Paper - The Anatomy of a 17.8 mm Human Embryo
| Embryology - 27 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Thyng FW. The anatomy of a 17.8 mm human embryo. (1914) Amer. J Anat. 17: 31-112.
| Online Editor |
|---|
| These historic 1914 drawings by Thyng are based on studies of the Harvard Embryological Collection while he was in Minot's Lab in 1907-08. He was also an anatomist at Northwestern University Medical School. The embryo external appearance and dimensions suggest that it is a Carnegie stage 19 embryo (Week 7, 48 - 51 days, 16 - 18 mm).
|
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
The Anatomy of a 17.8 mm Human Embryo
by Thyng, FW (1914)
The Department of Comparative Anatomy of Harvard Medical School
Eight Figures
This (original) work has been aided by a grant from the Elizabeth Thompson Science Fund.
Contents
|
|
|
Introduction
This work was undertaken at the suggestion of Prof. C. S. Minot, and carried on for the most part in his laboratory during the years 1906 and 1907 when the author held an Austin Teaching Fellowship in Histology and Embryology at the Harvard Medical School. Its completion, unfortunately delayed by other work, has been accomplished in the Anatomical Department of the University and Bellevue Hospital Medical College. During the progress of the work many helpful suggestions have been received from Professors Minot and F. T. Lewis of Harvard, and Prof. H. D. Senior of Bellevue, for which I am very grateful. I also desire to express my gratitude to Mr. W. T. Oliver of Lynn, Massachusetts, for the careful manner in which he has reproduced in finished form my original drawings.
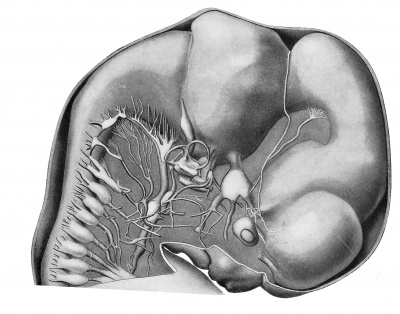
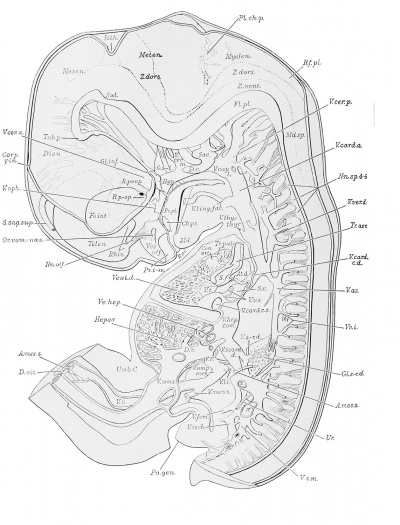
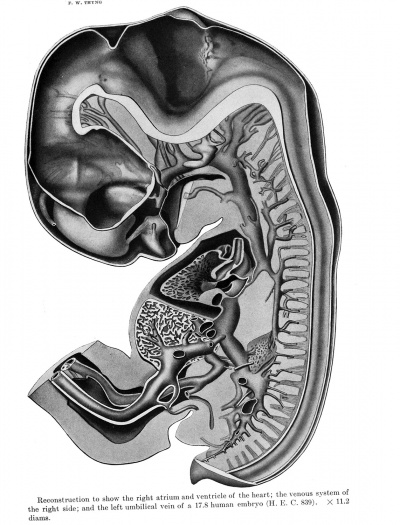
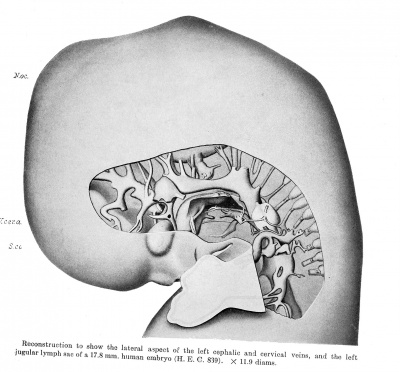
The reconstructions upon which this work is based, were made from transverse sections of Embryo 839 of the Harvard Embryological Collection, chiefly by the modified graphic reconstruction method of His. The shading usually has been inferred from a study of the sections, but in a few instances wax models were made of regions requiring a fuller interpretation.
This embryo (extra-uterine) measured in formalin 17.8 mm, greatest length, with a neck breech of 16.7 mm. The greatest length in 80 per cent alcohol was 13.6 mm. In previous papers in which this embryo was referred to (Thyng '08, and Lewis and Thyng '08), the latter measurement was given.
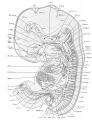
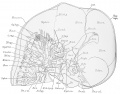
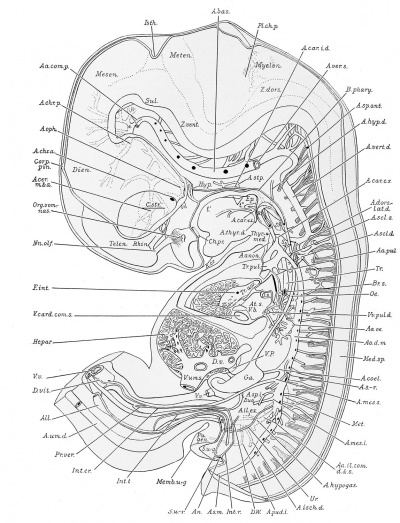
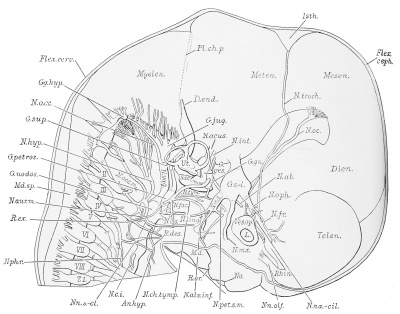
External Features
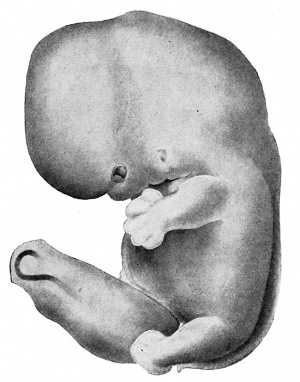
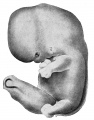
The external features of this embryo are seen in profile view in text figure 1, a reproduction of figure 104 in Minot's ('10) "Laboratory text-book of embryology," also in part in plates 3 and 5. The neck-bend is approximately a right angle; the cephalic flexure is also very nearly a right angled bend, so that the oral aperture is in close proximity to the cardiac region. The dorsal flexure has disappeared almost entirely, only a slight elevation persisting to mark its earlier position. Above this elevation there is a shallow depression, said to disappear in the course of development.
A distinct groove, extending transversely l)etween the medial angles of the developing eyes, separates the forehead from the root of the nose. The maxillary process of either side has joined the adjacent lateral and median nasal processes, obliterating the naso-optic grooves. The nares are open, but separated by a rather low, broad sei)tum. A triangular space still intervenes between the globular processes so that the median region of the upper lip is not well differentiated. The median groove between the ventral ends of the mandibular arches has been obliterated, but differentiation between the chin and lip regions has not occurred. The line of fusion between the first and second branchial arches is marked ventrally by a transverse groove, dorsal to which is seen the fossa conchae. The grooves between the other branchial arches have disappeared. 'A reproduction of figure 104, page 153 of "Laboratory Textbook of Embryology," Charles Sedgwick Minot, edition of 1910, published by P. Blakiston's Son and Company, Philadelpliia.
The limb buds extend nearly perpendicularly to the longitudinal axis of the body. The upper project slightly beyond the ventral border of the body, and show a differentiation of arm, forearm, and clearly outlined digits. The latter protrude slightly beyond the border of the hand-plate. Upon the lower limb buds are slight indications of developing toes.
Circular thickenings of the epidermis on the lateral body walls mark the developing mammae. In section these thickenings appear slightly convex on the surface, and project into the underlying mesenchyma. The umbilical cord as it leaves the body wall bends towards the right.
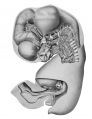
Digestive System
Oral cavity
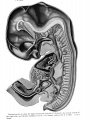
The primitive oral cavity appears in median sagittal section in plate 1, but is represented more fully in plate 4, a portion of the tongue having been cut away. It is a short, dorso-ventrally compressed passage consisting of a roof and a floor, the epithelia of which meet laterally at the angle of the mouth. The external aperture of the primitive oral cavity, the rima oris, (R.or.) is indicated as seen from the exterior in plates 2, 3 and 5; as seen in median sagittal section in plates 1 and 4. It is a narrow, horizontal expanded orifice, concave dorsally where it is bounded by the fused maxillary and median nasal processes, convex ventrally where it is formed by the united mandibular processes.
Dorsally the oral cavity communicates with the pharynx, the division between the two being marked in the median line on the roof by the stalk of the hypophysis (Hyp.) While the roof, as thus bounded, is of considerable extent, the floor is very limited, consisting of merely the anlage of the lower lip and teeth.
The lip-grooves are just beginning to indent the oral epithelium. The lower lip-groove is seen in sagittal section in plate 1. Into the anterior part of the roof of the prhnitive oral cavity open the large primitive choanae (Ch.pr.) of the olfactory vesicles, separated from one another by a primitive nasal septmii. The roof also presents on either side of the median line, a prominent longitudinal ridge, the palate process (Pr.pl.). This process of the right maxillary arch is clearly shown in plate 4 where a portion of the tongue has been removed. It begins at the intermaxillary process iPr.i.m.) and extends dorsally, lateral to the choana (Ch.pr.) along the primitive oral cavity and the cephalic part of the pharynx. Its free ventral border nearly reaches the floor of the pharynx in the region of the alveolo-lingual groove. The roof of the oral cavity between the palate processes thus forms a high arch which receives the dorsum of the developing tongue. It is evident that the primitive oral cavity now comprises a portion which will be cut off later by the union of the palate processes and nasal septum, to fonn in part the nasal cavities of the adult.
The hypophysis (Hyp.), alluded to above, consists of a distal spade-like portion, connected to the oral epithelium in the median line by a slender stalk with reduced lumen. It is represented in side view in plate 4, in median sagittal section in plate 1. Its flattened body impinges upon the ventral surface of the infundibulum, on either side of which it projects dorsally as a short, blunt process. On its cephalic surface there is a distinct ridge or fold, continuous with the anterior surface of the stalk.
Pharynx. A left lateral view of the entodermal wall of the pharynx is seen in plates 2 and 5. The interior, as it appears in median sagittal section, is represented in plate 1. It is a broad, dorso-ventrally compressed canal which narrows rapidly in passing caudally to divide into trachea (Tr.) and oesophagus (Oe.). The epithelium of the roof and floor of the pharynx meet to form an external ridge, which extends from near the angle of the mouth to the lateral border of the oesophagus. Corresponding with the ridge there is an internal furrow.
The tongue (plate 1) is a comparatively broad elevation of the floor of the pharynx, composed of a large cephalic part (t') intimately fused with a smaller caudo-lateral division (t"), the root. A surface view of the dorsum of the tongue would show the line of fusion of these two parts to be a V-shaped groove with the apex of the V pointing caudad. In plate 1 , the apex and right hmb of this groove, sulcus tenninalis, are represented, the apex marking the place of origin of the median thyreoid gland. Laterally the tongue is bounded by deep alveolo-lingual grooves which converge cephalad so as to separate its tip from the subjacent mandible.
According to Kallius ('01) the anterior anlage of the tongue is derived chiefly from the dorsum of the ventral ends of the mandibular processes, the so-called lingual folds, the tuberculum impar of His contributing only a small part thereto. Hammar, however, believes ('01), that the tuberculum impar is a transitory structure, and that the tip and body of the tongue are formed by a considerable area of the floor of the oral (pharyngeal) cavity. In regard to the development of the root of the tongue there is some disagreement in that His derived it ('85) from the ventral ends of the second and third visceral arches, while Born ('83) and Hammar ('01) limit it to the second arch.
Posterior to the root of the tongue, and fused with it, there is a broad, bilobate elevation (Ep.) which represents the epiglottis. It is a derivative of the third visceral arch (Born '83, and Hammar '01).
The first pharyngeal pouch (Ph.P.l, plate 5), the cavity of which ultimately, will form the tuba auditiva and cavum tympani, is seen at this stage to be an extensive, lateral, pointed, evagination of the pharyngeal wall, extending somewhat dorsally toward the depression of the primary meatus acusticus externus. It presents three surfaces, dorsal, cephalo-ventral, and caudoventral. The dorsal surface, which cannot be seen in the drawing, is triangular in outline; medially it passes over into the dorsolateral wall of the pharynx. Dorsal to it is the cochlear division of the otocyst. The caudal boundary of this surface is the posterior tympanal ridge. The cephalic boundary is marked by a ridge overlying the tubo-tympanal sulcus of Moldenhauer ('77). This ridge extends from the tip of the first pharyngeal pouch in a cephalic and medial direction to the oral epithelium between the hypophysis and the angle of the mouth. The cephalo-ventral surface of the first pharyngeal pouch is concave. A dorso-ventral ridge, representing the entodernial part of the first closingplate, separates it from the caudo-ventral surface. Upon the caudo-ventral surface near the tip of the pouch there is a slight groove. This, the 'tensor groove' of Hammar ('02), marks the place of formation of the tensor tendon.
The second pharyngeal pouch (Ph.P.2, plate 2) appears on either side as a low evagination from the lateral pharyngeal wall. It is situated just caudal to the first pouch and projects towards the cephalic aspect of the glossopharyngeal nerve. According to Hammar ('03) this evagination represents only the dorsal part of the primary pouch. A deeply staining cyst is present in this embryo on either side of the pharynx, w^hich evidently belongs to either the ectodermal or entodernial part of the second branchial groove. The left cyst is situated just lateral to the left glossopharyngeal nerve, while the right is just cephalad of the right glossopharyngeal nerve and in close relation to the second pouch. Piersol ('88) found that in rabbit embryos there were formed, in the development of the second pharyngeal pouches, two epithelial tubes on either side, one from the entoderm and the other from the ectodenn, both of which subsequently atrophied. The former, however, persisted longer than the latter. Hammar ('03) described and figured a structure in human embryos protruding above the margin of the tonsilar pouch. In early stages this was connected with the ectoderm, and hence he concluded that it was of ectodermal origin. Fox ('08) did not find any ectodermal remnant in this region of the pig embryo, but described and figured a long filiform process continuous with each of the second pharjmgeal pouches.
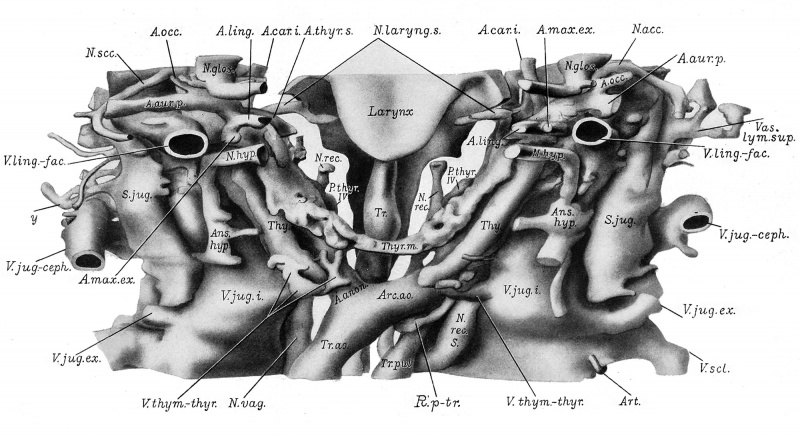
The third pharyngeal pouches (plates 2 and 6) have lost their connection with the pharynx. They are now represented each by a compact cylinder (Thy., plates 2 and 6) in the side of the neck, and which contains only a slight lumen. The cylinders converge caudally toward the median line and end approxhnately at the level of the aortic arch (Arc.ao.). The right and left cylinders become, eventually, the corresponding lobes of the thymus gland with the exception of the cephalic extremities which are compact epithelial masses (not marked off in the figures) differing in structure from the rest of the anlagen. The cephalic portion of each cylinder is closely applied to the lateral wall of the common carotid artery, and is the part described by Katschenko ('87) as the nodulus thymicus, and by Fox ('08) as the carotid gland. The recent work of Hammar ('11) substantiates the view that this part eventually becomes separated from the thymic cylinder and (coming to lie at the caudo-dorsal border of the lateral part of the thyreoid gland) forms with its fellow of the other side the caudal pair of para-thyreoids. The cephalic ends of the thymic cylinders also show two hollow projections, a medial one, extending toward the pharynx and ending blindly, dorsal to the common carotid artery ; and a more lateral process extending cephalad and ending blindly alongside the ventro-medial surface of the vagus nerve where the latter is crossed on its lateral side by the hypoglossal. The former or medial of these processes is evidently the remains of the thymo-pharyngeal duct. The lateral process seems to be the remains of the cervical sinus fused with the third pharyngeal pouch, as maintained for corresponding structures by Katschenko ('87), Fox ('08), and Hammar ('11), and not an outgrowth from the thymus as conjectured by Prenant ('94) and Bell ('05). Katschenko from a study of this structure in the pig, maintained that it formed a considerable portion of the head of the thymus, a view since corroborated by Prenant ('94) who, however, considers it of entodermal origin. Fox ('08) found that in the cat it apparently atrophied early in development, and that in later stages of the rabbit it had largely, if not entirely disappeared. He is inclined to think that, when it does persist, it does not form an integral part of the thymus, but merely an associated structvire.
The fourth pharyngeal pouch of either side, exclusive of the so-called ultimobranchial (postbranchial) body (Greil '05) is represented by a solid epithelial mass (P-thijr. IV) seen in plates 2 and 6. These masses, which represent the cephalic pair of parathyreoids, are situated dorsal to the lateral lobe of the thyreoid and are entirely separate from the pharynx. That on the right side is bilobate and somewhat removed from the prohferating entoderm of the thyreoid, but on the left the two are intimately connected.
The thyreoid gland (plates 2 and 6) is distinctly U-shaped, with the concavity of the U directed cephalad. The level of the slender connecting bar of the median thyreoid (Thyr.m.) is seen in plates 1 and 6. The arms of the U, derivatives in part of the ultimobranchial bodies (which are still discernible although intimately connected with the median thyreoid) are widened considerably dor so-ventr ally, and terminate at a level corresponding with the cephalic ends of the thymic cylinders. Numerous proliferating cords of cells make its surface somewhat irregular. Its connection on the left with the parathyreoid anlage of the fourth pharyngeal pouch has been referred to above.
Salivary glands
The parotid and submaxillary glands are shown in plates 2 and 5. The parotid gland is represented by a small, solid cord of cells (Gl.p.) partly constricted off from the ridge leading caudad from the angle of the mouth and overlying the sulcus buccalis. The submaxillary gland is represented by a solid cord of cells (Gl.s.) but is larger than the parotid anlage. It projects from the caudal part of the floor of the alveolo-lingual groove into the underlying mesenchyma which for some space around the gland consists of closely packed cells for the development of a capsule. Its extremity is broader than the stalk, and shows slight indications of proliferating buds. The anlagen of the sublingual glands have not developed at this stage. That they are the latest of the salivary glands to develop, has been noted by His ('85), Chievitz ('85), Hammar ('01), Paulet ('10) and others.
Oesophagus
The oesophagus is comparatively long at this stage. The entodermal part of the tube only is figured in plates 1 and 2. This consists of an epithelium, containing four or more layers of nuclei. Numerous irregular cavities, as seen by Schultze ('97), and others, are found within the epithelium. They occur in scattered situations, and are separated from the surrounding mesenchyma usually by a single layer of columnar or cuboidal cells. In no case were they found to connect with the lumen of the oesophagus. They are apparently vacuoles as maintained by Forssner ('07), and Johnson ('10). A granular coagulum was invariably found within them, but Kreuter ('05) who has studied these structures concludes that a degeneration of cells does not occur. For a general account of these structures see Lewis ('12).
Stomach
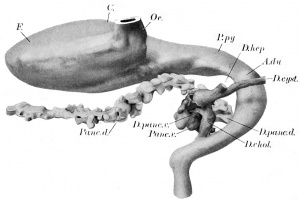
The stomach has assumed practically the adult position. It is represented by the entodermal lining only, in plate 1 and text figure 2 (Ga., C) . Its primitive dorsal border representing the greater curvature has revolved to the left, while its ventral border, now^ identified by the lesser curvature, faces toward the right. Its entodennal lining is an epithelium containing four or more layers of nuclei. It presents at the cardiac end a prominent, dorsally directed pouch, the fundus (F.), which according to Keith and Jones ('01) develops as a localized outgrowth. That this outgrowth is from the left side of the primitive stomach is evident, a relation evidenced in the adult by the reflection line of the lieno-gastric ligament. The body (C) extends caudad and ventrally, passing into the attenuated pars pylorica (P.py.) which ends at a dilated portion of the duodenum (A.du.), the duodenal antrum of Retzius ('57). This, according to Lewis ('12) always marks the position of the pylorus. The external surface of the epithelium is for the most part smooth, but the internal surface is indented by slight grooves representing the beginning of the gastric pits.
Intestine
Most of the small intestine and all of the large are shown in plates 1 and 2 (Duo., Int.t., Int.cr., Int.r.). The duodenal division of the small intestine (Duo.) leading from the pyloric end of the stomach passes transversely across the median line from the left to right. Here it bends dorsally and receives the duct of the dorsal pancreas (D.panc.d.), and the bile duct (D.chol.) (text fig. 2) . The small intestine (hit.t.) then extends in a caudal and ventral direction, a little to the right of the median line, into the umbilical cord. In the umbilical cord it is bent twice in the sagittal plane at approximately 90°. On the left side of the second or cephalic bend (^1.2) it is continuous with the yolk-stalk (D.vit.). The portion of the small intestine beyond the second bend returns toward the body, cephalad of the part described, and terminates at the caecum.
The caecum forms a considerable dilatation and ends in the vennifonn process (Pr.ver.) which projects ventrally and to the left.
The colon (Int.cr.) extends from the caecum, dorsally in the median plane, crossing to the left of the duodenum. Opposite the caudal extremity of the Wolffian body it turns caudally, and at an arbitrary point becomes the rectum (Int.r.). It is evident that the primitive U-shaped loop of intestine has undergone in this embryo a rotation of approximately 180°.
The epithelium of the duodenum a short distance caudad to the bile duct presents on its left side one prominent diverticulum, directed cephalad. Indications of similar outgrowths occur at twelve other places along the portion of the small intestine within the umbilical coelom. The lumen of the du(xlenum, beginning somewhat cephalad of the duct of the dorsal pancreas and extending caudad to the duodenal diverticulum above mentioned, is subdivided into two or three parts by a proliferation of the epithelium in a manner similar to that already described by Tandler ('00) in a human embryo of 14.5 mm. This observation has been confirmed by Forssner ('07), Johnson ('10) and Lewis ('12). Beyond this proliferation the lumen of the small intestine is either a small cylindrical passage or a slight slit.
The yolk-stalk has a lumen for a short distance beyond its connection with the epithelium of the intestine. It then becomes a cord of degenerating cells, showing here and there traces of a lumen. Whether the yolk-stalk is still connected with a rudimentary yolk-sac could not be determined as these parts were cut away in the embryo.
The lumen of the colon as it leaves the caecum, is a very small cylindrical passage, but as it nears the rectum, it becomes a transversely directed slit (corresponding to the shape of the intestine which is compressed dorso-ventrally) .
The cephalic portion of the rectum is a little larger in diameter than the colon, while the terminal part is more attenuated. The latter is circular in transverse section and the lumen is reduced to a very small cylindrical passage, which, however, does not connect with the shallow external depression (An.) between the protruding genital folds.
Liver
The liver (see plates 1, 2 and 4, Hepar) occupies the greater part of the cephalic and ventral regions of the abdominal cavity. The right lobe is much the larger, and extends from the cephalic end of the abdominal cavity on the right, caudad to a point on a level with the crossing of the duodenum by the colon. It is joined to the dorsal abdominal wall on the right of the dorsal mesogastrium, at the ventral region of the right suprarenal gland, by the plica venae cavae of Ravn ('89) (caval mesentery) .
The caudate lobe is located between the caval mesentery and the lesser omentum. It projects somewhat toward the left into the bursa omentalis. The position of the quadrate lobe can be determined from plate 2. It is situated to the right of the vesica fellea (Ves.fel.), lying between this and the umbilical vein (V.um.s., plate 4).
The hepatic duct (D.hep., plate 2, and text figure 2) takes origin from the hepatic trabeculae of the medial surface of the right lobe, ventral to the entrance of the portal vein (V.P.). It extends dorsally and to the left for a short distance and then caudad, uniting with the small cystic duct (Dxyst) to form the ductus choledochus (D.chol.).
The cystic duct has a slender lumen, and leads in a nearly dorso-ventral direction from a small, distal dilatation, the gallbladder (Ves.fel., plate 2). The latter is closely apphed to the ventro-medial surface of the right lobe of the liver.
The common bile duct is considerably larger in diameter than either the hepatic or cystic ducts. It has a well-defined lumen, and extends caudad through the lesser omentum in an S-shaped course to open into the duodenum. This it does upon its left side, a short distance beyond the pyloric end of the stomach. The epithelium of the gall-bladder and of the cystic and common bile ducts, is devoid of knob-like buds and diverticula, met with in these situations in other embryos.
Pancreas
The pancreas of this embryo has been described, and pictured in a previous paper (Thyng '08), but for the sake of completeness it will be described briefly in this connection. It consists of two parts, a dorsal and a ventral pancreas (Panc.d. and v., text figure 2). The dorsal pancreas is considerably the larger, and extends distally into the mesogastrium. It is essentially a long, irregular, hollow mass of epithelium with proliferating branches of varying length which in turn often give off hollow buds. Its duct (D.panc.d.) is larger than that of the ventral pancreas, contains a well-defined lumen, and opens into the left side of the duodenum nearer the stomach than the bile duct.
The ventral pancreas is in close relation with the proximal part of the dorsal anlage, the two having anastomosed ventral to the portal vein, and on the left of the common bile duct. Like the dorsal pancreas, the ventral also shows a branching condition of its epithelium. The duct of the ventral pancreas (D.panc.v.) is short, and opens into the bile duct (Dxhol.) near its entrance into the duodenum.
Respiratory System
The olfactory pits are described with the sense organs.
The entodermal lining of the larynx and trachea are represented as seen in ventral view in plate 6; from the left side in plate 2; and in the median sagittal section in plate 1. In plate 1 the anlage of the right lung is seen from the left side.
Larynx. The larynx, which opens from the pharynx by a T-shaped aperture, is placed immediately caudal to the epiglottis. The pedicle of the T or interarytaenoid notch, extends dorsoventrally between the arytaenoid protuberances, and is bounded laterally by the ary epiglottic folds. A median raph6 (R.) extends dorso-ventrally across the larynx, in a somewhat caudal direction, so as to close it temporarily.
Trachea. The trachea passes caudally to bifurcate into the two bronchi. The root of the left bronchus is shown in section in plate 1 (Br.s.) but, the greater portion of this bronchus and the corresponding lung have been removed.
Lungs. The entodermal outpocketings of the right lung and its pulmonary vessels (A.pul. and Vp.pul.d.) are represented in plate 1, as seen through the mediastinum. An eparterial (tracheal) bronchus is present on the right. It is situated dorsal to the right pulmonary artery, and a branch of the latter passes cephalad of this bronchus. The oesophagus (Oe.) passes between the developing lungs.
Pleural cavity. Each pleural cavity is closed off completely from the pericardial by the pleuro-pericardial membrane, but still communicates with the abdominal cavity. The aperture on the left is very small.
A blind prolongation of the right pleural cavity begins medially and dorsally to the anlage of the root of the right lung. From here it extends caudally and somewhat ventrally along the right side of the oesophagus to the diaphragm. It then passes between the oesophagus and diaphragm and ends at a situation approximately corresponding to the level of the most caudal extension of the right pleural cavity. This diverticulum is ventral to the main pleural cavity and evidently corresponds to the space, termed by Broman ('04) the infracardial bursa. The infracardial bursa in this case differs from that described by Broman in that the primitive connection with the pleural cavity has been retained. The retention of this connection is of interest in that such a recess of the right pleural cavity exists normally in animals possessing an infracardial lobe of the right lung, and may be expected to occur occasionally in man.
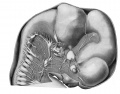
Urogenital System
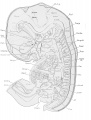
The parts of the urogenital system are shown in plate 2 in which the urogenital sinus, the left Wolffian body and duct, the left metanephros and ureter, the left genital ridge, and Miillerian duct are shown from the left side.
Wolffian ridge and body. The Wolffian ridge extends almost the entire length of the abdominal cavity, its anterior threefourths being occupied by the Wolffian body. The ridge tapers off bluntly at its cephalic end ; caudad of the area of mesonephritic tubules (T.W.) it dwindles to a slight elevation upon the abdominal wall in which the Wolffian duct (D.W.) passes to the urogenital sinus (S.u.-g.). Under the influence of the rapidly growing liver and suprarenal glands the ridge has moved laterally from its primitively dorso-medial position.
Wolffian duct. The Wolffian duct (D.W.) passes through the ventro-lateral region of the Wolffian body receiving the tributary mesonephric tubules (T.W.). From the caudal end of the Wolffian body it extends through the remainder of the Wolffian ridge to end by piercing the dorso-lateral wall of the urogenital sinus (S.u.-g.). Close to its entrance into the sinus there is a slight dorsal enlargement of the duct which suggests the first anlage of the seminal vesicle. It should be noted, however, that, according to the investigations of Pallin ('01), the vesiculus seminalis first ap])ears at a much later stage of development.
Müllerian duct. The Müllerian duct (D.M.) lies parallel with the Wolffian, being a little ventral to it. Its cephalic end opens freely into the abdominal cavity. The opening which occurs near the cephalic end of the Wolffian body, shows something of a fimbriated condition (Fim.). In the caudal direction the Miillerian duct seems to terminate in a blind pointed end close to the Wolffian duct. The point of termination is about on a level with the junction of the middle and caudal thirds of the Wolffian body.
Bladder
The ventral segment of the cloaca is divisible into two parts, a cephalic portion [B) which will ultimately form the bladder and a caudal portion (S.u.-g.) the urogenital sinus.
The region of the bladder anlage adjoining the urogenital sinus is expanded on either side to produce a dorso-lateral ridge. Into each ridge at the caudal extremity of the bladder anlage opens the corresponding ureter. The portion of the bladder remote from the urogenital sinus is continued as the allantois into the caudal wall of the umbilical cord. The proximal portion of the allantois consists of a solid cord of cells, the urachus, but distally isolated portions of the original lumen are found.
Urogenital sinus
The cephalic region of the urogenital sinus receives the Wolffian ducts. The orifices of these are placed on either side, a short distance from the median hne. The remainder of the sinus extends into the median caudal part of the genital tubercle (Pa.gen.) as a laterally compressed structure. In profile view this part of the sinus appears somewhat triangular in outline. Its dorsal region encloses a cavity, but its latero-ventral walls are approximated to form an incomplete raphe, the urogenital membrane (Mem.u.-g.). This membrane is broken down in its extreme caudal part so that the urogenital sinus opens to the exterior.
The lower part of the sinus becomes the vestibule in the female, while in the male it forms the main portion of the urethra. The female urethra and the proximal part of the male urethra being formed by a canal differentiated later between the bladder anlage and the urogenital sinus.
Metanephros
The metanephros (Met., pi. 2; see also pi. 1) is situated dorsal to the middle part of the Wolffian body, extending approximately from a level of the twelfth thoracic to the second lumbar nerve, and appears externally as an oval, lobulated body. It consists of approximately eight branches arising from the cephalic extremity of the ureter, the renal pelvis. Each branch ends in a bilobated ampulla which is surrounded by condensed 'nephrogenic tissue.' The ureter as it leaves the developing metanephros, extends at first caudally and medially in the ventral part of the dorsal body wall. It then passes ventrally in the lateral parieties and opens into the extreme lateral wall of the anlage of the bladder. The ureteral orifices are slightly cephalad of those by which the Wolffian ducts communicate with the urogenital sinus.
Genital ridges
The ovaries are two compact, longitudinal protuberances projecting from the medial borders of the Wolffian bodies. They taper at either extremity, more gradually at the cephalic than the caudal. The cephalic end of the left ovary (G.R.) is partly hidden from view in plate 2 by the Wolffian body, but it does not extend quite to the cephalic pole of the latter. The caudal end of the ovary is on a level with that of the mesonephros by w^hich it is hidden in the drawing. The cephalic region of each ovary is deeply marked by infolding of the germinal epithelium.
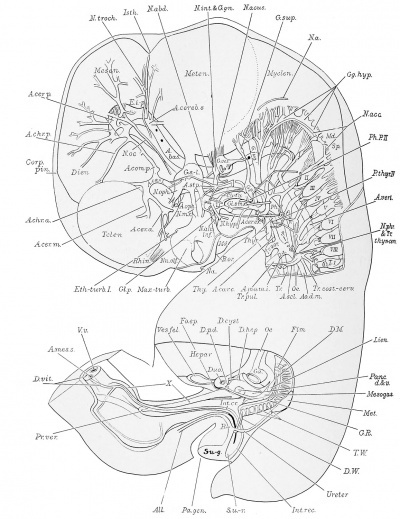
Ductless Glands
The right suprarenal is shown in plate 4, the spleen in plate 2. The thyreoid, thymus, and parathyreoid glands are described in connection with the pharynx (pp. 38-40) and shown in plates 2 and 6.
Suprarenal gland. The suprarenal glands (Crl.s.-r.) are somewhat oval bodies of considerable size, developing in the ventral region of the dorsal body wall between the Wolffian body and the dorsal aorta. Their cephalic extremities lie at the level of the ninth thoracic nerves, i.e., a short distance cephalad of the caudal extension of the pleural cavities, of which they form, in part, the caudo-lateral wall. Their caudal extremities lie on a level with the twelfth thoracic nerves, i.e., slightly beyond the cephalic extremity of the Wolffian body.
Each gland has developed in the path of the cephalic part of the subcardinal vein of that side, and the vein has been subdivided to fomi the sinusoidal channels now found within it. Sinusoids in the suprarenal glands of maniinals were demonstrated histologically by Minot ('00).
The cells composing the cortex are arranged in peripheral layers, surrounding a loose reticular core. These comprise what is usually termed the interrenal part of the suprarenal glands, from their resemblance to the interrenal bodies of Selachians. These interrenal bodies are being invaded on their medial surfaces by numerous scattered clumps of deeply staining cells, derived from the sympathetic ganglia, often termed the sympatho-chromaffine organs.
A deeply staining, oval mass of cells is present on either side of the inferior mesenteric artery, ventro-lateral to the aorta. Caudally these bodies reach the level of the proximal part of the common iliac arteries, while cephalad they are continued by scattered, smaller groups of cells to the caudal extremities of the suprarenal glands. They have a rich blood supply, and are intimately related to the sympathetic system in their locality. They unquestionably correspond to the aortic bodies discovered by Zuckerkandl ('01)., It seems very probable to the author that each of these main groups of cells has been partially isolated from the suprarenal gland of its side by the development of the large dorso-ventral segment of the supra-ureteral venous channel (plate 4) which now intervenes between them. Further evidence of this interpretation is the fact that a closer relation between the two exists on the left side in this em_bryo, where the vein in question is much smaller than its companion on the right.
Spleen
The spleen (Lien.) is clearly recognizable as a small, protuberance of the mesogastrium, containing dense m.esenchyma. The tissue directly ventral to this protuberance, is permeated by a vascular network, supplied by the splenic artery, and drained by the splenic vein (V.li., plate 4).
Nervous System
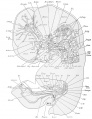
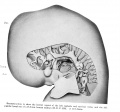
Brain
The surface of the brain is represented from the left in plate 2, and from the right in plate 3. The brain is shown in median sagittal section from the right side in plates 1 and 4, By the cephalic flexure (Flexxeph.) which occurs in the mesencephalon (Mesen.) the fore-brain is bent at an acute angle to the hindbrain. The cervical flexure (Flexxerv.) is nearly a right angle.
Telencephalon. The telencephalon (Telen.) is sharply marked off dorso-laterally from the diencephalon (Dien.) by a deep external groove and a corresponding internal ridge or fold, the velum transversum. This fold forms the caudal boundary of the 'interventricular foramen (Fo.int.). From this fold the line of demarcation extends ventrally just behind the optic evaginations to the postoptic recess (R.po.-op.). The telencephalon thus bounded contains a median cavity, the anterior part of third ventricle. The latter communicates with the lateral ventricles by comparatively large crescentic openings, the interventricular foramina (Fo.int.). The median cavity is bounded anteriorly by the lamina terminalis. The large oval hemispheres* represent the dorsal zones of the telencephalon. Each protrudes considerably beyond the lamina terminalis and presents orally two internal depressions with corresponding external swellings. These are the developing anterior and posterior olfactory lobes [Rhin.). Caudal and dorsal to the olfactory area the wall of each hemisphere is much thickened to form the corpus striatum (C.str.) which appears as a prominent swelling on the ventral surface in front of the praeoptic recess [R.p.-op.). Externally the position of the corpus striatum is marked by a shallow depression, the developing lateral fossa. The praeoptic recess is a slight groove passing transversely across the lamina terminalis into the optic stalk of either side. It represents the cephalic extension of the sulcus limitans, His ('92), Johnston ('09) and others. The ventral zones of the telencephalon comprise the area between the praeoptic and the postoptic recesses, which marks the place of later development of the optic chiasma. In regard to the area of evagination of the optic vesicles it is somewhat questionable whether this belongs primarily with the tel- or di-encephalon.
Diencephalon. The bomidary between the diencephalon and the mesencephalon (Mesen.) is a slight constriction extending nearly transversely across the brain toward the tuberculmn posterius (Tub. p.). An ill defined furrow, sulcus limitans, extends cephalo-caudad along the internal surface of the diencephalon separating the dorsal zone above from the ventral zone or hypothalamus below. The roof of the diencephalon is thin; near its caudal limit there is a slight evagination which represents the first appearance of the epiphysis (Corp.pin.).
The ventral part of the dorsal zone caudad of the interventricular foramen is thickened, forming on the medial surface a low ridge, the developing optic thalamus. Dorsal to the thickening, the internal surface of each dorsal zone presents a prominent concavity. The lateral wall of the hypothalamus is thickened. Caudad of the recessus postopticus (R.po.-op.) the cavity of the diencephalon extends into a small median evagination from the floor-plate. The evagination is the anlage of the infundibular gland (Gl.inf.). The thickened knob-like termination of the gland is embraced by the forked distal end of the hypophysis (Hyp.). It becomes the neural lobe of the adult pituitary.
Mesencephalon
The conventional boundary between the mesencephalon (Mesen.) and metencephalon (Meten.) is the constricted portion or isthmus (Isth.). As already stated a slight constriction extending nearly transversely across the brain toward the tuberculum posterius (Tuh.p.) divides it from the diencephalon. The roof -plate is comparatively thin, and bears no trace of the longitudinal ridge which has been described as occurring later. The floor-plate is considerably thicker than the roof-plate. The dorsal and ventral zones are distinct. The cavity of the mesencephalon (cerebral aqueduct) is more expanded in the caudal two-thirds of the mesencephalon, a large oval concavity appearing on the internal surface of each dorsal zone. The ventral zones are considerably thickened so that in the region of the oculo-motor nerve they project ventrally below the floorplate. Externally the surface of the mesencephalon is smooth except for slight dorso-lateral depressions on either side indicating the area of division into anterior and posterior colliculi.
Isthmus
The isthmus (Isth.) usually is described as the constricted portion between the mesencephalon and the metencephalon. It was considered by His ('92), and given in the Basel nomenclature ('95) as a distinct segment of the rhombencephalon which formed a marked ring somewhat narrower dorsally than ventrally.
In the corresponding area of the brain of this embryo there is on the internal surface of each ventro-lateral wall, a distinct transverse groove. The two grooves unite ventrally in a recess or sulcus (Sul.) caused by a depression of the floor-plate, perceptible on the external surface as a distinct elevation (E.i-p.) the eminentia interpenduncularis of His ('92). This sulcus was noted by Burckhardt ('91) and considered by him to be of general occurrence. It was observed in the human embryos by His ('92), and named by him the isthmus groove. Kupffer ('03-'05) named it the sulcus intraencephalicus posterior. The author believes that the transverse grooves mentioned above are the ventral continuations of the caudal mesencephalic neuromere; also that the adjacent brain wall should be regarded as the caudal part of the mesencephalon rather than a distinct division of the rhombencephalon.
Metencephalon
The metencephalon (Meten.) is the division of the primitive rhombencephalon from which the pons and cerebellum are developed. Its separation from the myelencephalon (Myelen.) is indicated, in part, by an internal transverse ridge (Pl.ch.p.) and a corresponding external groove, representing the developing plica chorioidea posterior.
The roof-plate is enormously expanded so that it forms not only the roof, but the greater part of the lateral wall of the metencephalon. The remaining or cephalic part of the lateral wall is formed on either side by a thickened band which is joined caudally to the ependymal roof-plate by a thinner intermediate layer known as the rhombic lip. This band on either side represents the corresponding dorsal zone (Z.dors.). It extends obliquely cephalad and medially to merge into the slightly thickened roof-plate, caiidad of the isthmus. It will form the corresponding lateral portion of the cerebellmn. The vermis is believed to be developed from the slightly thickened roof-plate intervening between these thickenings.
The ventral zones (Z.vent.) of the metencephalon, which form the pons, are deep longitudinal bands separated by a thick median raphe which represents the floor-plate. They extend caudad to the angle made by the pontal flexure where they blend with the corresponding zones of the medulla.
Myelencephalon. The myelencephalon (Myelen.) is the remaining portion of the brain, which arches over the cervical flexure (Flexxerv.) and joins the spinal cord. The roof-plate is exceedingly thin. It is widest in the region of the plica choroidea posterior where it forms the roof and the dorsal half of the lateral wall. Its caudal extension gradually tapers out to pass into the narrow roof-plate of the spinal cord. It becomes the caudal part of the posterior medullary velum.
The dorsal zones are thick. In the region near the spinal cord they are nearly vertical, but, by becoming progressively oblique, their internal surfaces form the lateral region of the ventricular floor at the cephalic end of the myelencephalon. The ventral zones, as in the metencephalon, are thick longitudinal bands on either side of the median line. In the floor of the ventricle a median longitudinal groove extends between them. Ventral to the groove a thickened raphe, floor-plate, unites the ventral zones. As the raphe approaches the region of the spinal cord it gradually becomes thinner.
Spinal cord The spinal cord (Md.sp.) is represented in median sagittal section in plates 1 and 4. The surface is shown, in part, in plates 2 and 3. It has a narrow slit-like cavity, somewhat expanded dorsally. The lateral walls of the cord have clearly marked ventral and dorsal zones which are continued into the corresponding zones of the myelencephalon.
Cranial nerves
The cranial nerves of the right side are displayed in plate 3. and those of the left are shown in plates 2 and 5. Plate 6 shows some of the cranial nerves on both sides.
Nn. olfactorii. Numerous nerve fibers (Nn.olf., plate 1) extend from the dorsal and medial surfaces of the nasal epithelium, and from the vomero-nasal organ (Org.vom.-nas.) to the olfactory area (Rhin.) of the telencephalon. In plates 2 and 3 the trunk formed by these nerves is represented as a stump. Among the fibers are numerous groups of cells which are not represented in the reconstruction. These cells perhaps have migrated from the nasal epithelium along the nerve fibers.
N. opticus. Fibers are present, extending from the retinal layer of the optic vesicle along the optic stalk to the corresponding ridge (optic) of the brain.
N. oculomotorius. The oculomotor nerve (iV.oc, plates 2, 3 and 5) issues from the ventro-lateral wall of the mesencephalon (Mesen.) by numerous small rootlets. It extends ventrally and cephalad, passing lateral to the posterior cerebral artery (A.cer.p.), and medial to the cavernous sinus (S.cav.) and the ophthalmic nerve (N.oph.). From the ophthalmic nerve it acquires a small sensory branch. Caudad of the optic stalk it gives off a branch to the anlage of the superior rectus muscle, but its main trunk is continued to the partially differentiated anlage for the inferior and medial recti and inferior oblique muscles.
N. trochlearis. The trochlear nerve (N.troch., plates 2 and 3) issues from the roof of the isthmus and extends ventrally in a sinuous course to the orbit. In its course it passes just cephalad of the superior cerebellar artery (A.cereh.s., plate 2) and lateral to the anterior cerebral vein. In the orbital region it passes dorsal to the anlage of the lateral rectus muscle, and medial to the frontal ramus (N.fr., pi. 3) of the ophthalmic nerve. It terminates in the anlage of the superior oblique muscle. The trochlearis receives a small sensory branch from the ophthalmic nerve (N.oph., pi. 3).
N. trigeminus. The trigeminal nerve (plates 2, 3, 4 and 5) is composed of sensory and motor components. The sensory fibers arise from the large semilunar ganglion (G.s-L), which lies lateral to the cavernous sinus (Sxav., plates 4 and 5), and form a large trunk which enters the latero-ventral surface of the metencephalon (Meten.) . The motor fibers issue from the metencephalon at a point slightly ventro-cephalad of the sensory root. They form a trunk of considerable size which crosses the medial surface of the semilunar ganglion to join the mandibular nerve (plate 4). The peripheral fibers leave the semilunar ganglion as three main branches, the ophthalmic (N.oph.), the maxillary (iV.?nx.), and the mandibular (N.vicL).
The ophthalmic nerve passes to the orbit. Dorsal to the optic stalk it gives a branch to both the oculomotor and trochlear" nerves, then divides into naso-ciliary and frontal nerves. The frontal (N.fr.) passes dorsal to the superior rectus and superior oblique muscles, and breaks up into several branches of which the supraorbital may be recognized by its dorsal direction. The naso-ciliary (N.na.-cil.) passes ventral to these muscles, and can be followed into the cephalic part of the corresponding lateral nasal process. The maxillary nerve (Njnx.) soon after leaving the semilunar ganglion becomes a bundle of loosely connected fibers extending into the maxillary process ventral to the optic vesicle (Ves.op.).
The mandibular nerve (N.md.) receives, in addition to the sensory fibers from the semilunar ganglion, the motor part of the trigeminal nerve. It divides into a small cephalic and a large caudal trunk. The former or buccal nerve at first extends cephalad in company with the infra-orbital branch of the stapedial artery (A.stp., pi. 2). Soon leaving this the buccal nerve crosses the anlage of the parotid gland (GI.p.), and passing cephalad, furnishes branches to the epithelium near the angle of the rima oris. The larger, caudal trunk of the mandibular as it crosses the tubo-tympanal ridge, divides into three branches;, the auriculo-temporal, the inferior alveolar, and the lingual. The latter near its origin is joined by the chorda tympani branch (N.ch-ty7?ip., plate 3; ch.-ty., plate 2) of the facial nerve.
The auriculo-temporal has two roots of origin which embrace a branch of the stapedial artery. It extends at first ventrally and laterally, and then bends dorsally, giving off twigs to the epithelium, in the neighborhood of the tuberculum tragicum.
The inferior alveolar (N.alv.inf.) crosses the lateral border of the pharynx, and follows the lateral side of Meckel's cartilage beneath the pharyngeal floor. It soon gives off the mylohyoid branch which passes ventrally, lateral to Meckel's cartilage, to the anlage of the mylohyoid muscle. The inferior alveolar then continues cephalad in the mandibular process of the mandibular arch where it divides into a dorsal and a ventral branch. The former supplies the oral epithelium of the corresponding side, the latter or mental nerve supplies the ectoderm on the Ventrolateral surface of the mandibular process.
The lingual nerve (N.ling.) is formed by the union of mandibular and chorda tympani fibers (Nxh.tymp.) medial to Meckel's cartilage. It extends cephalad for a distance between the cartilage and the alveolo-lingual ridge where the submaxillary gland (Gl.smx.) has developed. Here it passes into the anlage of the submaxillary ganglion (plate 3). From the ganglion it issues as several bundles which curve medially around the alveolo-lingual ridge into the lateral part of the tongue. In this situation branches extend cephalad between the ridge and the hypoglossal nerve and, after repeated subdivisions, are ultimately distributed to the epithelium of the tongue.
N. abducens. The abducens (N.ab.) issues from the ventral wall of the metencephalon (Meten.) by several rootlets which are hidden in the drawings by the overlying auditory and facial nerves. A caudal aberrant root is present on either side which extends from the region of the glossopharyngeal and vagus nerves to join the abducens. Similar aberrant roots of the abducens have been observed by Elze ('07) and represented by Bremer ('08). The abducens extends ventrally and somewhat laterally towards the orbital region. It passes dorsal to the internal carotid artery, obliquely across the medial side of the cavernous sinus and ophthalmic vein. The abducens then turns laterally between the vein and the oculomotor nerve to enter the anlage of the lateral rectus muscle.
N. facialis. The motor root of the facial nerve (plate 3) issues from the ventrolateral wall of the metencephalon (Meten.) and passes to the geniculate ganglion (G.gn.) by which its fibers are enveloped for a short distance. The sensory root (pars intermedia, N.int.) arises from the geniculate ganglion and enters the metencephalon immediately caudo-lateral to the motor root. The mixed facial trunk which emerges at the caudo-ventral border of the geniculate ganglion (G.gn.) represents the posttrematic ramus of the first pharyngeal pouch. It takes a caudal and lateral direction. Having given off the chorda tympani (N.ch.tymp., pi. 3, ch.ty., pi. 2) it ends in several small branches.
The chorda tympani nerve leaves the facial trunk at an acute angle and extends cephalad, ventral to the auditory pouch, to join the lingual branch of the mandibular nerve as described above. From the geniculate ganglion a small nerve (N.pet.s.m.) extends at first ventrally, and then bends sharply cephalad medial to the mandibular nerve and the tubo-tympanal ridge. It is the great superficial petrosal nerve. As it bends cephalad it gives off eaudally a short twig (anastomotic with the tympanic plexus) to meet the tympanic branch (N.tym.) of the glossopharyngeal nerve: The nerve arising from this junction (the anlage of the tympanic plexus) is the small superficial petrosal.
N. acusticus. The ganglion acusticum and otic vesicle are shown in plate 3. In plates 2 and 5 the otic vesicle has been removed. The ganglion acusticum is partially differentiated into cochlear and vestibular divisions. The vestibular part (G.ves.) lies cephalad of the utriculo-saccular division of the otic vesicle and lateral to the cochlear part of the ganglion, so that the latter is mostly hidden from view in plate 3. Four nerve trunks proceed from the vestibular division, a cephalic which divides into two rami to supply the ampullae of the superior and lateral semicircular canals ; a caudal which passes medial to the utriculosaccular division of the otic vesicle to supply the posterior semicircular canal; and two intermediate branches, the cephalic of which extends to the developing utricle, the caudal to the saccule.
From the cochlear division short fibers extend ventrally to the cochlear duct (D.c). The central fibers of the ganglion acusticum (N.acus.) enter the caudo-ventral wall of the metencephalon slightly dorsal and caudal to the sensory root of the facialis.
N. glosso-pharyngeus. The motor fibers of the glosso-pharyngeal nerve (plates 2, 3 and 6) issue from the myelencephalon just ventral to the entering sensory roots. The latter are hidden partially from view in plate 3 by the ductus endolymphaticus (D.end.) and crus commune, but are exposed in plate 2, The small ganglion superius (G.sup.) lies at the medial side of the ampulla of the posterior semicircular canal, separated from it by a narrow zone of the developing otic capsule. It extends slightly dorsal to the ampulla, and apparently involves only the more posterior and medial of the fibers of the nerve.
The ganglion petrosum (G.petros.) is a large ganglion, the caudo-lateral surface of which is closely applied to the trunk and ganglion nodosum of the vagus which somewhat overlap it. The medial surface of this ganglion is in contact with the internal carotid artery and the accompanying sympathetic fibers. From the cephalic part of the ganglion arises the tympanic nerve (N.tym.) which extends cephalad lateral to the internal carotid artery and nearly parallel with it. It passes dorsal to the remains of the second pharyngeal pouch and to the auditory (first pharyngeal) pouch, and lateral to the stapedial artery close to its origin. A little cephalad of the stapedial artery it is joined by a slender branch from the great superficial petrosal as described above (p. 57). The small superficial petrosal, resulting from this communication, I have been able to trace as far as a point near the caudo-ventral border of the semilunar ganglion.
Beyond the ganglion petrosum the glossophar^mgeal nerve skirts closely around the caudal aspect of the remains of the second pharyngeal pouch and reaches the ganglion nodosum. At the ventral border of the lateral pharyngeal wall it receives a branch (not shown in the figure) from the superior cervical sympathetic ganglion. This branch, extending around the medial side of the internal carotid artery, joins the dorsal aspect of the glossopharyngeal. The glossopharyngeal nerve, having passed on the cephalic aspect of the ganglion nodosum, leaves the vagus, but gives to it a communicating branch (also omitted from the reconstruction) which contributes fibers to the pharyngeal branch of the vagus. The glossopharyngeal then extends medially, bifurcating into a lateral or pharyngeal branch and a medial or lingual branch. The pharyngeal branch (plate 6) sends a twig to the developing stylo-pharyngeus muscle, and continues cephalad, for a short distance, along the pharynx. The lingual branch which is somewhat larger than the pharyngeal, also gives off a small branch to the pharynx, and then passes in a cephalomedial direction to the lateral -side of the caudal part of the tongue where it splits up into many branches.
The glossopharyngeal trunk which extends peripherally from the ganglion nodosum, is usually considered as the posttrematic ramus of the second branchial cleft.
N. vagus. The vagus (N.vag., plates 2, 3 and 6) is a large, mixed nerve, its main sensory component being derived from two ganglia, the jugular (G.jug.) and the nodosum (G.nodos.). The vagus acquires additional sensory fibers from an irregular series or chain of ganglionic masses (Gg.hyp.) situated caudad of the jugular ganglion and dorsal to the accessory nerve (N.acc). These irregular clumps of cells without doubt represent hypoglossal ganglia, but the exact number of ganglia formed by them is uncertain. Prentiss ('11) in dissected pig embryos frequently found fibers from two or three of such ganglia passing ventrally to join the corresponding motor roots of the hypoglossal nerve. Such fibers, however, were not found in this embryo. The chain of ganglia (Gg.hyp.) on the left side (plate 2) is continuous with the first cervical ganglion which is very slender in its middle part.
Small motor rootlets issuing from the myelencephalon ventral to the entering sensory fibers, together with fibers from the accessory, furnish the motor components of the vagus. The vagus, therefore, from its sensory as well as from its motor composition, is a compound nerve, as has been maintained by many investigators.
Each vagus nerve extends caudad upon the medial side of the internal jugular vein as far as the common cardinal vein. It then occupies the angle between the oesophagus and trachea until the latter bifurcates. From here each nerve, as it continues caudad, passes dorsally to the bronchus of its own side where it gives off pulmonary branches. More caudally the nerves are in close relation to the oesophagus, forming a coarse plexus superficial to the anlage of the external muscular layer. From the oesophagus the vagi spread onto the stomach and give branches to the neighboring viscera.
An auricular branch of the vagus has not been identified on either side of this embryo. It is possibly a nerve of late development. In rare instances its absence has been noted in the adult.
A short, rather ill-defined pharyrgeal branch is present on either side (but not reconstructed). Each arises irom the medial side of the cephalic part of the ganglion nodosum, and extends to the pharynx, passing ventral to the proximal part of the internal carotid artery. -To the formation of this nerve on either side the glossopharyngeal seems to contribute fibers as mentioned above. These branches of the vagus are apparently late in becoming formed into definite trunks.
The superior laryngeal nerve (N.Ls., pi. 3; N.laryng.s., pi. 6) arises at the ganglion nodosum, slightly caudad of its middle. It is represented in plate 3 as a stump, but is shown in part in plate 6. It extends ventrally and medially, dorsal to the distal part of the common carotid artery, and caudad of the detached (from pharynx) end of the thymico-pharyngeal duct, to divide into two branches, internal and external.
The internal branch passes medially over the cephalic border of the lateral lamina of the thyreoid cartilage, and divides into dorsal and ventral branches. The dorsal extends in the mesenchyma between the entodermal portions of the oesophagus and larynx. The ventral extends caudad, medial to the lateral lamina of the thyreoid cartilage. The external branch of the superior laryngeal is smaller than the internal, and can be traced caudally along the lateral aspect of the thyreoid cartilage for a short distance.
Froriep ('85) has shown conclusively that the superior laryngeal nerve is the posttrematic ramus of the third branchial cleft.
Another branch (R.p-tr.) of the left vagus arises from the ganglion nodosum directly caudad of the place of origin of the superior laryngeal. It is a small strand which extends caudally, at first between the vagus and the lateral lobe of the thyreoid gland, and dorsal to the thymus and left common carotid artery. Having passed the caudal extremity of the thymus gland it is in relation dorsally with the vagus, medially with the aortic arch, and laterally with the internal jugular vein. The nerve finally winds around the fourth aortic arch, and extends cephalad lateral to the recurrent of that side. It passes dorsal to the parathyreoid of the fourth pharyrgeal pouch and turning cephalad, appears at first dorsal and then dorso-medial to the lateral lobe of the thyreoid gland. It ultimately becomes exhausted by giving off fibers which extend ventrally in the region of the termination of the external ramus of the superior laryngeal nerve.
The corresponding nerve on the right takes origin from the dorso-medial side of the right ganglion nodosum, where the hypoglossal nerve crosses the vagus laterally. As it extends caudad it lies close to the medial side of the vagus, and it is conceivable that in later development the two nerves might be inclosed in a common connective tissue sheath. It finally winds around the right fourth aortic arch (A. subclavia) in the concavity of the larger recurrent nerve, and returns cephalad, passing lateral to the parathyreoid of the fourth pharyngeal pouch, but having the same relation to the lateral lobe of the thyreoid as the corresponding nerve on the right. It has a shorter course in the neck than that of the right side, the right fourth aortic being situated considerably more cephalad than the left.
From their site of origin, and from their relation to the fourth aortic arches and to the parathyreoids of the fourth pouches, it can scarcely be doubted that these nerves represent the posttrematic rami of the fourth pharyngeal pouches.
These nerves are obviously comparable to the branches of the vagus identified by Froriep ('85) in cow embryos (8.7 to 8.8 mm. in length) as the posttrematic rami of the fourth pharyngeal pouches, and figured in Taf. I, figs. I and II, II, 115. Froriep, however, could find this nerve only in the young stages.
Lewis ('06) found in the 12 mm. pig embryo a small nerve running beside the postbranchial body which he thought might be comparable to the nerve described by Froriep as the posttrematic ramus for the fourth pouch.
Elze ('07) identified this posttrematic branch of the vagus in a human embryo of about 7 m_m., (p. 427, text figs. 7-8), but could not find the nerve in two older human embryos (II and III), measuring (greatest length) 9.5 and 11 mm. respectively. Hence, like Froriep, he concluded that in man the existence of this posttrematic ramus of the vagus is transitory.
In this embryo it is seen that these posttrematic rami become closely associated with the recurrent, especially on the right side where the pulmonary aortic arch atrophies, and the author believes that they occasionally, at least, persist in the adult. The support for this conclusion is based not only upon their presence in an embryo of this stage of development (17.8 mm.), but upon the observations of Wrisberg. Wrisberg (Henle's Anatomie des Menschen, Bd. 3, p. 441, 1868) observed in three cases a reduplication of the right recurrent nerve. The extra branch was much smaller than the normal, and it accompanied the latter upwards between oesophagus and trachea, in much the same way as occurs in this embryo.
The recurrent nerves are displayed in plate 6. The right recurrent nerve (A^rec), arises from the vagus at the caudal border of the right fourth aortic arch (A. subclavia), and passes cephalad in the neck, dorsal to the parathyreoid of the fourth pharyngeal pouch (P.-thyr.IV). Here it gives off some oesophageal branches, and then continues to the medial side of the right lamina of the thyreoid cartilage where it becomes exhausted by giving off branches which turn ventrally to the anlagen of the laryngeal muscles.
The left recurrent nerve (N.rec.s.) arises from the vagus at a more caudal level than the right, viz., at the caudal border of the pulmonary arch (ductus arteriosus). Winding around this arch ventro-dorsally, it extends cephalad. In the neck it has relations similar to those of the corresponding nerve on the right, with the exception that it passes medial to the parathyreoid IV instead of dorsal.
It has been suggested by Froriep ('85) that the recurrent nerves may well be considered as trunks formed by the fusion of branchial nerves for clefts which fail to develop. This interpretation, again advanced by Lewis in 1906, gains support from the relation of the posttrematic rami IV to the recurrent nerves in this embryo, especially on the right side where the two nerves a^e brought close together, the caudal aortic arches having atrophied.
One of the inferior cardiac branches of the right vagus is seen in plate 3. It (N.ci.) arises from the dorso-medial wall of the vagus caudad of the place of origin of the recurrent nerve. It extends caudad in the angle between the oesophagus and the trachea, being dorsal to the vagus and medial to the internal jugular vein. It eventually becomes in part incorporated in the deep cardiac plexus, a part rejoining the vagus. Slightly caudad of the above nerve another inferior cardiac ramus from the vagus extends medially to the anlage of the deep cardiac plexus. It is hidden in the reconstruction by the overlying vagus. An inferior cardiac nerve was found on the left side corresponding to the right nerve shown in plate 3, but it has been omitted in the reconstructions. It arises from the left vagus just after it has given off its recurrent ramus, hence at a considerably more caudal level than the right inferior cardiac. It extends caudad in the angle between the oesophagus and the trachea to the deep cardiac plexus.
N. accessorius. The accessory nerve (N.acc, plates 2 and 3) is form.ed by a series of small rootlets which emerge from the lateral surface of the spinal cord and myelencephalon. The most caudally placed root of the series issues at the level of, and in close proximity to, the dorsal root of the second cervical nerve. In this embryo the place of emergence of the most caudally placed root is similar on the two sides, and has been noted in consequence of its variation in other human embryos. Streeter ('04) places the level at the third or fourth cervical, but finds that sometimes it is more caudally placed.
The main trunk of the nerve arches cephalad and ventrally under the hypoglossal ganglia (Gg.hyp.) to become incorporated with the vagus ventral to the jugular ganglion. .At the cephaloventral border of the ganglion nodosum of the vagus, the ramus externus (R.ex., plate 3) of the accessory nerve curves laterally and dor sally around the lateral side of the internal jugular vein (plate 6) to the developirg sterno-cleido-m.astoid m.uscle. From here it passes around the cephalic part of the adjacent lymph sac to reach the anlage of the trapezius muscle. In the angle between the internal jugular vein and the lymph sac (S.jug.) it communicates with the great auricular nerve.
N. hypoglossus. Numerous, small, hypoglossal rootlets issue from the ventral wall of the myelencephalon on either side, and each group converges to form four trunks. These, after crossing the lateral side of the vertebral artery (A.vert.), further join to form the two main roots of the hypoglossal nerve (N.hyp., plates 2 and 3).
On the right side, in addition to the four trunks seen on the left, a vestigial root joins the fourth trunk (plate 3). Such vestigial roots of the hypoglossal nerve have been found by Bremer ('08) to occur frequently in embryos of man, pig, sheep and dog, and almost constantly in the turtle and chick.
It will be noted that the caudal of the four hypoglossal roots on the left (plate 2) passes through an arterial fenestra of the vertebral artery. A similar, but more extensive anastomosis of arterial branches with the vertebral, enclosing the hypoglossal roots has been shown by Elze ('07), (Taf. 15, fig. 2). They show how, by means of island-formation, the vertebral artery in the adult may come to pass between the roots of the hypoglossal nerve or, in rare cases, extend lateral to the entire nerve. The hypoglossal ganglia have been considered with the description of the vagus.
The hypoglossus receives a branch from the first cervical nerve (one from the second cervical in addition on the right), and extends ventrally between the vagus and the internal jugular vein. At the ventral border of the vagus, a little caudad of the termination of the linguo-facial vein, it gives off its ramus descendens. The latter extends caudad, ventral to the internal jugular vein, and joins the descendens cervicalis to form the ansa hypoglossi. The hypoglossus then continues cephalad, medial to the linguofacial vein and the developing submaxillary gland (plate 5) to divide in the tongue into muscular branches.
Spinal nerves
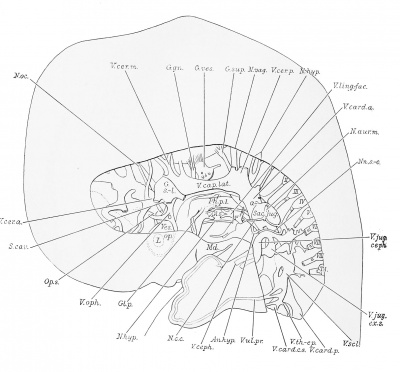
Plates 1 and 4 show portions of the ventral divisions (rami anteriores) of the spinal nerves (Nn.sp.); plate 3 gives a lateral view of the right cervical nerves and plexus; and plates 2 and 5, a lateral view of the left cervical nerves and plexus. In plates 5 and 6 the relations of these nerves to the jugular lymph sac (S.jug.) are shown. It will be noted that some of the ganglia are still connected by ganglionic bridges.
Nn. cervicales. The cervical nerves divide just beyond the union of the dorsal and ventral roots into dorsal and ventral primary divisions (rami posteriores and anteriores). The dorsal rami are shown as stumps with the exception of the great occipital branch (N.occ.m.) from the second, a piece of which has been added. The ventral primary divisions run ventrally and caudally, and the second to the sixth inclusive, extend laterally to the vertebral artery (A. vert.).
The first cervical nerve has a long slender ganglion (plates 2 and 3), the dorsal part of which overlies the accessory nerve (N.acc). The ganglion shows signs of atrophy in its middle part, especially on the left side. As the nerve extends ventrally it is in close contact for a distance with the vertebral artery (plates 1 and 2), which it crosses on the medial side. A short communicating branch connects the first cervical nerve with the ventral primary division of the second cervical. The fibers of this communicating branch join the hypoglossal nerve, and are thought to assist in forming the ramus descendens hypoglossi.
The ventral primary division of the second cervical nerve behaves differently on the two sides. On the left (plate 2) beyond the communicating branch with the first cervical nerve, it passes caudad to join a branch from the third. The junction occurs dorsal to the internal jugular vein and forms the descendens cervicalis which extends ventrally between the internal jugular vein and jugular lymph sac (plates 5 and 6). The descendens cervicalis joins the ramus descendens hypoglossi to form the ansa (An.hyp.).
On the right side (plate 3), beyond the communicating branch with the first cervical nerve, the ventral primary divisions of the second cervical bifurcates. One branch joins the third cervical to form the descendens cervicalis, while the other passes ventrally between the vagus and internal jugular vein to join the hypoglossus. The fibers passing direct to the hypoglossus, therefore, reach the ansa by way of the ramus descendens hypoglossi.
The ventral prunary division of the third cervical nerve also differs on the two sides. On the right (plate 3) distal to the communicating branch with the second cervical nerve, it divides into a cephalic and a caudal branch. The caudal branch furnishes medially the ramus descendens cervicalis; and laterally a branch which unites with one from the fourth cervical to form the supra-clavicular nerves (N?i. s-cL).
On the left side the ventral division of the third cervical (plate 2) also divides into two branches. The more cephalic of these first gives off medially a branch which passes between the vagus and jugular lymph sac to the ramus descendens cervicalis. Then, continuing ventrally between the dorsal portions of the internal jugular vein and adjacent lymph sac, it furnishes (plates 5 and 6) the great auricular (N.aur.vi.), small occipital, and cutaneous colli (N.c.c.) nerves. The great auricular nerve pierces the dorsal part of the cephalic segment of the saccus jugularis, and here communicates with the ramus externus of the accessory nerve. The small occipital and cutaneous colli nerves appear a little more caudally between the two cephalic segments of the lymph sac. At the place of origin of the small occipital and the cutaneous colli, the third cervical nerve has a communicating branch with the fourth cervical, probably to assist in the formation of the supraclavicular nerves (Nn.s-cl.) as on the right side. The more caudal branch of the ventral ramus of the third cervical nerve extends caudad, and gradually tapers out. Possibly this branch would have joined, eventually, the phrenic nerve (N.phr.).
The fourth cervical nerve on either side (plates 2 and 3) contributes to the formation of the supraclavicular nerves (Nn.s-cL), which extend ventrally along the cephalic border of the jugulocephalic vein (plate 5). It also sends a branch to the phrenicus [N.phr.), and to the ventral ramus of the fifth cervical. The root which the phrenic derives from this nerve (plate 2) passes between the two terminal branches of the thyreo-cervical artery.
The ventral primary division of the fifth cervical nerve gives off the upper root of the long thoracic nerve, and then joins with the sixth cervical to form the upper trunk of the brachial plexus (plate 3). Just before joining, however, the fifth cervical nerve gives off its phrenic root which, extending caudad, dorsal to the thyreo-cervical artery (plate 2), meets the branch from the fourth cervical nerve. Each phrenic nerve (N.phr.), thus formed, accompanies the internal mammary artery (A.mam.i.) dorsoventrally around the caudal border of the terminal portion of the subclavian vein (V.scl., plate 5). The nerve here leaves the artery, and at first extends in the somatopleure along the lateral side of the anterior cardinal vein, then between this vein and the pleural cavity, and finally in the pleuro-pericardial membrane, lateral to the common cardinal. The nerves ultimately reach the anlage of the diaphragm on either side of the common hepatic vein (the right phrenic being for some distance in close relation to the latter).
The ventral primary division of the sixth cervical nerve, dorsal to where it is joined by that of the fifth, gives off the middle root of the long thoracic nerve.
The ventral primary division of the seventh cervical nerve furnishes the lower or caudal root of the long thoracic nerve, and becomes the middle trunk of the brachial plexus.
The ventral primary division of the eighth cervical and that of the first thoracic nerves unite to form the lower trunk of the brachial plexus.
Sense Organs
Eye
The eye-ball is distinctly outlined, and the anlagen of the eyelids form two prominent arches. The optic vesicle (Ves.op.), its stalk (Op.s.), and the lens (L.) of the right eye are shown in plate 3; the same structures on the left in plate 5.
The optic stalk is slender and the lens is now devoid of a cavity. Into the loose mesenchyma, occupying the vitreous chamber extend the hyaloid artery and its branches. Other blood vessels reach this chamber through the circular fissure at the margin of the lens.
Ear
The fossa conchae (text fig. 1) is a depression rather broad and shallow dorsally, but narrow and deep ventrally. The mandibular border of the fossa is formed by a fold especially prominent in its ventral part where it presents a conspicuous projection. This, the tuberculum tragicum, extends laterally and somewhat toward the fossa. The ridge at the hyoid border of the fossa is more prominent than that on the mandibular border and is bounded caudally by the retroauricular groove. At the ventral end of the ridge is a well marked protuberance which overhangs the fossa directly opposite the tuberculum tragicum. It represents the tuberculum antitragicum. The portion of the ridge dorsal to this tubercle is the developing helix. In the central part of the floor of the fossa is the low elevation which represents the tuberculum membrane tympani of Hammar ('02).
The lateral aspect of the right otic vesicle is represented in plate 3 and the medial in plate 4. The stumps of the nerves which innervate it are seen in plate 2. The utricular (Ut.), saccular (Sac), and cochlear divisions (D.c), are not sharply differentiated from one another, and the coalesced lamellae of the lateral canal have not been absorbed as yet. The ductus endolymphaticus widens into the saccus, and extends for a considerable distance dorsally. A peculiar anomaly occurs in this otocyst; a short hollow diverticulum projecting from the utricle just dorsal to the ampulla of the lateral semicircular canal. This diverticulum joins the crus commune of the superior and posterior semicircular canals without, however, opening into it. This diverticulum, does not occur upon the left side, but a similar condition in other human embryos of the Harvard Collection, has been observed by the author.
Nose
The nares are represented in plates 3 and 5, and the ectodermal linirg of the olfactory vesicles in plates 1, 2 and 4.
The nares (Na.) are open, but separated by a broad septum, From the naris each olfactory pit (Ves.olf.) extends dorso-caudally as a small tube, oval in transverse section. Each tube shortly expands to form the main nasal cavity which opens into the fore part of the roof of the mouth by a large primitive choana (Ch.pr.).
In lateral view (plate 2) the epithelial wall of the olfactory vesicle presents cephalad a large concavity partially subdivided by a slight external ridge (unfortunately not shown in the drawing) into a shallow dorsal depression, the nasoturbinal (agger fiasi), and a more extensive ventral one (Max-turb.), the maxilloturbinal (concha inferior). The maxilloturbinal is limited ventrally by a pronounced, somewhat dorsally curled fold of the vesicular wall, containing within the meatus nasi inferior.
At the dorsal part of the choanal extremity of the olfactory vesicle there is seen in a lateral view a slight indentation (Eth.iurh.I), representing the developing ethmoturbinal I (concha media). The ethmoturbinal I, although it now forms in part the lateral wall of the olfactory vesicle, has been shown by Peter ('01) to be a derivative of the septal wall. The ridge appearing between the ethmo- and maxillo-turbinal overlies the meatus nasi medius. The palatal process forms the lateral boundary of the choanal extremity of the olfactory vesicle.
On the medial wall of the olfactory vesicle (plates 1 and 4) is seen the small, tubular outgrowth (Org.vom.-nas.) for the vomero-nasal organ (Jacobson's), rudimentary in man.
The nasolacrimal duct (not shown in the reconstructions) begins at the medial angle of the eye in an expanded end, disconnected from the ocular epithelium, and extends medially to end near the epithelium of the meatus nasi inferior. It is a solid, irregularly branching, cord of cells, entirely surrounded by mesenchyma. Outgrowths from the ocular extremity for the superior and inferior lacrimal ducts have not developed.
Vascular System
Heart
The heart in its relation to the surrounding organs is shown in plates 1 and 4. The section shown in plate 1 passes to the left of the atrial and ventricular septa, and therefore, opens the left atrium and ventricle. In plate 4 the section passes through the cavities of the right side. The ventricular trabeculae are represented somewhat diagrammatically.
The sinus venosus (S.v., pi. 4) receives the common hepatic (V. hep.com.) and the right and left common cardinal veins (Vv.card.c.d. and s.), and opens into the right atrium.
Upon the dorsal wall of the right atrium (At.d., plate 4) is the sagittally directed, sinu-atrial orifice. The latter forms a narrow slit, bounded laterally by the right and left sinus valves (V.V.S.). The cephalic ends of the valves converge and meet in a ridge. The continuation of this ridge, which can be traced for some distance along the cephalic part of the interior of the atrium, is the septum spurium.
The caudal extremity of the left sinus valve meets the right side of the septum primum at the caudal part of the right atrium. Just where these two structures meet ventrally there is a ridge or tubercle, which probably represents the caudal end of the future septum secundum. Born ('89) goes into no detail with regard to the earlier stages in the developm.ent of the septum secundum, which is shown completely formed in his figure 29. From the relation which the tubercle bears to the septum primum and the left sinus valve it can scarcely be doubted that it would eventually . form part of the adult limbus fossae ovalis.
A large portion of the right sinus valve subsequently disappears but its caudal part will persist to form the valvulae venae cavae inferioris and sinus coronarii. On the right of the atrio-ventricular orifice a portion of the developing tricuspid valve (V.t.) is seen.
The principal outlet for the right ventricle (Vent.d.) is now the truncus pulmonalis (Tr.puL), although the presence of the small interventricular opening (F.int., pi. 1) still allows some blood to pass out by way of the aorta. The aortic septum (S.) is practically complete at this stage, and is seen in plate 4, separating the root of the aorta from the conus arteriosus iCon.art.) of the right ventricle.
The left atrium (At.s., pi. 1) is partially separated from itsfellow on the right by the septum primum. A quadrilateral opening in the septum., the ostium secundum (O.s.), places the two atria in direct communication. Leading into the left atrium on its dorsal side two pulmonary veins are shown (Vv.pul.d.). Between the left atrium and ventricle there is a marked protuberance of the fused endocardinal cushions, which represents in part the anlage of the bicuspid valve. The vessel (V. card. com. s.) seen in section immediately caudad of the left atrium is the part of the left common cardinal which will become the coronary sinus.
The left ventricle is still in communication with the right by a small opening (F.int.) the foramen interventriculare which is directed obliquely dorso-ventrally from left to right. This eventually becomes closed towards the right ventricle by complete fusion between the aortic and interventricular septa. When this has occurred, the aorta (Tr.aor.) will become the only outlet of the left ventricle. The ventricular wall immediately ventral to the anlage of the tricuspid valve (V.t.) separates the left ventricle from the right atrium, and has been named by Hochstetter ('98) the septum atrio-ventriculare. This septum, together with that formed by the fusion of the aortic (S.) and interventricular septa, form the septum membranaceum ventriculorum of the adult heart.
Arteries
The arteries of the right side are shown in plate 1 from the left side. The arteries of the left side of the head and neck are shown in plate 2.
Systemic derivatives of the aortic arches. The truncus aorticus (Tr. aor., plate 1) leaves the left ventricle of the heart, extends in the dorsal mesocardium across the pericardial cavity, and im.mediately divides into two ventral aortae. The left ventral aorta gives off the short fourth left aortic arch which becomes part of the arcus aortae, and continues cephalad as the !eft common carotid. It is shown in this plate as a stump, but is displayed fully in plate 2.
The left common carotid extends cephalad between the thymic and thyreoid anlagen to the region of the larynx, where it divides into a dorsal and a ventral branch, the internal and the external carotid arteries, respectively.
The external carotid (A. car. ex.) represents the continuation of the ventral aorta. It is a short stem which terminates by dividing; into five branches, viz.: superior thyreoid, occipital, lingual, external maxillary and posterior auricular. The superior thyreoid (A.thyr.s. and d., plates 1 and 6) runs medially skirting the cephalic extremity of the lateral lobe of the thyreoid gland, and at once breaks up into small branches. The occipital (A.occ, plates 2 and 6) takes a cephaFc and dorsal direction around the lateral side of the internal jugular vein, and gives off the sternomastoid branch. The lingual (A.ling., plates 2 and 6) runs medially to the tongue, giving off laterally a branch to the submaxillary gland. The external maxillary (A.rnax.ex., plates 2 and 6) extends ventrally along the mandibular arch. The posterior auricular (A.aur.p., plates 2 and 6) is a large vessel which, having given off the stylo-mastoid branch curves laterally behind the primary external acoustic meatus and passes dorsally. The ascending pharyngeal and temporal arteries have not been identified. The internal maxillary, as such, is still wanting in this embryo; it is foreshadowed by the stapedial branch of the internal carotid to be described later.
The left internal carotid (A. card.) comprises the third aortic arch, the left dorso-lateral aorta cephalad of the third arch, and a terminal portion or branch from the first aortic arch. It curves at first dorsally and cephalad, and then extends directly cephalad, dorsal to the roof of the pharynx. It passes medial to the cranial nerves and bifurcates, lateral to the diencephalon (Dien.) into caudal and cephalic divisions. The former is the posterior communicating artery (A.com.p.). This extending caudad gives off the posterior chorioidal artery, and joins the left posterior cerebral (A.cer.p.). The other terminal division of the internal carotid artery divides into two branches, the anterior chorioidal (A.chr.a.), and a stem common to the middle cerebral (A.cer.m.) and the anterior cerebral (Axer.a.) arteries. Besides the terminal branches two other branches of the left internal carotid are shown here, the ophthalmic and the stapedial. The ophthalmic artery (A.oph.) arises from the internal carotid artery medial to the ophthalmic nerve (N.oph.). It extends cephalad and laterally, ventral to the optic stalk which it penetrates, becoming the central artery of the retina.
The stapedial artery (A.stp., pi. 2) arises from the internal carotid near the middle of the pharynx, and passing through the left stapedial cartilage, runs as shown in plate 2, towards the semilunar ganglion (G.s.-L). A short distance from the ganglion it divides into two branches, dorsal and ventral. The dorsal or supraorbital branch passes lateral to the semilunar ganglion into the region of the orbit. The ventral division soon bifurcates, and the resulting branches pass to the medial and lateral sides, respectively, of the mandibular nerve. The medial branch, having communicated with the lateral by a branch passing ventral to the mandibular nerve, first accompanies the buccal branch of the mandibular and later the maxillary nerve, as the infraorbital artery. The lateral branch passes between the roots of origin of the auriculo-temporal nerve and,' having communicated with the medial branch, becomes the inferior alveolar which accompanies the nerve of the same name (N. ah. inf.).
The development and importance of the stapedial artery have been demonstrated admirably by Tandler ('02). He finds that this artery represents a persistent portion of the dorsal part of the second aortic arch, and an intermediate segm.ent of the first arch; the two being united by a longitudinal anastomosis. In certain mammals the stapedial artery persists throughout life, but in man an anastomosis between the external carotid artery and the mandibular ramus of the stapedial results in the formation of the adult internal maxillary artery to which the stapedial transfers its branches. In this embryo the anastomosis is yet to form; there can be no doubt, however, that it will join the lateral branch of the ventral division of the stapedial between the auriculo-temporal nerve and the branch of communication between the lateral and medial branches. As a result of such an anastomosis the lateral branch of the ventral division, together with the dorsal division (supraorbital of Tandler), would become the middle meningeal. The middle meningeal and inferior alveolar would then spring from the internal maxillary. The original main trunk of the stapedial would persist in part as the carotico-tympanic branch of the internal carotid and the superior tym.panic branch of the middle meningeal. It is extremely probable that the medial branch of the original ventral division of the stapedial is represented in the adult by the accessory meningeal branch of the internal maxillary.
The right ventral aorta (plate 1) takes a cephalic direction from the truncus aorticus. It soon gives off a short dorsal branch, the fourth aortic arch, which becomes part of the right subclavian. The portion of the right ventral aorta proximal to the fourth arch becomes the innominate artery (A. anon.), the smaller distal continuation represents the right common carotid. The relations of the latter are essentially the same as those just given for the corresponding artery on the left. A branch, however, from the proximal part of the right internal carotid, accompanies the hypoglossal nerve. This, the hypoglossus artery (A.hyp.d., plate 1) has been noted in the human embryo by Zimmennan ('89), Tandler ('02) and Ingalls ('07), and in the rabbit by Hochstetter ('90). The hypoglossus artery of the left side had disappeared in this embryo. The hypoglossus arteries represent the second pair of primitive intersegmental arteries from the dorso-lateral aortae, later becoming temporary branches of the internal carotids.
The dorso-lateral aortae have ceased to exist as complete trunks, for their continuity has been interrupted in the segment of each intervening between the third and fourth aortic arches.
The segm.ents cephalad of the third pair of aortic arches consist of two symmetrical vessels which enter into the formation of the internal carotid arteries, as already stated. The segments caudad of the fourth pair of arches, consist of two very unsymmetrical vessels which converge and unite, opposite the second pair of thoracic nerves, to form the dorso-median aorta (Ao.d.m.).
The caudal segment of the right dorso-lateral aorta is still large in the neighborhood of the fourth aortic arch, with which it participates in the formation of the right subclavian artery (A.scl.d.). The remainder of this segment of the right dorsolateral aorta is reduced to a fibrous cord containing only a trace of a lumen. The caudal segment of the left dorso-lateral aorta is large and contributes to the formation of the permanent arcus aortae.
Although the intermediate segments of the dorso-lateral aortae have lost all connection with the third pair of aortic arches, they still persist as a pair of vestigial arterial tubes (*S.) projecting from the cephalic aspect of the fourth aortic arches.
The fourth aortic arches extend from the place of origin of the common carotids, upon the right and left sides, to the vestigial tubes above mentioned. They are both short, wide trunks, of which the right occupies a more cephalic position. The right fourth aortic arch eventually forms the proximal part of the right subclavian artery. The left participates in the formation of the arcus aortae.
Pulmonary arteries. The truncus pulmonalis (Tr.puL, plates 1, 2 and 4) begins in the conus arteriosus (Con.art.) of the right ventricle which is almost completely separated from the tnmcus aorticus (vestibule) . Of the pulmonary arches, only the left persists in its entirety. The truncus pulmonalis is now continued directly into the left pulmonary arch which opens into the left dorso-lateral aorta in common with the left fourth arch.
The two small pulmonary arteries (Aa.puL, plate 1) extend caudad from the main pulmonary trunk, one to each developing lung. The right vessel is reconstructed more completely than the left, and represents not only the right pulmonary artery, but the proximal portion of the right pulmonary arch, as has been shown by Bremer ('02). The left pulmonary artery is shown only as a stump. It represents the left pulmonary artery proper. The segment of the pulmonary arch (D.a., plate 2) between the left pulmonary artery and the left dorsal aorta is the ductus arteriosus.
Derivatives of the occipital and cervical segmental arteries. The right hypoglossus (intersegmental) artery (see p. 75) and the proximal parts of the cephalic six (five on the right side) pairs of cervical intersegmental arteries have disappeared. The distal parts of these intersegmentals have now become branches of the large vertebral arteries (A. vert.), fonned from the series of post-costal anastomoses between them. The seventh intersegmental artery (sixth on the right side) has retained its connection with the dorso-lateral aorta of its own side to form a portion of the stibclavian and the proximal part of the corresponding vertebral artery. The vertebral arteries extend cephalad medial to the cervical nerves, with the exception of the first pair which they accompany towards the spinal cord. The sudden change in direction of the vertebral arteries opposite the suboccipital pair of nerves arises from the fact that the portion of these arteries between the atlas and occiput develops from the first cervical intersegmental spinal rami. The vertebral arteries pass ventral to the myelencephalon and medial to the hypoglossal and vagus nerves. Opposite the glossopharyngeal the arteries of the two sides unite to form an enormous median vessel, the basilar (A .has.) . The basilar artery extends cephalad in the median line between the notochord and the rhombencephalon. Between the trochlear nerves (N.troch.) it divides into two lateral branches, the posterior cerebral arteries. Each posterior cerebral artery (A.cer.p.) communicates ventrally with a branch (posterior communicating) of the internal carotid artery (A.car.i.) to participate in the formation of the circulus arteriosus. It also dispenses several branches to the lateral and dorsal surfaces of the di- and mesencephalon (Dien. and Mesen.), a considerable branch to the mesencephalon passing through the rootlets of the oculomotor nerve (N.oc, plate 2). The left superior cerebellar artery (A.cereh.s.) arising from the basilar is seen in plate 2, just caudad of the trochlear nerve. Its branches overlie the isthmus and the metencephalon. The other branches of the basilar are seen in part in plate 1, those on the left side having been cut away.
The left subclavian artery (A.scL, plate 2) is a branch of the corresponding seventh cervical intersegmental artery. Beyond the origin of the vertebral artery (A. vert.), the thyreo-cervical (Tr.thyr.-cerv.) arises from the cephalic aspect of the subclavian; the costo-cervical (Trxost.-cerv.) from the dorsal; and the internal mammary (A.mam.i.) from the ventral aspect. The costocervical divides, as usual, into the deep cervical and superior intercostal; the others can be identified by their direction. The remaining portion of the subclavian trunk extends ventro-laterally into the upper limb.
On the right side (plate 1) there is a common anomaly, the sixth (not the seventh) cervical intersegmental being connected with the right dorso-lateral aorta. By means of the right dorsolateral aorta and a short precostal anastomosis the sixth intersegmental is connected with the seventh.
Consequently the right subclavian comprises three elements (exclusive of the parts derived from the fourth aortic arch and dorso-lateral aorta). These are: (1) the root of the sixth cervical intersegmental artery; (2) a longitudinal precostal anastomosis connecting the sixth and seventh intersegmentals; and (3) a portion of the seventh intersegmental. From the first element arises the right vertebral artery (A.vert.d.), the root of which represents a part of the sixth cervical segmental. From the third element the right superior intercostal arises on the caudal aspect; the thyreo-cervical on the cephalic; the deep cervical on the dorsal, and the internal mammary on the medial aspect. The deep cervical is derived from the dorsal ramus of the seventh intersegmental artery. The internal mammary represents the real continuation of the ventral ramus of the seventh cervical intersegmental. The continuation of the subclavian beyond the point of origin of the internal mammary appears to represent a lateral branch of the ventral ramus of the seventh intersegmental. By the anomaly found on the right side of this embryo, the deep cervical and superior intercostal arteries come to have separate origins from the subclavian as sometimes occur in the adult.
Parietal arteries of the trunk. From the level of the second pair of thoracic nerves, where the dorso-lateral aortae unite, the single dorso-median aorta (Ao.d.m.) continues throughout the trunk to end as the middle sacral (A.s.m.). The dorsal intersegmental arteries arise in regular pairs from the dorso-median aorta. Those of the right side are shown in plate 1, the corresponding vessels on the left having been omitted.
The dorsal intersegmental arteries curve laterally and dorsally, passing lateral to the sympathetic, and either pass dorsally to become muscular branches or continue medially as spinal rami into the vertebral canal. In the latter situation the spinal rami pass laterally to the spinal cord to form the posterior spinal arteries or join to form a bilateral ventral longitudinal anastomosis which extends from the vertebral artery (directly caudad of the myelencephalon) along the spinal cord into the tail process. Some of the spinal rami bifurcate contributing thereby a branch to each anterior and posterior spinal artery of that side.
That the embryonic right anterior spinal anastomosis eventually joins its fellow of the opposite side to form the single anterior spinal artery of the adult was first indicated by His ('86). Recently the matter has been treated in greater detail by Sterzi ('04), and Evans ('09 and '12). The latter observers conclude that fusion between the two stems does not occur, but that the single adult artery represents the selected channel persisting from the alternative paths offered by the two longitudinal vessels and the plexiform connections between them.
In the thorax and abdomen the ventral rami of the dorsal intersegmentals become the aortic intercostal and lumbar arteries, respectively. Some of the f orm.er are represented as short stumps in plate 1. The ventral rami of the first and second thoracic pairs of dorsal intersegmentals have lost their connection with the dorso-median aorta. They are now connected with the subclavian arteries by the precostal anastomosis which forms the adult superior intercostal.
Visceral arteries. The ventral visceral arteries and the lateral visceral arteries of the right side are indicated in plate 1.
In the thorax several small, ventral visceral arteries (Aa.oe.) extend from, the aorta through the mediastinum to the oesophagus. According to Broman ('08) these oesophageal arteries are of secondary formation, the primary segmental arteries having early disappeared.
The coeliac artery (A.coel.) leaves the aorta slightly caudad of the 11th thoracic pair of dorsal intersegmental arteries. It extends between the medial surfaces of the anlagen of the suprarenal glands, and entering the mesogastrium at once gives off the left gastric artery which turns cephalo-ventrally. The coeliac then continues in the mesogastrium (hidden in the reconstruction by the stomach, Ga.), and having given off the splenic artery it becomes the hepatic. The former artery extends in the mesogastrium to the splenic anlage; the latter enters the lesser omentum and divides into the hepatic artery proper and the gastroduodenal. The hepatic proper can be seen in the reconstruction, entering the liver just ventral to the portal vein (V.P.). It gives off the small cystic branch (not shown in the reconstruction) which accompanies the cystic duct. The gastro-duodenal artery can be traced distally between the left side of the duodenum and the proximal part of the dorsal pancreas, Tandler ('03) finds that the coeliac artery in a 17 mm. human embryo arises from the aorta slightly above the level of the 20th intersegmental (12th thoracic) artery.
The superior mesenteric artery (A.mes.s.) takes origin from the aorta approximately on a level with the 12th thoracic pair of dorsal intersegmental arteries. It extends ventrally between the caudal ends of the suprarenal glands (plate 4) into the dorsal mesentery. It courses to the left of the duodenum and passes dorsal to the vena omphalomesenterica. In the dorsal mesentery it gives off numerous small branches, and crosses on the right of the large intestine. In the umbilical cord, after again crossing the gut (on the left side of the small intestine), it follows the yolk-stalk. In a 17 mm. human embryo studied by Tandler ('03) the origin of this artery is opposite the 21st intersegmental (1st lumbar) artery.
On a level with the 2nd lumbar pair of dorsal intersegmental arteries the inferior mesenteric (A.mes.i.) arises from the aorta. It is a small ventral visceral branch, extending ventrally and somewhat caudally between the aortic bodies (of Zuckerkandl) into the dorsal mesentery. In the dorsal mesentery it gives off several branches which ramify in the region of the colon. Tandler (-03) found this artery in a 17 mm. human embryo arising from the aorta at the level of the 23d intersegmental (3d lumbar) artery.
That the arteries of the gut migrate cephalo-caudad was demonstrated by Mall ('91 and '97), but comparison of the origins of these arteries in this embryo with those described by Tandler ('03) in an embryo of approximately the same length, indicates that the rate of migration is somewhat variable, or we may have to do merely with variations in the point of origin, such as have been demonstrated for the superior mesenteric artery.
Just caudad of the pleural cavities and on a level with the 10th thoracic pair of dorsal intersegmental arteries, there appears a left lateral visceral artery. It extends from the aorta laterally and caudad first along the medial border of, and then through, the suprarenal gland to the Wolffian body. Apparently this vessel becomes the left inferior phrenic artery which furnishes a superior suprarenal branch in the adult. Another pair of lateral visceral (mesonephric) arteries (A.s.-r.) presumably the middle suprarenal of the adult leaves the aorta near the origin of the superior mesenteric artery. This pair of vessels extends laterally in a cephalo-dorsal direction to the anlagen of the suprarenal glands.
Beginning just caudad of the place of origin of the superior mesenteric artery the left components of four successive pairs of lateral visceral (mesonephric) arteries are seen in the reconstruction. The cephalic two have a common stem of origin from the aorta.
The first passes through the caudal part of the suprarenal gland to the Wolffian body. It presumably becomes the inferior suprarenal branch of the left renal.
The second skirts the caudal part of the suprarenal anlage^ passing dorsal to the left suprarenal vein (left subcardinal) , and ends in the region medial to the metanephros of that side. It may be concluded, therefore, that this artery (including the common trunk referred to above) becomes the left renal.
The corresponding artery (renal) on the right arises from the aorta dorsal to the left renal vein (renal anastomosis) and extends to the Wolffian body, passing dorsal to the subcardinal segment of the inferior vena cava.
Broman ('08) finds that the renal arteries take origin from the 21st or 22nd aortic segment.
The third mesonephric artery is small and, hence, of little moment. The corresponding one on the right passes in a strand of mesenchjana through the inferior vena cava (i.e., the supraureteral channel).
The fourth (A.sp.i.) is of considerable caliber. It takes origin at the level of the 21st dorsal intersegmental arteries, dorsal to the termination of the left suprarenal vein, and extends caudad„ lateral to the anlage of the corresponding spermatic vein, to the Wolffian body. In the W^olffian body it supplies several glomeruli,, lying adjacent to the most prominent part of the genital ridge.
The corresponding artery on the right arises from the aorta dorsal to the renal anastomosis. It extends ventral to the vena cava inferior (i.e., the supra-ureteral channel) and caudad along the lateral side of the anlage of the right spermatic vein to the Wolffian body.
These arteries, from their relations, the author believes, are the embryonic representatives of the internal spermatic (ovarian) arteries of the adult.
Broman ('08) finds that usually the spermatic arteries arise from the 22nd aortic segment. Iliac arteries. Each common iliac artery (Aa.il.com.) arises from the lateral wall of the aorta directly opposite the third lumbar pair of dorsal intersegmental arteries, and on a level with the 2nd lumbar nerves. At first of small caliber the common iliacs soon increase notably in diameter. The right common iliac artery is the more fully displayed in the reconstruction (plate 1). It extends laterally and caudad, passing ventral to the caudal segment of the inferior vena cava, medial to the metanephros, and dorsal to the ureter and Wolffian body (compare plate 2). This relation of the common iliac artery, which is preserved in the adult, is probably that of a dorsal intersegmental artery.
The right common iliac artery terminates in two branches, the external iliac (A.il.ext.), a medium sized artery which accompanies the corresponding vein, and the arteria hypogastrica (A.hypogas.).
The external iliac artery first extends laterally and gives off a branch directed cephalad, the inferior epigastric (not shown in the reconstruction). It then continues caudad in the proximal part of the posterior limb-bud to finally join the A. ischiadica.
The hypogastric artery is only a short trunk which divides, lateral to the ureter, into dorsal and ventral branches (the socalled anterior and posterior divisions of adult anatomy). The dorsal branch appears in the drawing only as a short stump, but probably here continues as the superior gluteal artery. The ventral branch soon divides into the large umbilical artery (A.um.d.) and a short trunk common to the sciatic and internal pudendal arteries (A.isch.d. axid A.pud.i.). The umbilical artery extends ventrally in the lateral body wall across the lateral side of the anlage of the bladder into the caudal wall of the umbilical cord.
The sciatic (inferior gluteal and A. comitans n. ischiadica) is at first dorso-lateral to the corresponding vein (V.isch., plate 4) with which it continues into the posterior limb-bud. The tap between it and the femoral has already occurred.
The internal pudendal is at first ventral to the sciatic vein but soon bends ventrally into the cephalo-lateral part of the genital papilla where it becomes the artery of the clitoris or penis, according to sex.
Veins
The general distribution of the veins of the right side of the body is shown in plate 4. Plate 5 is a left lateral, and plate 6 a ventral view of part of the anterior cardinal system.
Anterior and commoyi cardinal system. Between the dorsomedial surfaces of the cerebral hemispheres (plate 4) appears the paired anlage of the superior sagittal sinus (S. sag. sup.). This vessel together with numerous other tributaries arising on the lateral surface of the tel-, di-, and mes-encephalon (Telen.,Dien., and Mesen.) contributes to form the anterior cerebral vein (V.cer.a.) which joins the cephalic end of the cavernous sinus (S.cav.). The ophthalmic vein (V.oph.) which is represented more fully in plate 5, joins the cavernous sinus ventral to the entrance of the anterior cerebral vein.
Venules arising on the lateral surface of the metencephalon (Meten.) unite to form the middle cerebral vein (V.cer.m.) which passes around the caudal border of the semilunar ganglion •(G.s-l.) to enter the cavernous sinus.
The cavernous sinus is medial to the semilunar ganglion, and represents a persistent portion of the primitive anterior cardinal vein. The original anterior cardinal from the cavernous sinus to the site of the future jugular foramen, has disappeared, and a temporary vein, the vena capitis lateralis (V.cap.lat.), has formed lateral to the otocyst and the adjacent cranial nerves. The vena capitis lateralis is joined at the caudal aspect of the otocyst by the posterior cerebral vein (V.cer.p.) which takes its origin from a capillary plexus overlying the caudo-lateral surface of the myelencephalon (Myelen.).
From the point at which it receives the posterior cerebral vein the right anterior cardinal (internal jugular) takes a direct course towards the region of the heart. Dorsal to the heart it unites with the azygos vein (V.az.) to form the right common cardinal (V.card.c.d.). The right common cardinal, which becomes the proximal part of the adult vena cava superior, empties through the sinus venosus (S.v.) into the right atrium (At.d.).
The extra-cranial part of the right anterior cardinal is represented in the adult by the right internal jugular and innominate veins, and by the part of the superior cava distal to the point of entrance of the vena azygos.
The extra-cranial part of the left anterior cardinal (plate 5) at this time is symmetrical in size and position with its fellow of the right side. Just before entering the sinus venosus the left common cardinal (V.card.c.s.) turns to the right and occupies the sulcus coronarius on the diaphragmatic surface of the heart. It is shown in the latter situation in plates 1 and 4.
The vertebral veins of which the right (V.vei'.d.) is reconstructed in plate 4, take the major share in the drainage of the cervical intersegmental veins. They open on either side into the dorsal aspect of the common cardinal near the termination of the azygos and hemiazygos respectively.
The above description of the extra-cranial portions of the anterior cardinal veins includes only the main venous channels draining the sinuses of the developing dura mater. In the opinion of the author the true 'anterior cardinals' are not represented by these channels alone, but by the m.ain trunks of the vertebral veins as well. This opinion is supported by the occurrence in a pig embryo of 7.8 mm. (Thyng '11) of a series of fenestrae in the dorsal portion of the anterior cardinal vein (fig. 2) which appears to foreshadow the segregation of the dorsally placed vertebral vein from the main ventral channel (corresponding to the anterior cardinal as described above). A vertebral vein, thus formed, would receive the cervical intersegmental veins as does the vertebral in this embryo.
The right and left linguo-facial veins (V.ling-fac.) (Grosser '01, Lewis '09, and others) are shown in plates 4 and 5, respectively, and the terminal parts of both are seen in plate 6. Each arises in tributaries from its own side of the tongue, mandible and face. The trunk, thus formed, enters the ventral wall of the internal jugular vein of its own side, near the p'ace where the latter is crossed medially by the hypoglossal nerve (N.hyp.). Earlier in development each linguo-facial vein enters the ventral wall of the anterior cardinal at a more caudal level, i.e., immediately cephalad of the pericardial cavity. Or it may open into the common or even posterior cardinal vein (Lewis '09, p. 34).
Immediately cephalad of the pericardial cavity there now opens into the ventral wall of each anterior cardinal a vein (V.thym.-thyr., plate 6) arising from a venous plexus of the thymic and thyreoid anlagen. For this vein the term, vena thymico-thyreoidea, does not seem inappropriate.
The right and left thymico-thyreoid veins, it seems to the author, undoubtedly unite to form the transverse anastomosis which eventually becomes the left innominate vein. Szawlowski ('91) arrived at a similar conclusion from a study of these veins in the human embryo, as did Anikew ('09) who investigated the question in pig embryos and in a human embryo of 17.5 mm. The vena anonyma sinistra when fully formed, would thus receive tributaries from the caudal part of the thyreoid anlage, the vv. thyreoidese inferiores and ima of the adult.
Dorsal to the vena thymico-thyreoidea the right anterior cardinal receives a vein, arising in a plexus situated at the cephalic end of the thyreoid gland, and the caudo-dorsal wall of the pharynx. It is possible that this vessel may represent the middle thyreoid vein.
The subclavian veins, of which the left is shown in plate 5, (V.scL), are formed by the union of the thoraco-epigastric (V.th.-ep.) and brachial veins. The latter vein begins as the primitive ulnar (V.ul.pr.) vv^hich forms a venous loop at the circumference of the hand-plate. Each subclavian (plate 6) joins the dorso-lateral aspect of the internal jugular vein of its own side somewhat caudal to the place of entrance of the vena thymico-thyreoidea. The plexiform termination of the subclavian (plate 5) , the foramina of which transmit branches of the brachial plexus, would indicate that this vein is still migrating in the cephalic direction.
The terminations of the external jugular veins (V.jug.ex.) are distinct in plate 6. The terminal part of the left external jugular vein (V.jug.ex.s., plate 5) enters the internal jugular on a level with the caudal extremity of the jugular lymph sac (Sac.jug.). It is also connected, more dorsally, by a considerable channel with the terminal part of the subclavian vein at about the same level. The connection with the internal jugular is (usually) temporary, in which case the communication with the subclavian represents the future permanent outlet of the adult vessel. Sometimes a reverse condition occurs. Distally each external jugular vein is connected through a capillary plexus, caudad of the fossa conchae (shown, in part, in plate 5) with the linguo-facial vein and its tributaries from the hyoid arch.
The cephalic vein (Vxeph.) of which only a part is represented here, occupies the radial border of the arm, and passes superficial to the clavicle (which is just beginning to ossify) to join the external jugular. A similar condition of this vein has been represented for a human embryo of 22.8 mm. by Lewis ('09, fig. 4), and for a human embryo of 20 mm. by Evans ('12, fig. 478). The portion of the cephalic vein superficial to the clavicle (V.jug.ceph.) has been named the jugulo-cephalic vein. It usually atrophies since the cephalic commonly acquires a new connection with the axillary. The jugulo-cephalic occasionally persists in the adult, in which case the cephalic remains partially or entirely tributary to the external jugular vein.
The proximal end of the external jugular vein receives ventrally the anterior jugular vein (plat»e 5), proceeding from a superficial venous plexus of the neck. A similar venous connection between the external and anterior jugular veins has been represented in a human embryo of 22.8 mm. by Lewis ('09, fig. 4). Since the anterior jugular vein is in the adult normally a tributary of the external, it may be supposed that that part of the external jugular vein which now opens into the internal jugular, finally becomes the terminal part of the anterior jugular.
Posterior cardinal system. The primitive posterior cardinals " in great part have lost their identity since they have been reduced to sinusoidal channels by the developing Wolffian bodies. These channels, some of which gain prominence have been reduced further or interrupted by the developing metanephros and suprarenal gland.
Minot ('98) demonstrated that the posterior cardinal veins in pig embryos become subdivided by the mesonephric tubules into sinus-like channels, and in 1900 he introduced additional histological evidence of the presence of these channels to which he applied the name 'sinusoids.' It is also noteworthy that Hochstetter ('93) expressed doubt whether in man the azygos and hemiazygos veins could be considered in their entirety as remnants of the posterior cardinals.
Only the right posterior cardinal derivatives have been reconstructed (plate 4) .
The middle sacral component (V.s.m.) of the right posterior cardinal begins in the tail-process and extends cephalad ventrolateral to the arteria sacralis media (A.s.m., pi. 1), receiving as tributaries the right caudal intersegmental veins.
At the level of the fourth and fifth lumbar intersegmental veins the right vena sacrahs media is connected to its companion vein on the left by a relatively large, transverse anastomosis (x'), passing ventral to the middle sacral artery. Other anastomoses of the middle sacral veins exist more caudally.
It is by virtue of this large transverse anastomosis (which becomes the terminal part of the left common iliac vein of the adult) that the left middle sacral and the veins of the left lower extremity, come to drain into the inferior vena cava.
Two venous channels (sub- and supra-ureteral) extend cephalad from the right middle sacral, enclosing between them an area of mesenchyma through which passes the ureter ( Ur.) . The smaller, sub-ureter al channel having at first passed dorsal to the Wolffian body, with the sinusoids of which it is intimately connected, bends medially to terminate in the caudal part of the persistent portion of the right subcardinal vein (V.scard.d.) described below. This channel from its relation to the ureter and right Wolffian body, corresponds perfectly with Hochstetter's 'Urnierenvene.' It is of importance as Hochstetter ('93) pointed out, in that from * its cephalic portion and the tributary mesonephric sinusoids there probably arises the right spermatic vein.
The channel extending from the vena sacralis media, dorsomedial to the ureter (supra-ureteral), is much larger than the sub-ureteral. It extends cephalad, lateral to the aorta, and receives as tributaries the last two thoracic and first four lumbar intersegmental veins. Opposite the 1st lumbar spinal nerve it bends ventrally, and is continued as a large dorso-ventral channel (compare Sabin '09, fig. 11), which terminates in the caudal extremity of the part of the subcardinal which still persists. This dorso-ventral segment of the supra-ureteral channel, extends medial to the metanephros and Wolffian body from both of which it receives tributary veins. One (or more) of these tributaries undoubtedly forms the right renal vein (or veins) . The supra-ureteral channel forms a considerable portion of the definitive inferior vena cava.
The subcardinal veins are derived from Wolffian sinusoids in the region ventral to the mesonephric arteries (Lewis '02).
x4.t this stage the right subcardinal (V.scard.d.) begins at the renal anastomosis (x), which is formed by the confluence of the sub- and supra-ureteral channels from both sides of the body. From the renal anastomosis the right subcardinal vein extends cephalad, ventral to the right suprarenal gland (Gl.s.-r.d.), as a component of the vena cava inferior.
By the development of the suprarenal gland the cephalic part of the V. subcardinalis (which previously opened into the posterior cardinal vein on a level now represented by the cephafic extremity of the gland) has been reduced to sinusoidal channels. The cephalic end of the portion of the right subcardinal vein, now persisting, represents the anlage of the right suprarenal vein (V.s.-r.d.) which connects, as does the azygos, with the suprarenal .sinusoids.
It should be stated here that the presence of sinusoids in the suprarenal glands of mammals and other vertebrates, has been demonstrated histologically by Minot ('00).
Also significant in this connection are the observations of Lewis ('02, p. 236) upon the veins in question in a rabbit embryo, 11 mm. in length. He says "at the upper end of the veins, on either side, cardinal and subcardinal anastomose in condensed mesenchyma probably connected with the suprarenal anlage." The large renal anastomosis (x), referred to above, unites the two subcardinals. It crosses the median line, ventral to the aorta, a little caudad of the superior mesenteric artery (A.wes.s.) of which a short piece has been added in plate 4. It persists as the terminal pari of the left renal vein.
Simultaneously with the transformation of the subcardinal veins that part of the right subcardinal, situated ventral to the developing suprarenal gland has tapped the hepatic sinusoids as described by Lewis ('02). In this manner there has developed that segment of the vena cava inferior, intervening between the subcardinal and common hepatic (V.hepxo7n.) veins.
The entire vena cava inferior, as shown in plate 4, is composed, therefore, of four parts. These are the supra-ureteral channel; a segment of the original right subcardinal (V .scard.d.) intermediate between the renal anastomosis and the termination of the anlage of the right suprarenal vein (V.s.-r.d.); a large channel passing through the plica venae cavae formed by the tapping of the hepatic sinusoids by the right subcardinal, and the teniiinal part, the vena hepatica communis (V. hep.com.), which empties into the right atrium through the sinus venosus (S.v.).
The course of the vena azygos (V.az.) along the lateral side of the aorta has been interrupted in the region of the suprarenal gland, so that it now receives the first ten thoracic intersegmental veins (the first three indirectly, for they unite to form a common trvmk, the vena intercostalis suprema dextra).
The vena azygos, a little cephalad of the termination of the vena intercostalis suprema, also receives a small ventral tributary, the cephalic remnant of an earlier mesonephric sinusoid, figured in pig embryos, by Lewis ('03), Davis ('10), and Thyng ('11). This vessel has been designated by Davis ('10) the ventro-lateral vein of the mesonephros.
At the level of the 10th thoracic intersegmental vein the vena azygos communicates with the hemiazygos by a transverse anastomosis, passing dorsal to the aorta. Slightly cephalad of this anastomosis tributaries from the suprarenal gland (referred to above) join the azygos.
Although the left posterior cardinal system has not been reconstructed, the following observations are here recorded for the sake of completeness.
The sub-ureteral channel (Urnierenvene of Hochstetter) has been obUterated caudally. The supra-ureteral channel is not only much smaller than its companion on the right, but is subdivided caudally by numerous mesenchymal septa, indicative of commencing atrophy.
The left subcardinal vein, caudad of the renal anastomosis has degenerated, but its cephalic extremity is connected with the suprarenal sinusoids as on the right. It will ultimately form the left suprarenal vein, a tributary to the left renal.
The vena hemiazygos still opens into the left common cardinal. It is connected with the azygos by the anastomosis at the level of the 10th thoracic spinal nerve, mentioned above.
Portal system. The left umbilical vein (V.um.s.) is represented in plate 4. In the liver it communicates with the hepatic sinusoids, its blood passing chiefly through an especially large sinusoid, the ductus venosus (D.v.) which joins the left side of the common hepatic vein (vena cava inf erioris) .
The trunk (V.vit.) fomned by the fused vitelline veins (Begg '12) passes through the coelom in a separate strand of mesentery (plate 2). In the dorsal mesentery of the duodenum it is joined by the superior mesenteric vein (V.mes.s.) of which only thestump is here shown. The trunk formed by this union, the vena omphalo-mesenterica (V.07np.-mes.), now bends cephalad, and passes dorsal to the anastomosis of the dorsal and ventral pancreatic outgrowths. After receiving the lienal vein (V.li.) it becomes the vena portae (V.P.) which enters the liver where it joins the hepatic sinusoids, and, by means of a considerable channel, the right side of the ductus venosus.
The hepatic sinusoids discharge into the vena cava inferior (common hepatic segment) by way of the hepatic veins (Vv.hep.).
Lymphatics
The jugular lymph sacs (S.jug.), comparable to those discovered by Sabin ('02) are seen in plate 6. The left also appears in plate 5. They are large, nearly syimnetrical sacs somewhat constricted into segments, and are situated one on either side, immediately lateral to the internal jugular vein. Through the sacs pass branches of the cervical plexus.
Saccus jugularis sinister. The cephalic end of the left sac communicates with the internal jugular vein through a small channel (a). There is also a large caudal connection between the sac and vein which cannot be seen in the reconstruction. The latter opening is in the caudo-medial wall of the sac near the temporary opening of the external jugular vein (V.jug.ex.) into the internal jugular. Incomplete valves, dorsally and ventrally placed, guard the opening.
The branches of the cervical plexus which pass through this sac are seen in plate 5. Through the cephalic extremity extends the great auricular (N.aur.m.). A little more caudally there issues a branch of the third cervical which is joined lateral to the sac by a branch of the fourth cervical nerve. The trunk thus formed, immediately gives off the small occipital, and then bends ventrally across the sac as the N. cutaneous colli.
The vasa lymphatica superficialia (Vas.lym.sup.), arising from the cephalo-lateral portion of the left lymph sac, are seen plainly in plate 6. They extend laterally and dorsally into the subcutaneous tissue. A prolongation from the caudal extremity of the left sac overlies the lateral aspect of the terminal part of the subclavian vein.
Saccus jugularis dexter. The cephalic end of the right lymph sac does not communicate with the internal jugular vein as does the left. Caudally at a level corresponding approximately with the large communication on the left, the right sac opens into the internal jugular. The opening in this case is a very small slit-like aperture between two valves, a lateral and a medial. The lateral valve is adjacent to the permanent temiination of the external jugular vein, while the medial projects into the cavity of the internal jugular.
A prolongation of the sac overlies the lateral surface of the proximal portion of the subclavian vein as occurred on the left.
Saccus mesentericus. There is a plexus of vessels, situated immediately ventral to the aorta, which extends for the most part between the proximal parts of the superior and inferior mesenteric arteries. The cephalic part of this plexus lies between the subcardinal segment of the inferior vena cava and the left suprarenal vein (left subcardinal) , the caudal portion lies dorsal to the renal anastomosis. In places the diameter of the plexus is equal to, or even greater than, that of the adjacent aorta. Although the lumina of the vessels are closely packed with corpuscles, it is difficult to connect the vessels with any of the definitely formed veins. Nevertheless, it would be impossible at least without reconstructions to say that such connections do not exist. From the relations of the plexus, given above, there seems little doubt that it corresponds to the anlage of the mesenteric lymph sac, discovered in rabbit embryos by Lewis ('02 and '05).
Baetjer ('08) who has investigated the development of this sac in a series of pig embryos, concludes that it originates in a series of small veins which separate from the renal anastomosis.
Cisterna chyli. A series of anastomosing venous channels is found on the right and left side, dorso-lateral to the aorta. The cephalic end of this plexus apparently connects with the azygos and hemiazygos veins respectively. Many of the channels open into the supra-ureteral venous channels, previously described. The intersegmental arteries in this locality extend dorsal to these channels, although offshoots of the latter frequently anastomose between the successive pairs of arteries, dorsal to the aorta. These vessels may represent the anlage of the cisterna chyli, for they correspond in position to the anlage as described by Sabin ('09).
Sacci lymphatici posterior es. Definite posterior lymph sacs in relation to the sciatic veins have not been found, but numerous small tributaries, entering the proximal part of the veins may foreshadow them. Sabin ('09) states that they first appear in an embryo of 20 mm., as a plexus of small veins.
Plates
- Embryo 17.8 mm Links: Fig 1 | Fig 2 | Plate 1a | Plate 1b | Plate 2a | Plate 2b | Plate 3a | Plate 3b | Plate 4a | Plate 4b | Plate 5a | Plate 5b | Plate 6 | Harvard Collection | Carnegie stage 19
Reference
Thyng FW. The anatomy of a 17.8 mm human embryo. (1914) Amer. J Anat. 17: 31-112.
Cite this page: Hill, M.A. (2024, April 27) Embryology Paper - The Anatomy of a 17.8 mm Human Embryo. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_The_Anatomy_of_a_17.8_mm_Human_Embryo
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G
1914 Human Embryo 17.8 mm Harvard Collection
Plate Legend
allantois An.hyp. (A}is.hyp.), ansa hypoglossi A 71., anus Ao.dors.Iat., aorta dorsalis lateralis Ao.d.m., Ao. dorsalis mediana Arc.ao., arcus aortae A. anon., arteria anonynia A.aur.p., A. auricularis posterior A.bas., A. basilaris A.car.c.d., A. carotis comnuinis dextra A.car.c.s., A. carotis communis sinistra A.car.ex., A. carotis externa A.car.i.d., A. carotis interna dextra A.car.i.s., A. carotis interna sinistra A.cer.a., A. cerebri anterior A.cer.m., A. cerebri media A.cer.p., A. cerebri posterior A.cereb.s., A. cerebelli superior A.chr.a., A. chorioidea anterior A.chr.p., A. chorioidea posterior A.coel., A. coeliaca Aa.com.p., Aa. communicantes posteriores A.hyp.d., A. hypoglossa dextra Aa.il.com.d. and s., Aa. iliacae communes, dextra et sinistra A.il.ex., A. iliaca externa A.isch.d., A. ischiadica dextra (A. glutea inferior et a. comitans n. ischiadici) A.ling., A. lingualis A.mam.i., A. mammaria interna A.7nax.e.T:., A. maxillaris externa A.mes.s., A. mesenterica superior A.mes.i., A. mesenterica inferior A.occ, A. occipitalis Aa., Aa. oesophageae A.oph., A. ophthalmica A.pud.i., A. pudenda interna Aa.pul., Aa. pulmonales A.s.m., A. sacralis media A.scL, A. subclavia A.sp.i., A. spermatica interna A.stp., A. stapedia .4..s-r., A. suprarenalis A.tJiijr.d., A. thyreoidea dextra A.thip-.s., A. thyreoidea sinistra A.nni.d., A. umbilicalis dextra A.vert.d., A. vertebralis dextra A.vert.s., A. vertebralis sinistra At.d., atrium dextrum ^^s., At. sinistrum
B., vesicula urinaria Br.s., bronchus sinister B.phary., bursa pharyngea
Ch.pr., choana primitiva Con. art., conus arteriosus Corp.pin., corpus pineale Co7'p.str., corpus striatum
Dien., diencephalon D.a., ductus arteriosus D.c, D. cochlearis D.cyst., D. cysticus D.end., D. endolymphaticus D.hep., D. hepaticus D.p.d., D. pancreatis dorsalis D.V., D. venosus D.vit., D. vitellinus D.M., D. Mulleri D.W., D. Wolffii Duo., duodenum
Em.i-p., eminentia interpeduncularis Ep., epiglottis Eth.-turb.I., ethmo-turbinale I
Fim., fimbriae Flex.ceph., flexura cephalica Flex.cerv., flexura cervicalis Fl.pl., floor-plate Fo.ep., foramen epiploicum Fo.int., foramen interventriculare (Monroi) Fo.iv., foramen interventriculare
G.gn., ganglion geniculatum Gg. hyp., Gg. hypoglossa G.jiig., G. jugularc G. nodes., G. nodosum G.petros. (G.p.), G. petrosum G.s-l., G. semilunaris G.sup., G. superius G.ves., G. vestibularis Ga., gaster G.R., genital ridge Gl.inf., glandula infundibularis Gl.p., Gl. parotidis Gl.smx.iGl.s.), Gl. submaxillaris Gl. s-r.d., Gl. suprarenalis dextra
Hyp., hypophysis Int.cr., intestinum crassum
Int.r. (Int.rec.), intestinum rectum Int.t., intestinum tenue Isth., isthmus L., lens
Md., mandible Max.-turb., maxillo-turbinale Md.sp., medulla spinalis Mem. u.-g., membrana urogenitalis Mesen., mesencephalon Mesogas., mesogastrium Met., metencephalon Myelen., myelencephalon
Na., naris N.a., aberrant nerve of myelencephalon N.abd. (N.ab.), nervus abducens N.acc, N. accessorius N.alv.inf., N. alveolaris inferior N.acus., N. acusticus N.aur.m., N. auricularis magnus N.c.i., N. cardiacus inferior N.ch.tymp. (Ch.ty.), N. chorda tympani N.c.c, N. cutaneous colli N.fac, N. facialis N.fr., N. frontalis N.glos., N. glossopharyngeus N.hyp., N. hj'poglossus N.int., N. intermedins N.laryng.s. (N .I.S.), X. laryngeus superior N.l. (l.), N. lingualis N.md., N. mandibularis N.mx., N. maxillaris N.na.-cil., N. nasociliaris N.occ.m., N. occipitalis major N.oc, N. oculomotorius Nn.olf., Nn. olfactorii N.oph., N. ophthalmicus N.pet.s.m., N. petrosus superficialis major N.phr., X. phrenicus N.7-ec.s., X. recurrens sinister Nn.sp., Nn. spinales Nn.s.-cl.iNn.s-c), Xn. supraclavicu lares (common trunk) N.troch., N. trochlearis N.tyvi., N. tympanicus N.vag., X. vagus
Oc, oesophagus Op.s., optic stalk Or.vom.-nas., organon vomero-nasale O.S., ostium secundum
Panc.d.and v., pancreas dorsale et ven trale Pa.geti., papilla genitalis P.-thyr.IV ., parathyreoidea IV Ph.P.l, pharyngeal pouch 1 Ph.P.2, pharyngeal pouch 2 (region only) Ph., pharynx Pl.ch.p., plica chorioidea posterior Pr.i-m., processus intermaxillaris Pr.pl., processus palatinus Pr.ver., processus vermiformis
R., raphe R.des., ramus descendens (hypoglossi) R.ex., ramus externus (accessorii) R.p-tr., ramus posttrematicus (of fourth pharyngeal pouch) R.po-op., recessus postopticus R.p-op., recessus preopticus Rhin., rhinencephalon R.or., rima oris Rf.pl., roof-plate Sac, sacculus Sac.jug. (S.jug.), saccus lymphaticus jugularis
S., septum aorticum (aortico-pulmo nale) S.u.-7\, septum uro-rectale S.cav., sinus cavernosus S. sag. sup., S. sagittalis superior S.u.-g., S. uro-genitalis S.V., S. venosus S (plate 1) vestigial portions of dorsolateral aortae Sul., sulcus (mesencephali?)
T.I., ganglion thoracicale primum Telen., telencephalon Thy., thymus Thyr., glandula thyreoidea Thyr.m., gl. thyreoidea media /' and t", anterior and posterior parts of the tongue 7V., trachea Tr.aor., truncus aorticus Tr.cost.cerv., Tr. costocervicalis Tr.pul., Tr. pulmonalis Tr.thyr.-cerv., Tr. thyreocervicalis Tiib.p., tuberculum posterius T.W., tubuli Wolffii
Umb.c, umbilical cord Ur., ureter Ut., utriculus
Vas.lymp.sup., vasa lymphatica super ficialia Vv.v.s., valvulae sinus venosi V.b., valvula bicuspidalis V.t., V. tricuspidalis V.az., vena azygos V.cap.lat., V. capitis lateralis V.card.a., V. cardinalis anterior V.card.c.d., V. cardinalis communis dextra F. card. c. s. (F. card. com. .s), V. cardinalis communis sinistra V.card.p., V. cardinalis posterior V.ceph., V. cephalica V.cer.a., V. cerebralis anterior V.cer.m., V. cerebralis media V.cer.p., V. cerebralis posterior V.fem., V. femoralis Vv.hep., Vv. hepaticae V. hep.com., V. hep. communis V.isch., V. ischiadica V.jug.-ceph., V. jugulo-cephalica V. jug. ex., V. jugularis externa V.jug.i., V. jugularis interna V.li., V. lienalis V.ling.-fac, V. linguo-facialis V.mes.s., V. mesenterica superior V.oph., V. ophthalmica V.om,p.-mes., V. omphalo-mesenterica F.P., V. portae Vv.pul.d., Vv. pulmonales dextrae V.s.m., V. sacralis media V.scard.d., V. subcardinalis dextra V.scl., V. subclavia F.s-r.d., V. suprarenalis dextra V.th.-ep., V. thoracico-epigastrica V.thy.-thyr., V. thymico-thyreoidea V.ul.pr., V. ulnaris primitiva V.um.s., V. umbilicalis sinistra V.ver.d., V. vertebralis dextra V.V., Vv. vitellinae (fused) Vent.d., ventriculus dexter Ves.fel., vesica fellea Ves.op., vesicula optica
X, renal anastomosis (V. renalis sinistra) x', transverse iliac anastomosis (V. iliaca communis sinistra)
A', strand of mesentery containing the fused vitelline veins y, branch of right jugular lymph sac
Z.dors., zona dorsalis Z.vent., zona ventralis 1 to 8, ganglia cervicales Abas.
Plate 1
Reconstruction to illustrate chiefly the interior of the brain and the spinal cord; the digestive system and its appendages; the arterial system; the left atrium and ventricle of the heart, and m part the urogenital system of a 17.8 mm. human embryo (H. E. C. 839). X 11.2 diams.
Plate 2
This plate consists of two reconstructions. The upper shows a left lateral view of the brain and cervical cord with the nerves in situ, the aortic arch, and other arteries of the left side of the head and neck. It also represents the oral, nasal and pharyngeal epithelia; the left thymic and thyreoid anlagen; and illustrates in a measure the relation of the nerves and arteries to these epithelial structures. The lower reconstruction shows the pancreas and spleen within the mesogastrium (a portion of the stomach having been removed) ; the left genital ridge, and the left meso- and metanephros with their ducts opening independently into the urogenital division of the cloaca (H. E. C. S39). X 11.2 diams.
Plate 3
Reconstruction showing the right side of the brain and the cervical cord ; the right cranial and cervical nerves; the internal ear, and the optic vesicle of a 17.8 mm. human embryo (H. E. C. 839). X 11.9 diams.
Plate 4
Reconstruction to show the right atrium and ventricle of the heart; the venous system of the right side; and the left umbilical vein of a 17.8 human embryo (H. E. C. 8.39). X 11.2 diams.
Plate 5
Reconstruction to show the lateral aspect of the left cephalic and cervical veins, and the left [jugular lymph sac of a 17.8 mm. human embryo (H. E. C. 839). X 11.9 diams.
Plate 6
Bibliography
Anikew, a. 1909 Zur Frage liber die Entwickelung tier Vena anonj-ma sinister. Anat. Anz., Bd. 34, S. 24-29.
Baetjer, W. A. 1908 The origin of the mesenteric sac and thoracic duct in the embryo pig. Am. Jour. Anat., vol. 8, pp. .303-317, with one plate figure.
Begg, A. S. 1912 The anomalous persistence in embryos of parts of the periintestinal rings formed by the vitelline veins. Am. Jour. Anat., vol. 12, pp. 103-110.
Bell, E. T. 1905 The development of the thymus. Am. Jour. Anat., vol. 5, pp. 29-62, pis. 1-3.
Born, G. 1883 Ueber die Derivate der embryonalen Schlundbogen und Sch lundspalten bei Saugethieren. Arch, fiir mikr. Anat., Bd. 22, S. 271 318, Taf. 10-11.
- 1889 Beitrage zur Entwickelungsgeschichte des Siiugethierherzens. Arch, fiir mikr. Anat., Bd. 33, S. 284-378, Taf. 19-22.
Bremer, J. L. 1902 On the origin of the pulmonary arteries in mammals. Am. Jour. Anat., vol. 1, pp. 137-144.
- 1908 Aberrant roots and branches of the abducent and hypoglossal nerves. Jour. Comp. Neur., vol. 18, pp. 619-639.
Broman, I. 1904 Die Entwickelungsgeschichte der bursa omentalis. S. 1 602, Taf. 1-20; Wiesbaden.
- 1908 tjber die Entwickelung und "Wanderung" der Zweige der Aorta abdominalis beim IMenschen. Anat. Hefte, Heft 110, S. 407-549.
Burckhart, R. 1891 Untersuchungen am Hirn und Geruchsorgan von Triton und Ichthyophis. Zeitschr. fiir wiss. Zool., Bd. 52, S. 369-403, Taf. 21-22.
Chievitz, J. H. 1885 Beitrage zur Entwickelungsgeschichte der Speicheldriisen. Arch, fiir Anat. und Phys., anat. Abth., S. 401-436, Taf. 19.
Davis, D. M. 1910 Studies on the chief veins in early pig embryos and the origin of the vena cava inferior. Am. Jour. Anat., vol. 10, pp. 461-472, pis. 1-6.
Elze, C. 1907 Beschreibung eines menschlichen Embryo von zirka 7 mm. grosster Lange. Anat. Hefte, Heft 106, S. 411^92, Taf. 14-20.
Evans, H. M. 1909 On the development of the aortae, cardinal and umbilical veins, and the other blood vessels of vertebrate embryos from capillaries. Anat. Rec, vol. 3, pp. 498-518.
- 1912 The development of the vascular system. Keibel and Mall's human embryology, vol. 2, pp. .570-709; Philadelphia and London.
Forssner, H. 1907 Die angeborenen Darm- und Osophagusatresien. Anat. Hefte, Heft 102, S. 1-163, Taf. 1-9.
Fox, H. 1908 The i^haryngeal pouches and their derivatives in the Mammalia. Am. Jour. Anat., vol. 8, pp. 187-250.
Froriep, a. 1885 Ueber Anlagen von Sinnesorganen am Facialis, Glosso pharyngeus and Vagus, u.s.w. Arch, fiir Anat. und Phys., anat. Abth., S. 1-55, Taf. 1-2.
Greil, a. 1905 tjber die Anlage der Lungen, sowie der ultimobranchialen (postbranchialen, suprapericardialen) Korper bei anuren Amphibien. Anat. Hefte, Heft 89, S. 445-506, Taf. 46-50.
Grosser, O. 1901 Zur Anatomie und Entwickelungsgeschichte des Gefass systemes der Chiropteren. Anat. Hefte, Heft 55, S. 205-424, Taf. 13-25.
- 1907 Die Elemente des Kopvenensystems der Wirbeltiere Verb. d. Anat. Ges. 21, S. 179-192.
Hammar, J. A. 1901 Notiz liber die Entwickelung der Zunge und der Mund speicheldriisen beim Menschen. Anat. Anz., Bd. 19, S. 570-575.
- 1902 Studien liber die Entwickelung des Vorderdarms und einiger angrenzenden Organe. Abt. I. Allegemeine Morphologie der Schlundspalten beim Menschen. Entwickelung des Mittelohrraumes und des ausseren Gehorganges. Arch, flir mikr. Anat., Bd. 59, S. 471628, Taf. 26-29
- 1903 Abt. II. Das Schicksal der zweiten Schlundspalte. Zur vergleichenden Embryologie und Morphologie der Tonsille. Arch, fur mikr. Anat., Bd. 61, S. 404-458, Taf. 21-22.
1911 Zur groberen Morphologie und Morphologenie der Menschen thymus. Anat. Hefte, Heft 129, S. 203-242, Taf. 27-32.
Henle, J. 1868 Handbuch der systematischen Anatomie des Menschen. Bd. 3, Braunschweig. His, W. 1880-1885 Anatomie menschlicher Embryonen. Text und Atlas, Leipzig.
1886 Zur Geschichte des menschlichen Rlickenmarkes und der Ner venwurzeln. Abh. math.-phys. Kl. Sachs. Akad. Gesellsch. d. Wiss., Bd. 13, S. 479-544 mit 1 Tafel.
1892 Zur allgemeinen Morphologie des Gehirns. Arch, flir Anat. und Phys., anat. Abth., S. 346-383.
1895 Die anatomische Nomenclatur. Arch, flir Anat. und Phys., anat. Abth., Suppl., S. 1-180.
Hochstetter, F. 1890 Ueber die Entwickelung der A. vertebralis beim Kanninchen, nebst Bemerkungen liber die Entstehung der Ansa Vieussenii. Morphol. Jahrbuch, Bd. 16, S. 572-586, Taf. 21.
- 1893 Beitrage zur Entwickelungsgeschichte des Venensystems der Amnioten. Morphol. Jahrbuch, Bd. 20, S. 543-648, Taf. 21-23.
- 1898 Ueber die Pars membranacea septi. Vortrag gehalten in der wissenschaftlichen Aerztegesellschaft zu Innsbruck. Wiener Klinische Wochenschrift, 11, S. 247-248.
Ingalls, N. W. 1907 Beschreibung eines menschlichen Embryos von 4.9 mm. Arch, fiir mikr. Anat., Bd. 70, S. 506-576, Taf. 29-31.
Johnson, F. P. 1910 The development of the mucous membrane of the oesophagus, stomach, and small intestine in the human embryo. Am. Jour. Anat., vol. 10, pp. 521-561.
Johnston, J. B. 1909 The morphology of the forebrain vesicle in vertebrates. Jour. Comp. Neur., vol. 19, pp. 457-540.
Kallius, E. 1901 Beitrage zur Entwickelung der Zunge. Verh. Anat. Ges. 15, S. 41-42.
Kastschenko, N. 1887 Das Schicksal der embryonalen Schlundspalten bei Saugethieren. Arch, fur mikr. Anat., Bd. 30, S. 1-26, Taf. 1-2.
Keith, A., and Jones, F. W. 1901 A note on the development of the fundus of the human stomach. Proc. Anat. Soc. Gr. Brit, and Ire., pp. 34 38 (in Jour. Anat. and Physiol., vol. 36, 1902).
Kreuter, E. 1905 Die angeborenen Verschliessungen und Verengerungen des Darmkanals in Lichte der Entwickelungsgeschichte. Deutsche Zeit schr. fiir Chir., Bd. 79, S. 1-89.
VON Kupffer, K. 1903-05 Die Morphogenie des Centralnervesystems. Hertwig's Handbuch der Entwickelungslehre der Wirbelthiere, Bd. 2, Teil 3; Jena, 1906.
Lewis, F. T. 1902 The development of the vena cava inferior. Am. Jour. Anat., vol. 1, pp. 229-244, pis. 1-2.
- 1903 The gross anatomy of a 12 mm. pig. Am. Jour. Anat., vol. 2, pp. 211-225, pis. 1-4.
- 1905 The development of the lymphatic system in rabbits. Am. Jour. Anat., vol. 5, pp. 95-111.
- 1906 The mixed cerebral nerves in mammals. Jour. Comp. Neur., vol. 16, pp. 177-182, pi. 17.
- 1909 On the cervical veins and lymphatics in four human embryos. Am. Jour. Anat., vol. 9, pp. 33-42.
- 1912 The form of the stomach in human embryos, with notes upon the nomenclature of the stomach. Am. Jour. Anat., vol. 13, pp. 477-503.
- 1912 Development of the oesophagus (pp. 355-368), and development of the small intestine (pp. 381-393). Keibel and Mall's human embryology, vol. 2; Philadelphia and London.
Lewis, F. T., and Thyng, F. W. 1908 The regular occurrence of intestinal diverticula in embryos of the pig, rabbit and man. Am. Jour. Anat., vol. 7, pp. 505-519.
Mall, F. P. 1891 A human embryo of twenty-six days old. Jour. Morph., vol. 5, pp. 459-477, pis. 29-30.
- 1897 Development of the human coelom. Jour. Morph., vol. 12, pp. 395-453.
MiNOT, C. S. 1898 On the veins of the Wolffian bodies in the pig. Proc. Bost.Soc. Nat. Hist., vol. 28, pp. 265-274, pi. 1.
- 1900 On a hitherto unrecognized form of blood circulation. Proc.Bost. Soc. Nat. Hist., vol. 29, pp. 185-215.
- 1910 Laboratory text-book of embryology. Second edition; Philadelphia.
Mouldenhaur, W. 1877 Die Entwickelung des mittleren und des ausseren Ohres. Morphol. Jahrbuch., Bd. 3, S. 106-151, Taf. 6-9.
Pallin, G. 1901 Beitrage zur Anatomie und Embryologie der Prostate und Samenblasen. Arch, fiir Anat. und Phys., anat. Abth., S. 135-176, Taf. 5-8.
Paulet, J. L. 1910 Kopf- und bucconasale Bildung eines menschlichen Em brj'o von 14.7 mm. Scheitelsteisslange. Arch, fiir mikr. Anat., Bd. 76, S. 658-681, Taf. 25-26.
Peter, K. 1901 Die Entwickelung des Geruchsoigans, u.s.w. Hertwig's Handbuch der Entwickelungslehre der Wirbelthiere, Bd. 2, Teil 2; Jena, 1906.
PiERSOL, G. A. 1888 liber die Entwickelung der embryonalen Schlundspalten und ihre Derivate bei Saugethieren. Zeitschr. flir wiss. Zool., Bd. 47, S. 155-189, Taf. 1&-17.
Prenant, a. 1894 Contribution a I'etude du developpement organique et histologique du thymus, de la glande thyroide, et de la glande caro tidienne. La Cellule, tome 10, pp. 87-184, planches 1-4.
Prentiss, C. W. 1910 The development of the hypoglossal ganglia of pig embryos. Jour. Comp. Neur., vol. 20, pp. 265-282.
Ravn, E. 1889 Ueber die Bildung der Scheidewandzwischen Brust- und Bauch hohle in Siiugethierembryonen. Arch, filr Anat. und Phys., anat.
Abth., S. 123-154, Taf. 9-10. Retzius, a. 1857 Bemerkungen liber das Antrum Pylori beim Menschen und einigen Thieren. Arch, fiir Anat. Phys. und wiss. Med., S. 74^87.
Sarin, F. R. 1902 On the origin of the lymphatic system from the veins, and the development of the lymph hearts and the thoracic duct in the pig. Am. Jour. Anat., vol. 1, pp. 367-389.
- 1909 On the development of the lym))lmtic system in human embryos, with a consideration of the morphology of the system as a whole. Am.Jour. Anat., vol. 9, pp. 43-91.
ScHULTZE, O. 1897 Grundriss der Entwickelungsgeschichte des Menschen und der Saugethiere. 8. 1-468; Leipzig.
SouLiE, A., ET Badier, E. 1907 Recherches sur le developpement du larynx chez I'homme. Journ. de I'Anat. et de la Phys., vol. 43, pp. 137-240, planches 10-14.
Sterzi, G. 1904 Die Blutgefjisse des Rilckenmarks. Anat. Hefte, Heft 74, S. 3-364, Taf. 1-4.
Streeter, G. L. 1904 The development of the cranial and spinal nerves in the occipital region of the human embryo. Am. Jour. Anat., vol. 4, pp. 83-116, pis. 1-4.
SzAWiiOWSKi, J. 1891 Zur Morphologie der Venen der oberen Extremitat und des Halses. Dokt.-Diss., St. Petersburg.
Tandler, J. 1900 Zur Entwickelungsgeschichte des menschlichen Duodenum. Morphol. Jahrbuch, Bd. 29, S. 187-216.
- 1902 Zur Entwickelungsgeschichte der Kopfarterien bei den Mammalia. Morphol. Jahrbuch, Bd. 30, S. 275-373, Taf. 3-5.
- 1903 Zur Entwickelungsgeschichte der menschlichen Darmarterien. Anat. Hefte, Heft 71, S. 189-210.
Thyng, F. W. 1908 Models of the pancreas in embryos of the pig, rabbit, cat and man. Am. Jour. Anat., vol. 7, pp. 489-503.
- 1911 The anatomy of a 7.8 mm. pig embryo. Anat. Rec, vol. 5, pp. 17-45.
Zimmerman, W. 1889 Rekonstruktion eines menschlichen Embryos von 7 mm. Lange, aus der 4 Woche. Verh. d. Anat. Ges., 3, S. 139-142.
Zuckerkandl, E. 1901 Ueber Nebenorgane des Sympathicus im Retropcrito naealraum des Menschen. Verh. d. Anat. Ges. 15, S. 95-107.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 27) Embryology Paper - The Anatomy of a 17.8 mm Human Embryo. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_The_Anatomy_of_a_17.8_mm_Human_Embryo
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G