2015 Group Project 3
| 2015 Student Projects | ||||
|---|---|---|---|---|
| 2015 Projects: Three Person Embryos | Ovarian Hyper-stimulation Syndrome | Polycystic Ovarian Syndrome | Male Infertility | Oncofertility | Preimplantation Genetic Diagnosis | Students | ||||
| 2015 Group Project Topic - Assisted Reproductive Technology | ||||
| This page is an undergraduate science embryology student and may contain inaccuracies in either description or acknowledgements. | ||||
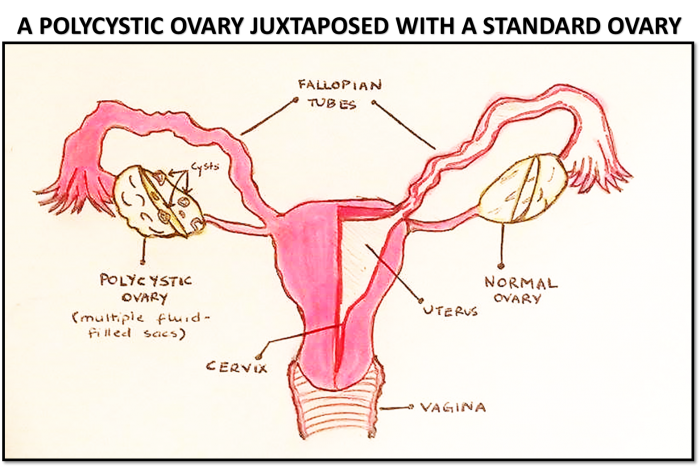
Polycystic Ovarian Syndrome
Introduction
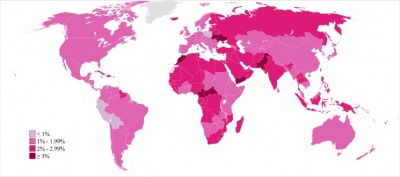
Female Infertility refers to the failure to conceive after one year of regular unprotected intercourse in females [3]. In 2010, infertility affected 48.5 million couples world wide and the areas of highest prevalence included North Africa and Middle East, South Asia, Central/Eastern Europe and Central Asia and Sub-Saharan Africa, as shown by the graph on the right [1]. As there are several causes of female infertility, we will be focusing on infertility that is caused by Polycystic Ovarian Syndrome (PCOS), the most common cause of infertility that is medically treatable [3].
Causes
PCOS is a disorder of heterogeneous origin with unknown aetiology [2]. Studies have shown that both genetic and environmental factors contribute to the development of PCOS in women however there is limited research into the actual interactions between these two factors [4].
Genetic Factors
Despite the lack of information regarding its aetiology, there is increasing evidence for a genetic involvement in the endocrine disorder. Studies have shown that genes play an important role in contributing towards the clinical and biochemical features of the disease. This evidence includes the family clustering of cases, greater unanimity between monozygotic twins than heterozygotic twins and the ability to inherit endocrine and metabolic features of PCOS [2].
In one study, 115 sisters of women with PCOS were examined and results showed that half of these sisters had high testosterone levels, suggesting that hyperandrogenemia is a common trait with a dominant pattern of inheritance. These results strongly supported the idea of the familial clustering of biochemical features such as hyperandrogenism in first degree relatives [5].
Several genes have been considered to play an important role in the development of PCOS such as CYP11A, the insulin gene, the follistatin gene and a region near the insulin receptor gene. Yet, the evidence supporting most of these genes has been refuted by larger studies, except for the region near the insulin receptor gene [5]. Among these, the gene CYP11a is the one that is strongly associated with hyperandrogenism in women with PCOS. [6] While it doesn't isolate this gene as the sole cause of PCOS, variation of the genotype at this locus contributes to production of excess androgen [7].
Studies have also shown that there is strong evidence for linkage between the alleles at the Variable Number Tandem Repeats (VNTR) which is situated 5' to the insulin gene and PCOS. The data from the study suggests that it is a very susceptible region in PCOS and also may play a role in the development of hyperinsulinaemia [8].
Environmental Factors
The prevalence of PCOS in populations where there has been a constant gene pool indicates that environmental factors play a significant role in the aetiology of PCOS [4]. The role of environmental factors in PCOS has been extensively investigated through family studies, especially in first degree relatives of those affected by PCOS [5].
Obesity and Diet
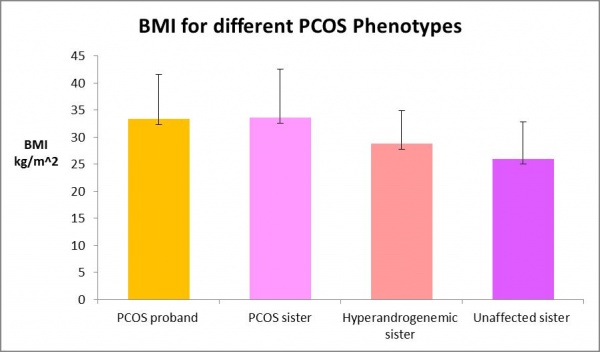
Obesity is the major environmental factor driving the development of PCOS in susceptible individuals. The rising rates of PCOS have been paralleled by the widespread emergence of obesity and type 2 diabetes which are prevalent in developed countries,[4]. Several family studies have shed light on the role of obesity in PCOS developpement, primarily one which studied the phenotypic characteristics among 80 PCOS probands and their 115 sisters. Proband was defined as the first family member to be affected by the genetic disorder. Results showed that PCOS probands and PCOS sisters had higher BMIs than hyperandrogenic (HA) and unaffected sisters (UA) with HA sisters defined as those who have elevated androgen levels but with regular menstrual cycles. These results are shown graphically on the figure shown on the right. These results suggested that mean BMI decreased as the phenotype improved and prompts further studies to looks at the contribution of adiposity towards PCOS development in genetically susceptible women [5]. These findings were supported by another study which reported that affected sisters had higher body weights than unaffected ones [9]. The role of obesity in the intrauterine environment was also shown to be important after studies suggested that PCOS women with increased weight at birth were associated with overweight mothers [10].
The risk of developing PCOS from obesity is compounded by poor dietary choices and lack of physical activity which has begun to characterise the lifestyle in developed countries [5]. Energy dense food and larger sizes in portion act as major contributors towards the increasing incidence of obesity. The quality and type of nutrition can also have an effect on the PCOS phenotype while interacting with different genotypes [4].
Medication
Studies have shown that valproic acid in medication used to treat epilepsy and bipolar disorders contributes to the development of PCOS. Women with these disorders may develop the typical features of PCOS such as polycystic ovaries, anovulation, hyperandrogenism and obesity [11]. Recent studies also suggest that the weight gain from medication is significant as it can lead to the development of PCOS [5].
Pathogenesis
Cyst Formation
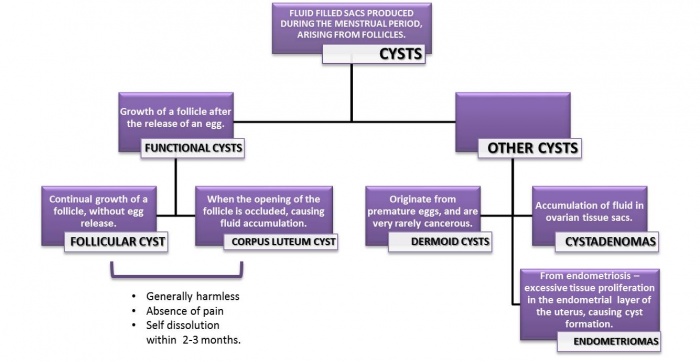
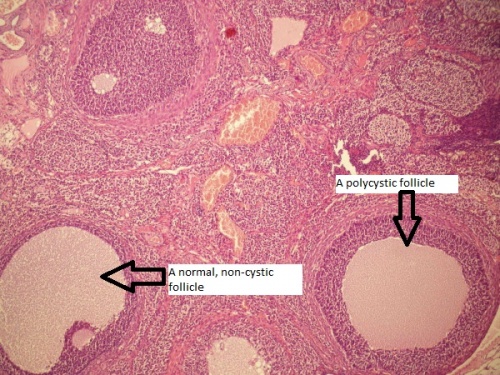
The pathogenesis of polycystic ovarian syndrome is poorly understood, however it is believed that insulin resistance and excess androgens such as testosterone play a fundamental role. The below image details the various types of cysts that can form in the ovary and uterus. In PCOS patients, cysts arise due to irregular periods, and thus an imbalance of hormone levels. [12]
Underlying factors such as obesity and genetic predispositions likely contribute to the onset of polycystic ovarian syndrome through the development of a combination of hyperinsulinemia and hyperandrogenemia.[13]
Hyperinsulinemia
Hyperinsulinemia is a term that defines the presence of excessive amounts of insulin in the blood relative to the amount of glucose. [14] This phenomenon is typically associated with obesity and excess adipose tissue, both of which are associated with insulin resistance. In order to overcome this resistance, insulin secretion is increased in an attempt to normalise blood glucose levels, thereby leading to hyperinsulineamia. [15] Excessive levels of insulin typically cause disruption to the hypothalamus-hypophysis-ovary axis whilst insulin resistance in ovarian tissues leads to decreased metabolic signalling. However, steroidogenic activity is largely unaffected by the impaired metabolic signalling, thereby paving the way androgens to become more effect and paving the way towards hyperandrogenemia. [16]
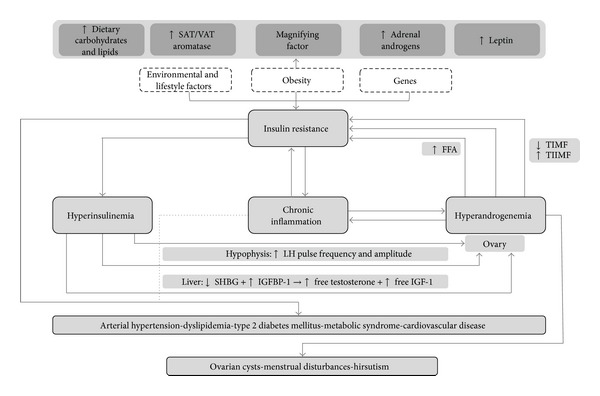
Hyperandrogenemia
Hyperandrogenemia is a hallmark of the development of polycystic ovarian syndrome, with 80% of women presenting with excess androgen also having polycystic ovaries.[17] The disruption of the hypothalamus hypophysis ovary axis as a result of hyperinsulinemia increases the release of GnRH and LH, leading to increased androgen production in the ovarian theca cells. Hyperandrogenemia decreases the feedback sensitivity of both estradiol and progesterone within gonadotropic hypothalmic cells. [16] A deficiency of FSH is observed and correlates with the increased secretion of Gonadotrophin-releasing hormone (GnRH) and Luteinizing hormone (LH). As a result of this deficiency, graunlosa cell aromatisation to oestrogens as well as follicle maturation and ovulation will be drastically impaired. [18]
As a consequence of the impairment of follicle maturation, small antral follicles will accumulate within the periphery of the ovary and develop a cystic morphology. Ovulation will be unable to occur due to the absence of mature follicles, culminating in infertility. [19]
Hyperandrogenism
Animal Models
Studies conducted on female rhesus monkeys have supported evidence for the foetal origins of the clinical features of PCOS [20]. It has been shown that in human females, an excess of androgen exposure at any stage from the development of the ovaries to the onset of puberty manifests as characteristic features of PCOS, such as resistance to insulin and LH hypersecretion. After exposure to levels of testosterone matching those of fetal males, in utero, female rhesus monkeys were found to display the clinical and biochemical features typical of PCOS such as hypersecretion of LH and abnormal insulin action. These results correlated to those of similar studies conducted on sheep, where the pregnant ewe was exposed to excessive levels of testosterone and LH secretion and abnormality in ovarian cycles were again prominent features [21]. While these studies indicate that similar biochemical and clinical manifestations in humans originate from excess androgen exposure of the female fetus, it must be noted that it is very unlikely for any excess androgen production to be passed across the placenta from the mother to her daughter. This is because mechanisms such as androgen binding proteins and placental metabolism of androgen prevent excess androgen entering the circulation of the fetus [22].
Cell Culture Models
While studies have shown that the adrenal could be a source of excess androgen such as the adrenal [23], they were followed by various studies using cell culture models which demonstrated that the ovary was the major source of excess androgen [7] [24]. In vitro studies have shown that the production of steroids is abnormal in theca cells [23]. In women affected by PCOS, stimulation by human chorionic gonadotrophin (hCG) is followed by an increase in thecal steroid production after suppression of LH by a GnRH analogue [24]. Theca cells cultured from polycystic ovaries produce 20 times as much androstenedione as those from normal ovaries [7] with increased expression of mRNA for enzymes responsible for making steroids also reported in following studies. [25] These results have prompted many studies on genes encoding these steroidogeneic enzymes.
Signs and Symptoms
The symptoms of PCOS are different for each women. The three cardinal signs of PCOS are hyperandrogenism, polycystic ovaries and ovarian dysfunction [27]. Common clinical symptoms include [28] ;
|
Along with obesity, PCOS is also associated with other metabolic issues such as insulin resistance, hyperinsulinemia and type 2 diabetes mellitus. It has also been associated with cardiovascular problems and cancers of the breast and endometrium [29].
Diagnosis
A major diagnostic tool for PCOS is a pelvic ultrasound, but PCOS diagnosis cannot be completely dependent on just pelvic ultrasounds. A patient with PCOS may not necessarily have polycystic ovaries, and those with ovarian cysts may not necessarily have PCOS. A range of diagnostic assessments and associated symptoms have to be thoroughly investigated.
Medical history and examination
Clinical questions are asked or filled out in the form of a questionnaire to assess and assist in the diagnosis of PCOS. Studies show that those with a history:
- infrequent and irregular menstruation
- hirsutism (related to high levels of androgens in the blood – hyperandrogenism)
- obesity
- acne production
are at a higher predictive risk of PCOS. But questions regarding failed pregnancy attempts and history of nipple discharge proved no correlation in diagnosing PCOS. [30]
Blood Test
A patient with elevated levels of androgens, a medical condition called hyperandrogenism, is very highly likely to have PCOS. [32] Androgens, such as testosterone, and free androgen index (FAI) are the two tests best used in diagnosing hyperandrogenism. Androgen excess in PCOS is a result from increased synthesis and release of ovarian androgens. Dehydroepiandrosterone sulfate (DHEA-S) levels are used as a biomarker for testing androgen levels, as it is made exclusively by the adrenal glands. DHEA-S levels of above 1890 micromol/L is extremely suggestive of hyperandrogenism. [33] Free androgen index (FAI) is the ratio of total testosterone measurement to levels of sex hormone-binding globulin (SHBG). Those with PCOS will have high FAI, as this indicates a high ration of free testosterone in the body. Other blood tests are also done to help exclude other predisposing factors such as levels of Thyroid Stimulating Hormone (TSH) and Follicle-stimulating hormone (FSH). An elevated ratio of LSH to FSH, of above 2:1 [34], indicates a possibility, but not definitively, of PCOS. The effect of insulin on blood can also be tested for insulin resistance. Insulin resistance enhances androgen production and increases free androgen levels by reducing SHBG. This can used as an indication marker for the possibility of PCOS A study in 2014 showed that Anti-Müllerian hormone (AMH) is found to be elevated in patients with PCOS [35], giving rise to its potential clinical implication in diagnosing PCOS. AMH is exclusively produced in ovarian follicles, with individual AMH concentration reflecting the quantity of remaining primordial follicles and measure of ovarian reserve.
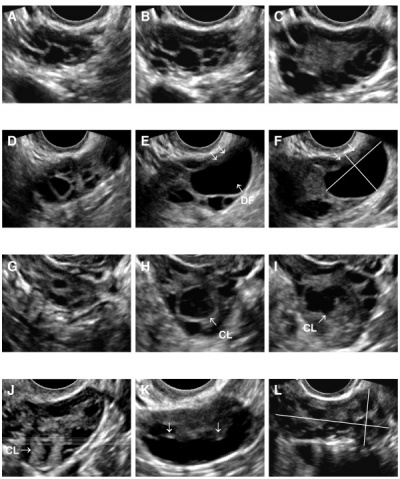
Gynecologic ultrasonography
Gynecologic ultrasonography is a pelvic ultrasound, specifically to the female pelvic region, with regards to the uterus, ovaries and the fallopian tube. The examination can be performed by transabdominal ultrasonography, with a full bladder, or transvaginal ultrasonography with a vaginal transducer. Typically transvaginal imaging shows a better and clearer image than transabdominal ultrasonography, as it uses higher frequency imaging, but is limited to areas it can reach. In the case of PCOS, both techniques can provide an image of the ovaries. Studies suggest that if more than 25 follicles are seen in an ovary, predominantly in the periphery, in women between 18-35 years, it is described to have a polycystic ovarian morphology. [36]
Magnetic Resonance Imaging
Ovaries can also be seen on MRIs. On MRIs, PCOS is characterized by numberous peripheral cysts where the ovaries affected to be slightly larger than normal. Although MRI is seen to be effective in ovarian imaging, the finding of peripheral ovarian cysts in MRIsmay not be PCOS specific. [37] But studies have seen that MR imaging in adolescence uncover distinct differences between young patients with and without PCOS [38], with also great diagnostic sensitivity with patients between the age of 21-30 [39], providing assistance of imagery in patients whom may be opposed to transvaginal ultrasounds.
Prevention and Treatment
There currently is no cure for PCOS, nor can it be prevented. This can be largely attributed to genetics, and because it can progress without being noticed for an extended period (generally until puberty begins). Treatment maybe involve lifestyle changes or hormonal augmentation in regulating periods, hirsutism, and acne, in addition to regulating androgen levels.
Current Treatments
| CURRENT TREATMENT | DISADVANTAGES |
| General Practitioners tend to prescribe oral anti-oestrogens, most commonly the contraceptive pill, to restore ovulation. | This method only has a 50% success rate in PCOS patients. It does not directly increase fertility rates, but restores the menstrual cycle. [40] |
| Lifestyle changes are highly encouraged by medical practitioners. Diet and exercise regimes can be designed with a dietitian or nutritionist. [41] | Exercise and diet plans do not directly increase fertility. They are designed to enhance weight loss, which in some patients can encourage normal hormone levels from the pituitary and thyroid. [41] |
| Clomiphene citrate administration (generally in pill form, as prescribed by a GP). Such pills stimulate the pituitary gland to release GnRH to simulate ovulation (with a 72% success rate). [9] | Clomiphene can cause multiple side effects, most notably abnormal uterine bleeding and pelvic pain to due enlargement of the ovaries.
Thus, it cannot be as frequently prescribed as the contraceptive pill, and careful patient monitoring is imperative [9]. |
| Higher fertility rates are observed in treatments combining clomiphene citrate and metformin (79% success) [9]. | Similar disadvantages as clomiphene, above. |
Note that the success rates for current PCOS treatments vary between patients. Some individuals may require more than a simple diet and exercise plan and may need various medicines and even ovarian drilling (detailed below) to encourage fertility. Oral contraceptives do not have a significant impact on fertility rates, but rather regulate the menstrual cycle and in turn, hormone levels (oestrogen and progesterone).
Laparoscopic Ovarian Drilling
Laparoscopic Ovarian drilling (LOD) is a surgical method of treating polycystic ovaries and is typically used when an infertile woman does not respond to administration of clomiphene citrate. [10]. The most common form of LOD involves electrocautery through the use of an insulinated uniupolar needle electrode. The exact method of action of LOD is poorly understood. The most accepted hypothesis however is that the breakdown of ovarian follicles and stroma via heat, laser or mechanical drilling brings about a decrease in androgen and inhibin levels, followed by a rise in the levels of FSH. Inflammatory growth factors produced in response to the thermal injury caused by laser drilling can enhance the action that FSH has on folliculogenesis. Improved blood flow often results from LOD which serves to drastically improves the delivery of gonadotrophins to the ovary.[10].
LOD can be an appealing alternative to treatment with gonadotrophins, as the latter is typically time consuming, expensive and requires constant monitoring in order to be effective.[42]
The overall effectiveness of LOD as a standalone treatment for PCOS is still questionable. Several studies have shown that there is no evidence of significant differences in pregnancy and live births in women undergoing LOD and those undergoing other medical treatments. [10].
<html5media height="300" width="400">https://www.youtube.com/watch?v=JH-Yx1QWUBk</html5media>
Ovarian Drilling by Laparoscopy for PCOS [43]
New Trials
Some studies have found that regular Selenium supplements can encourage fertility among PCOS patients. The program, which is still in its early trial period, requires women to consume selenium tablets for 8 weeks. This study has found a significant decrease in dehydroepiandrosterone (DHEA) levels in patients, thus reducing excessive hair growth associated with PCOS. A direct link to increased fertility is yet to be established, and thus further testing is required.[44]
PCOS and Conceiving

Although PCOS can significantly hinder a woman's chances of conceiving, many women with the condition do give birth to healthy children. This is largely due to modern day medical treatments, as detailed above. The following is a blog post written by 'Tarryn,' the founder of PCOS Diet Support (www.pcosdietsupport.com):
- I’m Tarryn and I was diagnosed with PCOS in January 2010. I always suspected something was amiss as my menstrual cycle has always been irregular. In 2001 or 2002 I consulted a Gynaecologist who did an ultrasound and told me that I don’t ovulate every month and put me on the pill. He made no mention of PCOS.
- When hubby and I decided to start trying for a baby in 2009, I just knew something was wrong. So, I went to my doctor who did some blood tests and diagnosed me with PCOS. Although I had my suspicions already, I was devastated.
- I decided I had to do something and went on a low GI diet and exercised regularly. I fell pregnant 9 months later. I had been taking my basal body temperature every morning and the only time I ovulated in 9 months, I fell pregnant with our beautiful daughter.
- Toward the end of 2012, we decided we’d like to give our little girl a brother or sister. I was very careful with my diet and supplements. It took 5 months to fall pregnant but we are now a family of 4. Our little boy was born in December 2013.
- It is possible to overcome PCOS! I’m still determined to continue the fight against PCOS and keep it under control with diet and lifestyle changes. [45].
Glossary
Cyst - a general term for a fluid filled sac that grows abnormally on various organs (i.e. ovarian, kidney cysts).
FSH - Follicle Stimulating Hormone. Stimulates the production of eggs or sperm in the human gonads; produced by the anterior pituitary.
Hirsutism - An abnormal growth of hair on a woman's face or body.
hCG - Human Chorionic Gonadotrophin. A hormone secreted by the placenta in the early stages of pregnancy. It encourages the production of other hormones such as progesterone by ovarian cells in order to maintain pregnancy.
GnRH - Gonadotrophin Releasing Hormone, responsible for the release of LH and FSH from the pituitary.
LH - Luteinizing Hormone. Produced by the anterior pituitary, stimulating ovulation.
LOD - Laparoscopic Ovarian Drilling (detailed in the Treatment section).
PCOS - Polycystic Ovarian Syndrome
mRNA - Messenger Ribonucleic acid; a transcribed version of a section of DNA that is later translated to produce a protein.
DHEA - Dehydroepiandrosterone sulfate; a steroid hormone produced by various organs (reproductive organs, the brain and the adrenal glands).
Proband - The first family member who is affected by a genetic disorder and seeks medical attention.
References
- ↑ 1.0 1.1 <pubmed>23271957</pubmed>
- ↑ 2.0 2.1 2.2 <pubmed>21896560</pubmed>
- ↑ 3.0 3.1 3.2 <pubmed>26150870</pubmed>
- ↑ 4.0 4.1 4.2 4.3 <pubmed>17185788</pubmed>
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 5.6 <pubmed>9843997</pubmed>
- ↑ <pubmed>9147642</pubmed>
- ↑ 7.0 7.1 7.2 <pubmed>7962289</pubmed>
- ↑ <pubmed>9455828</pubmed>
- ↑ 9.0 9.1 9.2 9.3 <pubmed>11994352</pubmed> Cite error: Invalid
<ref>tag; name 'green' defined multiple times with different content - ↑ 10.0 10.1 10.2 10.3 <pubmed>9343501</pubmed> Cite error: Invalid
<ref>tag; name 'blue' defined multiple times with different content Cite error: Invalid<ref>tag; name 'blue' defined multiple times with different content Cite error: Invalid<ref>tag; name 'blue' defined multiple times with different content - ↑ <pubmed>8413434</pubmed>
- ↑ 12.0 12.1 Mayo Clinic Staff Ovarian Cysts Causes 13th August 2014 Date Retrieved: 22nd October 2015. http://www.mayoclinic.org/diseases-conditions/ovarian-cysts/basics/causes/con-20019937
- ↑ <pubmed>PMC4556908</pubmed>
- ↑ <pubmed>PMC4114053</pubmed>
- ↑ <pubmed>PMC2782313</pubmed>
- ↑ 16.0 16.1 16.2 <pubmed>PMC4334071</pubmed>
- ↑ <pubmed>PMC3872139</pubmed>
- ↑ <pubmed>PMC3453528</pubmed>
- ↑ <pubmed>PMC4562327</pubmed>
- ↑ <pubmed>18406243</pubmed>
- ↑ Padmanabhan V, Evans NP, Taylor JA, Robinson JE1997 Prenatal exposure to androgens leads to the development of cystic ovaries in the sheep. Biol Reprod 56:194
- ↑ <pubmed>12098657</pubmed>
- ↑ 23.0 23.1 <pubmed>7962325</pubmed>
- ↑ 24.0 24.1 <pubmed>9302378</pubmed>
- ↑ <pubmed>10852468</pubmed>
- ↑ <pubmed>PMC3395821</pubmed>
- ↑ <pubmed>14688154</pubmed>
- ↑ Women's Heath, U.S. Department of Health and Human Services ,[ http://www.womenshealth.gov/publications/our-publications/fact-sheet/polycystic-ovary-syndrome.html#d ], 'Polycystic ovary syndrome (PCOS) fact sheet'
- ↑ <pubmed>PMC4341818</pubmed>
- ↑ <pubmed> 17872783 </pubmed>
- ↑ <pubmed>18638401</pubmed>
- ↑ <pubmed> 20591140 </pubmed>
- ↑ <pubmed> 18844715 </pubmed>
- ↑ <pubmed> 14737959 </pubmed>
- ↑ <pubmed> 24821925 </pubmed>
- ↑ <pubmed> 24345633 </pubmed>
- ↑ <pubmed> 8888256 </pubmed>
- ↑ <pubmed> 23292744 </pubmed>
- ↑ <pubmed> 19241321 </pubmed>
- ↑ <pubmed>26194691</pubmed>
- ↑ 41.0 41.1 <pubmed>26289303</pubmed>
- ↑ <pubmed>22696324</pubmed>
- ↑ Mishra, R., (2014, September 8) Ovarian Drilling by Laparoscopy for PCOS. Retrieved from https://www.youtube.com/watch?v=JH-Yx1QWUBk
- ↑ <pubmed>26267328</pubmed>
- ↑ 45.0 45.1 <Tarryn About PCOS Diet Support Date Retrieved: 22nd October 2015. http://www.pcosdietsupport.com/about-pcos-diet/>
- 2015 Course: Week 2 Lecture 1 Lecture 2 Lab 1 | Week 3 Lecture 3 Lecture 4 Lab 2 | Week 4 Lecture 5 Lecture 6 Lab 3 | Week 5 Lecture 7 Lecture 8 Lab 4 | Week 6 Lecture 9 Lecture 10 Lab 5 | Week 7 Lecture 11 Lecture 12 Lab 6 | Week 8 Lecture 13 Lecture 14 Lab 7 | Week 9 Lecture 15 Lecture 16 Lab 8 | Week 10 Lecture 17 Lecture 18 Lab 9 | Week 11 Lecture 19 Lecture 20 Lab 10 | Week 12 Lecture 21 Lecture 22 Lab 11 | Week 13 Lecture 23 Lecture 24 Lab 12 | 2015 Projects: Three Person Embryos | Ovarian Hyper-stimulation Syndrome | Polycystic Ovarian Syndrome | Male Infertility | Oncofertility | Preimplantation Genetic Diagnosis | Students | Student Designed Quiz Questions | Moodle page
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 21) Embryology 2015 Group Project 3. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/2015_Group_Project_3
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G