Musculoskeletal System - Tendon Development: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 17: | Line 17: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* '''Jitterbug/Filamin and Myosin-II form a complex in tendon cells required to maintain epithelial shape and polarity during musculoskeletal system development'''{{#pmid:30213743|PMID30213743}} "During musculoskeletal system development, mechanical tension is generated between muscles and tendon-cells. This tension is required for muscle differentiation and is counterbalanced by tendon-cells avoiding tissue deformation. Both, Jbug/Filamin, an actin-meshwork organizing protein, and non-muscle Myosin-II (Myo-II) are required to maintain the shape and cell orientation of the Drosophila notum epithelium during flight muscle attachment to tendon cells. Here we show that halving the genetic dose of Rho kinase (Drok), the main activator of Myosin-II, enhances the epithelial deformation and bristle orientation defects associated with jbug/Filamin knockdown. Drok and activated Myo-II localize at the apical cell junctions, tendon processes and are associated to the myotendinous junction. Further, we found that Jbug/Filamin co-distribute at tendon cells with activated Myo-II. Finally, we found that Jbug/Filamin and Myo-II are in the same molecular complex and that the actin-binding domain of Jbug/Filamin is necessary for this interaction. These data together suggest that Jbug/Filamin and Myo-II proteins may act together in tendon cells to balance the tension generated during development of muscles-tendon interaction, maintaining the shape and polarity of the Drosophila notum epithelium." | |||
* '''mTORC1 Signaling is a Critical Regulator of Postnatal Tendon Development'''{{#pmid:29215029|PMID29215029}} "Tendons transmit contractile forces between musculoskeletal tissues. Whereas the biomechanical properties of tendons have been studied extensively, the molecular mechanisms regulating postnatal tendon development are not well understood. Here we examine the role of mTORC1 signaling in postnatal tendon development using mouse genetic approaches. Loss of mTORC1 signaling by removal of Raptor in tendons caused severe tendon defects postnatally, including decreased tendon thickness, indicating that mTORC1 is necessary for postnatal tendon development. By contrast, activation of mTORC1 signaling in tendons increased tendon cell numbers and proliferation. In addition, Tsc1 conditional knockout mice presented severely disorganized collagen fibers and neovascularization in the tendon midsubstance. Interestingly, collagen fibril diameter was significantly reduced in both Raptor and Tsc1 conditional knockout mice, albeit with variations in severity." | * '''mTORC1 Signaling is a Critical Regulator of Postnatal Tendon Development'''{{#pmid:29215029|PMID29215029}} "Tendons transmit contractile forces between musculoskeletal tissues. Whereas the biomechanical properties of tendons have been studied extensively, the molecular mechanisms regulating postnatal tendon development are not well understood. Here we examine the role of mTORC1 signaling in postnatal tendon development using mouse genetic approaches. Loss of mTORC1 signaling by removal of Raptor in tendons caused severe tendon defects postnatally, including decreased tendon thickness, indicating that mTORC1 is necessary for postnatal tendon development. By contrast, activation of mTORC1 signaling in tendons increased tendon cell numbers and proliferation. In addition, Tsc1 conditional knockout mice presented severely disorganized collagen fibers and neovascularization in the tendon midsubstance. Interestingly, collagen fibril diameter was significantly reduced in both Raptor and Tsc1 conditional knockout mice, albeit with variations in severity." | ||
Revision as of 00:22, 8 January 2020
| Embryology - 26 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
This page describes skeletal tendon development, during formation of the connective tissue connection muscle to bone.
The syndetome is the embryonic structural origin of tendons from the somite and originates from the dorsolateral edge of the sclerotome. Expression of the basic helix-loop-helix (bHLH) transcription factor scleraxis (SCX) in early progenitor cells is thought to be key regulator in the formation of tendon and ligament tissues.[1] Scleraxis may also have additional roles in other tissues such as in early heart valve development.[2]
The origins of some muscles and tendons in the head differ from those found in the remained of the body.
See also notes Connective Tissue Development.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Tendon development | Tendon embryology |syndetome | myotendinous junction development |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Molecular
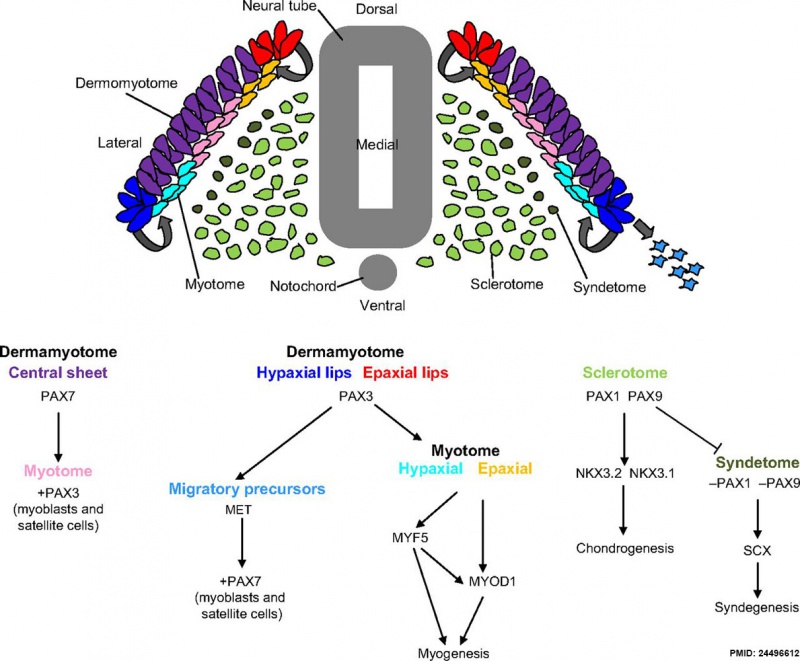
Mesoderm Development and Pax[9]
dark green - syndetome originates from the dorsolateral edge of the sclerotome, as Pax1 and Pax9 are downregulated and scleraxis (Scx) upregulation leads to syndegenesis. Pax, paired homeobox; MYOD1, myogenic differentiation antigen 1; MYF5, myogenic factor 5; NKX, NK homeobox; SCX, scleraxis.
Scleraxis
Scleraxis (SCX) is a member of the basic helix-loop-helix (bHLH) transcription factor family. It is expressed in early mesoderm progenitor cells and may regulate the formation of tendon and ligament tissues.[1]
Scleraxis may also have additional roles in other tissues such as in early heart valve development.[2]
- Cytogenetic location: 8q24.3
- Links: NCBI databases - Scleraxis | OMIM 609067
Tenomodulin
Tenomodulin (TNMD, Tnmd) is a marker of tendon differentiation, its expression has been shown to be regulated by the transcription factors Scleraxis and Mohawk.[10] May also affect the tendon stem/progenitor cells.
- Links: NCBI databases - Tenomodulin
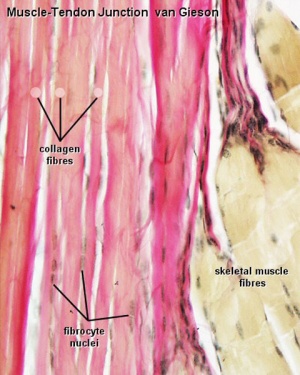
Histology
References
- ↑ 1.0 1.1 <pubmed>11585810</pubmed> Cite error: Invalid
<ref>tag; name 'PMID11585810' defined multiple times with different content - ↑ 2.0 2.1 Barnette DN, VandeKopple M, Wu Y, Willoughby DA & Lincoln J. (2014). RNA-seq analysis to identify novel roles of scleraxis during embryonic mouse heart valve remodeling. PLoS ONE , 9, e101425. PMID: 24983472 DOI.
- ↑ Manieu C, Olivares GH, Vega-Macaya F, Valdivia M & Olguín P. (2018). Jitterbug/Filamin and Myosin-II form a complex in tendon cells required to maintain epithelial shape and polarity during musculoskeletal system development. Mech. Dev. , 154, 309-314. PMID: 30213743 DOI.
- ↑ Lim J, Munivez E, Jiang MM, Song IW, Gannon F, Keene DR, Schweitzer R, Lee BH & Joeng KS. (2017). mTORC1 Signaling is a Critical Regulator of Postnatal Tendon Development. Sci Rep , 7, 17175. PMID: 29215029 DOI.
- ↑ Russo V, Mauro A, Martelli A, Di Giacinto O, Di Marcantonio L, Nardinocchi D, Berardinelli P & Barboni B. (2015). Cellular and molecular maturation in fetal and adult ovine calcaneal tendons. J. Anat. , 226, 126-42. PMID: 25546075 DOI.
- ↑ Brown JP, Finley VG & Kuo CK. (2014). Embryonic mechanical and soluble cues regulate tendon progenitor cell gene expression as a function of developmental stage and anatomical origin. J Biomech , 47, 214-22. PMID: 24231248 DOI.
- ↑ Schweitzer R, Zelzer E & Volk T. (2010). Connecting muscles to tendons: tendons and musculoskeletal development in flies and vertebrates. Development , 137, 2807-17. PMID: 20699295 DOI.
- ↑ Gilsohn E & Volk T. (2010). Slowdown promotes muscle integrity by modulating integrin-mediated adhesion at the myotendinous junction. Development , 137, 785-94. PMID: 20110313 DOI.
- ↑ Blake JA & Ziman MR. (2014). Pax genes: regulators of lineage specification and progenitor cell maintenance. Development , 141, 737-51. PMID: 24496612 DOI.
- ↑ Miyabara S, Yuda Y, Kasashima Y, Kuwano A & Arai K. (2014). Regulation of Tenomodulin Expression Via Wnt/β-catenin Signaling in Equine Bone Marrow-derived Mesenchymal Stem Cells. J Equine Sci , 25, 7-13. PMID: 24834008 DOI.
Reviews
Articles
Brent AE, Schweitzer R & Tabin CJ. (2003). A somitic compartment of tendon progenitors. Cell , 113, 235-48. PMID: 12705871
Search PubMed
Search Pubmed: Tendon Development | myotendinous junction development
Additional Images
Terms
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 26) Embryology Musculoskeletal System - Tendon Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Musculoskeletal_System_-_Tendon_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G