Group Project Topic - Assisted Reproductive Technology | ||||
| This page is an undergraduate science embryology student and may contain inaccuracies in either description or acknowledgements. | ||||
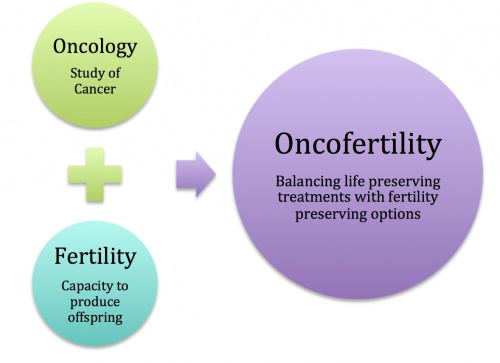
Oncofertility
Oncofertility refers to the medical field that bridges the specialties of oncology and reproductive endocrinology with the purpose of maximizing the reproductive potential of cancer patients and survivors. Infertility is an adverse side affect of cancer and cancer treatment. Previously, this has been tolerated since the main concern was treating patients with the main goal being their survival. However, over the past decade more people have been surviving cancer, and concern for fertility has garnered greater attention as reproductive ability is considered an imperative for many. Thus came about the notion of Oncofertility as an attempt to spare or restore fertility function in men and women suffering from cancer. [1]
The aim of this page is to explore infertility as a cause of cancer due to the cancer treatments that are utilised to treat cancer and as a result impair fertility. Following this different fertility preservation methods will be discussed that can be implemented in males and females.
Oncofertility timeline
| Date | Event |
| 1838 | Blastema Theory – Muller demonstrates that cancer contain cells. His student found the cells were derived from other cells. |
| 1846 | Anesthesia invented, allowing doctors to remove entire tumors during surgery along with the lymph nodes. Later Paget (surgeon) reported cancers spread through metastasis |
| 1856 | J. Marian Sims incised the cervix through surgery to make the opening larger to address infertility |
| 1878 | Thomas Beatson finds by removing a rabbits ovaries, their breast stop producing milk. This lead to new hormonal drugs to treat prostate and breast cancer. |
| 1896 | X-ray discovered by Roentgen. 3 years later, radiation used to diagnose and treat cancer. |
| Late 1800s to early 1900s | More doctors were using surgery to treat infertility and more women sought medical advice due to their inability to conceive. Success rates are unknown. Options available included unblocking fallopian tubes through surgery, artificial insemination (husband or donor sperm) or ovarian transplantation (grafting portion of fertile woman’s ovary into infertile woman). |
| 1915 | Cancer induced in rabbits by applying coal tar to its skin. Now more than 100 carcinogens have been discovered. |
| 1940s | John Rock and Miriam Menkin fertilized four human eggs In Vitro |
| Mid 1900s | Scientists find cancer can be due to carcinogens, radiation, viruses or inherited. These can damage DNA. |
| 1960s | Mammograms develop to help detect breast cancer |
| 1970s | Scientists discover Oncogenes (cause uncontrolled cell growth) and Tumour Suppressor Genes (normally cause growth inhibition, when mutated lead to uncontrolled cell growth)
Medical imaging replaces explorative surgery. |
| 1978 | First baby, Louise Brown is born through In Vitro fertilization
Alex Lopata in Australia stimulates ovarian cycles by using clomiphene citrate |
| 1980s | IVF is no longer experimental, and is now an accepted reproductive technique. First Australian IVF baby delivered, Candice Elizabeth Reed. |
| 1982 | The first baby is born via Intrauterine Insemination. Media came into use for culturing embryos. |
| 1983 | Pregnancies achieved by using donor oocyte, donor embryo, and frozen embryo. In-vitro maturation is introduced. Oocytes retrieved through vaginal route. Robert Casper and team use hCG to aid the luteal phase during assisted ovarian cycles. |
| 1984 | First birth from a frozen embryo. First baby born through surrogacy. |
| 1986 | First pregnancy from sperm surgically retrieved.
First pregnancy via Zygote intrafallopian transfer (ZIFT) |
| Late 1900s | Surgery, chemotherapy and radiation combined as appropriate approaches to treat cancer. More targeted cancer treatments came about such as growth signal inhibitors, apoptosis inducing drugs and endogenous angioinhibitors (first one not approved til 2004). |
| 1992 | First pregnancy after Intracytoplasmic sperm injection (ICSI) |
| 1996 | Successful pregnancy after the use of ICSI from cryopreserved sperm |
| 2000s | Antiangiogenic Chemotherapy – combines angioinhibitors and chemotherapy (in clinical trials). Targeted treatments continue to advance for example with the use of nanotechnology and RNA profiling.
Success of human ovarian tissue transplant after frozen storage in 2000 |
| 2004 | First birth after orthotopic transplantation of crupreserved ovarian tissue. First single blastocyst transfer. |
| 2005 | The term “Oncofertility” is coined
Oncofertility consortium first created in North America. At this stage sperm banking is offered routinely to boys/men before cancer treatment. Women had no options given to them yet. |
| 2007 | National Institutes of Health Roadmap Grant for Biomedical Research given to help fund the Oncofertility consortium |
| 2010 | First pregnancy from vitrified blastocysts. |
| 2015 | Online registry launched in Australia to provide a patient history of their cancer treatment and reproductive journey to help survivors plan a family. |
Infertility
Infertility refers to an inability to conceive after having regular unprotected sex. Infertility can also refer to the biological inability of an individual to contribute to conception, or to a female who cannot carry a pregnancy to full term.
Sexual dysfunction is a common consequence of cancer treatment, affecting at least half of men and women treated for pelvic malignancies and over a quarter of people with other forms of cancer. Problems are usually linked to damage to nerves, blood vessels, and hormones that underlie normal sexual function. Innovations in cancer treatment such as robotic surgery or more targeted radiation therapy have not had the anticipated result of reducing sexual dysfunction [5]. Some new and effective cancer treatments, including aromatase inhibitors for breast cancer or chemoradiation for anal cancer also have very severe sexual morbidity. Men frequently have erectile dysfunction (ED) related to damage to the autonomic nervous system and/or reduced circulation of blood to the penis. Hormonal impairment of sexual function is less common. Women, in contrast, are able to overcome damage to autonomic nerves if genital tissues remain structurally intact and estrogenized. Female sexual dysfunction is frequently associated with sudden premature ovarian failure or the direct effects of radiation fibrosis or scar tissue which causes pain with sexual activity. [6]
In terms of oncofertility, cancers and more specifically cancer treatments, lead to infertility. Rather than the cancer itself causing infertility, it is the chemotherapy, targeted and biologic (immune) therapies, radiation therapy and surgery that contribute to fertility loss.
Infertility Causes in Cancer
Targeted drugs
These drugs attack cancer cells differently from standard chemotherapy drugs. Use of these medicines has increased a lot in recent years, but little is known about their effects on fertility or problems during pregnancy. Bevacizumab (Avastin), an angiogenesis inhibitor is one exception – studies have found that this drug can cause ovarian failure, and some women’s ovaries never recover. Another group of drugs that are of concern are targeted drugs called tyrosine kinase inhibitors (TKIs) such as Imatinib (Gleevec), which cause birth defects in lab animals. At this time the recommendation is that women/men talk to their doctors before becoming pregnant while taking TKIs. [7]
Radiation
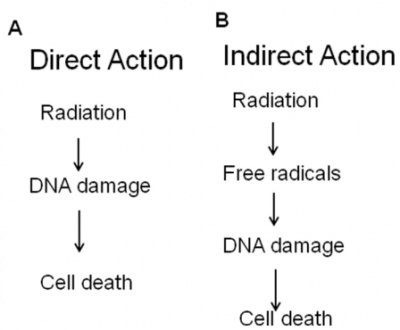
Radiation treatments use high-energy rays to kill cancer cells. Radiation works by damaging the DNA and ultimately the genes in cells. As depicted in the flow diagram, radiation can interact directly with DNA causing damage or indirectly by forming free radicals through ionisation of the water components within the cell which then damage the DNA causing double stranded or single stranded breaks. DNA controls how cells grow, divide and develop. When radiation damages the DNA of cells, the cells are no longer able to grow, divide or perform their ordinary function and over time, the cells die. Therefore, radiation can be used as a treatment for cancer. [8]
However, radiation can also compromise fertility such as in the following scenarios:
- Causing damage to a woman’s ovaries, especially when treating cancers in the abdominal or pelvic region. For a woman in this scenario the amount of radiation absorbed by the ovaries will determine if she becomes infertile. High doses can destroy some or all of the eggs in the ovaries and might cause infertility or early menopause. Even if the radiation is not directed right at the ovaries, the rays can bounce around inside the body and still cause damage the ovaries.
- When radiation is directed inside the vagina, the ovaries also absorb a high dose of radiation.
- Radiation to the uterus can cause scarring, which restricts flexibility and blood flow to the uterus. These problems can limit the growth and expansion of the uterus during pregnancy, and increase the risk of miscarriage, low-birth weight infants, and premature births.
- In both men and women, when radiation is directed at the brain it can affect the pituitary gland thus affecting fertility. The pituitary gland normally signals the ovaries and testis to make hormones, so interfering with these signals can affect ovulation and sperm production respectively. This may or may not affect fertility depending on the focus and dose of the radiation.
- Women who are still fertile when they start getting radiation treatments should avoid becoming pregnant until treatment is completed because radiation can also harm the foetus. [9]
- Men undergoing radiation treatment directed at their testis can also have their fertility compromised. Radiation at high doses kill spermatogonia i.e. undifferentiated male germ cells that produce sperm. Radiation is aimed directly at the testicles to treat some types of testicular cancer e.g,. seminoma, which is a type of cancer of the testicle, which involves radiation to the groin area, in close proximity to the remaining testicle.
- Radiation to treat a tumour in a males abdomen or pelvis can also affect the testis. [9] [10]
Surgery
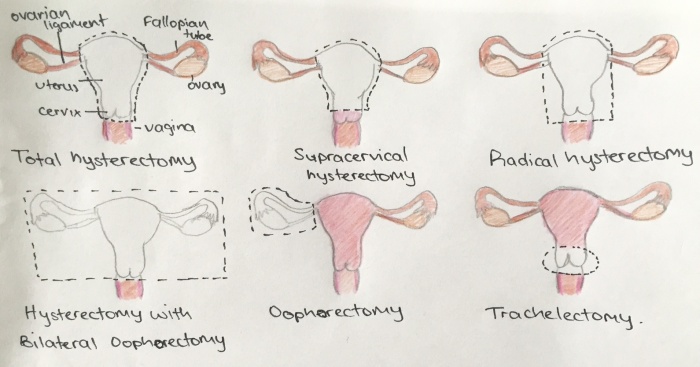
Surgery on certain parts of the reproductive system as a cancer treatment causes infertility, as described in text and depicted in the diagrams below.
Surgery in women
Hysterectomy: Removal of the uterus either through the vagina or through an incision made in the abdomen, such as in the case of endometrial cancer. When the uterus is removed the woman is no longer able to carry a child.
Oophorectomy: Refers to the surgical removal of the ovaries. This may occur in conjunction with a hysterectomy or alone such in the case of ovarian cancer. Without ovaries, a woman can’t get pregnant because she no longer has oocytes to mature and release at ovulation. [11].In some women with early stage ovarian or cervical cancer, surgeons try to save one ovary, if possible, to preserve oocytes, which might still allow a woman to conceive through artificial means. Keeping at least one ovary also preserves the hormones that prevent menopause symptoms like hot flashes and vaginal dryness [11].
Trachelectomy: Performed on women with small cervical cancers. In this technique the cervix is removed but the uterus is spared, allow a women to bear a child.
In some scenarios, surgery can cause scarring in the fallopian tubes. These scars may block the tubes and prevent oocytes from traveling through the fallopian tubes to be fertilised by the sperm and implant into the uterus.
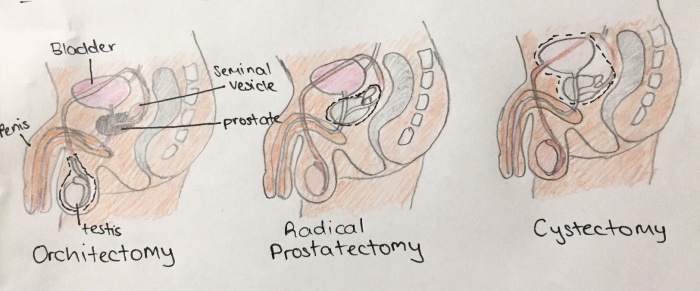
Surgery in men
Orchiectomy: Involves the removal of a testicle and is performed on males as a treatment for testicular cancer. As long as a man has one healthy testicle, he can continue to produce sperm after surgery. (Less than 5% of men develop cancer in both testicles.) However, some men with testicular cancer have poor fertility because the remaining testicle may carry some abnormalities. [12]
In metatstatic prostate cancer, orchiectomy may be performed on both testicles as a form of castration. This stops testosterone production in order to reduce the rate of growth of prostate cancer cells. This is referred to as a bilateral orchiectomy. These men cannot father children unless they have banked sperm before surgery. [13]
Radical prostatectomy: Removal of the prostate gland and seminal vesicles for men who have prostate cancer that has not spread beyond the gland. The prostate is removed through a cut in the abdomen or in the perineum (the area behind the testicles and in front of the anus), as a result the male can no longer produce semen. Surgery to remove the prostate also can damage the nerves that control erection, causing erectile dysfunction (ED). The patient can not conceive a child through sex as a result of both outcomes mentioned. [14] .
Cystectomy: Surgery to treat some bladder cancers is much like a radical prostatectomy, except the bladder is also removed along with the prostate and seminal vesicles. The testicles still make sperm, but in an invasive surgery the vas deferens may also be cut. Therefore, there is no ejaculate with sperm at sexual intercourse. [15] .
Surgery that interferes with erection and ejaculation: Some surgical treatments can also damage nerves that are required for an erection and to ejaculate sperm. They include removing lymph nodes in the pelvis during surgery for testicular cancer and some surgeries for colon cancers which may damage nerves in the process. Sometimes surgery can completely paralyze the prostate and seminal vesicles, which normally squeeze and relax to move the semen as a man’s climax begins. After these operations, a man still makes semen, but it doesn't come out of the penis at orgasm. Instead, it either shoots backward into his bladder which is called retrograde ejaculation or does not go anywhere. In cases of retrograde ejaculation, medicines can sometimes restore normal ejaculation of semen. The seminal vesicles contract, the internal valve at the bladder entrance closes, and semen is ejaculated from the penis at orgasm. For example in the USA, ephedrine sulfate is the most common medicine used to restore normal ejaculation. Because it does not help everyone and may only work for a few doses, ephedrine sulfate is usually prescribed only for the fertile week of the woman’s cycle. To improve this condition several methods are available. Fertility specialists can gather sperm from these men using several types of treatments including electrical stimulation of ejaculation or sperm aspiration surgery. [16] [17] .
Chemotherapy
Chemotherapy is the use of anti-cancer drugs on the body to destroy and kill cancer cells. The severity and types of anti-cancer drugs used is largely dependant on the type of cancer cells and degree of aggressiveness. Chemotherapy is also commonly used in conjunction with radiation therapy. [19] Chemotherapy is the treatment that will have the most profound impact on fertility. The damage that chemotherapy has to cells can stop eggs from being released, reduce the number of stored eggs, alter hormone release and cause amenorrhea. [20]
Chemotherapy drugs kill cells that are undergoing the process of dividing into 2 new cells – known as mitosis. Because cancer cells are rapidly dividing cells, and divide much more often then regular cells, they are more likely to be targeted and killed by the chemotherapy drugs. Each specific chemotherapy drug used kill cells in a different way, and the response is varied across the types of chemotherapy drugs. Some common mechanisms used to kill cancer cells are by damaging the part of the cell ‘s control centre that makes it divide – this often includes altering and/or disabling the checkpoint system in the cell cycle to ensure mitosis cannot complete. Other chemotherapy drugs work by interrupting the chemical processes involved in cell division. [21]
Chemotherapy's killing of healthy cells is what can lead to infertility in some patients as cells necessary for the production of offspring are destroyed in the process of also killing dangerous tumor cells.
What are Cancer Cells?
Healthy, normal cells do not become aggressive cancerous cells instantly or overnight. The transformation into a cancer cell is a gradual and ongoing change in which cells become their own functioning and active indestructible cell. [22]
Normal cells, in their functioning and division processes, respond to many signals and cellular checkpoints controlled by hundred of genes. These control mechanisms are in place to regulate the cells division, life span and growth – as well as preventing mutated or damaged cells from dividing and thus furthering damaged cells. When these checkpoints are not met chromosomes may be lost, rearranged, or copied too many times, often giving the cell further ability to develop mutations. [22]
Cancer cells develop mutations in the genes that regulate the control checkpoints, according to findings from the Cancer Genome Project, most cancer cells possess over 60 mutations to their genome. Normal cells do, however often have multiple mutations and are still able to function normally – almost as if the mutation did not exist. [23]
- Some mutations researches have discovered as common in developed cancer cells are the gene for the signaling protein Ras, as well as the tumor suppressor genes – the genes that suppress cell proliferation, such as the p53 gene.
| <html5media height="384" width="352">File:How Cancer Cells Divide.mp4</html5media> |
[24] This video describes how normal cells become cancer cells. It looks into tumor formation and how cancer cells come into existence. |
Cancer cells originate in tissues and as they continue to grow and divide, they diverge further and further away from cell regulations and thus normal cell functioning. As they grow, these cells become increasingly resistant to the controls that maintain normal tissue, and as such, they divide increasingly more rapidly than their progenitors and become less dependent on signals from other cells. Allowing these cells to divide uncontrollably.
- This uncontrolled cell division is problematic for the body because destructive and dangerous mutations cannot be prevented from spreading throughout the body like they would be in healthy cells.
Cancer cells, through their lack of functioning regulation and control genes, thus have the ability to evade programmed cell death – which normally would occur if a cell became abnormal or mutated. Making cancer cells ultimately immortal. They have the ability to escape destruction from the body’s defences and go on to develop their own blood supply and invade into other regions of the body – further spreading their cancerous capabilities. [23]
Cancer is uncontrolled cell growth – with the body being incapable of destroying these cells on its own accord. It is thus necessary to introduce external mechanisms, often vicious and aggressive, such as chemotherapy and radiation to kill these cells which can also cause infertility.
How Does Chemotherapy Work?
Chemotherapy used to treat cancer mainly uses drugs known as cytostatic, which aim to stop the uncontrollable dividing of cancer cells. There are a few different types of chemotherapy – all used aiming to achieve different outcomes in the treatment of cancer.
- Curative Chemotherapy → The most popular type of chemotherapy used, and often tried first in many cases. This model of chemotherapy aims to eliminate all cancer cells and removes the cancer completely and permanently.
- Adjuvant Chemotherapy → Is predominantly aimed at cancer cells that may be left in the body after surgery to remove a cancerous tumor has occurred, but the cells cannot be detected.
- Neoadjuvant Chemotherapy →This type of chemotherapy typically is done before surgery. Some cancerous tumors are too big and complex to operate on, so patients with these types of tumors will undergo neoadjuvant chemotherapy to shrink the tumor as much as possible before surgery.
- Palliative Chemotherapy → When it is no longer possible to remove all of the cancer cells from an individual, chemotherapy may still be used to reduce the extent of the symptoms, slow down the growth of the cancer and to avoid further complications. The use of palliative chemotherapy is often debated due to the side effects of chemotherapy being weighted against the benefit received from palliative chemotherapy, which in some cases can be almost none.[25]
| center|>File:Angiogenesis.mov</html5media> | File:Angiogenesis.mov [26]
A cancer tumor forms in a bed of healthy cells. The animation goes on to show how the tumor recruits blood vessels and how metastasis occurs. |
Methods of Administration
The most common method of administering chemotherapy is intravenously. Cytostatics can however be taken in a variety of forms, and often a combination of a range of different applications will be utilized for patients. Venous administration is useful, as the drug is in the bloodstream it is able to reach all parts of the body quicker - reaching cancer cells that may not have been detected in any examinations or tests.
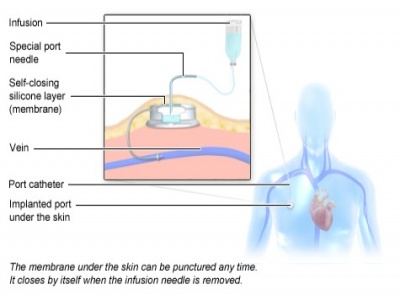
People receiving chemotherapy over a long extended time period, may also have a device known as a port set up. The port is a small device/container inserted under the skin that connects to a major vein and administers the drug.
Chemotherapy will operates in cycles based on various factors of the drug, the patient, and the response of the tutor. [27]
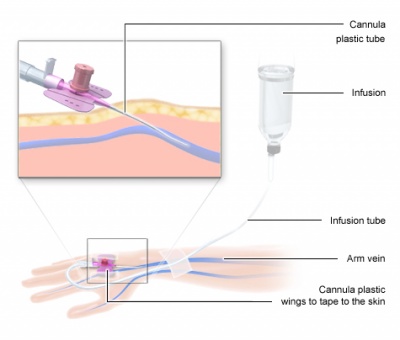
Types of Chemotherapy Drugs
There are various types of chemotherapy drugs, all used for different purposes and often specific to a certain type of cancer or certain type of patient. They are often divided into several groups based on how they work, their chemical structure, and their relationship to another drug. Cancer drugs are not however limited to one type of group, and often work in various ways and as such will belong to many groups. [30]
Groups of Chemotherapy Drugs
|
Type |
Description |
Examples |
| Alklyating Agents | Alkylating agents directly damage the DNA to prevent the cell from reproducing and dividing. The drugs work in all phases of the cell cycle and can be used to treat a wide variety of cancers, including leukemia, lymphoma, Hodgkin disease, multiple myeloma, and sarcoma, as well as cancers of the lung, breast, and ovary. [30]
They have 3 different mechansims of operation, however all function to achieve the same result - the disruption of the cell's DNA, preventing replication and thus causing cell death.
|
The different classes of drugs that alkylating agents are divided into include; [30]
|
| Antimetabolites | Antimetabolites interfere or disrupting the DNA and RNA growth by substituting out the normal building blocks of the DNA. Metabolite are the organic compounds that are synthesised, broken down in cells or recycled during metabolism. Examples include vitamins and amino acids, as well as urea - waste which is excreted (as urine).
Antimetabolites in a cell are mistaken for metabolites, and as such are processed in a manner analogous to the metabolite they resemble. The antimetabolite then prevents the cel from functioning. They are mainly used to treat cancers such as leukemias, cancers of the breast, ovary and the intestinal tract. [30] |
Some common antimetabolites include;
|
| Anthracyclines | Anthracyclines are anti-tumor antibiotics that interfere with the enzymes involved in DNA replication. The drugs work in all phases of the cell cycle and can be used for a wide variety of cancer types.
|
Examples of Anthracyclines include;
|
| Topoisomerase inhibitors | These type of drugs inhibit the function of the enzyme topoisomerase (I and II).
Topoisomerase are DNA enzymes that control the topology of coiled DNA during transcription and replication of genetic material. The two major types are Topo I and II. By inhibiting this enzyme the cell can no longer survive. [32] |
Currently there are only 2 approved Topoisomerase inhibitor drugs, namely topotecan and irinotecan, however there are many more currently undergoing clinical trials. |
| Mitotic inhibitors | Mitotic inhibitors are derived from natural products and often are plant alkaloids and other products. The drugs prevent mitosis in the M phase of the cell cycle, but also have the ability to damage the cell in all cycles by hindering enzyme's production of proteins needed for cell replication.
These drugs can be used for a variety of cancers, including breast, lung, myelomas, lymphomas, and leukemias, however the drugs have been known to cause nerve damage - which often limits the amount of usage, and hence the effectiveness of the drug. [30] |
Some examples of Mitotic Inhibitors include;
|
Side Effects of Chemotherapy
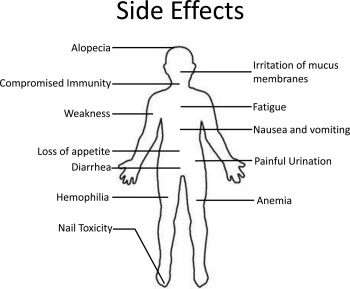
The common downside or obstacle of treating cancer cells effectively is current chemotherapy treatments operate with severe side effects. Cytostatic's function not only by killing the cancer cells, but healthy cells that may divide quickly – and it is this killing of healthy cells that cause often severe side effects.
It is very common for healthy, and often extremely necessary cells to become killed and attacked in the process. Some cells commonly destroyed while a patient undergoes chemotherapy treatment include hair cells, blood producing cells in bone marrow, cells lining mucous membranes including areas of the pharyngeal region and the digestive system.[34]
Some common side effects experienced can include; [35]
- Fatigue
- Pain
- Mouth and throat sores
- Diarrhea
- Nausea and vomiting
- Constipiation
- Blood disorders
- Nervous system effects
- - Tingling
- - Burning
- - Weakness/numbeness of hands/feet/limbs
- - Weak/achy muscles
- - Loss of balance
- - Trembling
- Changes in thinking/memory
- Appetite loss
- Hair loss
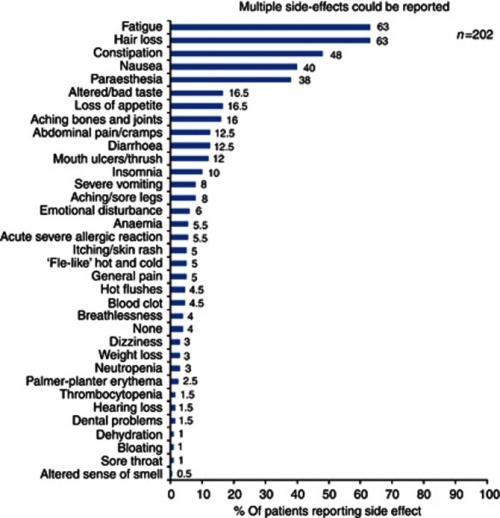
Chemotherapy largely does leave a patient exposed to a wide region of adverse effects and complications that may arise as a result of the compromised system. Patients have to undergo careful and systematic monitoring of their health, limiting any further issues as best as possible. It is a tough balance using drugs and therapy to kill cancer cells and tumors, while preserving the health of the patient, and retaining vital factors for the future, including fertility. Oncofertility largely focuses and addresses these issues.
The severity of chemotherapy side effects varies considerably from person to person, and depending on the type of drug used and the dose. Every case must be dealt with individually. Most side effects disappear when treatment stops, however some side effects can have a long term significance. Different chemotherapy drugs have different effects on the fertility of women depending on the duration of treatment and dosage.
Late Side Effects
Damage done any major organs, including the heart, kidneys, lungs or liver can also cause complications in the future for chemotherapy patients. Brain and neurological damage received can also open a pathway to many complications in the future for the patient. Nervous system changes can continue to develop after treatment, and have the ability of causing further problems many years down the track – these are known as late effects, and are often extremely complicated and problematic for cancer survivors. This can often be the case with fertility viability also. [35]
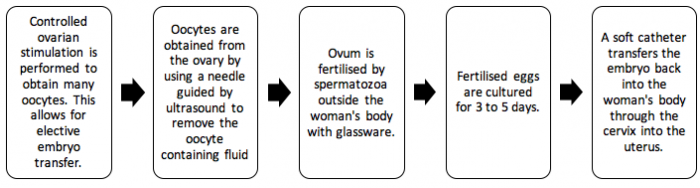
Fertility preservation
As discussed previously, cancer and cancer treatments are a cause of lost fertility. Fertility is important among many men and women and as a result there are various fertility preservation techniques being studied and implemented to help patients. Drugs are available to treat infertility however, the most common fertility preservation techniques involve the use of artificial reproductive technologies.
Fertility Drugs
Clomiphene or clomiphene citrate is recommended for women with irregular ovulation, the most common fertility side effect of cancer therapy. This drug reactivates the ovulation cycle by acting as an inhibitor of the estrogen receptors in the brain. This blocking effect tricks the body into bumping up levels of follicle-stimulating hormone (FSH) and luteinising hormone (LH). FSH causes the oocytes to mature in the ovaries and prepare them for release. LH triggers the release of one or more mature oocytes from the ovary follicles. [37]
Fertibella helps mothers to conceive their child in a natural. safe and easy way. Fertibella includes ingredients that are absolutely essential for a healthy and quick conception, such as folic acid, progesterone, selenium and iron. Furthermore, Studies have shown that women who followed this treatment were 33 percent more successful within one month than the control group [38].
Follistim is used to treat infertility in women with ovulation problems, but who do not have ovarian failure. Unlike the previous treatments that are to be administered orally, Follistim needs to be injected under the skin or into a muscle. The same product can be used to stimulate the production of sperm in men [38] .
Luteinising hormone (LH) and follicle-stimulating hormone (FSH) are types of gonadoptrophins. LH and FSH directly stimulate ovary to produce and mature oocytes. Gonadotrophins are normally used for women with polycystic ovary syndrome (PCOS) who have not responded to other drugs or for women undergoing IVF. Gonadotrophins are also used for women donating their eggs and for egg freezing procedures [38] . Follicle stimulating hormone, can be taken as a course of injections over about 12 days. The injections of FSH will be followed by a final injection of human chorionic gonadotrophin (hCG). hCG signals the release of an oocyte(s) after they have developed. hCG is structurally similar to LH and has the same physiological effect on the ovaries causing final maturation and oocyte release. [38]
Risks of fertility drugs
Fertility drugs, like any drugs , come with potential risks and side effects such as adverse drug reactions, multiple births, ovarian hyper-stimulation syndrome (OHSS), birth defects , ectopic pregnancy or ovarian cysts. [39] Multiple births also occurs in IVF. Mothers who have multiples births, have more complications during pregnancy such as gestational diabetes, hypertension and miscarriages. One way to reduce the risk of multiple births is to use single embryo transfer [39] .
OHSS – Ovarian Hyperstimulation Syndrome occurs when the ovaries become filled with fluid, which is then released into the uterus during ovulation. This release of fluid causes several complications including blood clots or kidney failure. This is due to taking mild fertility drugs such as clomiphene. One way to reduce this risk is to take a lower dose of fertility drugs and to monitor the fertility cycle closely [39] . Clomid (clomiphene) side effects are mild for most people. Because most of the estrogen receptors are blocked, this leads to some of side effects such as headaches, vaginal dryness and hot flashes. Most of the other side effects are caused by the ovaries becoming slightly enlarged [40]
Ectopic pregnancy is a serious condition when an embryo implants outside the uterus such as in the fallopian tube which is the most common site for this condition or in the ovary. It is important to note that the chances of an ectopic pregnancy would be higher in women having IVF, especially those with problems affecting their tubes. [39]
Fertility preservation in women
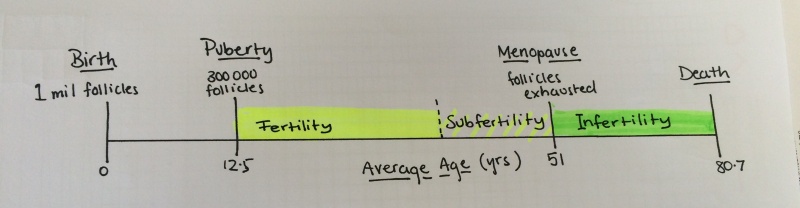
Fertility preservation options are difficult to implement in women as they have a limited number of follicles, are varying degrees of maturity based on a women's fertility timeline depicted above, leading to obstacles to preserving and maturing oocytes. The options available for females (some more successful to date than others) include:
|
Technique |
Description |
| Cryopreservation of immature oocytes | Experimental studies have shown that immature oocytes might freeze better due to the less developed and less fragile nature of immature oocytes, thus potentially handling the freezing and thawing processes better than mature oocytes. Immature oocytes can be collected at any time with no hormone stimulation needed. Because of this, researchers are also looking at whether immature oocytes can be harvested, matured in the lab, and then frozen. This keeps the woman from having to get hormone stimulation and then wait for oocytes to mature naturally in the women's body. Immature oocytes are removed through a needle guided through the vagina and into the ovary by the help of ultrasound. Immature oocytes are sucked into the needle and then frozen or matured and frozen. When the woman is ready, her immature oocytes are thawed, matured in the lab (if not done before freezing), fertilized, and then implanted in her uterus. This method is still considered to be experimental. Few reports have been published so far showing this method results in live births. [41] [42] [43] |
| Oocyte Freezing: | 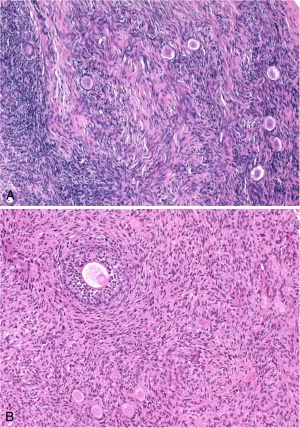 Ovarian tissue extracted after ovarian stimulation [44] Less is known about egg freezing than embryo freezing, but the methods and success rates have greatly improved in the past several years and it’s being done more often in the US. Some fertility centers have reported success rates much the same as using unfrozen eggs, especially in younger women. It is important to note that if you have frozen eggs, it’s important to stay in contact with the cryopreservation facility to be sure that any yearly storage fees are paid and your address is updated. Once a couple is ready to have a child, the frozen eggs are sent to their fertility specialist.[45] |
| Donor Oocytes: | Donated oocytes are used for fertilisation by a couple when the female is unable to use her own oocytes and does not have any cryopreserved ones. Women who wish to donate their oocytes can apply to egg donation programs where they undergo screening and interviews to ensure the woman is an appropriate donor. Once a donor is selected, they are matched to a recipient. She then begins administering injections to suppress her menstrual cycle in order to synchronise it with the recipient. Next, the donor injects gonadotropins to stimulate her ovaries which encourages many oocytes to maturity for retrieval. Meanwhile the recipient receives oestrogen and progesterone to prepare her endometrial lining for implantation. The donor receives hCG to trigger ovulation when ready and the oocytes are aspired through a needle. The oocytes can be fertilised with the partner’s sperm (or donor sperm) and the embryo transferred at approximately day 3. [46] |
| Embryo Freezing: | Also referred to as embryo cryopreservation is considered the better option for women who are married or in stable relationships. In this process the ovaries are stimulated to form mature oocytes with gonadotropins. The oocytes are aspirated and fertilized with their partner’s sperm through ICSI or can be fertilized in the lab through IVF (vitro fertilization) The process of collecting oocytes for embryo freezing is similar to oocyte freezing where under light anaesthetic, oocytes are collected during surgery, with the aid of an ultrasound guiding a needle to aspirate the oocytes. The oocytes are fertilized, then frozen and stored. Several embryos are stored to increase the chance for success. Most women start a cycle of hormone shots within 3 days of starting their menstrual cycle and continue them for 2 to 3 weeks until many oocytes are mature. [47] |
| Embryo Donation: | When couples undergo fertility treatment such as IVF normally multiple embryos are created. Not all the embryo's are utilised and therefore a decision needs to be made on what happens to the remaining embryos once the couple has completed their family. In this case couples have the option of donating the remaining embryo's to infertile couples who are unable to start a family. The couple undergoes screening and can then match with a recipient. Embryos can be thawed when they are ready for use and implanted into the recipient. [48] |
| Ovarian tissue storage: | In this technique, laparoscopy is used to take a biopsy of the ovary or to completely remove the ovary for preservation. The histological image demonstrates the ovarian tissue retrieved through this technique. The most follicles are found in the cortical tissue which is made into strips and cryopreserved. Once the patient is free from cancer, the thawed strips can be transplanted back into the patient’s ovary via grafting, or in the case of autotransplantation, the tissue is reimplanted into a different pelvic site, or extrapelvic site e.g. the forearm or abdominal wall. In the later case, pregnancy is only achieved via oocyte harvesting followed by IVF. Whole ovary transplantation is more difficult and requires immediate revascularisation. [49]
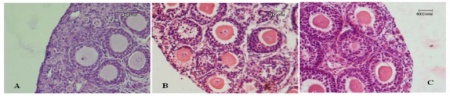 Representation of morphology of follicles after ovarian tissue vitrification [50] |
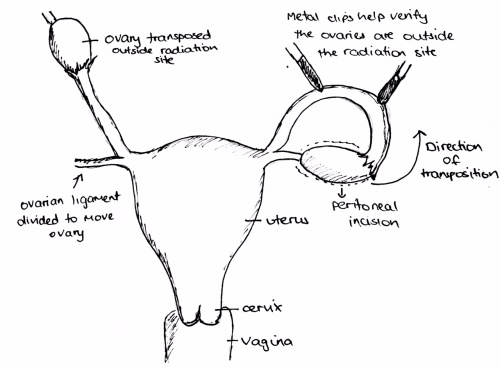
| Laparoscopic Ovarian Suspension Before Irradiation: | In many patients the use of radiotherapy is an essential part of treatment. The effect of radiotherapy on fertility depends on the location and extent of the disease as well as the dose of radiation being administered. It is especially detrimental to fertility in women with genitourinary or low intestinal tumours. To preserve fertility doctors may perform ovarian transposition by laparotomy to an extrapelvic site where radiation can be avoided. [49]
This involves moving the ovaries away from the target zone of radiation treatment such as pelvic radiation. Surgeons move the ovaries above and to the side of the central pelvic area. The rate of success for this procedure is measured by the percentage of women who regain their menstrual periods, not by being able to have a live birth. Typically, 50 % of the women start menstruation again. [51] [52] |
| Radical trachelectomy | Radial trachelectomy is considered as an option for women with cervical cancer who have very small and localised tumours. In this procedure, the cervix is removed but the uterus and the ovaries spared with uterus connecting to the upper part of the vagina. A special band or stitch is wrapped around the bottom of the uterus to act as the cervix and a small opening allows blood from the female’s period to flow out and sperm to enter the uterus to fertilize an oocyte. Trachelectomy is as successful in treating cervical cancer in women as radical hysterectomy. It is important to note that women can become pregnant after the surgery, but they are at risk of miscarriage and premature birth because the opening to the uterus may not close as strongly or tightly as before. [53] , [54] |
| Fertility-sparing surgery (Oopherectomy): | Fertility sparing surgery is used for young women with ovarian cancer in only one ovary. [55] This type of the cancer must be slow growing and less likely to spread all over the body such as borderline, low malignant potential, germ cell tumours, or stromal cell tumours. This typically includes grades 1 and some grade 2 epithelial ovarian cancers. In this situation, surgeons remove just one ovary with cancer and leave the healthy ovary and the uterus in place. Studies have shown that this does not affect long-term survival, and allows future fertility. The remaining ovary should be removed later if there is a risk of recurring cancer. This can be done after the woman has finished having children. [56] |
| Ovarian Suppressor agents: | This is also called "GnRH agonist treatment". As mentioned previously, chemotherapy treatment in cancer patients is involved in decreased fertility in women. In an analysis by Clowse et al. (2009) is was found that patients on Gonadotropin-releasing hormone (GnRH) agonists during chemotherapy had improved ovarian function and therefore an increased ability to get pregnant after chemotherapy. Therefore, the aim of this treatment is to shut down the ovaries during cancer treatment to help protect them from the damaging effects of treatment. This reduces the activity in the ovaries during treatment and will reduce the number of oocytes that are damaged, so women can have normal menstrual cycles after treatment. [57] GnRH agonist is a long acting hormonal drug that can be used to make a woman go into menopause for a short period of time. GnRH treatment is given each month for the duration of the cancer treatment. Studies explain that this method of treatment would help prolong fertility in some women, especially those 35 years old and younger, but results are not clear and more research is needed. If this treatment is used, it’s best done with a back-up method of preserving fertility such as embryo freezing. [58] |
| Adoption | Adoption involves parenting a non-biological child. Adoption takes place through public agencies or by a private arrangement or even by international public agencies. Most of these organisations do not exclude cancer survivors as potential parents but they need a letter from their doctor stating their healthy lifespan. Some agencies require a certain period of being off treatment and cancer-free before a cancer survivor can apply for adoption. Costs of adopting vary greatly from $4,000 (for a public agency) to $50,000 (some international adoptions) [59] |
| Surrogacy: | Surrogacy is an option for women who cannot carry a pregnancy, either because they no longer have a healthly uterus, or would be at high risk for a health problem if they got pregnant. There are 2 types of surrogate mothers:
A "gestational carrier" is a healthy female who receives the embryos as a result of fusion of the egg and sperm of the intended parents. The gestational carrier does not contribute her own egg to the embryo and has no genetic relationship to the baby. [60] A "traditional surrogate" is usually a woman who becomes pregnant through artificial Insemination or IVF with the sperm of the man in the couple who will raise the child. Through IVF the woman’s oocyte is fertilized with the man’s sperm in the lab and implanted in the surrogate. Therefore, the woman is the genetic mother of the baby. [60] |
Fertility preservation in men
Depending on the sexual maturity of a male, different fertility preservation options may be prescribed:
|
Technique |
Description |
| Sperm Banking | This is usually the first choice for males who are still capable of producing semen. However, this strategy isn't suitable for all patients as most males will not produce sperm suitable for cryopreservation until the age of about 12-13 [49]. This technique is a well-established method, easy and successful way for those who have gone through puberty to store sperm. This method is usually used for men before cancer treatment. Once the sperm bank obtains the sample, they test it to see how many sperm cells it contains (sperm count), what percentage of them are able to swim (motility), and how many have a normal shape (morphology). The sperm cells are then frozen and stored. [61]
Through cryopreservation sperm may be stored indefinitely within liquid nitrogen at a fee. The spermatozoa which has been collected can be used when the patient is ready to conceive for Intracytoplasmic Sperm Injection (ICSI), Artificial insemination or in the use of IVF. [49] |
| Electro-ejaculation |  Electroejaculator and ejaculatory probe [62] |
| Collecting sperm from urine | This is used when the the nerves that are necessary to ejaculate semen or close the valve at the bottom of the bladder are damaged during cancer surgery. In this case, a male still makes sperm but it does not come out of the penis at orgasm and it goes backward into the bladder which is know as "retrograde ejaculation". Therefore, fertility specialists collect sperm from the urine of these men and use them to fertilize their female partner’s oocytes in the lab through IVF. [64] |
|
Sperm donors |
Or Donor insemination (DI) is the process of conceiving a baby using donated sperm. Donated sperm can be used in intrauterine insemination (IUI) or IVF. Donor insemination is a very simple and low expense method for men who are infertile after cancer treatment to become a father. Most rganisations only collect sperm from young men with standard physical health, family health history, educational and emotional history, and conduct genetic testing. These sperm donors are also examined for sexually transmitted diseases, including HIV and hepatitis B and C viruses. Sperm donors might remain anonymous or can be in contact with the child later in life. [65] . |
| Testicular biopsy | This process is suitable for young boys who have not yet undergone puberty and therefore spermatogenesis. As a result, they do not have sperm available for cryopreservation for future utilisation. In this case, sample tissue from the testicles is collected with a thin needle during surgery and cryopreservation of immature testicular tissue which contains spermatogonial stem cells can be undertaken. Tissue is removed through biopsy prior to cancer treatment and then cryopreserved. It can then be used in the future after thawing, and allowing spermatogonial stem cells to proliferate and then autotransplanting the stem cells as is depicted in the diagram below or for In-Vitro maturation and sperm collection for IVF or ICSI. Results in this technique have been promising where spermatogonia in research has survived for future use and initiated spermatogenesis. [66] The thawed tissue can be implanted to the young men's testicles or stem cells might be taken out and injected into their testicles, which might allow them to make their own sperm. [67]
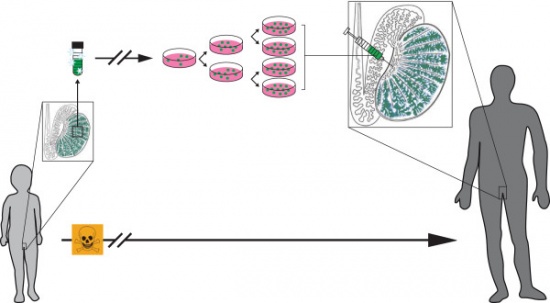 Testicular tissue cryopreservation and autotransplantation [68] |
| Testicular sperm extraction and epididymal sperm aspiration | Epididymal sperm aspiration and testicular sperm extraction (TESE) are experimental fertility options for collecting sperm in men who do not have mature sperm cells in their semen, after cancer treatments. In epididymal sperm aspiration, a tiny opening is made in the epididymis and sperm are taken out with a needle. In TESE, tiny pieces of tissue are removed from the testicles and checked for sperm cells. With either technique, if mature sperm are found, they can be used for IVF or ICSI or frozen for future use. [69] |
| Radiation shielding | Fertility can be protected in men and women who are getting radiation treatments that focus’ harmful rays on areas of the body. A lead barrier, or shield, can be placed over the patient's body to keep harmful radiations from affecting the pelvis, testicles and ovaries. Risk of harming the sperm for men getting radiation to the areas near testicles is uncertain, thus doctors suggest men avoid getting a woman pregnant during and for some weeks after radiation treatment. [9] |
| Adoption | See above in 'Fertility preservation in women' |
Artificial Insemination
Artificial insemination, also known as Intra-Uterine Insemination (IUI) involves the placing of sperm within the uterus with the use of a plastic catheter. In this procedure, fast-moving sperm are separated from sluggish or non-moving sperm. A patient is usually monitored for ovulation onset through hormone levels and ultrasound of the ovaries for the crucial time of sperm deposition. IUI can only begin if it has been confirmed that fallopian tubes are open and healthy. In this process, the purified sperm sample is put right into the woman’s uterus through a tiny, flexible tube. By increasing the number of sperm in the uterine cavity, and bypassing poor ejaculation the chance of pregnancy is increased by 2 to 3 fold. Furthermore, the chance for pregnancy is further enhanced by combining IUI with controlled ovarian hyper-stimulation. [70] [65] .
Depending on the particular fertility problem, patients may need to use fertility drugs alongside the IUI treatment. IUI with the use of fertility drugs is called "a stimulated cycle", because the drugs stimulate ovulation. If drugs are not used it's called "an unstimulated cycle", or natural cycle. It is important to note that IUI is more effective than intracervical insemination ICI. This is because by placing the sperm higher in the female reproductive tract, more sperm are likely to reach the oocyte in the fallopian tube and there is a higher chance of success for fertilisation. Intracervical insemination (ICI) is one of the oldest and most common artificial insemination procedures, dating back as far as the 1880s. [71] [72]
IUI is not effective for couples with:
- Tubal blockage or severe tubal damage
- Ovarian failure (menopause)
- Severe male factor infertility
- Severe endometriosis [73] .
In-Vitro Fertilisation
In-Vitro Fertilisation (IVF) refers to the fertilisation of an oocyte outside of the body within glass tubes. Other IVF related procedures include Intracytoplasmic Sperm Injection (ICSI), In Gamete Intra-Fallopian Transfer (GIFT), Zygote Intra-Fallopian Transfer (ZIFT).
When a women begins treatment she is first given FSH injections which she will self-administer. This stimulates the ovaries to produce an increased amount of follicles. When the appropriate amount of follicles are available, hCG is administered to trigger ovulation. The oocytes are collected with a needle guided by an ultrasound, and at this stage the partner will also need to supply sperm. The oocytes and sperm are then placed together in for fertilisation. After about 5 days, some embryos are placed in the uterus using a catheter via the cervix while others can be frozen for future use. [74]
The flow chart below lists the main steps involved in the IVF process:
Intracytoplasmic Sperm Injection
In Intracytoplasmic Sperm Injection (ICSI) a single sperm is chosen, and then inserted into an oocyte to fertilise it through the use of a needle. Once the oocyte is fertilised it is cultured and the blastocyst is transferred into the woman's uterus as in the case of IVF.[70] This technique is described further in the marmoset model below.
In Gamete Intra-Fallopian Transfer
In Gamete Intra-Fallopian Transfer (GIFT) involves placing mature oocytes and sperm in the fallopian tube unfertilised where they can undergo natural fertilisation in the natural location.[70]
Zygote Intra-Fallopian Transfer
Zygote Intra-Fallopian Transfer (ZIFT) combines both the standard IVF procedure and GIFT technique by fertilising the oocyte In-Vitro and then placing the embryo in the fallopian tube to take it's natural course towards implantation. [70]
Fertility preservation counselling
While there are various fertility preservation options available, it does not indicate whether they are being regularly implemented in medical practice. In a study by Reynolds et al. (2015) endocrinologists were surveyed with a 36 item survey to determine fertility preservation practice for cancer patients.
They found that 98% of endocrinologists who responded were counseling women diagnosed with cancer about the fertility options available. Cryopreservation of oocytes and embryos was the most common, offered by all providers, however managed differently. For example, in women with breast cancer, 86% of respondents used letrozole in the women positive for estrogen receptor breast cancer, when undergoing controlled ovarian stimulation (COS). This is essential in minimizing exposure to estrogen. Men were also managed differently among practioners, 86% were informed about sperm banking, and 22% were advised against it if they had already undergone rounds of chemotherapy.[75]
Through this study, it is evident that awareness in fertility preservation is increasing and improving. However, it is clear not all patients are advised in a universal manner.
Animal Models
Mice Model
Fertility preservation options for women are more difficult to address compared to men. This is because options such as ovarian cryopreservation and autotransplantation may reintroduce metastatic deposits and oocytes grown in vitro are generally low quality and difficult to preserve. Therefore, Xu et al. (2006) conducted a study using a 3D cultures system on tissue-engineered follicles from mice and found that they produced live, fertile offspring. Mice are used to model follicles and birth rates as it is unethical to conduct these experiments on humans. The 3D culture system was used as opposed to the current culture systems that use 2D substrates because they stimulate the appropriate physiological conditions more accurately.
In this study, immature follicles were isolated from prepubertal female F1 hybrid mice and and sperm obtained from CD1 male breeders mice. An alginate hydrogel matrix was then prepared as a scaffold to grow the follicles on, by encapsulating the follicle in an alginate bead. This culture system can be altered to mimic growth factors and hormones involved in normal oocyte maturation and provide structural support to maintain oocyte-somatic cell interactions. Following culture, follicles are transferred to maturation media that contains hCG and the oocytes then isolated. IVF was performed on CD1 pseudopregnant female mice and the zygotes transferred to mice oviducts.
Using this animal model, it was found that using a 3D system is more effective than 2D in stimulating physiological conditions. This system resulted in significant live birth rates thus providing an opportunity for ovarian follicle storage and maturation. [76]
Marmoset Model
Takahashi et al. (2014) conducted a study to model ICSI in the common marmoset, a small primate used as a model for biomedical translational research. The marmoset model is used as it has physiological similarity to humans and a short gestation period. ICSI is studied as a technique which avoids the need to obtain a large amount of high quality sperm from infertile males.
The female marmosets underwent ovarian stimulation and follicles were then stimulated using FSH and hCG. The animals underwent anaesthesia and follicles were aspired. Following this, oocytes underwent In Vitro maturation, and then IVF or ICSI. Sperm was collected from suitable male marmosets and divided into two groups for IVF or ICSI. In ICSI, an oocyte is help using a holding pipette and the zona pellucida drilled using piezo pulses. A single sperm is aspirated and injected into the cytoplasm as in image A below. In IVF, oocytes are transferred into a medium containing sperm and incubated. The blastocysts such as in image B below, from both techniques are cultured and transferred to surrogate mothers.
This study concluded that the embryos produced using this technique can develop to healthy blastocysts and neonates. The fertilisation rate was found to be higher in ICSI embryos compared to IVF but no significant developmental differences in rate were observed. [77]
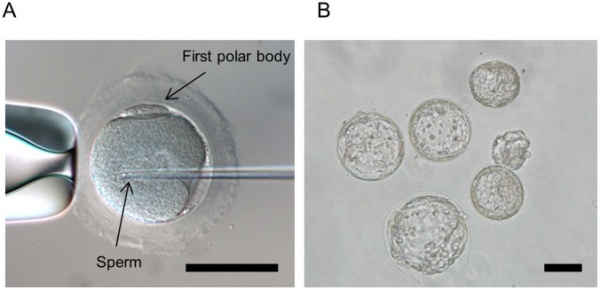
Oncofertility limitations
With oncofertility comes a list of limitations and risks:
- The amount of time available in which to employ fertility preservation methods in between cancer detection and cancer treatment.
- Only a limited amount of embryos will be available for selection from in the case of embryo storage.
- Gonadotropins leads to high oestrogen levels which is concerning in cancers such as oestrogen positive breast cancer.
- Ovarian tissue storage is an invasive procedure requiring general anaesthetic and has a 1 in 12 000 mortality rate.
- Ovarian tissue re-implantation carries the risk of a second surgery and re-implanting micro deposits of cancer
- Ovarian transplantation is limited by the small ovarian artery, short vascular pedicle length and in the case of failure a compromised ovary with no second chance of transplantation. [49]
- Multiple pregnancies as a result of IVF run the risk of maternal complications such as hypertension, diabetes, metal malpresentation and postpartum depression. The babies are at higher risk or prematurity, death and disabilities.
- IUI can not be used for tubal infertility as ovum can not reach uterine cavity. [78]
- Timely patient referral and coordination between specialists for patient care limits access to fertility options.
- Lack of knowledge especially in men to a fertility threat at the time of cancer diagnosis.
- Oocyte retrieval is difficult compared to sperm because it requires surgery, is limited in numbers and varies in maturity. [1]
- Ethical and moral issues surrounding the use of fertility preservation methods.
- Sperm Banking is not useful for fast-growing cancers, has high costs and many sperm banks do not accept samples from men who have HIV or hepatitis B (risk of infectious disease) [61]
- Testicular tissue removed from a boy with cancer before treatment could re-introduce cancer cells and cause disease again. [79]
- Couples donating embryos may not agree to have the same types of genetic testing as is usually done for egg or sperm donors, and they may not want to supply a detailed health history. [80]
- High cost of treatment.
- Many preservation methods are relatively new, still in research and have low success rates.
Glossary
Amenorrhea - an abnormal absence of menstruation
Biopsy- sample of tissue taken for examination
Blastocyst- Stage of embryo at approximately day 5 consisting of an outer (trophoblast) layer and inner (embryoblast) cell mass
COS- controlled ovarian stimulation- is a technique used in assisted reproduction involving the use of fertility medications to induce ovulation by multiple ovarian follicles.
Cytostatic- is a word some doctors and researchers use to describe the way some anti cancer drugs work
DI- Donor insemination- process of conceiving a baby using donated sperm
ED- erectile dysfunction- is the inability to develop and maintain an erection for satisfactory sexual intercourse or activity in the absence of an ejaculatory disorder such as premature ejaculation.
FSH - Follicle stimulating hormone- a hormone secreted by the anterior pituitary gland which promotes the formation of ova or sperm.
F1 hybrid mice- They are produced by crossing mice of two different inbred strains. Although they are heterozygous at all loci for which their parents have different alleles, they are similar to inbred strains in that they are genetically and phenotypically uniform
GIFT - In Gamete Intra-Fallopian Transfer-a method of assisting reproduction in cases of infertility that involves obtaining eggs from an ovary, mixing them with sperm, and inserting them into a fallopian tube by a laparoscope
GnRH - Gonadotropin releasing hormone- is a trophic peptide hormone responsible for the release of follicle-stimulating hormone (FSH) and luteinizing hormone (LH) from the anterior pituitary.
Hodgkin's disease- a malignant though often curable disease of lymphatic tissues typically causing painless enlargement of the lymph nodes, liver, and spleen
ICI- Intracervical insemination (ICI) is one of the oldest and most common artificial insemination procedures, dating back as far as the 1880s. Similar to intrauterine insemination (IUI), it involves placing sperm directly into the woman's reproductive tract to improve the chances of pregnancy
ICSI - Intracytoplasmic Sperm Injection- IVF technique used to treat male infertility and involves direct injection of one sperm into an oocyte
IUI - Intrauterine Insemination- is a fertility treatment that involves placing sperm inside a woman's uterus to facilitate fertilization
IVF - In Vitro Fertilisation- is a reproductive technology in which an egg is removed from a woman, joined with a sperm cell from a man in a test tube (in vitro)
LH - Luteinizing hormone- a hormone secreted by the anterior pituitary gland that stimulates ovulation in females and the synthesis of androgen in males
Myeloma- a malignant tumour of the bone marrow
Ovulation- release of eggs from the ovaries
OHSS- Ovarian Hyper-stimulation Syndrome- is a medical condition affecting the ovaries of some women who take fertility medication to stimulate egg growth
Pseudopregnant- a condition which resembles pregnancy: as a : pseudocyesis b : an anestrous state resembling pregnancy that occurs in various mammals usually after an infertile copulation.
sarcoma- a malignant tumour of connective or other non-epithelial tissue
TESE-testicular sperm extraction - experimental fertility options for collecting sperm in men who do not have mature sperm cells in their semen, after cancer treatments
ZIFT - Zygote Intra-Fallopian Transfer-) is an infertility treatment used when a blockage in the fallopian tubes prevents the normal binding of sperm to the egg. Egg cells are removed from a woman's ovaries, and in vitro fertilised.
References
- ↑ 1.0 1.1 <pubmed>20498666</pubmed>
- ↑ <pubmed>20740081</pubmed>
- ↑ <pubmed>24163793</pubmed>
- ↑ <pubmed>20811828</pubmed>
- ↑ <pubmed>26217165</pubmed>
- ↑ <pubmed>16304430</pubmed>
- ↑ American Cancer Society, Targeted Cancer Therapy, Retrieved 8 September, 2015 http://www.cancer.org/treatment/treatmentsandsideeffects/treatmenttypes/targetedtherapy/targeted-therapy-toc
- ↑ 8.0 8.1 <pubmed>22408567</pubmed>
- ↑ 9.0 9.1 9.2 <pubmed>8243379</pubmed>
- ↑ Medical Research Council, The influence of radiation on fertility in man, Radiobiology Unit, Harwell, Didcot, Oxon, retrieved on 8 September 2015, http://www.birpublications.org/doi/abs/10.1259/0007-1285-53-628-271
- ↑ 11.0 11.1 <pubmed>15302291</pubmed>
- ↑ American Cancer Society, Surgery for testicular cancer, Retrieved 8th September, 2015, http://www.cancer.org/cancer/testicularcancer/detailedguide/testicular-cancer-treating-surgery
- ↑ <pubmed>9761805</pubmed>
- ↑ American Cancer Society, Surgery for prostate cancer, Retrieved 8th September, 2015, http://www.cancer.org/cancer/prostatecancer/detailedguide/prostate-cancer-treating-surgery
- ↑ Cancer Council NSW, Surgery for invasive bladder cancer, Retrieved 8th September, 2015, http://www.cancercouncil.com.au/58014/b1000/bladder-cancer-10/surgery-for-invasive-bladder-cancer/
- ↑ <pubmed>4068047</pubmed>
- ↑ American Cancer Society, How cancer treatment can affect ejaculation, retrieved 8th September, 2015, http://www.cancer.org/treatment/treatmentsandsideeffects/physicalsideeffects/sexualsideeffectsinmen/sexualityfortheman/sexuality-for-men-with-cancer-ejaculation-and-treatment
- ↑ Rose, Jeanne, 'Chemotherapy Hastens Deterioration Near End of Life', 'Gazette Review' 31 July 2015, http://gazettereview.com/2015/07/chemotherapy-hastens-deterioration-near-end-of-life/.
- ↑ Cancer Council Australia, Cancer Council of Australia - About Cancer, June 2015, retrieved 10th September, 2015, http://www.cancer.org.au/about-cancer/treatment/chemotherapy.html
- ↑ Flinders Fertility, Oncofertility, retrieved 10th September, 2015, http://www.flindersfertility.com.au/Treatments-Services/Oncofertility-Booklet
- ↑ Cancer Research UK, How Chemotherapy Kills Cancer Cells, retrieved 10th September, 2015, http://www.cancerresearchuk.org/about-cancer/cancers-in-general/treatment/chemotherapy/about/how-chemotherapy-works
- ↑ 22.0 22.1 Science Museum, How Do Healthy Cells Become Cancerous? retrieved 10th September, 2015, http://www.sciencemuseum.org.uk/WhoAmI/FindOutMore/Yourbody/Whatiscancer/Whathappensincancer/Howdohealthycellsbecomecancerous.aspx
- ↑ 23.0 23.1 Scitable by Nature Education, Cell Division and Cancer, retrieved 10th September, 2015, http://www.nature.com/scitable/topicpage/cell-division-and-cancer-14046590
- ↑ Biodigital, 3D Medical Animation - What is Cancer?, October 14, 2008, retrieved 10th September, 2015, https://www.youtube.com/watch?v=LEpTTolebqo
- ↑ Better Health, Cancer Treatments - Chemotherapy, October 2012, retrieved 12th September 2015, http://www.betterhealth.vic.gov.au/bhcv2/bhcarticles.nsf/pages/Cancer_treatments_chemotherapy
- ↑ Dennis Liu, Bert Vogelstein, Satoshi Amagai, Drew Berry, Angiogenesis, HHMI BioInteractive, Retrieved on 9 October 2015, http://www.hhmi.org/biointeractive/angiogenesis
- ↑ Pubmed Health, How Does Chemotherapy Work? March 15, 2012, retrieved 12th September, 2015, http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0072611/
- ↑ Informed Health Online [Internet]. Cologne, Germany: Institute for Quality and Efficiency in Health Care (IQWiG); 2006- How does chemotherapy work? 2012 Mar 15, http://www.informedhealthonline.org/how-does-chemotherapy-work.2280.en.html.
- ↑ Informed Health Online [Internet]. Cologne, Germany: Institute for Quality and Efficiency in Health Care (IQWiG); 2006- How does chemotherapy work? 2012 Mar 15, http://www.informedhealthonline.org/how-does-chemotherapy-work.2280.en.html.
- ↑ 30.0 30.1 30.2 30.3 30.4 30.5 American Cancer Society, Types of Chemotherapy Drugs, June 2, 2015, retrieved 4th October, 2015, http://www.cancer.org/treatment/treatmentsandsideeffects/treatmenttypes/chemotherapy/chemotherapyprinciplesanin-depthdiscussionofthetechniquesanditsroleintreatment/chemotherapy-principles-types-of-chemo-drugs
- ↑ Emory University, A Closer Look at Mechanisms of Alkylating Agents, 6 May 2013, retrieved 4th October, 2015, http://www.cancerquest.org/genotoxic-chemotherapy-drugs.html
- ↑ <pubmed>10388070</pubmed>
- ↑ <pubmed>23243460</pubmed>
- ↑ American Cancer Society, Chemo Side Effects, retrieved 6th September, 2015, http://www.cancer.org/treatment/treatmentsandsideeffects/treatmenttypes/chemotherapy/understandingchemotherapyaguideforpatientsandfamilies/understanding-chemotherapy-chemo-side-effects
- ↑ 35.0 35.1 Cancer.net, Side Effects of Chemotherapy, retrieved 6th October, 2015, http://www.cancer.net/navigating-cancer-care/how-cancer-treated/chemotherapy/side-effects-chemotherapy
- ↑ <pubmed>23652312</pubmed>
- ↑ BabyCenter Australia Medical Advisory Board, Fertility drug: clomiphene citrate (clomifene, clomid)Retrieved 9th September, 2015, http://www.babycenter.com.au/a6186/fertility-drug-clomiphene-citrate-clomifene-clomid
- ↑ 38.0 38.1 38.2 38.3 BabyCenter Australia Medical Advisory Board, Fertility drugs for women, Retrieved 9th, September 2015, http://www.babycenter.com.au/a4090/fertility-drugs-for-women
- ↑ 39.0 39.1 39.2 39.3 Human Fertilisation and Embryology Authority, Risks of fertility treatment, Retrieved 9th, September 2015, http://www.hfea.gov.uk/fertility-treatment-risks.html#wrapper
- ↑ About health, Clomid (Clomiphene) Side Effects and Risks, Retrieved 9th September, 2015, http://infertility.about.com/od/clomid/tp/clomid_side_effects.htm
- ↑ <pubmed>11941534</pubmed>
- ↑ <pubmed>19778481</pubmed>
- ↑ <pubmed>21048443</pubmed>
- ↑ <pubmed>23510640</pubmed>
- ↑ 45.0 45.1 The Practice Committees of the American Society ,Mature Oocyte Cryopreservation: a guideline, retrieved 18 October, 2015, http://www.acog.org/Resources-And-Publications/Committee-Opinions/Committee-on-Gynecologic-Practice/Oocyte-Cryopreservation
- ↑ Centre for Human Reproduction, Egg Donation, 2015, retrieved 9th October, 2015, https://www.centerforhumanreprod.com/egg-donation/how-it-works/
- ↑ IVF Australia, Freezing Embryos, Retrieved on 7th October, 2015, http://ivf.com.au/fertility-treatment/ivf-treatment/frozen-embryo-transfer#success-rates-with-frozen-embryos
- ↑ IVF Australia, Embryo Donation, 2013, retrieved 9 October, 2015, http://ivf.com.au/fertility-treatment/donor-program/embryo-donation
- ↑ 49.0 49.1 49.2 49.3 49.4 <pubmed>24669162</pubmed>
- ↑ <pubmed>25250122</pubmed>
- ↑ <pubmed>16369371</pubmed>
- ↑ Togas Tulandi, MD, MHCM, Ovarian transposition before pelvic radiation Retrieved 6th October, 2015, http://www.uptodate.com/contents/ovarian-transposition-before-pelvic-radiation
- ↑ <pubmed>26095894</pubmed>
- ↑ <pubmed>26026821</pubmed>
- ↑ <pubmed>26255458</pubmed>
- ↑ <pubmed>25628110</pubmed>
- ↑ <pubmed>19281314</pubmed>
- ↑ <pubmed>17462639</pubmed>
- ↑ Resolve, The National Infertility Association, FAMILY BUILDING OPTIONS/Adoption retrieved 9th October, 2015, http://www.resolve.org/family-building-options/adoption/?referrer=https://www.google.com.au/
- ↑ 60.0 60.1 IVF Australia, Surrogacy, Retrieved 8th October, 2015, http://ivf.com.au/fertility-treatment/donor-program/surrogacy
- ↑ 61.0 61.1 Cancer Research UK, Sperm collection and storage (sperm banking), Retrieved 25th September, 2015, http://www.cancerresearchuk.org/about-cancer/coping-with-cancer/coping-physically/sex-sexuality-and-cancer/Sperm-collection-and-storage
- ↑ <pubmed>23774799</pubmed>
- ↑ <pubmed>6451670 </pubmed>
- ↑ Memorial Sloan Kettering, Cancer Center, Cancer and Fertility: Information for Men, retrieved 24th September, 2015, https://www.mskcc.org/cancer-care/patient-education/cancer-and-fertility-information-men
- ↑ 65.0 65.1 Human Fertilisation and Embryology Authority, What is donor insemination (DI) and how does it work? Retrieved 25th September, 2015 http://www.hfea.gov.uk/fertility-treatment-options-donor-insemination.html
- ↑ <pubmed>25593971</pubmed>
- ↑ <pubmed>21481372</pubmed>
- ↑ <pubmed>25157677</pubmed>
- ↑ <pubmed>8671323</pubmed>
- ↑ 70.0 70.1 70.2 70.3 70.4 <pubmed>23074488</pubmed>
- ↑ <pubmed>25637621</pubmed>
- ↑ <pubmed>8425628</pubmed>
- ↑ Advanced Fertility Center of Chicago, Artificial insemination for infertility, Intrauterine insemination - IUI Advanced Fertility Center of Chicago, Retrieved 20th October, 2015 http://www.advancedfertility.com/insem.htm
- ↑ IVF Australia, IVF treatment, retrieved 23rd October, 2015, http://ivf.com.au/fertility-treatment/ivf-treatment#the-ivf-treatment-process-step-by-step
- ↑ <pubmed>26010087</pubmed>
- ↑ <pubmed> 17518643</pubmed>
- ↑ 77.0 77.1 <pubmed>24751978</pubmed>
- ↑ <pubmed>23074488</pubmed>
- ↑ <pubmed>21481372</pubmed>
- ↑ United Kingdom-BabyCentre Medical Advisory Board ,Egg and embryo donation, retrieved 18th October, 2015 http://www.babycenter.com.au/a1014397/egg-and-embryo-donation
- 2015 Course: Week 2 Lecture 1 Lecture 2 Lab 1 | Week 3 Lecture 3 Lecture 4 Lab 2 | Week 4 Lecture 5 Lecture 6 Lab 3 | Week 5 Lecture 7 Lecture 8 Lab 4 | Week 6 Lecture 9 Lecture 10 Lab 5 | Week 7 Lecture 11 Lecture 12 Lab 6 | Week 8 Lecture 13 Lecture 14 Lab 7 | Week 9 Lecture 15 Lecture 16 Lab 8 | Week 10 Lecture 17 Lecture 18 Lab 9 | Week 11 Lecture 19 Lecture 20 Lab 10 | Week 12 Lecture 21 Lecture 22 Lab 11 | Week 13 Lecture 23 Lecture 24 Lab 12 | 2015 Projects: Three Person Embryos | Ovarian Hyper-stimulation Syndrome | Polycystic Ovarian Syndrome | Male Infertility | Oncofertility | Preimplantation Genetic Diagnosis | Students | Student Designed Quiz Questions | Moodle page
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, April 19) Embryology 2015 Group Project 5. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/2015_Group_Project_5
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G