2015 Group Project 4
| 2015 Student Projects | ||||
|---|---|---|---|---|
| 2015 Projects: Three Person Embryos | Ovarian Hyper-stimulation Syndrome | Polycystic Ovarian Syndrome | Male Infertility | Oncofertility | Preimplantation Genetic Diagnosis | Students | ||||
| 2015 Group Project Topic - Assisted Reproductive Technology | ||||
| This page is an undergraduate science embryology student and may contain inaccuracies in either description or acknowledgements. | ||||
Male Infertility
Infertility is defined as the inability to achieve a clinical pregnancy after 12 months of unprotected sexual intercourse [1]. Male infertility is the inability for a male to successfully impregnate a fertile female. It is an ever increasing issue that affects one in six Australian couples as reported in the Australian Government Department of Health, National Women's Health Policy. [2] Of these couples who are considered infertile, one in five experience problems that lie solely with the male.
Due to the growing issue, this page will discuss the most common causes, diagnostic tools, and treatments of male infertility, and ultimately provide a scope of the topic to allow for further research to improve our current understanding of what infertility entails.
Spermatogenesis and Fertility
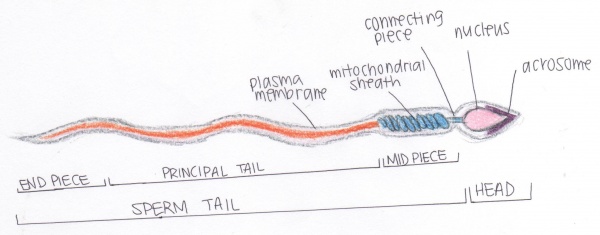
Structure of spermatozoa
The shape of spermatozoa are suitable for its transport to female gametes via the uterine tube. For this reason the nucleus of the spermatozoa is highly condensed, covered by an acrosome filled with enzymes for establishing contact to the female gamete. The enzyme within the acrosome degrades the zona pellucida of the oocyte (female gamete), allowing membrane fusion [3] . Spermatozoa also consists of a flagellum for progressive motility during its movement through the epididymal ducts and within the female reproductive organ. The motility is supported by the mitochondrial sheath found in the mid piece of the spermatozoa. [4]
Spermatogenesis
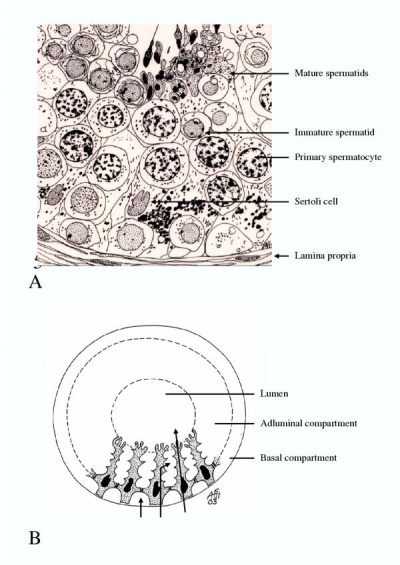
The complete process of male germ cell development is called spermatogenesis, male germ cells develop in the seminiferous tubules of the testes throughout life from puberty to old age. The product of spermatogenesis are mature male gametes called spermatozoa. There are three major stages in spermatogenesis:
1. Spermatogoniogenesis
2. Maturation of spermatocytes
3. Spermiogenesis (which is the cytodifferentiation of spermatids)
Spermatogoniogenesis is the process where spermatogonia multiplicate continuously in successive mitosis. However, the daughter cells will still be interconnected by cytoplasmic bridges and is only dissolved in advanced stages of spermatid development. The stage of meiosis is manifested through changes in the structure of the nucleus after the last spermatogonial division. Cells undergoing meiosis are called spermatocytes. As the process of meiosis comprises two divisions, cells before the first division are called primary spermatocytes and before the second division secondary spermatocytes [3]. During the prophase the duplication of DNA, the condensation of chromosomes, the pairing of homologuous chromosomes and crossing over take place. After division the germ cells become secondary spermatocytes. They do not undergo DNA-replication and divide quickly to the spermatids [3]. This results in four haploid cells, namely the spermatids. These differentiate into mature spermatids, a process called spermiogenesis which ends when the cells are released from the germinal epithelium. At this point, the free cells are called spermatozoa. During spermiogenesis three processes takes place; condensation of the nucleus, formation of acrosome cap filled with enzymes and the development of flagellum structures and their attachment to the head/mid piece of the developing spermatozoa.[3]
Physiology of fertility in Males
Normal reproductive functioning in males is controlled by gonadotropin releasing hormone (GnRH), androgens and gonadatropins. The correct metabolism and functioning of all three types of hormones is essential to the normal and efficient production of spermatazoa, as well as over all reproductive health. GnRH is synthesised and released by the hypothalamus, which stimulates the anterior pituitary to release two gonadatropins:
- Follicle stimulating hormone (FSH) responsible for spermatogenesis in the Sertoli cells
- Luteinizing hormone (LH) responsible for stimulating the release of androgens by the Leydig cells [5].
Testosterone, the primary androgen, is released into the testes and aids FSH by further promoting spermatogenesis. Furthermore, testosterone is vital to the normal development of many accessory reproductive organs, including the accessory glands. A negative feedback loop of testosterone and inhibin (secreted by Sertoli cells) acts on the anterior pituitary, either decreasing or stimulating the release of FSH and LH. [5]
Seminiferous Tubules
Seminiferous tubules special tubular structures in the testis that produce and release sperm continuously throughout the male life cycle. [6] The structure of the seminiferous tubules are complex stratified epithelium which is in invested by a capsule (tunica propria) of fibroelastic tissue and flattened fibroblasts. The spermatozoa within the tubules are not motile, it is believed that they are transported to efferent ducts by the continuous pushing motion provided by the production and release of new spermatozoa. It is also transported via the secretion of fluid from the tubules to the proximal region of the epididymal duct. [7] Problems in the seminiferous tubules can arise due to a blockage caused by ejaculatory duct obstruction. This can ultimately result in obstructive azoospermia, as described in "Male Infertility Disorders".
Male infertility disorders
Although infertility refers to the inability to conceive, there are numerous disorders that address particular reasons as to why this is the case. For males, the causes of infertility are endless and the most common factors have been discussed previously. Due to the range of aetiological factors, each one may affect a different aspect of the male's sperm including sperm count, morphology and motility rates. A fertile male is suggested to have normospermia [8] , in which the male's ejaculate contains normal sperm quality and quantity which are (based on the World Health Organisation (WHO)):
- Ejaculate volume of approximately 1.5 to 5 mL [9].
- Count of approximately 15 million to over 200 million spermatozoa per mL of ejaculate [9].
- Progressive motility of 32% or more spermatozoa [9].
- Normal morphology present in 4% of the ejaculate [9], in which normal form refers to the spermatozoa containing the 3 fundamental parts; a head, midpiece and tail.
Based on WHO's normal semen analysis, the specific types of male infertility disorders have been categorised accordingly.
Types of Male Infertility
| Type | Description |
|---|---|
| Oligospermia | Low spermatozoon count of less than 15 million sperm/mL of ejaculate [10] |
| Asthenospermia (asthenozoospermia) | Reduced motility of spermatozoa within the semen with a progressive motility of less than 20% [9] |
| Teratozoospermia | More than 95% of spermatozoa in the ejaculate has abnormal morphology [9] |
| Oligoasthenozoospermia | Combination of reduced motility of spermatozoa (asthenospermia) and low spermatozoa count (oligospermia) (referring to the statistics mentioned for each condition) |
| Obstructive Azoospermia | Absence of spermatozoa, despite normal spermatogenesis within the semen due to a blockage in the genital tract, obstructing the pathway for sperm to enter the penis from the testes [11] |
| Non-obstructive Azoospermia | Absence of spermatozoa within the semen due to the abnormal process or failure of spermatogenesis occurring, whereby sperm producing cells being damaged or destroyed [12] |
Causes of Infertility
Due to the increasing rates of male infertility worldwide, researchers have been focusing on aetiological factors for its treatment and prevention. There are numerous causes of male infertility, however, the most common causes are those that relate to the correct development and adequate supply of spermatozoa to result in pregnancy, or inefficient transport of spermatozoa. The three key parameters for assessing male infertility are spermatozoa count, viability and motility [13].
<html5media height="400" width="500">https://www.youtube.com/watch?v=QdIl1TjUvIQ</html5media>
Male Infertility [14]
Major Causes of Male Infertility
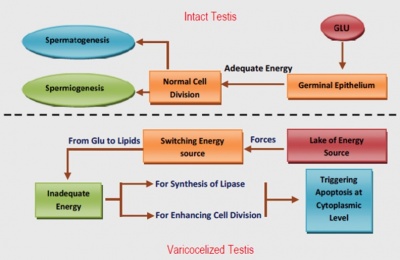
Varicocele
Varicocele is one of the leading causes of infertility in males and affects one third of individuals classified as infertile. Varicocele is the abnormal dilation of the internal spermatic veins and creamasteric veins from the panpiniform plexus as a result of back flow of blood. This downward flow of blood into the panpiniform plexus is due to the absence or presence of incomplete valves within the veins. [16] As previously mentioned, the three key markers of spermatozoa quality and of male infertility, spermatozoa viability, count and motility, are also heavily associated with varicocele. [17] Other causes of varicocele include an increase in programmed cell death (apoptosis), increased scrotal temperature of approximately 2.5 degrees Celcius and reduced androgen secretion leading to testosterone deprivation. [16] Testosterone is one of the hormones that play a major role in the correct physiological functioning of the male reproductive system. It is therefore evident that a deprivation of testosterone severely affects the rate of production of spermatozoa, their maturation as well as the male reproductive systems ability to effectively ejaculate semen (related to the development of accessory glands).
Male Reproductive Cancers
Male reproductive cancers, including prostate cancer and testicular cancer, have been shown to dramatically decrease the quality of semen prior to treatment, being comparable with that of infertile and subfertile men. [18] A link between testicular cancer and male infertility has been established by the identification of Testicular Dysgenesis Syndrome (TDS). The improper or abnormal development of the testicles associated with TDS has direct links to Sertoli and Leydig cell disfunction leading to failure of gonocyte maturation and therefore insufficient or low production of mature spermatozoa; one of the key indicators of male infertility. [19]. Furthermore, the presence of tumors in the male reproductive system have systemic effects including immunological and cytotoxic effects on the germinal epithelial leading to reduction in the quality of sperm produced and changes in the processes of spermatogenesis. [20] Finally, it has also been suggested that the fever and malnutrition associated with cancer may lead to alterations in spermatogenesis, a large decrease in spermatozoa concentration and evem azoospermia, the absence of motile spermatozoa. [21]
Chromosomal Abnormalities
Chromosomal Abnormalities are responsible for approximately 5% of all cases of male factor infertility and result in azoospermia (absence of spermatozoa) and oligozoospermia (low spermatozoa concentration). [22] Aneuploidy is the presence of an incorrect number of chromosomes and is the most common error of chromosomal abnormality resulting in infertility. [23] Klinefelter syndrome occurs in approximately 5% of severe oligozoospermic and 10% of azoospermic men and causes the cessation of spermatogenesis at the primary spermatocyte stage. [24] Another aneuploidy associated with male infertility is Y-chromosome microdeletions, present in 10-15% of azoospermic and 5-10% of severe oligozoospermic men, that can result in lack of spermatozoa in ejaculate (AZFa deletion), arrest of spermatogenesis at primary spermatocyte stage (AZFb deletion) and low concentration of spermatozoa (AZFc deletion). [25][26]
Damage to DNA
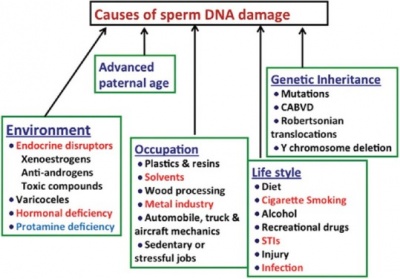
DNA damage in the germ cell population of males has been shown to be a contributing factor to many adverse clinical outcomes including poor semen quality, low fertilisation rates and impaired pre-implantation development; an outcome significant in the use of Assisted Reproductive Technologies when treating infertility. [28] The integrity of spermatzoa can be negatively impacted by deficits in the DNA repair pathways resulting in decrease in germ cell survival and the production of spermatozoa. [29] It has been demonstrated that common inherited variants within genes that encode enzymes utilised in the mismatch repair pathway have a negative relationship with the maintenance of genome integrity, meiotic recombination and even gametogenesis, therefore increasing the risk of DNA damage in spermatozoa and male infertility. [30] Finally, it has been demonstrated that an increase in age is associated with increased spermatozoa DNA damage resulting in a decline in semen volume, spermatozoa motility and morphology and over all semen quality. [31]
Lifestyle Factors
There are numerous lifestyle factors that are associated with a decrease in male fertility that often cause irreversible damage to processes in gametogenesis resulting in poor semen quality. Tobacco smoking has been seen to increase risk of male infertility by up to 30% due to the competitive binding of cadmium to DNA polymerase, replacing zinc and causing damage to the testes. [32] It was also suggested by the same study that excessive alcohol intake has an adverse affect on spermatozoa quality and chromosome number. [32] Another lifestyle factor that produces adverse clinical outcomes to male infertility is obesity and its association with hypogonadatropic hypogonadism; a condition characterised by a decrease in functional activity of the gonads (hormone production and therefore gametogenesis). [33] Studies conducted on animals demonstrates that a sensitivity to leptin in the hypothalamus as a result of obesity, decreases Kiss1 expression, therefore decreasing the release of gonadatropin releasing hormone (GnRH) and ultimately resulting in hypogonadatropic hypogonadism. [33] Studies have demonstrated vigorous physical exercise such as bicycle riding and horse riding, has been associated with urogenital disorders including erectile dysfunction, torsion of the spermatic cord and infertility. [34]
Immunological Infertility
Spermatogenesis commences at puberty after the body has developed a neonatal immune tolerance, therefore, without the necessary and correctly functioning physiological mechanisms such as the blood-testis barrier to separate the spermatozoa from the body's immune response, Sperm-reactive antibodies (SpAb) form and can be found attached to spermatozoa or within the semen. [35] [36] SpAb's have been found present in approximately 5-6% of infertile males.[35] [36] Various microbial pathogens can infect the testes via the circulating blood or the urogenital tract, which can result in orchitis (the inflammation of one or both testicles); characterised by the infiltration of leukocytes into the testes and damage of the seminiferous epithelium, ultimately contributing to male infertility. [37] The disruption of tight junctions within the epididymis, rete testes and even efferent ducts due to inflammation or trauma can result in the exposure of spermatozoa proteins to the immune system and therefore the formation of SpAb's. [35] The presence of SpAb's on the surface of spermatozoa contribute to infertility by causing agglutination in seminal plasma, reduced motility characterised by "shaking" of spermatozoa and even the reduced ability of spermatozoa to penetrate the cervical mucous of the female. [35]
Diagnosis
Male infertility is a widespread condition. There are different diagnostic techniques to detect male infertility, from medical histories, physical examinations to sophisticated tests such as blood tests, ultrasounds and semen analysis. Most cases, there are no obvious signs showing infertility. Sexual intercourse, erections and ejaculations occur usually without any difficulty; the quantity and sperm count of the ejaculated semen are not noticeable with the naked eye.
Physical examination
The physical examination focuses on the size and consistency of the genitals (testicles, epididymus and vas deferens) but also the overall body build. Noting the distribution of body hair and presence or absence of gynecomastia, which is the enlargement of male breasts due to the imbalance of hormones or hormone therapy. In some cases, by examining the size and consistency of the scrotum it is possible to palpate whether or not the epididymis may have hardened from a possible inflammation. Other cases may suggest obstruction within the ducts,this is determined by observing and examining the prostate size and consistency, checking for the presence of cysts or enlarged seminal vesicles.[13] Varicoceles are the most common abnormal finding in infertile men, typically diagnosed by physical examination of Valsalca manoeuvre. It is performed by forceful attempts of exhalation against closed airways by closing one's mouth and pinching their nose while pressing out. This strain increases their intrathoracic pressure and causes the venous return to the heart to decrease and increases the peripheral venous pressure.[38]
Varicoceles can be diagnosed by conducting Valsalva manoeuvre. [38] [17]
Classifications of Valsalva manoeuvre
| Grade | Description |
|---|---|
| Grade 1 | Varicocele (vein dilatation) only palpable during Valsalva manoeuvre on physical exam
|
| Grade 2 | Varicocele palpable on physical exam without Valsalva manoeuvre
|
| Grade 3 | Varicocele visible through the scrotal skin without performing Valsalva manoeuvre
|
Semen Analysis
Although the semen parameters of fertile men can vary, semen analysis is an initial and crucial laboratory test when determining male infertility.[13] Every 2 to 4 weeks, at least two semen samples should be collected. 2 to 4 days prior to the collection is the abstinence period; this is important as it will increase the sperm destiny by 25%. Semen samples are obtained by masturbation or by using a latex free, spermicide free condom during intercourse.
Testicular Colour Doppler Ultrasound
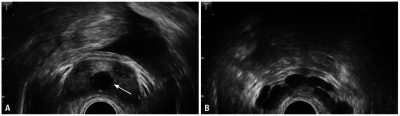
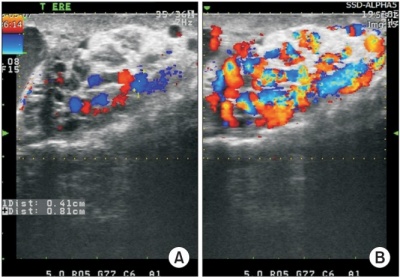 Color Doppler ultrasonography of varicocele. Maximal venous diameters in the pampiniform plexus were measured during resting (A) and during a Valsalva maneuver (B) in the standing position. [39] |
High resolution color Doppler ultrasound is a noninvasive means of simultaneously imaging and evaluating the blood flow to the testes in infertile men. An ultrasound machine that has a Doppler mode can see blood reverse direction in a varicocele with a Valsalva, increasing the sensitivity of the examination [39]. It is not generally performed as a routine examination, however physical examination may miss intrascrotal abnormalities readily detected by dopple ultrasound. Non-palpable intrascrotal abnormalities includes testicular and epididymal lesions and tumour.[38] It allows the identification of minimal ectasia of the scrotal veins and minimal retrograde venous flow. Ultrasonography and particularly Colour DopplerUltrasound appear to be the most reliable and practical methods for diagnosing subclinical varicocele. Colour Doppler Ultrasound can be used to measure the size of the pampiniform plexus and blood flow parameters of the spermatic vein. However, the reliability of the Colour Doppler Ultrasound to diagnose varicoceles remains controversial; the diagnostic criteria remain poorly defined, with considerable variation between investigators and researchers. Reflux is an important criterion for the diagnosis of varicocele. The change in color is subjective and unreliable for the diagnosis of reflux in the Colour Doppler Ultrasound examination and should be quantified with spectral Doppler analysis [39]. |
Risk Factors and Prevention
Below are a list of the most common risk factors that are associated with male infertility. Preventative measures involve the sufferer of male infertility to address these listed issues and possibly seek therapeutic services to maintain a healthy lifestyle. As infertility may be idiopathic (unknown cause), these risk factors should still be prevented to ensure the condition does not worsen.
Risk Factors of Male Infertility
| Risk Factors | Description |
|---|---|
| Smoking | Semen quality is significantly affected by cigarette smoke. Light smoking has been associated with asthenozoospermia and heavy smoking has been associated with asthenozoospermia, teratozoospermia and oligozoospermia. [40] |
| Alcohol Consumption | Alcohol abuse in men has been associated with impaired production of testosterone and therefore infertility. [41] One study demonstrated that a typical weekly alcohol consumption of ~40 units resulted in a 33% decrease is spermatozoa concentration. [42] Alcohol abuse adversely affects spermatozoa morphology and production ultimately causing asthenozoospermia and therefore reducing the quality of semen. [41] |
| Overweight/Obesity | An increase in waist circumference is associated with impaired semen parameters in infertile men. [43] A high body mass index (BMI) is negatively associated with normal spermatozoa morphology, spermatozoa concentration and motility, total spermatozoa count and percentage of vital spermatozoa, therefore negatively affecting male fertility. [44] |
| Psychiatric Considerations | Stress has been demonstrated to have a negative affect on fertility, reducing testosterone levels and spermatogenesis. [45] |
| Physical trauma | It has been demonstrated that physical traumas and vigorous exercise (often a combination of the two) can result in adverse urogenital disorders such as torsion of the spermatic cord, penile thrombosis, hematuria and infertility. [34] |
Treatments
Current treatments for male infertility aim to eliminate the causative factors mentioned above. These may involve improving the male's fertility using drug therapies or surgical procedures, however many assisted reproductive technologies have been introduced and have proven successful. Both methods of treatment have shown evidence of efficacy, thus having great implications on infertile couples worldwide.
Non-surgical Treatments
In order to effectively treat male infertility, it is imperative to correctly identify the specific cause and contributing factors. Currently, the different treatment strategies used or investigated tend to the specific aetiological factors for male infertility. Apart from theoretically allowing natural conception, these treatments also have an implication on the assisted reproductive technologies (ARTs) that are currently available.
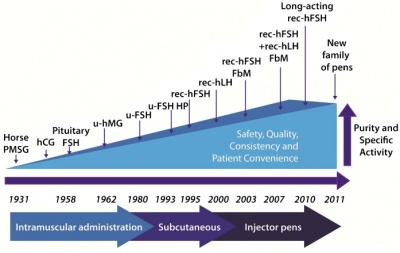
Injectable Hormones & Fertility Drugs
Hormonal imbalance is a non-obstructive cause for male infertility. The efficiency of spermatogenesis depends on stimulation and regulation mainly by gonadotropins, GnRH and testosterone, without which may cause infertility. Males that have a deficiency in these hormones are being targeted by research involving injectable hormones such as human chorionic gonadotropin (hCG) and human menopausal gonadotropin (hMG), and Clomiphene citrate, a fertility drug. hCG and hMG are gonadotropins that are used to treat male hypogonadotropic hypogonadism (MHH), a condition associated with infertility causing an underproduction of sperm or testosterone, or both [47]. These gonadotropins have been utilised in infertile males to stimulate the synthesis of testosterone and sperm directly, bypassing the pituitary gland that normally releases gonadoptropins LH and FSH. LH triggers Leydig cells to release testosterone, and FSH plays a vital role in spermatogenesis maintenance as it promotes Sertoli cell maturation [48]. The associated image demonstrates the development and availability of gonadotropins for commercial use.
Additionally, clomiphene citrate also increases secretion of GnRH from the hypothalamus, and FSH and LH from the pituitary gland by blocking feedback inhibition of serum estradiol [48]. Normally, males have more testosterone levels than estrogen however those with MHH and consequent infertility, may have the opposite [49]. This was investigated in a study conducted in 2013 by Hussein et al. showing that hCG, hMG and clomiphene citrate are suitable treatments particularly for azoospermia, increasing levels of FSH, LH and total testosterone [48]. Therefore the administration of these substances may correct abnormal hormone levels that contribute to male infertility, thus stimulates spermatogenesis to increase spermatozoa count, motility and viability.
Antioxidants
There has been increasing evidence that infertility may be directly linked to oxidative stress, thus various antioxidants have been experimented with to determine their efficacy as a treatment. Reactive oxygen species (ROS) formed during oxidation plays a vital role in sperm function, particularly in capacitation, acrosome reaction, hyperactivation and sperm-oocyte fusion [50]. In low concentrations, ROS are essential for the synthesis of energy, and contribute to signal transduction pathways within the cell. Usually ROS levels are regulated by natural antioxidants within the seminal plasma [50]. However an influx of ROS and/or a deficiency in antioxidants due to abnormal sperm or environmental stress, can lead to oxidative stress. Spermatozoal cell membranes contain high amounts of polyunsaturated fatty acids that consist of several electron-containing double bonds. The electrons of these fatty acids contribute to the formation of ROS and oxidative stress, thus causing a disruption in the flexibility of the spermatozoal membrane and diminishing the motility and sustainability of sperm [51]. This may result in sperm membrane lipid peroxidation, DNA fragmentation, and apoptosis [50].
The following are a few common antioxidants that have been proven to treat oxidative stress, and hence improves male fertility.
1. Carotenoids
- Naturally occurring pigments produced by plants, algae, and photosynthetic bacteria [52].
- Subtypes are divided into 2 different categories based on their chemical composition including carotenes that contain oxygen, and xanthophylls that only contain hydrocarbons [52]
- Main source of carotenoids in the human diet are from fruits and vegetables as they give them their yellow, red and orange pigments.
- Have been suggested as daily supplements for the human body, and act as treatments for various cancers and possibly infertility disorders [53].
- Their antioxidant activity of is performed by quenching (deactivating) singlet oxygen that is formed during photosnythesis by plants.
Two common carotenoids that have been strongly advised as treatments for male infertility include lycopenes and Astaxanthin, described below.
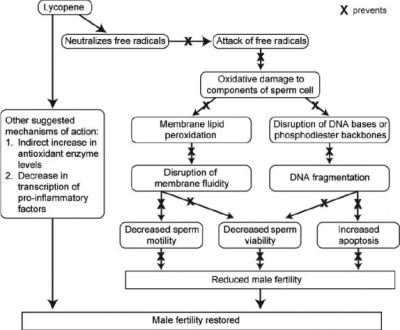
Lycopenes
- Type of carotene carotenoid that is found in various fruits and vegetables such as tomatoes and watermelon.
- Possesses strong antioxidant properties as it is one of the most effective quenchers of singlet oxygen [54].
- Have a role in neutralizing ROS and hindering their activity, achieved by their ability to donate an electron to free radicals [51].
- Inhibit lipid peroxidation allowing for spermatozoal membranes to be retained and protected from further damage.
- Suggested to increase natural antioxidant enzymes indirectly, and also decrease the production of pro-inflammatory agents.
Astaxanthin
- Keto-carotenoid produced naturally from the microalgae Hematococcus pluvialis [55]. it has been
- Suggested as an effective treatment and supplement for male factor infertility due to its higher antioxidant activity in comparison to vitamin E, a fat solube antioxidant found in soybean and margarine.
- An experimental trial to test Astaxanthin’s influence on sperm function was carried out in 2005 in 27 infertile men [56]. It was found that Astaxanthin allowed for increased motility concentration, improved sperm morphology and motility, and a decrease in ROS and Inhibin B (a regulator of spermatogenesis) levels.
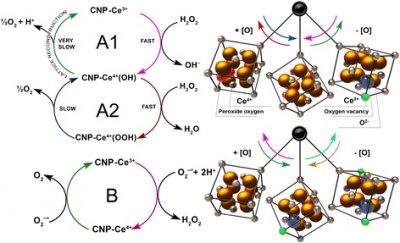
2. Cerium dioxide nanoparticles (CNPs)
- Cerium dioxide nanoparticles have been used extensively in the health care industry as potential pharmacological agents to treat various conditions from cancer to male infertility.
- They are formed by cerium combining to oxygen obtaining a strong crystalline structure [58].
- CNPs have the ability to interchange Ce 3+ and Ce 4+ ions that are present on its surface, leading to defects in oxygen within its crystal lattice structure. These regions on the surface of CNPs are ‘reactive sites’ to attract free radicals [57].
- A research team experimented on male rats to observe CNP effects on male health and infertility, providing further evidence that oxidative stress plays a key role in preventing proper spermatogenesis [57]. Therefore, the electronic structure of CNPs, and thus its antioxidant properties make this material a promising therapeutic for male infertility caused or affected by oxidative stress.
3. Vitamin E
- A fat – soluble antioxidant that exists in 8 chemical forms of different biological activity.
- The only form of vitamin E required by the human body is alpha-tocopherol [59], found in various foods such as wheat germ oil, sunflower seeds and oil, and almonds [60].
- The recommended dietary allowance (RDA) of vitamin E is 15 mg with an adult maximum of 1000 mg [60].
- Due to the ability for vitamin E to prevent the peroxidation of PUFA, it has extremely positive implications on infertile men as spermatozoa have high levels of these compounds.
- From previous studies, vitamin E (alpha – tocopherol) levels decreased to 66.54% and 66.04% in oligospermic and azoospermic males respectively compared to fertile men [61]. Therefore there is a positive association between alpha – tocopherol levels and sperm count and motility .
4. Vitamin C
- A water-soluble antioxidant that neutralizes free radicals and also prevents ROS synthesis[62].
- The human body does not produce or store vitamin C, so daily intakes of vitamin C – containing foods are required to maintain its levels internally.The RDA for vitamin C in male adults is 90mg/day [62].
- Foods with the highest vitamin C content include citrus fruits (oranges), kiwi fruit, broccoli and cauliflower.
- A study published in March 2015 demonstrated that infertile men administered with vitamin C had a significantly better sperm motility rate and morphology. Although it had little/no effect on sperm count, it is still a well recognizable and effective treatment for male infertility [63].
Traditional Chinese Medicine
More recently discovered treatments for male infertility involve the hollistic principles of traditional Chinese medicine (TCM). Disregarding the conventional medicines more commonly prescribed in today’s society, the effects of Chinese herbal therapy, massage and acupuncture, have been suggested to improve sperm motility and viability of infertile males [64]. Acupuncture and massage has been proven to alleviate stress, increase blood flow to reproductive organs, regulate the immune system, and improve dysfunctions in male infertility [64].
Additionally, Chinese herbal medicines have been widely used in experiments to prove their beneficial effects on treating infertility. The following are examples of a few herbal therapies that have been investigated.
Examples of Chinese Herbal Therapies
| Herb | Evidence |
|---|---|
| Yi Kang Decoction | 100 immune infertile males treated with this herb had greater sperm motility, agglutination, and overall increased pregnancy rates in comparison to prednisone, a steroid that reduces sperm antibody levels [65] |
| Hu Zhang Dan Shen Yin | 60 treated infertile men showed a higher antisperm antibody reversing ratio than prednisone, thus allows for greater sperm production [66] |
| Zhibai Dihuang | This herb was used to treat 80 cases of male immune infertility in the form of a pill, resulting in increased sperm motility and viability [67] |
Surgical Treatments
Varicocelectomy
Varicocele repair can be performed by either percutaneous radiographic embolization or surgery to correct male infertility [17]. The desired outcome of these procedures is to lower the temperature of the scrotum for normal spermatogenesis to occur.
Percutaneous radiographic embolization involves the catheterization of the internal spermatic vein and its occlusion using a sclerosant (injectable irritant) or solid embolic devices such as stainless steel coils [68]. The administration of the sclerosant and solid embolic devices are given at the level of the inguinal crease and ligament respectively to prevent the backflow of blood into the pampiniform plexus. This method is much less invasive than surgical procedures and has very high success rates, and low recurrence rates [68].
As for the surgical approach, these methods are far more invasive but variable in terms of success rates and recurrence. It is important to note that all of these varicocele repair methods, surgery and embolisation, aim to impede increasing temperature of the scrotum caused by the pampiniform plexus.
Surgical Approach to Varicocele Repair
| Surgical Method of Varicocele Repair | Description |
|---|---|
| Inguinal Surgery |
|
| Subinguinal Surgery |
|
| Retroperitoneal Surgery |
|
| Laparoscopic Varicocelectomy |
|
The short video provided describes the term 'varicocele' and outlines the process of a varicocelectomy using an inguinal and subinguinal incision.
<html5media height="400" width="500">https://www.youtube.com/watch?v=3crlbOiCO48</html5media>
Varicocelectomy [70]
Ejaculatory Duct Resection
Ejaculatory duct obstruction is a rare cause for infertile men. It is usually found in cases of severe oligospermia and azoospermia indicated by a low ejaculate volume and pH, and little or no fructose in seminal plasma [71]. To correct this in the minority of infertility patients, transurethral resection of ejaculatory ducts (TURED) can be performed. Firstly, a digital rectal exam will show a midline cystic lesion or dilated ejaculatory duct. The duct is instilled with methylene blue dye to open the duct and confirm the resection is in the system [71]. A study by Yurdakul, Gokce, Kilic and Piskin, concluded that 11 out of 12 azoospermic males with complete ejaculatory duct obstruction who received TURED had sperm in their ejaculation [72].
Male Infertility Treatments with Assisted Reproductive Technologies (ARTs)
It is known that males with fertility problems have little/no chance of conceiving a child with a woman. To address this issue many ARTs have been developed to allow for a successful pregnancy, which all involve the process of sperm retrieval. The following video demonstrates some common techniques that have been used to successfully retrieve sperm.
<html5media height="400" width="500">https://www.youtube.com/watch?v=c_nK2ZS_Mr0</html5media>
Sperm Retrieval Procedures [73]
Intrauterine Insemination (IUI)
Intrauterine insemination (IUI) is a simple procedure performed by a medical practitioner where washed sperm is injected directly into the uterus with a catheter. This allows the sperm to get as close to the egg as possible, increasing the chances of reaching it. This method is known as in vivo fertilisation as it is performed within the body of the female. It has been shown that if the woman rests for up to 15 minutes after insemination the chance of pregnancy is greater than if they are mobilised immediately after the procedure.[74] The optimal conditions for an IUI include; the female being less than age 30, the male having a total motile sperm count of more than 5 million per mL. A likely pregnancy will result from a cycle that produces two eggs of 16 mm or more and an oestrogen concentration of 500 pg/mL at the time of the procedure [75].
<html5media height="400" width="500">https://www.youtube.com/watch?v=ENrx7o_z9Ng</html5media>
IUI Treatment Procedure [76]
In Vitro Fertilisation (IVF)
Theoretically, all that is required for in vitro fertilisation is to combine the contents of a woman’s fallopian tubes and sperm, followed by re-inserting this mixture into the uterus. In practice, however, this process would be an oversimplification and not particularly successful. There are several major steps in the procedure that are necessary for pregnancy. The first step is hyperstimulation of the ovaries. The purpose of this step is to produce several oocytes to make sure there are enough suitable candidates for the procedure. This is achieved by injecting a GnRH antagonist and gonadotropins into the female. Careful monitoring of the concentrations of these hormones is essential for the safety and well-being of the patient and for the successful removal of adequate follicles [77]. Next, after the follicles have reached an appropriate level of development, final maturation induction is performed, typically by injection of hCG and GnRH agonist. This step is to replace the natural surge of LH that would normally mature the ovarian follicles.
Once the follicles have matured, they are retrieved from the ovaries by a process known as transvaginal oocyte retrieval [78]. This involves a needle guided by ultra-sound to pierce the vaginal walls, reaching the ovaries and finally aspiration of the mature oocytes and follicular fluid. Typically, 10-30 oocytes are removed under general anaesthesia [77]. The oocytes are then inspected and only those with the highest chance of successful pregnancy are chosen and the surrounding layer of cells is removed from the eggs. Semen is washed simultaneously by removing any seminal fluid and other proteins.
The next step is for the oocytes and semen to undergo co-incubation [79]. The sperm cells and oocytes are incubated in culture media at a ratio of 75 000:1. It is at this point that another ART may be used (ICSI) if the sperm count or motility is not optimal. Once fertilisation takes place, the egg is placed in special growth medium and left for approximately 2 days until the cell mass is around 6-8 cells. Following this the best 2-3 embryos are selected based on a morphokinetic scoring system to increase the chances of a successful pregnancy [78]. Characteristics tested include the if the growth of the cells is even, the number of cells and the level of fragmentation.
The best embryos are transferred to the patient with a plastic catheter to the uterus. More than one may be transferred to increase the chances of a successful pregnancy in older women or women who have infertility issues. In order to ensure the embryo grows normally and implants properly, the patient is given adjunctive medication. This involves injection of specific concentrations of progesterone and GnRH agonists which is performed to support the corpus luteum.
Intracytoplasmic Sperm Injection (ICSI)
Some ARTs allow for the male's genetic material to be passed onto the offspring, contingent upon a successful sperm extraction/retrieval such as intracytoplasmic sperm injection (ICSI). Although only a spermatozoon (single sperm) is required for this particular procedure, these treatment methods ultimately aim to "maximize the sperm retrieval yield" [48]. One of the most relevant clinical implications of ICSI is the ability to produce a viable embryo from an immature oocyte and a single spermatozoon injection; particularly due to the high proportion (15-20%) of oocytes retrieved when immature. [77] ICSI is typically performed during the co-incubation stage of IVF to make sure an oocyte is properly fertilised if the sperm is immobile [80]. The process involves several devices under a microscope, namely; micromanipulator, microinjectors and micropipettes). The following shows the simplified steps of performing ICSI, and is also outlined in the video provided.
- The female is administered various hormones to stimulate ovulation to release an oocyte, then the oocyte or several oocytes are removed and stored.
- Simultaneously, the males ejaculate is also collected and only a single spermatozoa may be required for fertilisation.
- Fertilisation is acheived by the directly injecting a spermatozoon into one stored oocyte using a fine needle.
- After 2 or 3 days, if fertilisation has successfully occured, the embryo will be transferred into the female's uterus to allow for implantation, and hopefully lead to pregnancy [77].
<html5media height="400" width="500">https://www.youtube.com/watch?v=h7uucZ7xpYs</html5media>
ICSI Procedure [81]
Current Research and Animal Models
The research involving male infertility most recently, is surrounding more innovative techniques to identify new and different targets to treat and diagnose the condition. A study performed in September, 2015 investigated the efficacy of using sperm chromatin structure assays to determine fertility in Nigerian men [82]. A total of 404 men consisting of fertile and unexplained infertile men underwent both semen analysis and sperm chromatin structure assays. Through the measurement of DNA fragmentation index, there was a more significant difference between infertile and fertile men when using sperm chromatin structure assaying [82]. Therefore this new diagnostic tool may be more accurate in determining the fertility potential of males. Not only does this allow for a more predictive indicator, but it may also lead future research to use modify these technologies and perhaps find the specific molecular and cellular pathways that are affected in each individual male.
Additionally, animal models have been used extensively and have resulted in major findings in treatments and pathways of male infertility. The following are examples of current research that have used animal models to make way for unique findings that have implications on future studies.
Rat Model
A study performed in August, 2015 investigated the potential for bone marrow mesenchymal stem cells to restore the internal reproductive testis in male rats with azoospermia. A total of 22 rat models underwent transplantation of rat bone marrow mesenchymal stem cells into the seminiferous tubules of the testis [83]. Observations were made to establish differences in the structure and composition of the cells in the seminiferous tubules. Results showed that these specific stem cells are able to differentiate into germ cells and have the potential to repair seminiferous tubules that are damaged in azoospermic rats [83].
Primate Model
Similar to the rat study, a population of monkeys known as rhesus macaques was used to test the effects of spermatogonial stem cells on infertility. These cells are fundamental to spermatogenesis, thus have a role in maintaining fertility of the male [84]. This particular research involved transplanting spermatogonial stem cells into the seminiferous tubules in the testes of 18 adult, and 5 prepubertal and infertile rhesus macaques, by ultrasound-guided injections [85]. There were significant improvements in adult test subjects as the level of spermatogenesis were of normal levels, meaning sperm count increased to normal levels. Through the use of large primate models, the potential for spermatogonial stem cells to rejuvenate spermatogenesis in seminiferous tubules has been established [85]. Future research regarding the functioning of sperm produced by spermatogonial stem cells will make ground breaking implications on fertility clinics and current ARTs that are available to infertile couples.
Glossary
ARTs - Assisted Reproductive Technologies
Aetiological factors - causative agents
Aneuploidy - the presence of an abnormal number of chromosomes in a cell
Cadmium - a soft, insoluble transition metal that is a byproduct of zinc production
Clomiphene citrate - a non-steroidal medication that induces infertility by increasing the release of GnRH, LH and FSH required for spermatogenesis
CNPs - Cerium dioxide nanoparticles
FSH - Follicle stimulating hormone
Gametogenesis - a biological process resulting in the formation of mature haploid male (spermatogenesis) and female (oogenesis) germ cells
GnRH - Gonadotropin releasing hormone
hCG - Human chorionic gonadotropin
hMG - Human menopausal gonadotropin
Hypogonadatropic hypogonadism - a condition characterised by a decrease in functional activity of the gonads
ICSI - Intracytoplasmic Sperm Injection
IUI - Intrauterine Insemination
IVF - In Vitro Fertilisation
Kiss1 - KiSS-1 Metastasis-Suppressor; a gene that codes for Kisspeptin, a G protein coupled receptor associated with hypogonadotropic hypogonadism
Klinefelter syndrome - genetic disorder whereby a male has an extra X chromosome
LH - Luteinizing hormone
Lipid peroxidation - the oxidation of lipids causing its degradation, usually caused by ROS
Progressive motility - the swimming of sperm from one place to another rather than in circles or twitching
Quenching - the deactivation of reactive oxygen forms
RDA - Recommended dietary allowance
ROS - Reactive oxygen species
Spermatogenesis - the production of development of new sperm
Sperm-reactive antibodies (SpAb) - antibodies present on the membrane of spermatozoa that result in adverse affects to reproduction and often infertility. [86]
TCM - Traditional Chinese medicine
Testicular Dysgenesis Syndrome (TDS) - a syndrome resultant of the disruption of embryonal programming and gonadal development during fetal life that is related to poor semen quality and testicular cancer, [87]
TURED - Transurethral resection of ejaculatory ducts
Varicocele - Abnormal dilation of the internal spermatic veins and creamasteric veins from the panpiniform plexus as a result of back flow of blood
References
- ↑ The World Health Organisation,. (2015). Human Reproductive Programme | Sexual and Reproductive Health. Retrieved 4 September 2015, from http://www.who.int/reproductivehealth/topics/infertility/definitions/en/
- ↑ The Department of Health,. (2011). Department of Health | Fertility and infertility. Health.gov.au. Retrieved 2 September 2015, from http://www.health.gov.au/internet/publications/publishing.nsf/Content/womens-health-policy-toc~womens-health-policy-experiences~womens-health-policy-experiences-reproductive~womens-health-policy-experiences-reproductive-maternal~womens-health-policy-experiences-reproductive-maternal-fert
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 <pubmed>14617369</pubmed>
- ↑ Holstein, A., & Roosen-Runge, E. (1981). Atlas of human spermatogenesis. Berlin: Grosse.
- ↑ 5.0 5.1 Germann, W., & Stanfield, C. (2005). Principles of human physiology. San Francisco: Pearson Benjamin Cummings.
- ↑ <pubmed> 5582405</pubmed>
- ↑ <pubmed> 26391090 </pubmed>
- ↑ <pubmed>PMC4156950</pubmed>
- ↑ 9.0 9.1 9.2 9.3 9.4 9.5 Escobar, J. (2013). New Semen Analysis Parameters - WHO - World Health Organization. Fertility Center in Irving and Arlington. Retrieved 20 October 2015, from http://ivfmd.net/new-world-health-semen-analysis-parameters/
- ↑ <pubmed>23757979</pubmed>
- ↑ <pubmed>PMC3583161</pubmed>
- ↑ <pubmed>PMC3583162</pubmed>
- ↑ 13.0 13.1 13.2 <pubmed>21243017</pubmed>
- ↑ Healthguru. (2008, January 4) Male Infertility (Getting Pregnant #3). Retrieved from https://www.youtube.com/watch?v=QdIl1TjUvIQ
- ↑ <pubmed> 26246871</pubmed>
- ↑ 16.0 16.1 Marmar, J. (2001). Varicocele and male infertility: Part II: The pathophysiology of varicoceles in the light of current molecular and genetic information. Human Reproduction Update, 7(5), 461-472. http://dx.doi.org/10.1093/humupd/7.5.461
- ↑ 17.0 17.1 17.2 17.3 17.4 17.5 Cocuzza, M., Cocuzza, M., Bragais, F., & Agarwal, A. (2008). The role of varicocele repair in the new era of assisted reproductive technology. Clinics, 63(3), 395-404. http://dx.doi.org/10.1590/s1807-59322008000300018 Cite error: Invalid
<ref>tag; name 'Cocuzzo' defined multiple times with different content Cite error: Invalid<ref>tag; name 'Cocuzzo' defined multiple times with different content Cite error: Invalid<ref>tag; name 'Cocuzzo' defined multiple times with different content Cite error: Invalid<ref>tag; name 'Cocuzzo' defined multiple times with different content Cite error: Invalid<ref>tag; name 'Cocuzzo' defined multiple times with different content - ↑ <pubmed>25837470</pubmed>
- ↑ <pubmed>21044369</pubmed>
- ↑ <pubmed>15192446</pubmed>
- ↑ <pubmed>11929007</pubmed>
- ↑ <pubmed>20103481</pubmed>
- ↑ <pubmed>16491264</pubmed>
- ↑ <pubmed>15509635</pubmed>
- ↑ <pubmed>11294825</pubmed>
- ↑ <pubmed>26385215</pubmed>
- ↑ 27.0 27.1 <pubmed> 26157295</pubmed>
- ↑ <pubmed>16793992</pubmed>
- ↑ <pubmed>18175790</pubmed>
- ↑ <pubmed>22594646</pubmed>
- ↑ <pubmed>22429861</pubmed>
- ↑ 32.0 32.1 <pubmed>16192719</pubmed>
- ↑ 33.0 33.1 <pubmed>21546379</pubmed>
- ↑ 34.0 34.1 <pubmed>15716187</pubmed>
- ↑ 35.0 35.1 35.2 35.3 <pubmed>12385832</pubmed>
- ↑ 36.0 36.1 <pubmed>2069684</pubmed>
- ↑ <pubmed>24954222</pubmed>
- ↑ 38.0 38.1 38.2 <pubmed>16903932</pubmed>
- ↑ 39.0 39.1 39.2 <pubmed>25685302</pubmed>
- ↑ <pubmed>17304390</pubmed>
- ↑ 41.0 41.1 <pubmed> 20090219</pubmed> Cite error: Invalid
<ref>tag; name 'PMID20090219' defined multiple times with different content - ↑ <pubmed>25277121</pubmed>
- ↑ <pubmed>24306102</pubmed>
- ↑ <pubmed>26067627</pubmed>
- ↑ <pubmed>22177463</pubmed>
- ↑ <pubmed>24714837</pubmed>
- ↑ <pubmed>26019400</pubmed>
- ↑ 48.0 48.1 48.2 48.3 <pubmed>22958644</pubmed>
- ↑ <pubmed>16422830</pubmed>
- ↑ 50.0 50.1 50.2 50.3 <pubmed>24675655</pubmed>
- ↑ 51.0 51.1 <pubmed>19439288</pubmed>
- ↑ 52.0 52.1 Higdon, J., & Drake, V. (2009). Carotenoids | Linus Pauling Institute | Oregon State University. Lpi.oregonstate.edu. Retrieved 5 October 2015, from http://lpi.oregonstate.edu/mic/articles/dietary-factors/phytochemicals/carotenoids
- ↑ <pubmed>12134711</pubmed>
- ↑ <pubmed>12899230</pubmed>
- ↑ Willett, E. (2015). Studies Show Astaxanthin May Improve Sperm Health & Fertilization Rates. Natural-fertility-info.com. Retrieved 7 October 2015, from http://natural-fertility-info.com/astaxanthin-for-sperm-health.html
- ↑ <pubmed>16110353</pubmed>
- ↑ 57.0 57.1 57.2 <pubmed>26097523</pubmed>
- ↑ Xu, C., & Qu, X. (2014). Cerium oxide nanoparticle: a remarkably versatile rare earth nanomaterial for biological applications. NPG Asia Materials, 6(3), e90. http://dx.doi.org/10.1038/am.2013.88
- ↑ Wen, J. (2006). The Role of Vitamin E in the Treatment of Male Infertility. Nutrition Bytes, 11(1), 1-6. Retrieved from http://escholarship.org/uc/item/1s2485fw
- ↑ 60.0 60.1 National Institutes of Health,. (2013). Vitamin E — Health Professional Fact Sheet. Ods.od.nih.gov. Retrieved 7 October 2015, from https://ods.od.nih.gov/factsheets/VitaminE-HealthProfessional/
- ↑ <pubmed>11225982</pubmed>
- ↑ 62.0 62.1 Evert, A., & Wang, N. (2015). Vitamin C: MedlinePlus Medical Encyclopedia. Nlm.nih.gov. Retrieved 7 October 2015, from https://www.nlm.nih.gov/medlineplus/ency/article/002404.htm
- ↑ <pubmed>26005963</pubmed>
- ↑ 64.0 64.1 <pubmed>23775386</pubmed>
- ↑ <pubmed>16705853</pubmed>
- ↑ <pubmed>16970170</pubmed>
- ↑ <pubmed>25632744</pubmed>
- ↑ 68.0 68.1 <pubmed>PMC2422968</pubmed>
- ↑ Tu, D., & Glassberg, K. (2010). Laparoscopic varicocelectomy. BJU International, 106(7), 1094-1104. http://dx.doi.org/10.1111/j.1464-410x.2010.09709.x
- ↑ Manipal Hospitals. (2015, May 19) Varicocelectomy | Testicular Diseases | Male Infertility | Urinary Problems | Manipal Hospitals. Retrieved from https://www.youtube.com/watch?v=3crlbOiCO48
- ↑ 71.0 71.1 Schroeder-Printzen, I. (2000). Surgical therapy in infertile men with ejaculatory duct obstruction: technique and outcome of a standardized surgical approach. Human Reproduction, 15(6), 1364-1368. http://dx.doi.org/10.1093/humrep/15.6.1364
- ↑ <pubmed>17899434</pubmed>
- ↑ Manipal Hospitals. (2015, May 19) Sperm Retrieval IVF | Male Infertility | Infertility Treatment | Manipal Hospitals. Retrieved from https://www.youtube.com/watch?v=c_nK2ZS_Mr0
- ↑ <pubmed>19875843</pubmed>
- ↑ <pubmed>18996517</pubmed>
- ↑ Indira IVF. (2014, July 27) What is IUI treatment for Pregnancy. Retrieved from https://www.youtube.com/watch?v=ENrx7o_z9Ng
- ↑ 77.0 77.1 77.2 77.3 <pubmed>26473112</pubmed>
- ↑ 78.0 78.1 <pubmed>22820320</pubmed>
- ↑ <pubmed>26460690</pubmed>
- ↑ <pubmed>26473111</pubmed>
- ↑ Mothercare Hosp. (2014, July 7) 3D Animation of how ICSI works. Retrieved from https://www.youtube.com/watch?v=h7uucZ7xpYs
- ↑ 82.0 82.1 <pubmed>26473109</pubmed>
- ↑ 83.0 83.1 <pubmed>26442294</pubmed>
- ↑ <pubmed>17141059</pubmed>
- ↑ 85.0 85.1 <pubmed>23122294</pubmed>
- ↑ <pubmed>8194608</pubmed>
- ↑ <pubmed>11331648 </pubmed>
- 2015 Course: Week 2 Lecture 1 Lecture 2 Lab 1 | Week 3 Lecture 3 Lecture 4 Lab 2 | Week 4 Lecture 5 Lecture 6 Lab 3 | Week 5 Lecture 7 Lecture 8 Lab 4 | Week 6 Lecture 9 Lecture 10 Lab 5 | Week 7 Lecture 11 Lecture 12 Lab 6 | Week 8 Lecture 13 Lecture 14 Lab 7 | Week 9 Lecture 15 Lecture 16 Lab 8 | Week 10 Lecture 17 Lecture 18 Lab 9 | Week 11 Lecture 19 Lecture 20 Lab 10 | Week 12 Lecture 21 Lecture 22 Lab 11 | Week 13 Lecture 23 Lecture 24 Lab 12 | 2015 Projects: Three Person Embryos | Ovarian Hyper-stimulation Syndrome | Polycystic Ovarian Syndrome | Male Infertility | Oncofertility | Preimplantation Genetic Diagnosis | Students | Student Designed Quiz Questions | Moodle page
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 25) Embryology 2015 Group Project 4. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/2015_Group_Project_4
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G