Genital - Female Development: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| (2 intermediate revisions by the same user not shown) | |||
| Line 12: | Line 12: | ||
There are also currently separate pages describing {{ovary}} | {{oocyte}} | {{uterus}} | {{vagina}} | {{menstrual cycle}} | {{X Inactivation}} | {{ChrX}} chromosome | {{Trisomy X}} | [[:Category:Female|Category:Female]] | There are also currently separate pages describing {{ovary}} | {{oocyte}} | {{uterus}} | {{vagina}} | {{menstrual cycle}} | {{X Inactivation}} | {{ChrX}} chromosome | {{Trisomy X}} | {{mammary gland}} | [[:Category:Female|Category:Female]] | ||
<br> | <br> | ||
| Line 24: | Line 24: | ||
* '''Development of the human penis and clitoris'''{{#pmid:30249413|PMID30249413}} "The human penis and clitoris develop from the ambisexual genital tubercle. To compare and contrast the development of human penis and clitoris, we used macroscopic photography, optical projection tomography, light sheet microscopy, scanning electron microscopy, histology and immunohistochemistry. The human genital tubercle differentiates into a penis under the influence of androgens forming a tubular urethra that develops by canalization of the urethral plate to form a wide diamond-shaped urethral groove (opening zipper) whose edges (urethral folds) fuse in the midline (closing zipper). In contrast, in females, without the influence of androgens, the vestibular plate (homologue of the urethral plate) undergoes canalization to form a wide vestibular groove whose edges (vestibular folds) remain unfused, ultimately forming the labia minora defining the vaginal vestibule. The neurovascular anatomy is similar in both the developing human penis and clitoris and is the key to successful surgical reconstructions." | * '''Development of the human penis and clitoris'''{{#pmid:30249413|PMID30249413}} "The human penis and clitoris develop from the ambisexual genital tubercle. To compare and contrast the development of human penis and clitoris, we used macroscopic photography, optical projection tomography, light sheet microscopy, scanning electron microscopy, histology and immunohistochemistry. The human genital tubercle differentiates into a penis under the influence of androgens forming a tubular urethra that develops by canalization of the urethral plate to form a wide diamond-shaped urethral groove (opening zipper) whose edges (urethral folds) fuse in the midline (closing zipper). In contrast, in females, without the influence of androgens, the vestibular plate (homologue of the urethral plate) undergoes canalization to form a wide vestibular groove whose edges (vestibular folds) remain unfused, ultimately forming the labia minora defining the vaginal vestibule. The neurovascular anatomy is similar in both the developing human penis and clitoris and is the key to successful surgical reconstructions." | ||
|} | |} | ||
{| class="wikitable mw-collapsible mw-collapsed" | {| class="wikitable mw-collapsible mw-collapsed" | ||
| Line 32: | Line 30: | ||
| [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | | [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | ||
Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=female+sex-determining ''female sex-determining''] | Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=female+sex-determining ''female sex-determining''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Müllerian+duct''Müllerian duct''] | ||
|} | |} | ||
| Line 39: | Line 37: | ||
|- | |- | ||
| {{Older papers}} | | {{Older papers}} | ||
* '''FOXL2 is a female sex-determining gene in the {{goat}}'''{{#pmid:24485832|PMID24485832}} "The origin of sex reversal in XX goats homozygous for the polled intersex syndrome (PIS) mutation was unclear because of the complexity of the mutation that affects the transcription of both FOXL2 and several long noncoding RNAs (lncRNAs). Accumulating evidence suggested that FOXL2 could be the sole gene of the PIS locus responsible for XX sex reversal, the lncRNAs being involved in transcriptional regulation of FOXL2. In this study, using zinc-finger nuclease-directed mutagenesis, we generated several fetuses, of which one XX individual bears biallelic mutations of FOXL2. Our analysis demonstrates that FOXL2 loss of function dissociated from loss of lncRNA expression is sufficient to cause an XX female-to-male sex reversal in the goat model and, as in the mouse model, an agenesis of eyelids. Both developmental defects were reproduced in two newborn animals cloned from the XX FOXL2(-/-) fibroblasts. These results therefore identify FOXL2 as a bona fide female sex-determining gene in the goat. They also highlight a stage-dependent role of FOXL2 in the ovary, different between goats and mice, being important for fetal development in the former but for postnatal maintenance in the latter. | |||
* '''Temporal and spatial dissection of Shh signaling in genital tubercle development.'''{{#pmid:19906863|PMID19906863}} "Genital tubercle (GT) initiation and outgrowth involve coordinated morphogenesis of surface ectoderm, cloacal mesoderm and hindgut endoderm. GT development appears to mirror that of the limb. Although Shh is essential for the development of both appendages, its role in GT development is much less clear than in the limb. Here, by removing Shh at different stages during GT development in mice, we demonstrate a continuous requirement for Shh in GT initiation and subsequent androgen-independent GT growth." | * '''Temporal and spatial dissection of Shh signaling in genital tubercle development.'''{{#pmid:19906863|PMID19906863}} "Genital tubercle (GT) initiation and outgrowth involve coordinated morphogenesis of surface ectoderm, cloacal mesoderm and hindgut endoderm. GT development appears to mirror that of the limb. Although Shh is essential for the development of both appendages, its role in GT development is much less clear than in the limb. Here, by removing Shh at different stages during GT development in mice, we demonstrate a continuous requirement for Shh in GT initiation and subsequent androgen-independent GT growth." | ||
| Line 222: | Line 222: | ||
:'''Links:''' {{atrial septal defects}} | :'''Links:''' {{atrial septal defects}} | ||
More than 80% of auto{{immune}} disease predominantly affects {{female}}s.{{#pmid:26713507|PMID26713507}} | |||
{{USA 1997-2009 Abnormalities Sex Ratio table}} | |||
== References == | == References == | ||
Latest revision as of 22:38, 28 December 2019
| Embryology - 3 May 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
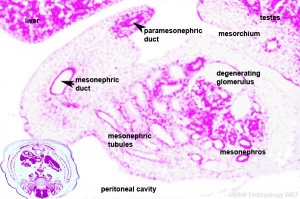
The male and female reproductive systems develop initially "indifferently", it is the product of the Y chromosome SRY gene that initially makes the "difference". The paramesonephric (Müllerian Duct) contribute the majority of female internal genital tract, while the mesonephric duct degenerates.
The mesonephric/paramesonephric duct changes are one of the first male/female differences that occur in development, while external genitaila remain indeterminate in appearance for quite a while.
There are many different issues to consider in the development of the genital system. Importantly its sex chromosome dependence, late embryonic/fetal differential development, complex morphogenic changes, long time-course, hormonal sensitivity and hormonal influences make it a system prone to many different abnormalities.
There are also currently separate pages describing ovary | oocyte | uterus | vagina | menstrual cycle | X Inactivation | X chromosome | Trisomy X | mammary gland | Category:Female
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: female sex-determining | Müllerian duct |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Textbooks
- Human Embryology (2nd ed.) Larson Chapter 10 p261-306
- The Developing Human: Clinically Oriented Embryology (6th ed.) Moore and Persaud Chapter 13 p303-346
- Before We Are Born (5th ed.) Moore and Persaud Chapter 14 p289-326
- Essentials of Human Embryology, Larson Chapter 10 p173-205
- Human Embryology, Fitzgerald and Fitzgerald Chapter 21-22 p134-152
- Developmental Biology (6th ed.) Gilbert Chapter 14 Intermediate Mesoderm
Movies
| Urogenital Sinus | Urogenital Septum |
| Uterus | Female External |
Development Overview
Three main stages during development, mesonephric/paramesonephric duct changes are one of the first male/female differences that occur in development, while external genitaila remain indeterminate in appearance for quite a while.
- Differentiation of gonad (Sex determination)
- Differentiation of internal genital organs
- Differentiation of external genital organs
The 2nd and 3rd stages dependent on endocrine gonad. Reproductive development has a long maturation timecourse, begining in the embryo and finishing in puberty. (More? puberty)
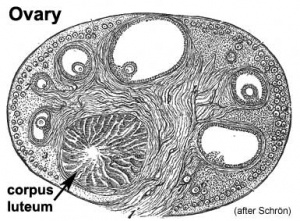
Gonad - Ovary
Primordial germ cell migration (mouse)
| Migration 1 | Migration 2 | Migration 3 |
- Links: Ovary Development | Puberty Development
Internal Genital
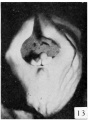
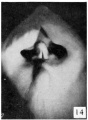
Historic Images of Genital Changes
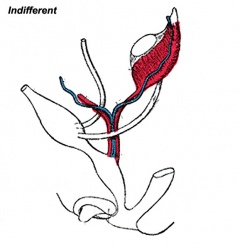
|
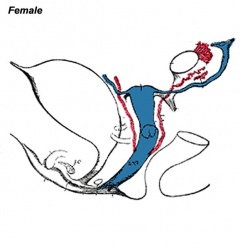
|
| Urogenital indifferent | Urogenital female |
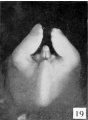
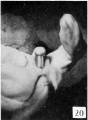
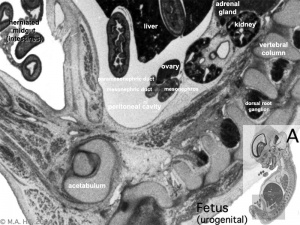
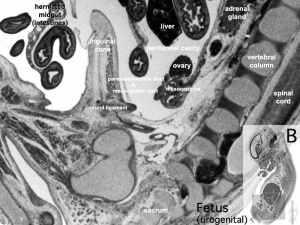
Fetal Week 10
|
|
| Ovary | Ovary |
|
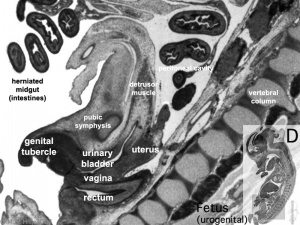
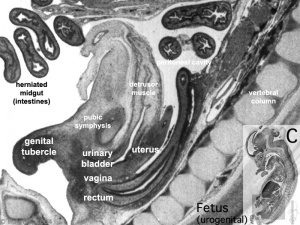
|
| Uterus | Uterus |
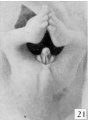
External Genital
Female Historic Descrition
Spaulding MH. The development of the external genitalia in the human embryo. (1921) Contrib. Embryol., Carnegie Inst. Wash. Publ. 81, 13: 69 – 88.
The entire process of development of the external genitalia in the female is accompanied by fewer pronounced morphological changes than occur in the male. It is noteworthy, however, that (in spite of this greater simplicity in structure) completion of development is more protracted, so that the final differentiation of the female genitalia, although brought about by comparatively minor changes, does not synchronize with the more complete transformation of the male (45 mm CR length), but instead, beginning with a slight change at a length of about 50 mm., extends as a gradual process throughout a considerable period of early fetal life.
From its beginning until the stage of 21 to 25 mm the genital tubercle of the female closely resembles that of the male except for the shorter urethral groove. About this period the female shows the beginnings of the caudal decurvation, which is apparently brought about by an excess in the growth of the cavernous over the urethral regions of the phallus. At the same time the urethral folds have become compressed into plate-like caudal projections supporting the slightly overhanging glans, which in this way is more clearly defined than in the male. As has already been pointed out, the coronary sulcus is not formed in either sex until the embryos reach a length of about 45 mm. From 25 mm. to 45 mm. this caudal decurvation becomes a more and more pronounced characteristic of the female phallus, and for this reason is a diagnostic feature of increasing importance as development proceeds.
In the female important changes occur at about the stage of 45 to 50 mm CR length, and these likewise mark the termination of the phallus period. While much less extensive than the correlated changes in the male, they are nevertheless characteristic and indicate the appro.ximate assumption of the final form. The most pronounced modification occurring at this time is that the caudal ends of the labio-scrotal swellings grow towards each other and finally join in the midventral line to form the posterior commissure (50 mm. CR length). In this manner these originally paired swellings are transformed into a cranially open, horseshoe- shaped rim, inclosing the rest of the external genitalia and separating them from the anus.
The formation of the posterior commissure in the female thus synchronizes with the formation of the raphe in the male and may be considered as representing the advent of the final differentiation, and from now on we may refer to the genitalia by their adult terms. The labio-scrotal swellings form the labia majora. The glans and cavernous portion of the phallus may be considered as the chtoris, and the urethral folds as the labia minora. The inclosed primary urogenital opening may now be called the urethro-vaginal orifice. It must be pointed out, however, that the apphcation of these terms at this time is an arbitrary one, because the actual separation of the female phallus into these more definitely adult structures does not take place until some time after the close of the period included in the present study (100 mm CR). Strict accuracy would demand that, until such division had been completed by the formation of the frenula clitoridis, the inclusive term phallus be retained.
Because of the persistence of the urethral folds (labia minora) in the female and their failure to fuse together as they do in the male, the female phallus retains a more conical outline than does the penis of the male.
In female embryos of 60 to 100 mm CR length there is shown a partial development of the prepuce, although the complicated set of folds involved in its complete formation is not produced at this time and only partial growth of the glandular portion is completed at the close of this period. While the glandular prepuce is apparently formed in much the same way as it is in the male, its growth is markedly slower, and in embryos 100 mm long the glans is not completely surrounded by it.
In these older embryos (60 to 100 mm CR length) there is also some increase in the height of the labia majora, so that the inclosed portions are somewhat submerged in the rim thus formed, although this submergence is by no means as complete as it becomes in later fetal life. It should also be noted that in fetuses up to 100 mm CR the labia majora are still cranially separated and there is no indication that they play any part in the formation of the mons veneris.
Molecular
- Paramesonephric duct - Wnt-7a Pax-8 expression associated with the initial stages of paramesonephric duct development
- Ovary - Notum, Phlda2, Runx-1 and Msx1 genes
| Y and X Chromosomes | |
|---|---|
| Males | Females |
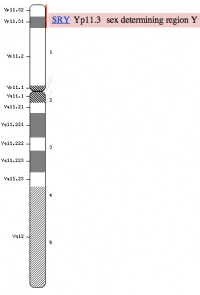
Y Chromosome
|
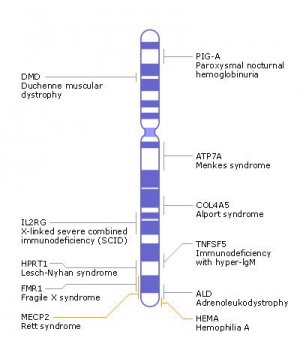
X Chromosome
|
|
|
X inactivation
|
Dax-1
DAX1 (NR0B1) is a nuclear receptor with a characteristic C-terminal ligand binding domain, but an atypical DNA binding domain, for review see[6] DAX1 protein is localized mainly in the nucleus and can bind to an RA responsive element (RARE).
- Links: OMIM 300473
Wnt-4
First identified in 1999 as a female specific gene[7], it has been recently shown to be an upstream regulator of Runx-1 expression.[8]
Abnormalities
Female Prevalence
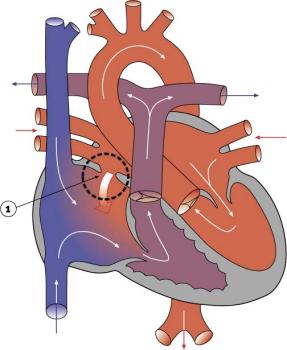
ICD-11 LB20 Congenital Anomaly of Atrioventricular Valves or Septum
|
The term atrial septal defects (ASD) describes a group of common (1% of cardiac) congenital anomolies defects occuring in a number of different forms and more often in Females. Patients have been reported living to old age with forms of this cardiac abnormality.[9]
- Links: atrial septal defects
More than 80% of autoimmune disease predominantly affects Females.[10]
| Male preponderance | Female preponderance |
|---|---|
|
|
| Cardiac defects | |
|
|
| Table data[11] Links: abnormal development | cardiovascular abnormalities | USA | Male | Female | cleft lip and palate | |
References
- ↑ Cunha GR, Robboy SJ, Kurita T, Isaacson D, Shen J, Cao M & Baskin LS. (2018). Development of the human female reproductive tract. Differentiation , 103, 46-65. PMID: 30236463 DOI.
- ↑ Baskin L, Shen J, Sinclair A, Cao M, Liu X, Liu G, Isaacson D, Overland M, Li Y & Cunha GR. (2018). Development of the human penis and clitoris. Differentiation , 103, 74-85. PMID: 30249413 DOI.
- ↑ Boulanger L, Pannetier M, Gall L, Allais-Bonnet A, Elzaiat M, Le Bourhis D, Daniel N, Richard C, Cotinot C, Ghyselinck NB & Pailhoux E. (2014). FOXL2 is a female sex-determining gene in the goat. Curr. Biol. , 24, 404-8. PMID: 24485832 DOI.
- ↑ Lin C, Yin Y, Veith GM, Fisher AV, Long F & Ma L. (2009). Temporal and spatial dissection of Shh signaling in genital tubercle development. Development , 136, 3959-67. PMID: 19906863 DOI.
- ↑ Wu X, Ferrara C, Shapiro E & Grishina I. (2009). Bmp7 expression and null phenotype in the urogenital system suggest a role in re-organization of the urethral epithelium. Gene Expr. Patterns , 9, 224-30. PMID: 19159697 DOI.
- ↑ McCabe ER. (2007). DAX1: Increasing complexity in the roles of this novel nuclear receptor. Mol. Cell. Endocrinol. , 265-266, 179-82. PMID: 17210221 DOI.
- ↑ Vainio S, Heikkilä M, Kispert A, Chin N & McMahon AP. (1999). Female development in mammals is regulated by Wnt-4 signalling. Nature , 397, 405-9. PMID: 9989404 DOI.
- ↑ Naillat F, Yan W, Karjalainen R, Liakhovitskaia A, Samoylenko A, Xu Q, Sun Z, Shen B, Medvinsky A, Quaggin S & Vainio SJ. (2015). Identification of the genes regulated by Wnt-4, a critical signal for commitment of the ovary. Exp. Cell Res. , 332, 163-78. PMID: 25645944 DOI.
- ↑ Matsumoto T, Tamiya E, Kanoh T, Takabe T, Kuremoto KI, Kamiyama T, Yamamoto S & Daida H. (2019). Atrial Septal Defect of the Ostium Secundum Type in A 101-Year-Old Patient. Int Heart J , , . PMID: 30799379 DOI.
- ↑ Liu K, Kurien BT, Zimmerman SL, Kaufman KM, Taft DH, Kottyan LC, Lazaro S, Weaver CA, Ice JA, Adler AJ, Chodosh J, Radfar L, Rasmussen A, Stone DU, Lewis DM, Li S, Koelsch KA, Igoe A, Talsania M, Kumar J, Maier-Moore JS, Harris VM, Gopalakrishnan R, Jonsson R, Lessard JA, Lu X, Gottenberg JE, Anaya JM, Cunninghame-Graham DS, Huang AJW, Brennan MT, Hughes P, Illei GG, Miceli-Richard C, Keystone EC, Bykerk VP, Hirschfield G, Xie G, Ng WF, Nordmark G, Eriksson P, Omdal R, Rhodus NL, Rischmueller M, Rohrer M, Segal BM, Vyse TJ, Wahren-Herlenius M, Witte T, Pons-Estel B, Alarcon-Riquelme ME, Guthridge JM, James JA, Lessard CJ, Kelly JA, Thompson SD, Gaffney PM, Montgomery CG, Edberg JC, Kimberly RP, Alarcón GS, Langefeld CL, Gilkeson GS, Kamen DL, Tsao BP, McCune WJ, Salmon JE, Merrill JT, Weisman MH, Wallace DJ, Utset TO, Bottinger EP, Amos CI, Siminovitch KA, Mariette X, Sivils KL, Harley JB & Scofield RH. (2016). X Chromosome Dose and Sex Bias in Autoimmune Diseases: Increased Prevalence of 47,XXX in Systemic Lupus Erythematosus and Sjögren's Syndrome. , 68, 1290-1300. PMID: 26713507 DOI.
- ↑ Michalski AM, Richardson SD, Browne ML, Carmichael SL, Canfield MA, VanZutphen AR, Anderka MT, Marshall EG & Druschel CM. (2015). Sex ratios among infants with birth defects, National Birth Defects Prevention Study, 1997-2009. Am. J. Med. Genet. A , 167A, 1071-81. PMID: 25711982 DOI.
Reviews
Sajjad Y. (2010). Development of the genital ducts and external genitalia in the early human embryo. J. Obstet. Gynaecol. Res. , 36, 929-37. PMID: 20846260 DOI.
Ribeiro SC, Tormena RA, Peterson TV, Gonzáles Mde O, Serrano PG, Almeida JA & Baracat EC. (2009). Müllerian duct anomalies: review of current management. Sao Paulo Med J , 127, 92-6. PMID: 19597684
Articles
Hashimoto R. (2003). Development of the human Müllerian duct in the sexually undifferentiated stage. Anat Rec A Discov Mol Cell Evol Biol , 272, 514-9. PMID: 12740945 DOI.
Search PubMed
Search Pubmed: Female Genital System Development | paramesonephric duct development | paramesonephric duct
Additional Images
Terms
| Genital System Terms (expand to view) |
|---|
Note there are additional glossaries associated with spermatozoa, oocyte renal.
|
| Other Terms Lists |
|---|
| Terms Lists: ART | Birth | Bone | Cardiovascular | Cell Division | Endocrine | Gastrointestinal | Genital | Genetic | Head | Hearing | Heart | Immune | Integumentary | Neonatal | Neural | Oocyte | Palate | Placenta | Radiation | Renal | Respiratory | Spermatozoa | Statistics | Tooth | Ultrasound | Vision | Historic | Drugs | Glossary |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, May 3) Embryology Genital - Female Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Genital_-_Female_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G