2017 Group Project 2
| 2017 Student Projects | |||
|---|---|---|---|
|
Kidney
The kidneys are two bean-shaped organs that play an important role in the renal system (also known as the urinary system). In the womb, the placenta is responsible for maintaining balance of water, nutrients, ion levels, etc. However, after birth, this responsibility is given to the kidneys. As such, they are vital for the everyday functions of the human body as they are the unit responsible for the filtration of blood and subsequent reabsorption of water and other nutrients according to what the body needs. The by-product of this process is urine, and it is excreted from the kidneys into the ureters which then deliver the urine to the urinary bladder.
Filtration is carried out in what is called the "nephron". In humans, the number of nephrons in each kidney ranges from 200,000 to 2.5 million. However, a typical kidney usually has approximately 900,000 to 1 million nephrons. Nephrons are the functional units of kidneys and are independent of each other. https://www.ncbi.nlm.nih.gov/pubmed/21604189
Kidney development in the embryo is known as nephrogenesis and has three stages: pronephros, mesonephros and metanephros. This process typically begins in week 4 of gestation and ends in week 36. About 1 in 500 babies are born with a kidney problem due to developmental abnormalities. https://www.kidney.org/atoz/content/detectkid
Developmentally, they arise from the intermediate mesoderm.
Anatomical Position
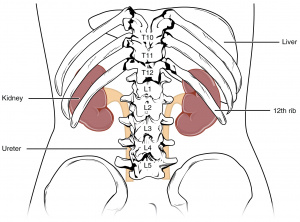
Unlike other abdominal organs, kidneys lie retroperitoneally in the abdomen, on either side of the vertebral column. They are typical located between the transverse processes from T12 to L3 of the vertebrae, however the right kidney sits slightly more superior due to the large size of the liver.
Kidney Structure
The kidneys have external coverings which involves complex layers of fascia and fat. From deep to superficial, the layers are as follows:
Renal capsule – Tough fibrous capsule
Perirenal fat – Collection of extra peritoneal fat
Renal fascia – Encloses the kidneys and the suprarenal glands
Pararenal fat – Mainly located on the posterolateral aspect of the kidney.
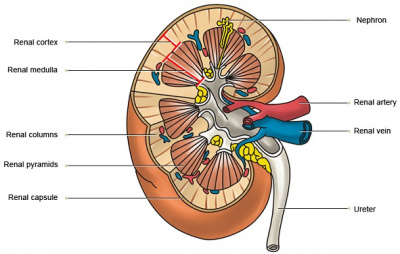
Their inner structure can be divided into 2 main areas: the outer cortex, and the inner medulla. The extension of the cortex into the medulla gives rise to renal pyramids, with the apex known as renal papilla. Each renal papilla is associated with structures known as minor calyx, which collects urine from the pyramids. Several minor calyces join to form a major calyx, where urine passes before it reaches the renal pelvis and into the ureter.
Timeline of Kidney Embryology
Kidney development (nephrogenesis) in humans begins in week 4 and commonly ends in week 36 of gestation. This is a brief timeline of the developmental processes.
| WEEK | DEVELOPMENT |
|---|---|
| 4 (early) | Pronephros begins (first stage of nephrogenesis) |
| 4 (late) | Mesonephros begins (second stage of nephrogenesis) |
| 5 | Metanephros begins (third last stage of nephrogenesis)
Development of renal vasculature begins |
| 6 | Ascension of kidneys from pelvis area begins |
| 8 | Functional kidney is formed. First nephrons are formed, a process that continues until week 36 |
| 9 | Kidneys complete their ascension and now sit just below the adrenal glands |
| 10 | Kidneys now ready to perform filtration |
| 15 | Renal vasculature development is completed |
| 36 | Nephrogenesis completed. No more nephrons formed from this point on. |
| Postnatal | Kidney and its structures continue to mature. |
Kidney development
Development of the kidney is called nephrogenesis and it arises from the intermediate mesoderm in the metanephric blastema.. Here are the three main stages of nephrogenesis which begins in week 4 of gestation and ends in week 36. It must be noted that whilst nephrogenesis does not continue beyond week 36, maturation of the kidney and its functional units does continue.
Nephrogenesis
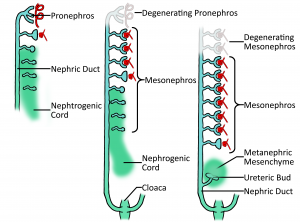
Nephrogenesis involves two transitory stages, pronephros and mesonephros, which end in a final stage (metanephros) giving us the final product of a functional kidney. It is not to say that kidney is ready and functional once metanephros is reached, but rather, it has reached the stage where its structures are set and can continue to mature into a proper functional kidney that can sustain life.
1. Pronephros
The earliest nephric stage in humans (week 4), arising from the intermediate mesoderm near the pharyngeal arches and extend from the 4th to the 14th somites and consists of 6-10 pairs of tubules. These spill into a pair (a 'pair' because there are two kidneys) of primary ducts (nephric or mesonephric duct) that are formed at the same level and go on to extend caudally. The pronephros is a transient structure that disappears completely by the 4th week of human embryonic life, its degradation can be seen in the diagram.
2. Mesonephros
This stage sees the continuation of the nephric duct caudally, with mesonephric tubules arising laterally from it. Together, these structures are known as the mesonephros and whilst still a transient structure, it has important excretory functions during early embryonic life (4—8 weeks). Gradually the top two thirds of the mesonephros go onto form the genitals, however the last third continues to form the functional kidney. This part of the mesonephros that goes on to form the kidney is known as the nephrogenic chord which is essentially mesenchymal tissue.
Towards the end of week 4 of development, the nephrogenic duct starts to move away from the nephric duct as seen in the diagram, whilst the nephric duct continues to grow caudally.
3. Metanephros
At the fifth week of development, a lateral projection called the ureteric bud develops from the nephric duct whilst the nephrogenic chord has now detached itself from the nephric duct to form the mentanephric blastema, which has been described as a cloud of mesenchymal cells[1]. After receiving relevant signalling, the metanephric blastema and ureteric bud interact by way of the blastema 'clouding around' the bud. This cloud takes the traditional kidney-bean shape and just gets bigger as time goes on.
Inside the metanephric blastema, the ureteric bud bifurcates to form the calyces, pelvis, ureter and collecting tubules of the kidney, where as blastema's mesenchymal cells go on to form numerous vesicles which develop into glomeruli and Bowman's capsules. Their interaction allows for the creation of the nephron. However, as general overview, the ureteric bud differentiates to create the renal tubule section of the kidney (reabsorption compartment of kidney), while the metanephric blastema's mesenchymal cells differentiate to create the renal corpuscle section (filtration compartment).
The permanent and functional kidney is now ready at week 8, although nephrons are still being made until approximately week 36 as they are incredibly important for life and therefore, there needs to be a lot of them.
[1]http://www.sciencedirect.com/science/article/pii/S1534580710002078
Nephron development
In a mature kidney, nephrons function as a complex epithelial network of blood filtration units which work to remove nitrogenous waste metabolites and regulate homeostasis of water and electrolytes in the body. Mammalian nephrons are generated exclusively during late embryonic and early postnatal development, with very limited cell turnover as opposed to intestine, stomach and skin epithelia which constantly renew throughout and individual's lifetime [1]. Although damaged nephrons are capable of regeneration, extremely damaged nephrons are lost and can not be replaced.
In the kidney, epithelial tubules develop from cell types of distinct embryonic origins using different cellular mechanisms (Little et al., 2010). Both the mesonephric blastema and ureteric bud contribute to the formation of the nephron and its two main units, the glomerulus and Bowman's capsule. The ureteric bud forms as an outgrowth of a pre-existing tubule and undergoes many rounds of branching to form the renal collecting system [2].
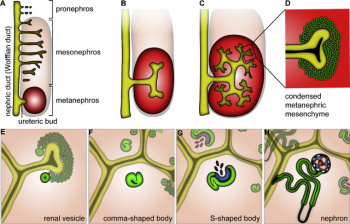
Stages in nephron formation:
1) Metanephric mesenchyme cells condense into a tight pre tubular aggregate near the ureteric bud.
2) The compacted cells then undergo a mesenchymal-to-epithelial transition, forming a sphere of polarised epithelia
3) The sphere of epithelial cells called the renal vesicle, elongates to form comma shaped bodies and primordial tubule called the 'S' shaped body
4) The proximal slit of the 'S' shaped tubules forms the glomerulus, and the distal pole connects to the tip of an adjacent bud tubule.
The ureteric bud is plays a main role in producing factors that promote mesenchymal survival, as well as secreting proteins that induce condensation of mesenchymal cells and differentiation into nephrons. One of the proteins that are produced by the ureteric bud is FGF-2, which was shown to prevent apoptosis in isolated rat mesenchyme and induce up regulation of the important transcription factor WT-1. Leukaemia inhibitory factor (LIF) combines with FGF-2 to form nephrons within 7 days in culture, and addition of TGFβ made by stromal cells speeds up the process to just 2-3 days [3].
WT-1 is a zinc finger protein, which is essential at various stages of renal development. It is also implicated to regulate genes such as Pax-2, syndecan, and E-cadherin, which are significant during the later stages on renal development. Absence of WT-1 prevents the metanephrogenic mesenchyme from differentiating into nephrons, leading to apoptosis [4].
Ascension
During metanephros, the ureteric bud forms the ureter. The ureter of each kidney descends from the kidney complex and connects to the urinary bladder. At week 6, the ureters ascend upwards as the torso of the foetus continues to extend. At week 9, they assume their permanent and proper anatomical position under the adrenal glands.
Genes Expressed
Current literature highlights two stages during organogenesis in which signalling molecules play a crucial role in the development of the functional kidney. The first is the outgrowth of the ureteric bud and the second instance being the inductive signal from the ureter that dictates differentiation of the mesenchyme. During these major signalling cascades, there are other more complex mechanisms which serve to ‘fine-tune’ this development through the regulation of cell proliferation, apoptosis, differentiation and motility. Without these growth factors and their respective receptors, outgrowth of the ureteric bud and differentiation of the mesenchyme will become severely affected.
The GDNF and RET pathway is the primary signalling complex that regulates ureteric bud growth. RET is expressed in the Wolffian duct from E8 to E11.5 and eventually in the ureteric bud as it emerges from the metanephric mesenchyme. During E13.5-17.5 expression of RET is confined to the growing tips of the ureteric bud epithelium. The RET protein acts cell autonomously by receiving a signal from the mesenchyme, that activates the proliferation and branching pathway of the ureteric bud epithelium.Targeted inactivation of RET results in a failure of the ureteric bud to emerge and respond to signals in the metanephric blastema.
Blood Supply
Although kidneys are relatively small as compared to other organs, they are responsible for filtering about 20% of the blood output from the heart. A constant and stable blood flow is important for the tissues to carry out respiration, thus ensuring the normal functioning of the kidneys. Renal infarction, that is, interruptions to the supply of oxygenated blood to the kidneys may result in kidney failure and subsequently the loss of kidney functions. A person with a damaged kidney is able to survive with the other functional kidney. In the case where both kidneys fail, dialysis or kidney transplant is needed to filter metabolic wastes from blood.
Angiogenesis and vasculogenesis
Blood vessels in kidneys are formed via angiogenesis and vasculogenesis. The first endothelial cells in early gestation are formed by vasculogenesis while those in later gestation are formed by both angiogenesis and vasculogenesis. The co-expression of Vascular Endothelial Growth Factor (VEGF) and its receptors (VEGF-R) during kidney organogenesis stimulates the development of renal blood vessels and is important in regulating vascular permeability. High levels of VEGF and VEGF-R are expressed in the kidneys during both embryonic development and adulthood. The high expression of VEGF and VEGF-R by the glomerular endothelium supports the hypothesis that VEGF and VEGF-R play an important role in the regulation of vascular permeability. When VEGF and VEGF-R levels are high, vascular permeability increases. Conversely, when VEGF and VEGF-R levels are low, the blood brain barrier permeability decreases. Hypoxia induces the production of VEGF by glomerular epithelial mass and the expression of VEGF-R by endothelial precursor cells. VEGF and/or anti-VEGF are believed to be therapeutically useful in treating many disorders.
Glomerulus
The glomerulus is a specialised network of blood capillaries that filters metabolic waste products in blood carried via the afferent blood arterioles. Metabolic waste products are filtered through fenestrae, which are small pores with diameter of 50nm to 100nm, on endothelial cells lining the glomerular capillaries. The resultant glomerular filtrate of water and soluble solutes is transported to the Bowman’s capsule and subsequently, to the renal tubule of the nephron to form urine. The glomerular capillaries converge into efferent arterioles in which filtered blood is carried away from the glomerulus. The juxtaglomerular cells lining the walls of the afferent arterioles secrete renin and regulate the volume and pressure of blood flow via the renin-angiotensin system. The efferent arterioles have high resistance that generates hydrostatic pressure that is sufficient for ultrafiltration within the glomerulus. The glomerulus serves as the connection between the vascular system and the nephron. The glomerulus and Bowman’s capsule form the filtration unit of the kidney known as the renal corpuscle. The glomerular filtration rate is the rate at which blood is completely filtered through the glomerulus and is a measure of the renal function.
Juxtaglomerular apparatus
The juxtaglomerular apparatus regulates the renal blood flow (volume and pressure) and glomerular filtration rate. It has three different types of cells, namely macula densa, juxtaglomerular cells and extraglomerular mesangial.
- Macula densa: The macula densa is a specialized area of the distal convoluted tubule where afferent arterioles enter the glomerulus and the efferent arterioles leave the glomerulus. The macula densa is able to detect changes in the levels of sodium chloride in the distal tubule of the nephron by the tubuloglomerular feedback loop.
- Juxtaglomerular cells: Juxtaglomerular cells are responsible for the synthesis, storage and secretion of renin in the kidneys. They line the walls of the afferent arterioles and regulate the volume and pressure of blood flow via the renin-angiotensin system. Juxtaglomerular cells secrete renin in response to (1) stimulation by macula densa cells when the concentration of sodium in tubular fluid decreases, (2) stimulation of β1 adrenergic receptors by epinephrine or norepinephrine and (3) a decrease in renal perfusion pressure. Poorly perfused juxtaglomerular cells activate the renin-angiotensin system.
Developmental abnormalities
As mentioned in the Introduction, every 1 in 500 newborns suffer from congenital abnormalities of the kidney and urinary tract (CAKUT). Studies have shown that certain CAKUT increases the risks of developing hypertension and cardiovascular diseases at adulthood.
An increase in anti-α smooth muscle actin (α-SMA),vimentin, and fibronectin expression in renal tissue [5] [6] as well as a decrease of the proximal tubule cubulin receptor [7] have been associated with disruptions in renal development. The proximal tubule receptor is important as it characterises the epithelial-mesenchymal transition (EMT) process, which is a physiological process that occurs during early embryogenesis, tissue repair, and pathology [8].
Vimentin and α-SMA are only expressed before the differentiation or transdifferentiation processes in epithelial cells. During this process, cells can proliferate, migrate and produce extracellular matrix. Thus, these proteins can be utilised as a marker of cell indifferentiation.
Studies have shown that the renin-angiotensis system (RAS) participates in renal development, and that exposure to RAS blockers resulted in the presence of acute kidney injury, chronic kidney disease, and tubular dysfunction in children. These studies demonstrated that inhibition of the RAS caused an increase in the relative interstitial area from the renal cortex, high levels of apoptosis, decreased cell proliferation, and impaired expression of growth factors in the kidney. Furthermore, Chen at al.'s treatment of neonatal rats with losartan for 2 days promoted down regulation of genes encoding cytoskeletal and extracellular matrix ECM components, which results in ECM malformation and cell-cell and cell-matrix interaction dysfunctions [9].
Although calcitriol is widely known for its important role in the homeostasis of calcium homeostasis and bone metabolism [10][11], recent studies have shown that it is also involved in The homeostasis of other cellular processes. These processes include the control of of autoimmunity, inflammatory process as well as blood pressure. Furthermore, calcitriol participates in the regulation of cell proliferation and differentiation processes, and the regulation of the renin gene [12].
Kidney developmental abnormalities are diverse and they correspond to defects at different stages of kidney development. Renal vascular anomalies are defects involving renal arteries and renal veins while fusion anomalies results in conditions such as fused pelvic kidney, crossed fused renal ectopia and horseshoe kidney.
Common congenital kidney defects include:
- Renal agenesis (absence of one or both kidneys)
- Multiple ureters (more than one ureter draining a kidney)
- Hypoplastic kidneys (underdevelopment of the kidneys)
- Dysplastic kidneys (abnormal development of kidneys which arises from tubules failing to branch out completely)
- Wilms tumours (kidney cancer)
- Patterning defects
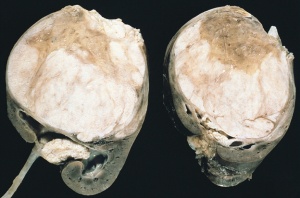
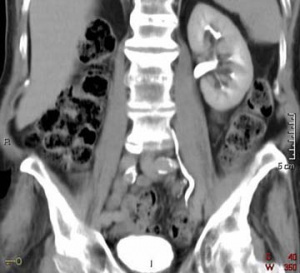
Horseshoe kidney
- Common congenital abnormality of the kidneys.
- The horseshoe kidney can be T-, horseshoe, or L-shaped, depending on the manner of fusion of the two kidneys. Occurs during development, when the left and right kidneys fuse at their lower poles by a parenchymal isthmus located ventral to the abdominal aorta, forming a "U" shape.
- Usually located in the lower lumbar position (L3 to L5), in front of the aorta and inferior vena cava, posterior to the inferior mesenteric artery [13]. The inferior mesenteric artery blocks the isthmus, preventing ascension of the kidneys and causing them to remain at a lower position.
- Shows a wide variation of arterial blood supply [14].
- During migration from the sacral region, the two metanephric blastemas can come into contact, mainly at the lower pole.
- The ureters pass in front of the zone of fusion of the kidneys.
- Despite the abnormality, kidneys and ureters are still able to function, however there is increased chance of developing upper urinary tract obstruction of infection.
- Horseshoe kidney is typically associated with other congenital defects, including Turners Syndrome, Wilms' Tumour, duplicated ureter, and Trisomy 18.
Wilms' tumour
- Most common intraabdominal cancer in children [15]; 9 out of 10 kidney cancers in children are Wilms' tumours
- Most Wilms' tumours are unilateral, meaning they occur in one kidney, in some cases it can occur bilaterally
- Transcription factor Wilms' Tumour 1 (WT-1) is known as a classic suppressor gene in Wilms' tumour [16]. Although the exact function of WT-1 is still unclear, research suggests that gemline mutations of WT-1 is the main cause for Wilms' Tumour.
- A classic Wilms' tumour has three main cell types (stromal, epithelia, blastomal), giving it a triphasic appearance
Renal agenesis
- Renal agenesis is a congenital abnormality occurring when there is a failure of development of the kidneys and ureter, induced by a lack of interaction between the ureteric bud and the metanephric mesenchyme
- This defect usually occurs around 5 weeks of embryonic life [17]
- Can occur in two forms: bilateral or or unilateral renal agenesis. Unilaterial
- Renal agenesis occurs when the ureteric bud fails to become a ureter, the renal pelvis and the collecting ducts and the mesenchyme to form nephrons [18]
- Renal agenesis patients frequently have extra-renal anomalies, such as cardiac, genital or gastrointestinal malformations [19].
- It is usually associated other congenital defects including oligohydramnios, as well as facial abnormalities including, wide set eyes and low-set ears, and a broad flat nose
Current Research
Can kidney disease be associated with nephron number?
Nephron development ceases around week 36 of gestation at the end of nephrogenesis, the body can not create new nephrons beyond that point. Due to the wide range of possible nephron numbers (250,000 - 2.5 million), many investigations have arisen to determine whether a lower nephron count predisposes a person to kidney disease later on in life.
https://www.ncbi.nlm.nih.gov/pubmed/16014104
https://www.ncbi.nlm.nih.gov/pubmed/16774009
https://www.ncbi.nlm.nih.gov/pubmed/21604189
https://www.ncbi.nlm.nih.gov/pubmed/19615565
https://www.ncbi.nlm.nih.gov/pubmed/28818273 Z5017644 (talk) 16:50, 31 August 2017 (AEST)
Questions for the future
General info on the renal system
References
- ↑ Barker, N., Bartfeld, S., and Clevers, H (2010). Tissue-resident adult stem cell populations of rapidly self-renewing organs. Cell Stem Cell. 7: 656–670
- ↑ Cebrián C., Borodo K., Charles N., Herzlinger D. A. (2004). Morphometric index of the developing murine kidney. Dev. Dyn. 231, 601–608
- ↑ Barasch, J., Yang, J., Ware, C.B., Taga, T., Yoshida, K., Erdjument-Bromage, H., Tempst, P., Parravicini, E., Malach, S., Aranoff, T., and Oliver, J.A. (1999b) Mesenchymal to epithelial conversion in ratmetanephros is induced by LIF. Cell 99, 377–386.
- ↑ Kreidberg, J.A., Sariola, H., Loring, J.M., Maeda, M., Pelletier, J., Housman, D., and Jaenisch, R. (1993). WT-1 is required for early kidney development. Cell 74, 679–691
- ↑ Balbi APC, Marin ECS, Francescato HDC, Costa RS, Coimbra TM. MAPK and angiotensin II receptor in kidney of newborn rats from losartan-treated dams. Pediatr Nephrol. 2008;23:1433–1444. doi: 10.1007/s00467-008-0830-1.
- ↑ Lamouille S, Jian XJ, Derynck R. Molecular mechanisms of epithelial–mesenchymal transition. Nat Rev Mol Cell Biol. 2014;3:178–196. doi: 10.1038/nrm3758.
- ↑ Lima WR, et al. ZONAB promotes proliferation and represses differentiation of proximal tubule epithelial cells. J Am Soc Nephrol. 2010;3:478–88. doi: 10.1681/ASN.2009070698
- ↑ Kalluri R, Weinberg RA (2009). The basics of epithelial-mesenchymal transition. J Clin Invest. 6:1420–1428. doi: 10.1172/JCI39104
- ↑ Chen Y, et al (2004) Neonatal losartan treatment suppresses renal expression of molecules involved in cell-cell and cell-matrix interactions. J Am Soc Nephrol. 5:1232–43. doi: 10.1097/01.ASN.0000123690.75029.3F
- ↑ Holick MF (2013). Vitamin D, sunlight and cancer connection. Ant. Canc. Ag Med. Chem. 13:70–82. doi: 10.2174/187152013804487308
- ↑ DeLuca HF. Overview of general physiologic features and functions of vitamin D. Am J Clin Nutr. 2004;80:1689–96
- ↑ Li YC, et al. 1,25-Dihydroxyvitamin D3 is a negative endocrine regulator of the renin-angiotensin system. J. Clin. Invest. 2002;2:229–238. doi: 10.1172/JCI0215219
- ↑ Moore KL: Clinically Oriented Anatomy, ed 3. Baltimore, Williams & Wilkins, 1992, p 223
- ↑ Yoshinaga K, Kodama K, Tanii İ, Toshimori K: Morphological study of a horseshoe kid- ney with special reference to the vascular system. Anat Sci Int 2002;77:134–139
- ↑ Davidoff, A. M. (2012). Wilms Tumor. Advances in Pediatrics, 59(1), 247–267. http://doi.org/10.1016/j.yapd.2012.04.001
- ↑ Call K. M. et al. Isolation and characterization of a zinc finger polypeptide gene at the human chromosome 11 Wilms' tumor locus. Cell 60, 509–20 (1990)
- ↑ Paolo Volpe, Valentina De Robertis, Nicola Volpe Unilateral renal agenesis, Visual Encyclopedia of Ultrasound in Obstetrics and Gynecology, www.VISUOG.org, 30th of March, 2013
- ↑ Kerecuk L, Schreuder MF, Woolf AS. Renal tract malformations: perspectives for nephrologists, Nat Clin Pract Nephrol , 2008, vol. 4 (pg. 312-325)
- ↑ Dursun H, Bayazit AK, Buyukcelik M, et al. . Associated anomalies in children with congenital solitary functioning kidney, Pediatr Surg Int , 2005, vol. 21 (pg. 456-459)