Spermatozoa Development: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 136: | Line 136: | ||
Gerbil{{#pmid:5477358|PMID5477358}} | Gerbil{{#pmid:5477358|PMID5477358}} | ||
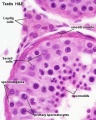
==Sertoli | ==Sertoli Cell== | ||
[[File:Testis_histology_2.jpg|thumb|alt=Histology Sertoli cell|Sertoli cells]] | [[File:Testis_histology_2.jpg|thumb|alt=Histology Sertoli cell|Sertoli cells]] | ||
The {{sertoli cell}} was named after Enrico Sertoli (1842 - 1910) an Italian (Milan) physiologist and histologist. | |||
* sustentacular cells of seminiferous tubules. | * sustentacular cells of seminiferous tubules. | ||
| Line 148: | Line 148: | ||
===Peritubular Myoid Cells== | |||
The {{peritubular myoid cell}} surrounds the seminiferous tubule. | |||
==Spermatozoa Structure== | ==Spermatozoa Structure== | ||
Revision as of 11:09, 3 June 2018
| Embryology - 16 Jun 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
This page introduces spermatogenesis the development of spermatozoa, the male haploid gamete cell. In humans at puberty, spermatozoa are produced by spermatogonia meiosis in the seminiferous tubules of the testis (male gonad). A second process of spermiogenesis leads to change in cellular organisation and shape before release into the central lumen of the seminiferous tubule. This overall process has been variously divided into specific identifiable stages in different species: 6 in human, 12 in mouse, and 14 in rat. Structurally, the seminiferous tubule epithelium is divided into a basal and an apical (adluminal) compartment by the blood–testis barrier (BTB). (More? Testis Development).
A second unique feature of this process is that during mitosis and meiosis the dividing cells remain connected by cytoplasmic bridges as the cells do not complete cytokinesis. This cellular organization is described as a syncytium, only ending with release into the central lumen of the seminiferous tubule, when the cell cytoplasm is discarded. Retinoic acid has been shown to be a key regulator of the development process. (More? Retinoic acid)
- In a healthy adult human male it takes about 48 days from meiosis to produce a mature spermatozoa, and he produces somewhere between 45 to 207 million spermatozoa per day, or about 1 to 2,000 every second. (More? Statistics)
Medicine Practical | Fertilization | Category:Spermatozoa
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Spermatozoa Development <pubmed limit=5>Spermatozoa Development</pubmed> |
Spermatozoa Movies
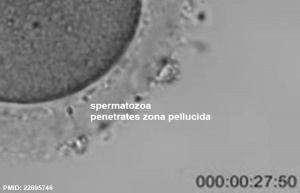
See also Week 1 movies.
|
|
| |||||||||||||
|
|
|
|
- Links: Week 1 movies | Movies
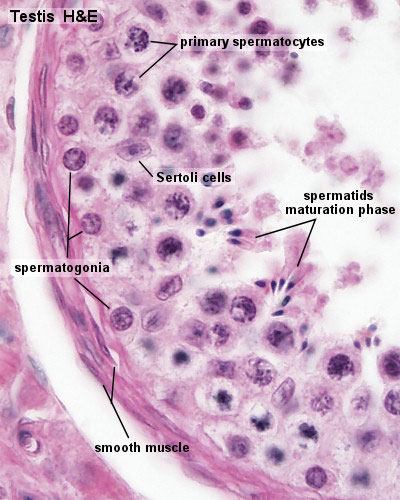
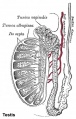
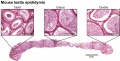
Seminiferous Tubule
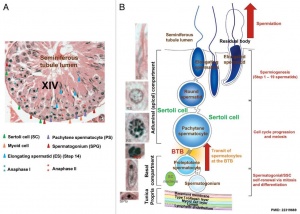
Seminiferous tubule cartoon[8]
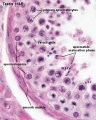
- Spermatogonia - are the first cells of spermatogenesis
- Primary spermatocyte - large, enter the prophase of the first meiotic division
- Secondary spermatocytes - small, complete the second meiotic division
- Spermatid] - immature spermatozoa
- Spermatozoa - differentiated gamete
- Spermatozoa development: primordial germ cell - spermatogonia - primary spermatocyte - secondary spermatocytes - spermatid - spermatozoa
- Links: Testis Histology | Testis Development
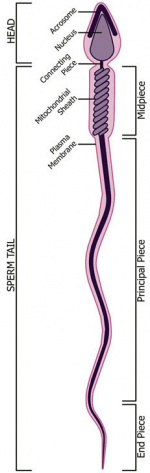
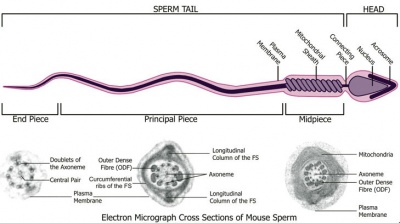
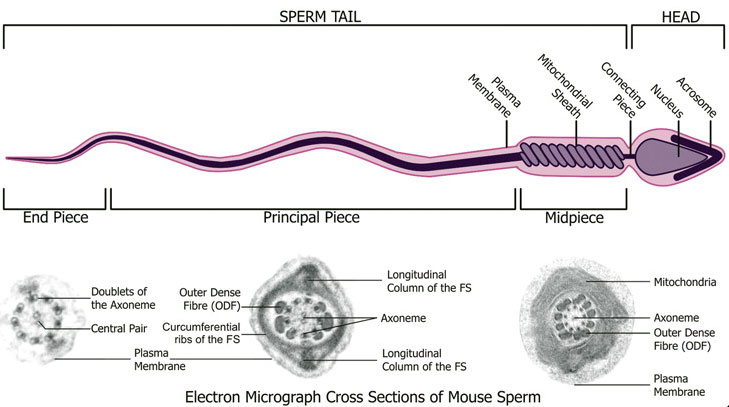
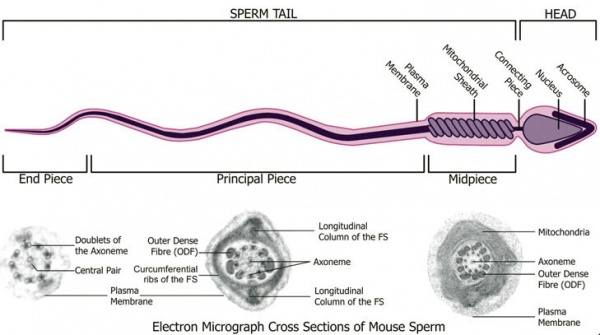
Spermatozoa Structure
Spermatozoa (mouse) cross-sections of tail (EM) and diagram[9]
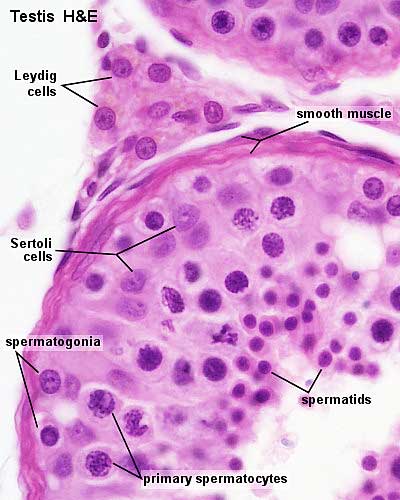
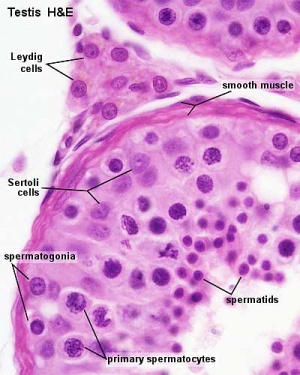
Other main cell types seen in the histological sections
- Sertoli cells- support cells seen within the seminiferous tubule
- Interstitial cells or Leydig cells - produce hormone
- Smooth muscle - surround seminiferous tubule and contribute to contraction of the tubule
Human Spermatozoa Development
- Spermatogenesis process of spermatagonia mature into spermatazoa (sperm).
- Continuously throughout life occurs in the seminiferous tubules in the male gonad- testis (plural testes).
- At puberty spermatagonia activate and proliferate (mitosis).
- about 48 days from entering meiosis until morphologically mature spermatozoa
- about 64 days to complete spermatogenesis, depending reproduction time of spermatogonia
- follicle stimulating hormone (FSH) - stimulates the spermatogenic epithelium
- luteinizing-hormone (LH) - stimulates testosterone production by Leydig cells
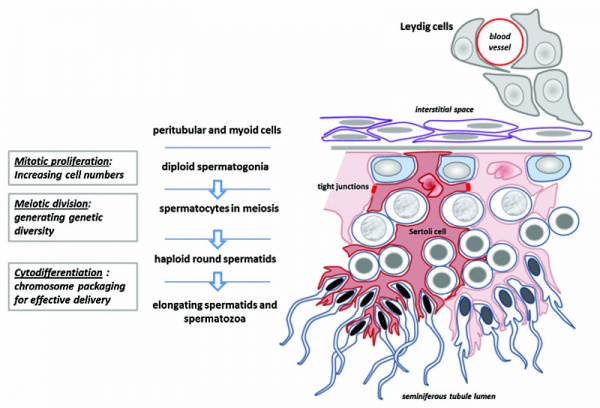
Spermatogonia
- spermatogonial stem cells (SSCs) diploid progenitor for spermatozoa.
- 1963 Clermont identified spermatogonia as Ap (pale) and Ad (dark) on basis of light microscope staining.[10]
- now also type B
- 60 years - Ap spermatogonia number decrease
- 80 years - Ad spermatogonia number decrease
Spermatogenic Cycle
Along the length of the seminiferous tubule spermatozoa develop in a cyclic manner over time progressing through a number of stages, called the spermatogenic cycle, see review.[11] The number of stages appears to differ between species, in mouse there are 12 stages (I – XII) and in the rat 14 stages.
In mouse, one spermatogenic cycle (12 stages) occurs over 8.6 days and four cycles (35 days) are required from spermatogonial stem cell to released spermatozoa.
Human[12]
Gerbil[13]
Sertoli Cell
The sertoli cell was named after Enrico Sertoli (1842 - 1910) an Italian (Milan) physiologist and histologist.
- sustentacular cells of seminiferous tubules.
- form a “blood-testis” barrier through junctional complexes
- separate the intra-tubular germinal epithelium into two compartments
- basal compartment - cells are exposed to the extra-tubular environment
- luminal compartment - cells are subject to an environment produced by Sertoli cells and germ cells
=Peritubular Myoid Cells
The peritubular myoid cell surrounds the seminiferous tubule.
Spermatozoa Structure
Spermatozoa (mouse) cross-sections of tail (EM) and diagram[9]
Acroplaxome
This structure forms the acrosome plate with intermediate filament bundles of the marginal ring at the leading edge of the acrosome. The acroplaxome site for Golgi-derived proacrosomal vesicles to tether and fuse and anchors the developing acrosome to the elongating spermatid head and may provide a scaffolding for the shaping of the spermatid nucleus.[14]
Acrosome
Derived from the Golgi apparatus in conjunction with transient specialized bundles of microtubules (manchette).
Nucleus
The spermatozoa nucleus undergoes extensive compression, and nuclear DNA chromatin remodelling by tightly packing with spermatozoa-specific protamines.[15]
| It is thought that the lysine-rich protein precursor (H1 histone) has evolved into the arginine-rich protamines.[16] | Three major spermatozoa nuclear basic proteins types:
|
| EM Human Spermatozoa Nucleus | |
|---|---|
|
|
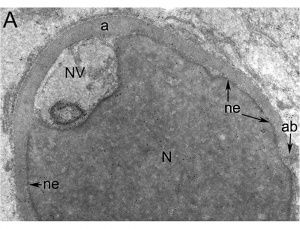
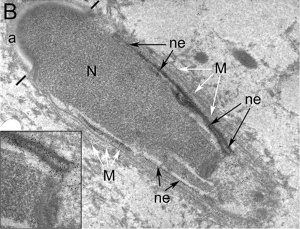
| Cap-phase spermatid nucleus[17] | Elongated spermatid nucleus[17] |
|
|
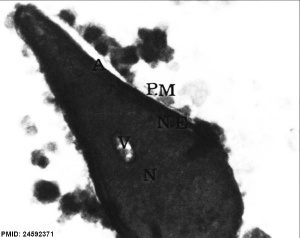
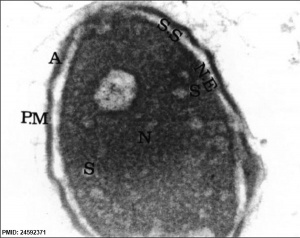
| Normal human spermatozoa[18] | Abnormal human spermatozoa[18] |
Axoneme
The stable mature microtubule-containing tail of the sperm.
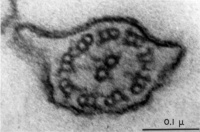
|
|
| Historic EM spermatozoon tail | Mouse cross-sections of tail[9] |
Centriole
Spermatozoa initially contains 2 centrioles (proximal, distal) and at fertilisation only a single (proximal) is present, which in most mammalian species is contributed to reconstitute the zygotic centrosome. Note that in rodents (rat, mice) both centrioles are lost and only a maternal centrosomal inheritance occurs.
- distal centriole - (perpendicular to membrane) required as the basal body generating the microtubule axoneme and is then lost (disintegration).
- proximal centriole - required after fertilisation for decondensing spermatozoa nucleus allowing development into the male pronucleus.
Manchette
A transient microtubule structure formed in spermatids involved in the process of: assembly of the mammalian sperm tail, mechanical shaping and condensation of the sperm nucleus. These microtubules are also invloved with specific transport, intramanchette transport, which has been likened to intraflagellar transport. This microtubular structure surrounds the nucleus of the developing spermatid and is thought also to assist in both the reshaping of the nucleus and redistribution of spermatid cytoplasm.
Mitochondria
Contained in the initial segment provide the energy for motility and may also enter the egg on fertilization, but are eliminated by a ubiquitin-dependent mechanism.
Perinuclear Theca
Located in the sperm head perinuclear region and contains a cytoskeletal element to maintain the shape of the sperm head and functional molecules leading to oocyte activation during fertilization.
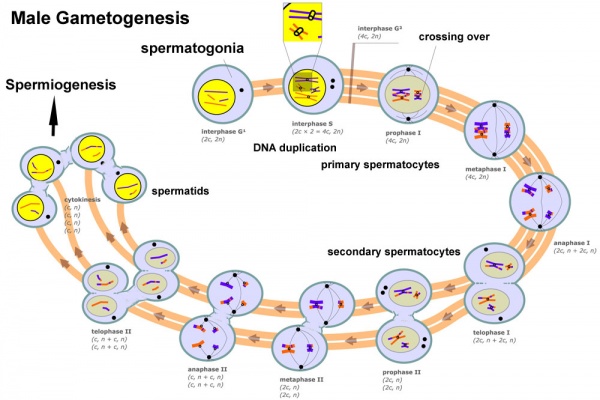
Meiosis
Spermatozoa maturation involves two processes meiosis and spermiogenesis. After puberty, new spermatozoa continue to be generated throughout life from a spermatogonia stem cell population in the testis.
Differences in Mammalian Meioses
| Female Oogenesis | Male Spermatogenesis | |
| Meiosis initiated | once in a finite population of cells | continuously in mitotically dividing stem cell population |
| Gametes produced | 1 / meiosis | 4 / meiosis |
| Meiosis completed | delayed for months or years | completed in days or weeks |
| Meiosis Arrest | arrest at 1st meiotic prophase | no arrest differentiation proceed continuously |
| Chromosome Equivalence | All chromosomes exhibit equivalent transcription and recombination during meiotic prophase | Sex chromosomes excluded from recombination and transcription during first meiotic prophase |
| Gamete Differentiation | occurs while diploid (in first meiotic prophase) | occurs while haploid (after meiosis ends) |
- Links: Cell Division - Meiosis
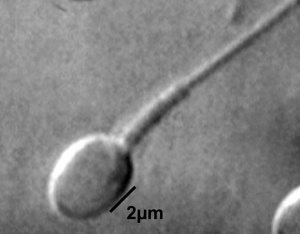
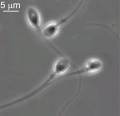
Mature Human Spermatozoa
Features:
Human Spermatazoa Statistics | Development Animation - Spermatozoa |
Spermatozoa Morphology
Morphology is a term used to describe the overall appearance of a cell or tissue and is often used to characterise changes in cellular state or activity. Historically, there have been studies comparing the overall appearance of spermatozoa between different species.[19] More recently, there have been several different ways of characterising the morphology of human spermatozoa developed mainly in relation to clinical reproductive technologies.
Integrated Sperm Analysis System (ISAS)
A semi-automated computer-aided system that measures spermatozoa head parameters length (L), width (W), area (A), perimeter (P), acrosomal area (Ac), and the derived values L/W and P/A. 20852650
- For each man a homogeneous population of distributions characterized seminal spermatozoa (7,942 cells: median values L 4.4 μm, W 2.8 μm, A 9.8 μm(2), P 12.5 μm, Ac 47.5%, L/W 1.57, P/A 1.27)
- Different men could have spermatozoa of significantly different dimensions.
- Head dimensions for swim-up spermatozoa from different men (4 812 cells) were similar to those in semen, differing only by 2%-5%.
- The values of L, W and L/W fell within the limits given by the World Health Organization (WHO).
- A subpopulation of 404 spermatozoa considered to fit the stringent criteria of WHO 'normal' seminal spermatozoa from both semen and swim-up were characterized by median values (and 95% confidence intervals) of L, 4.3 μm (3.8-4.9), W, 2.9 μm (2.6-3.3), A, 10.2 μm(2) (8.5-12.2), P, 12.4 μm (11.3-13.9), Ac, 49% (36-60), L/W, 1.49 (1.32-1.67) and P/A, 1.22 (1.11-1.35). These median values fall within the 95th centile confidence limits given by WHO, but the confidence intervals for L and W were larger.
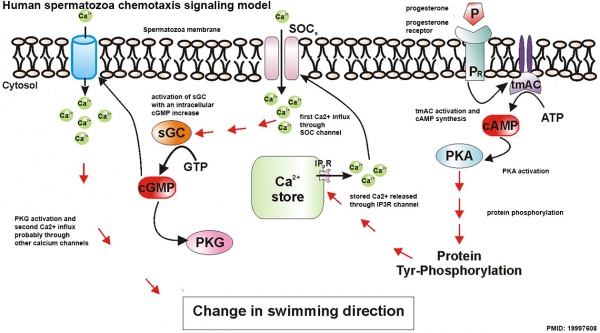
Spermatozoa Chemotaxis
Chemotaxis was first identified in marine species[20], which still remains today as a model system. While the signals may differ, the overall effect is to chemically attack spermatozoa to the oocyte to allow fertilisation to occur.
The following series of 2011 research articles have identified the spermatozoa calcium channel protein (CatSper) as the progesterone activated pathway involved in capacitated spermatozoa chemotaxis.
|
|
Human Spermatozoa Chemotaxis Model (2009)[24]
See also 2008 review.[25]
Human Spermatazoa Statistics
The following data is based normal human male values for reproductive ages between 20 to 50 years[26]:
- 45 to 207 million spermatozoa produced per day within the two testes
- 2,000 spermatozoa approx per second each day
- Compare this to adult human red blood cell production of about 250,000 million RBCs per day
- 182 million spermatozoa stored (epididymal reserves) up to per epididymis
- 440 million spermatozoa extragonadal stored
- 225 million extragonadal spermatozoa in the ductuli deferentia and caudae epididymides per ejaculation
- 23 million spermatozoa approx (all animals) per gram testicular parenchyma per day
- Transit times
- 0.72 day spermatozoa through the caput
- 0.71 days spermatozoa through the corpus
- 1.76 days spermatozoa through the cauda epididymidis
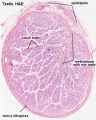
Sertoli Cell
Histology
Papanicolaou stain (Papanicolaou's stain, Pap stain) a multichromatic (five dyes) staining histological technique developed by George Papanikolaou, used to differentiate cells in smear preparations of various bodily secretions.
- Links: Testis Histology | Histology Stains
Male Abnormalities
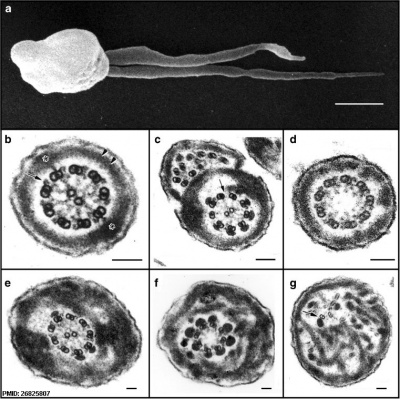
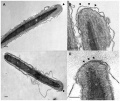
Human sperm pathologies[27] These electron micrographs show a range of tail structural abnormalities including (a) two tails. (b) shows a normal spermatozoa tail cross-section and c to g show a range of abnormal tail structures (open image to see details).
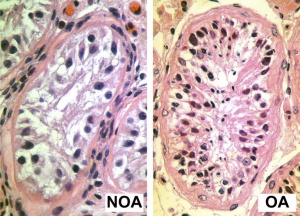
Johnsen score
A clinical score (1-10) for assessing spermatogenesis in a human testicular biopsy. Named after the author of the original article .[28]
| Johnsen score |
Description |
|---|---|
| 10 | complete spermatogenesis and perfect tubules |
| 9 | many spermatozoa present but disorganized spermatogenesis |
| 8 | only a few spermatozoa present |
| 7 | no spermatozoa but many spermatids present |
| 6 | only a few spermatids present |
| 5 | no spermatozoa or spermatids present but many spermatocytes present |
| 4 | only a few spermatocytes present |
| 3 | only spermatogonia present |
| 2 | no germ cells present |
| 1 | neither germ cells nor Sertoli cells present |
|
Reference: Johnsen SG. Testicular biopsy score count - a method for registration of spermatogenesis in human testes: normal values and results in 335 hypogonadal males. (1970) Hormones 1(1): 2-25. PubMed 5527187 | |
| Classification | Count (Millions/mL) |
|---|---|
| Azoospermia | 0 |
| Severe oligozoospermia | less than 1 |
| Moderate oligozoospermia | 1-5 |
| Mild oligozoospermia | 5-20 |
| Normal | greater than 20 |
Oligospermia
(Low Sperm Count) less than 20 million sperm after 72 hour abstinence from sex
Azoospermia
(Absent Sperm) blockage of duct network
Immotile Cilia Syndrome
Lack of sperm motility
Acephalic spermatozoa syndrome
Acephalic spermatozoa syndrome is characterized by the presence of very few intact spermatozoa and tailless sperm heads in the semen and leads to severe male infertility. Sad1 and UNC84 domain-containing 5 (SUN5) is a testis-specific nuclear envelope protein. A recent study has shown that mutations in SUN5 appear to affect the secondary structure of the protein and influence its folding and cellular localization.[29]
- Links: OMIM - SUN5
Additional Images
Human spermatozoa acrosomal protein SP-10[17]
Human spermatid electron micrograph[17]
Model capacitation-induced acrosome docking to sperm membrane[9]
Mouse spermiogenesis model[9]
Mouse- seminiferous tubule histology[9]
Rat Spermatogenesis figure[30]
Human spermatozoa - phospholipase C zeta localization[31]
Chemotaxis Model[24]
Labeled Chemotaxis Model[24]
Spermatogenesis androgen action[32]
Mouse spermatogenesis stages[33]
References
- ↑ Oliveira JB, Petersen CG, Massaro FC, Baruffi RL, Mauri AL, Silva LF, Ricci J & Franco JG. (2010). Motile sperm organelle morphology examination (MSOME): intervariation study of normal sperm and sperm with large nuclear vacuoles. Reprod. Biol. Endocrinol. , 8, 56. PMID: 20529256 DOI.
- ↑ Temporal trends in sperm count- a systematic review and meta-regression analysis
- ↑ Maree L, du Plessis SS, Menkveld R & van der Horst G. (2010). Morphometric dimensions of the human sperm head depend on the staining method used. Hum. Reprod. , 25, 1369-82. PMID: 20400771 DOI.
- ↑ Ho HC. (2010). Redistribution of nuclear pores during formation of the redundant nuclear envelope in mouse spermatids. J. Anat. , 216, 525-32. PMID: 20136667 DOI.
- ↑ Bastián Y, Roa-Espitia AL, Mújica A & Hernández-González EO. (2010). Calpain modulates capacitation and acrosome reaction through cleavage of the spectrin cytoskeleton. Reproduction , 140, 673-84. PMID: 20716611 DOI.
- ↑ Pellegrini M, Di Siena S, Claps G, Di Cesare S, Dolci S, Rossi P, Geremia R & Grimaldi P. (2010). Microgravity promotes differentiation and meiotic entry of postnatal mouse male germ cells. PLoS ONE , 5, e9064. PMID: 20140225 DOI.
- ↑ Cheng YH, Wong EW & Cheng CY. (2011). Cancer/testis (CT) antigens, carcinogenesis and spermatogenesis. Spermatogenesis , 1, 209-220. PMID: 22319669 DOI.
- ↑ Hunter D, Anand-Ivell R, Danner S & Ivell R. (2012). Models of in vitro spermatogenesis. Spermatogenesis , 2, 32-43. PMID: 22553488 DOI.
- ↑ 9.0 9.1 9.2 9.3 9.4 9.5 Borg CL, Wolski KM, Gibbs GM & O'Bryan MK. (2010). Phenotyping male infertility in the mouse: how to get the most out of a 'non-performer'. Hum. Reprod. Update , 16, 205-24. PMID: 19758979 DOI.
- ↑ Clermont Y. (1966). Spermatogenesis in man. A study of the spermatogonial population. Fertil. Steril. , 17, 705-21. PMID: 5920556
- ↑ Clermont Y. (1972). Kinetics of spermatogenesis in mammals: seminiferous epithelium cycle and spermatogonial renewal. Physiol. Rev. , 52, 198-236. PMID: 4621362 DOI.
- ↑ Chaturvedi PK & Johnson L. (1993). Architectural arrangement of stages of the spermatogenic cycle within human seminiferous tubules is related to efficiency of spermatogenesis. Cell Tissue Res. , 273, 65-70. PMID: 8364962
- ↑ Naylor GJ, McNamee HB & Moody JP. (1970). The plasma control of erythrocyte sodium and potassium metabolism in depressive illness. J Psychosom Res , 14, 179-86. PMID: 5477358
- ↑ Kierszenbaum AL & Tres LL. (2004). The acrosome-acroplaxome-manchette complex and the shaping of the spermatid head. Arch. Histol. Cytol. , 67, 271-84. PMID: 15700535
- ↑ Rathke C, Baarends WM, Awe S & Renkawitz-Pohl R. (2014). Chromatin dynamics during spermiogenesis. Biochim. Biophys. Acta , 1839, 155-68. PMID: 24091090 DOI.
- ↑ Saperas N & Ausió J. (2013). Sperm nuclear basic proteins of tunicates and the origin of protamines. Biol. Bull. , 224, 127-36. PMID: 23995738 DOI.
- ↑ 17.0 17.1 17.2 17.3 Westbrook VA, Schoppee PD, Vanage GR, Klotz KL, Diekman AB, Flickinger CJ, Coppola MA & Herr JC. (2006). Hominoid-specific SPANXA/D genes demonstrate differential expression in individuals and protein localization to a distinct nuclear envelope domain during spermatid morphogenesis. Mol. Hum. Reprod. , 12, 703-16. PMID: 17012309 DOI.
- ↑ 18.0 18.1 Iranpour FG. (2014). The effects of protamine deficiency on ultrastructure of human sperm nucleus. Adv Biomed Res , 3, 24. PMID: 24592371 DOI.
- ↑ Fawcett DW. (1970). A comparative view of sperm ultrastructure. Biol. Reprod. , 2, Suppl 2:90-127. PMID: 5521054
- ↑ Lillie FR. (1912). THE PRODUCTION OF SPERM ISO-AGGLUTININS BY OVA. Science , 36, 527-30. PMID: 17735765 DOI.
- ↑ Strünker T, Goodwin N, Brenker C, Kashikar ND, Weyand I, Seifert R & Kaupp UB. (2011). The CatSper channel mediates progesterone-induced Ca2+ influx in human sperm. Nature , 471, 382-6. PMID: 21412338 DOI.
- ↑ Johannessen JV. (1992). [Physicians and leadership]. Tidsskr. Nor. Laegeforen. , 112, 2950. PMID: 1412339
- ↑ Armon L & Eisenbach M. (2011). Behavioral mechanism during human sperm chemotaxis: involvement of hyperactivation. PLoS ONE , 6, e28359. PMID: 22163296 DOI.
- ↑ 24.0 24.1 24.2 Teves ME, Guidobaldi HA, Uñates DR, Sanchez R, Miska W, Publicover SJ, Morales Garcia AA & Giojalas LC. (2009). Molecular mechanism for human sperm chemotaxis mediated by progesterone. PLoS ONE , 4, e8211. PMID: 19997608 DOI.
- ↑ Kaupp UB, Kashikar ND & Weyand I. (2008). Mechanisms of sperm chemotaxis. Annu. Rev. Physiol. , 70, 93-117. PMID: 17988206 DOI.
- ↑ <pubmed>6772801</pubmed>
- ↑ Linck RW, Chemes H & Albertini DF. (2016). The axoneme: the propulsive engine of spermatozoa and cilia and associated ciliopathies leading to infertility. J. Assist. Reprod. Genet. , 33, 141-56. PMID: 26825807 DOI.
- ↑ Johnsen SG. (1970). Testicular biopsy score count--a method for registration of spermatogenesis in human testes: normal values and results in 335 hypogonadal males. Hormones , 1, 2-25. PMID: 5527187
- ↑ Shang Y, Yan J, Tang W, Liu C, Xiao S, Guo Y, Yuan L, Chen L, Jiang H, Guo X, Qiao J & Li W. (2018). Mechanistic insights into acephalic spermatozoa syndrome-associated mutations in the human SUN5 gene. J. Biol. Chem. , 293, 2395-2407. PMID: 29298896 DOI.
- ↑ Winawer SJ, Flehinger BJ, Buchalter J, Herbert E & Shike M. (1990). Declining serum cholesterol levels prior to diagnosis of colon cancer. A time-trend, case-control study. JAMA , 263, 2083-5. PMID: 2319669
- ↑ Aarabi M, Yu Y, Xu W, Tse MY, Pang SC, Yi YJ, Sutovsky P & Oko R. (2012). The testicular and epididymal expression profile of PLCζ in mouse and human does not support its role as a sperm-borne oocyte activating factor. PLoS ONE , 7, e33496. PMID: 22428063 DOI.
- ↑ Verhoeven G, Willems A, Denolet E, Swinnen JV & De Gendt K. (2010). Androgens and spermatogenesis: lessons from transgenic mouse models. Philos. Trans. R. Soc. Lond., B, Biol. Sci. , 365, 1537-56. PMID: 20403868 DOI.
- ↑ Phillips BT, Gassei K & Orwig KE. (2010). Spermatogonial stem cell regulation and spermatogenesis. Philos. Trans. R. Soc. Lond., B, Biol. Sci. , 365, 1663-78. PMID: 20403877 DOI.
Journals
- Spermatogenesis | PubMed - Spermatogenesis "Spermatogenesis is a new quarterly, peer-reviewed journal that will publish high-quality articles covering all aspects of spermatogenesis."
- WHO. WHO Laboratory Manual for the Examination and Processing of Human Semen. 5th ed. Geneva, Switzerland: World Health Organization; 2010. Online PDF
Reviews
Griswold MD. (2016). Spermatogenesis: The Commitment to Meiosis. Physiol. Rev. , 96, 1-17. PMID: 26537427 DOI.
Talwar P & Hayatnagarkar S. (2015). Sperm function test. J Hum Reprod Sci , 8, 61-9. PMID: 26157295 DOI.
Yoshida S. (2010). Stem cells in mammalian spermatogenesis. Dev. Growth Differ. , 52, 311-7. PMID: 20388168 DOI.
Hogarth CA & Griswold MD. (2010). The key role of vitamin A in spermatogenesis. J. Clin. Invest. , 120, 956-62. PMID: 20364093 DOI.
Ruwanpura SM, McLachlan RI & Meachem SJ. (2010). Hormonal regulation of male germ cell development. J. Endocrinol. , 205, 117-31. PMID: 20144980 DOI.
Hermo L, Pelletier RM, Cyr DG & Smith CE. (2010). Surfing the wave, cycle, life history, and genes/proteins expressed by testicular germ cells. Part 1: background to spermatogenesis, spermatogonia, and spermatocytes. Microsc. Res. Tech. , 73, 241-78. PMID: 19941293 DOI.
Hermo L, Pelletier RM, Cyr DG & Smith CE. (2010). Surfing the wave, cycle, life history, and genes/proteins expressed by testicular germ cells. Part 2: changes in spermatid organelles associated with development of spermatozoa. Microsc. Res. Tech. , 73, 279-319. PMID: 19941292 DOI.
Kaupp UB, Kashikar ND & Weyand I. (2008). Mechanisms of sperm chemotaxis. Annu. Rev. Physiol. , 70, 93-117. PMID: 17988206 DOI.
Eddy EM, Toshimori K & O'Brien DA. (2003). Fibrous sheath of mammalian spermatozoa. Microsc. Res. Tech. , 61, 103-15. PMID: 12672126 DOI.
de Rooij DG & Russell LD. (2000). All you wanted to know about spermatogonia but were afraid to ask. J. Androl. , 21, 776-98. PMID: 11105904
Clermont Y. (1972). Kinetics of spermatogenesis in mammals: seminiferous epithelium cycle and spermatogonial renewal. Physiol. Rev. , 52, 198-236. PMID: 4621362 DOI.
Articles
LEBLOND CP & CLERMONT Y. (1952). Definition of the stages of the cycle of the seminiferous epithelium in the rat. Ann. N. Y. Acad. Sci. , 55, 548-73. PMID: 13139144
NCBI Bookshelf
- StemBook [Internet]. Cambridge (MA): Harvard Stem Cell Institute; 2008 Regulation of spermatogonia
MBoC - Sperm | MBoC - Highly simplified drawing of a cross-section of a seminiferous tubule in a mammalian testis | MBoC - Cytoplasmic bridges in developing sperm cells and their precursors
- NCBI Bookshelf spermatozoa | spermatogenesis | spermiogenesis
Search
- Pubmed spermatozoa | spermatogenesis | spermiogenesis
Terms
Note there are additional glossaries associated with genital, spermatozoa, oocyte and renal.
- acroplaxome - structure forms the acrosome plate with intermediate filament bundles of the marginal ring at the leading edge of the acrosome. The sub-acrosomal layer located in the developing spermatozoa head perinuclear region, located between the inner acrosomal membrane and the nuclear envelope. The other part of the perinuclear region is the post-acrosomal sheath (PAS) at the post-acrosomal region.
- acrosome - Cap-shaped cellular structure formed from the golgi apparatus and contains enzymes to dissolve the oocyte (egg) zona pellucida for fertilisation.
- acrosome compaction - Acrosome reshaping process in final stages of spermatogenesis (spermatid to spermatozoa).
- acrosome reaction - Chemical change within the spermatozoa following binding to the zona pellucida, only acrosome reacted spermatozoa have an ability to fuse with oocytes.
- annulus - Cytoskeletal (septin) structure located between the midpiece and principal piece regions of the tail, thought to form a diffusion barrier between these two domains. PMID 20042538
- asthenozoospermia - (asthenospermia) Term for reduced sperm motility and can be the cause of male infertility.
- axoneme - (axonema) The basic structure in cilia and eukaryotic flagella and in the spermatozoa tail, consisting of parallel microtubules in a characteristic "9 + 2" pattern. This pattern describes 9 outer microtubule doublets (pairs) surrounding 2 central singlet microtubules, in humans 50 μm long. The motor protein dynenin move the outer microtubules with respect to the central pair, bending the cilia and generating motility. Note that prokaryotic bacteria have a similar process (flagellum) that uses an entirely different mechanism for motility.
- blood-testis barrier - (BTB) Formed by tight junctions, basal ectoplasmic specializations, desmosome-like junctions and gap junctions between adjacent sertoli cells near the basement membrane of the seminiferous epithelium. image - BTB | image - tight junction
- capacitation - term describing the process by which spermaozoa become capable of fertilizing an oocyte, requires membrane changes, removal of surface glycoproteins and increased motility.
- caput - proximal head of the epididymis, epithelium with stereocilia, involved in absorbing fluid to concentrate spermatozoa. Underlying smooth muscle aids movement. Epididymis three main parts : caput (head), corpus (body), cauda (tail).
- CatSper - cationic (Ca2+) channel of spermatozoa, progesterone activated involved in hyperactivation, acrosome reaction, and possibly chemotaxis.
- cauda - distal tail of the epididymis, region with a thin epithelium and the greatest quantity of smooth muscle. Epididymis three main parts : caput (head), corpus (body), cauda (tail).
- centriole - a microtubule organising centre. First required for axoneme formation (distal centriole) that is lost and a second for pronuclei formation (proximal) following fertilisation. Rodents loose both and only have maternal centrioles.
- connecting piece - linkage between the spermatozoa head and the midpiece of the tail. PMID 22767409
- corpus - elongated body of the epididymis, This has an intermediate thickness of epithelium and thicker smooth muscle layer than caput. Epididymis three main parts : caput (head), corpus (body), cauda (tail).
- cytoplasmic bridges - Transient cytoplasm connections between spermatids arising from one spermatogonium due to incomplete cytokinesis.
- diploid - (Greek, di = double + ploion = vessel) Having two sets of chromosomes, the normal state for all cells other than the gametes.
- end piece - Last portion of the spermatozoa tail region.
- epididymis - testis tubular structure connecting the efferent ducts to the ductus deferent and functions for the storage and maturation of spermatozoa. Epididymis three main parts : caput (head), corpus (body), cauda (tail). PMID27307387
- fibrous sheath - cytoskeletal structure surrounding the axoneme and outer dense fibers, defining the extent of the principal piece region.
- haploid - (Greek, haploos = single) Having a single set of chromosomes as in mature germ/sex cells (oocyte, spermatozoa) following reductive cell division by meiosis. Normally cells are diploid, containing 2 sets of chromosomes.
- interstitial cell - (Leydig cell) Male gonad (testis) cell which secrete the androgen testosterone, beginning in the fetus.
- interstitium - testis developmental region (space between testis cords) that generates Leydig cells and other less well characterized cell types.
- Johnsen score - a clinical score (1-10) for assessing spermatogenesis in a human testicular biopsy. Named after the author of the original article. PMID 5527187
- Leydig cell - (interstitial cell) Male gonad (testis) cell that secrete the androgen testosterone, beginning in the fetus. Fetal Leydig cells develop from coelomic epithelium and undifferentiated perivascular cells in the gonad–mesonephros border region. Adult Leydig cells appear after birth from stem/progenitor cells among peritubular and peri-vascular cells. Leydig cells were first histologically identified in 1850 by Franz von Leydig (1821 - 1908) a German scientist.
- meiosis - The cell division that occurs only in production of germ cells where there is a reduction in the number of chromosomes (diploid to haploid) which is the basis of sexual reproduction. All other non-germ cells in the body divide by mitosis.
- midpiece - (middle piece) spermatozoa tail initial segment of axoneme surrounded outer dense fibres then by mitochondria. Next in the tail is the principal piece then finally the end piece.
- mitosis - The normal division of all cells, except germ cells, where chromosome number is maintained (diploid). In germ cell division (oocyte, spermatozoa) meiosis is a modified form of this division resulting in reduction in genetic content (haploid). Mitosis, division of the nucleus, is followed by cytokinesis the division of the cell cytoplasm and the cytoplasmic contents. cytokinesis overlaps with telophase.
- outer dense fibres - (ODF, outer dense fibers) cytoskeletal structures that surround the axoneme in the middle piece and principal piece of the spermatozoa tail.
- primary spermatocyte - arranged in the seminiferous tubule wall deep (luminal) to the spermatogonia. These large cells enter the prophase of the first meiotic division. (More? meiosis)
- principal piece - Spermatozoa tail segment containing the plasma membrane calcium channels (CatSper1 and CatSper2) required for hyperactivation of motility. Region is partially separated from the midpiece by a barrier called the annulus.
- sertoli cells - (sustentacular cell) These cells are the spermatozoa supporting cells, nutritional and mechanical, as well as forming a blood-testis barrier. The cell cytoplasm spans all layers of the seminiferous tubule. The cells are named after Enrico Sertoli (1842 - 1910), and italian physiologist and histologist.
- sperm annulus - (Jensen's ring; Latin, annulus = ring) A region of the mammalian sperm flagellum connecting the midpiece and the principal piece. The annulus is a septin-based structure formed from SEPT1, 4, 6, 7 and 12. Septins are polymerizing GTPases that can act as a scaffold forming hetero-oligomeric filaments required for cytokinesis and other cell cycle roles.
- spermatogenesis - (Greek, genesis = origin, creation, generation) The term used to describe the process of diploid spermatagonia division and differentiation to form haploid spermatazoa within the testis (male gonad). The process includes the following cellular changes: meiosis, reoorganization of DNA, reduction in DNA content, reorganization of cellular organelles, morphological changes (cell shape). The final process of change in cell shape is also called spermiogenesis.
- spermatogenesis - (Greek, genesis = origin, creation, generation) The maturation process of the already haploid spermatazoa into the mature sperm shape and organization. This process involves reorganization of cellular organelles (endoplasmic reticulum, golgi apparatus, mitochondria), cytoskeletal changes (microtubule organization) and morphological changes (cell shape, acrosome and tail formation).
- spermatogonia - The cells located in the seminiferous tubule adjacent to the basal membrane that either divide and separate to renew the stem cell population, or they divide and stay together as a pair (Apr spermatogonia) connected by an intercellular cytoplasmic bridge to differentiate and eventually form spermatazoa.
- spermatozoa head - Following spermiogenesis, the first region of the spermatozoa containing the haploid nucleus and acrosome. In humans, it is a flattened structure (5 µm long by 3 µm wide) with the posterior part of nuclear membrane forming the basal plate region. The human spermatozoa is about 60 µm long, actively motile and divided into 3 main regions (head, neck and spermatozoa tail).
- spermatozoa neck - Following spermiogenesis, the second region of the spermatozoa attached to basal plate, transverse oriented centriole, contains nine segmented columns of fibrous material, continue as outer dense fibres in tail. In humans, it forms a short structure (1 µm). The human spermatozoa is about 60 µm long, actively motile and divided into 3 main regions (head, neck and tail).
- spermatozoa tail - Following spermiogenesis, the third region of the spermatozoa that has a head, neck and tail). The tail is also divided into 3 structural regions a middle piece, a principal piece and an end piece. In humans: the middle piece (5 µm long) is formed by axonema and dense fibres surrounded by mitochondria; the principal piece (45 µm long) fibrous sheath interconnected by regularly spaced circumferential hoops; the final end piece (5 µm long) has an axonema surrounded by small amount of cytoplasm and plasma membrane.
- spermatogonial stem cells - (SSCs) The spermatagonia cells located beside the seminiferous tubule basal membrane that either divide and separate to renew the stem cell population, or they divide and stay together as a pair (|Apr spermatogonia) connected by an intercellular cytoplasmic bridge to differentiate and eventually form spermatazoa.
- spermatozoon - singular form of of spermatozoa.
- sperm protein 56 - A component of the spermatozoa acrosomal matrix released to the sperm surface during capacitation.
- teratospermia - Clinical term for a spermatozoa with abnormal morphology (small, large, defects in the head, tail, and/or mid-piece) present in the semen or ejaculate.
- testis cords - developmental structure that give rise to the adult seminiferous tubules, the other developmental region is the interstitium.
- vasectomy - Clinical term for ligation of the scrotal portion of the ductus deferens.
See also: Spermatozoa Terms collapse table
| Other Terms Lists |
|---|
| Terms Lists: ART | Birth | Bone | Cardiovascular | Cell Division | Endocrine | Gastrointestinal | Genital | Genetic | Head | Hearing | Heart | Immune | Integumentary | Neonatal | Neural | Oocyte | Palate | Placenta | Radiation | Renal | Respiratory | Spermatozoa | Statistics | Tooth | Ultrasound | Vision | Historic | Drugs | Glossary |
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- World Health Organization - WHO Laboratory Manual for the Examination and Processing of Human Semen. 5th ed. Geneva, Switzerland: World Health Organization; 2010. Online PDF
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, June 16) Embryology Spermatozoa Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Spermatozoa_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G



![Human spermatozoa acrosomal protein SP-10[17]](/embryology/images/thumb/a/a8/Human_spermatozoa_acrosomal_protein_SP-10.jpg/111px-Human_spermatozoa_acrosomal_protein_SP-10.jpg)
![Human spermatid electron micrograph[17]](/embryology/images/thumb/8/8c/Human_spermatid_electron_micrograph.jpg/81px-Human_spermatid_electron_micrograph.jpg)
![Model capacitation-induced acrosome docking to sperm membrane[9]](/embryology/images/thumb/2/2e/Model_capacitation-induced_acrosome_docking_to_sperm_membrane.jpg/120px-Model_capacitation-induced_acrosome_docking_to_sperm_membrane.jpg)
![Mouse spermiogenesis model[9]](/embryology/images/thumb/4/4f/Mouse_spermiogenesis_model.png/120px-Mouse_spermiogenesis_model.png)
![Mouse- seminiferous tubule histology[9]](/embryology/images/thumb/0/04/Mouse-_seminiferous_tubule_histology.jpg/120px-Mouse-_seminiferous_tubule_histology.jpg)
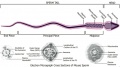
![Rat Spermatogenesis figure[30]](/embryology/images/thumb/3/3e/Spermatogenesis_cartoon_01.jpg/120px-Spermatogenesis_cartoon_01.jpg)
![Human spermatozoa - phospholipase C zeta localization[31]](/embryology/images/thumb/f/fd/Human_spermatozoa_phospholipase_C_zeta.jpg/120px-Human_spermatozoa_phospholipase_C_zeta.jpg)
![Chemotaxis Model[24]](/embryology/images/thumb/d/da/Human_spermatozoa_chemotaxis_model.jpg/120px-Human_spermatozoa_chemotaxis_model.jpg)
![Labeled Chemotaxis Model[24]](/embryology/images/thumb/8/8a/Human_spermatozoa_chemotaxis_labeled_model.jpg/120px-Human_spermatozoa_chemotaxis_labeled_model.jpg)
![Spermatogenesis androgen action[32]](/embryology/images/thumb/4/49/Spermatogenesis_androgen_action_cartoon.jpg/120px-Spermatogenesis_androgen_action_cartoon.jpg)
![Mouse spermatogenesis stages[33]](/embryology/images/thumb/5/51/Mouse_spermatogenesis_stage_cartoon.jpg/120px-Mouse_spermatogenesis_stage_cartoon.jpg)