Introduction

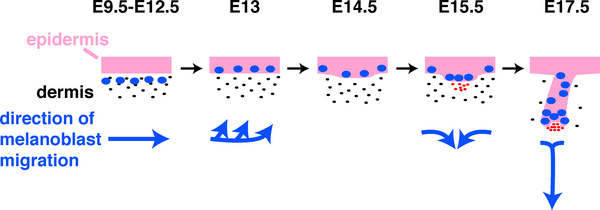
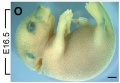
Mouse melanoblast distribution (E12.5 - E16.5)
[1]Melanocytes are located on the surface of the body in the skin and also associated with internal structures, such as the inner ear and eye. Melanocytes in the skin synthesise the pigment melanin that is then transferred to keratinocytes.
These cells are neural crest in origin and recent research suggests that skin melaocytes are derived from the same population that Schwann cells are derived. Schwann cells wrap around nerve axon processes outside of the central nervous system. These cells in development originate from neural crest cells migrating out along the developing nerve fibers and these cells differentiate to form myelin sheaths that surround the mature nerve. The cells are named after their original discoverer a German physiologist Theodor Schwann (1810 - 1882).
In mammals, these pigmented cells can also be found in other tissues such as the: eyes, ears, heart, and central nervous system meninges. Melanocyte cells are a key topic in medical research as they are the cell transformed in melanoma.
See Integumentary System Development
Some Recent Findings
- The exon junction complex (EJC) component Magoh regulates proliferation and expansion of neural crest-derived melanocytes.[2] "Defective melanoblast development and function underlies many disorders including Waardenburg syndrome and melanoma. Understanding the genetic regulation of melanoblast development will help elucidate the etiology of these and other neurocristopathies. Here we demonstrate that Magoh, a component of the exon junction complex, is required for normal melanoblast development. Magoh haploinsufficient mice are hypopigmented and exhibit robust genetic interactions with the transcription factor, Sox10. These phenotypes are caused by a marked reduction in melanoblast number beginning at mid-embryogenesis."
- Generation of human melanocytes from induced pluripotent stem cells[3] "We generated iPS cell lines from human dermal fibroblasts using the Yamanaka factors (SOX2, OCT3/4, and KLF4, with or without c-MYC). These iPS cell lines were subsequently used to form embryoid bodies (EBs) and then differentiated into melanocytes via culture supplementation with Wnt3a, SCF, and ET-3. Seven weeks after inducing differentiation, pigmented cells expressing melanocyte markers such as MITF, tyrosinase, SILV, and TYRP1, were detected. Melanosomes were identified in these pigmented cells by electron microscopy, and global gene expression profiling of the pigmented cells showed a high similarity to that of human primary foreskin-derived melanocytes"
- Schwann cell precursors from nerve innervation are a cellular origin of melanocytes in skin[4] "Current opinion holds that pigment cells, melanocytes, are derived from neural crest cells produced at the dorsal neural tube and that migrate under the epidermis to populate all parts of the skin. Here, we identify growing nerves projecting throughout the body as a stem/progenitor niche containing Schwann cell precursors (SCPs) from which large numbers of skin melanocytes originate. SCPs arise as a result of lack of neuronal specification by Hmx1 homeobox gene function in the neural crest ventral migratory pathway. Schwann cell and melanocyte development share signaling molecules with both the glial and melanocyte cell fates intimately linked to nerve contact and regulated in an opposing manner by Neuregulin and soluble signals including insulin-like growth factor and platelet-derived growth factor. These results reveal SCPs as a cellular origin of melanocytes, and have broad implications on the molecular mechanisms regulating skin pigmentation during development, in health and pigmentation disorders."
|
| More recent papers
|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
- This search now requires a manual link as the original PubMed extension has been disabled.
- The displayed list of references do not reflect any editorial selection of material based on content or relevance.
- References also appear on this list based upon the date of the actual page viewing.
References listed on the rest of the content page and the associated discussion page (listed under the publication year sub-headings) do include some editorial selection based upon both relevance and availability.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References
Search term: Melanocyte Development
<pubmed limit=5>Melanocyte Development</pubmed>
|
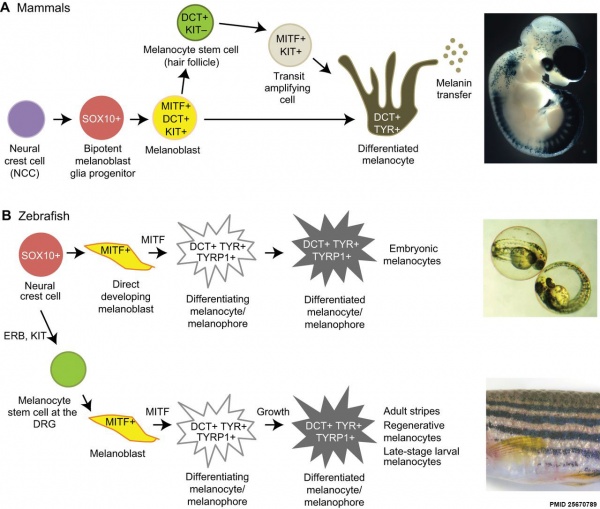
Overview of Melanocyte Development in Mammals and Zebrafish[5]
Skin Pigmentation
Ephilis (freckle)
(pl., ephilides; freckle) Clinical term describing a "freckle", that is a small brown or tan mark on the skin. These inherited features result from a copy of variant Melanocortin 1 Receptor (MC1R) gene and are common on fair skinned Celtic children. Melanocytes produce locally more melanin, this can also increase following exposure to ultraviolet radiation in sunlight.
- Links: OMIM MC1R
Cafe-au-lait spots
(French, cafe-au-lait = coffee with milk; café-au-lait macule; birthmark) describes the characteristic colour of the hyperpigmented skin patch. The common name (birthmark) reflects the presence at birth (congenital) or appearing in early infancy. The pigment is produced locally by melanocytes, that produce all skin pigmentation.
Appear commonly as a solitary feature, multiple café-au-lait macules are associated with various genetic syndromes including Neurofibromatosis type 1 and 2.
- Links: OMIM - NF1
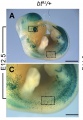
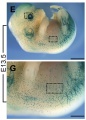
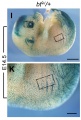
Mouse Melanocytes
Developing mouse ((bt9J/bt9J) melanoblast distribution in whole mount and trunk detail (green - β-galactosidase stained).[1]
Mouse Melanoblast Migration
See also Modeling melanoblast development[6]
- Links: Neural Crest Development
Zebrafish Melanocytes

Erbb3b gene is required to establish melanocyte stem cells in the embryo that are responsible for regenerating melanocytes after melanocytes are ablated in the larval zebrafish. Because this adult stem cell is not required for the development of embryonic melanocytes, we conclude that adult melanocyte stem cells develop in parallel to the embryonic tissues that they regulate.[7]
Inner Ear Melanocytes
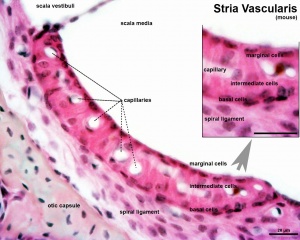
Melanocytes have been identified within the inner ear within the modiolus, vestibular and stria vascularis (intermediate cells).[8] ((see review [9])
The stria vascularis, in addition to blood vessels, consists of 3 main cell types:
- Marginal cells - epithelial origin - line the lumen of the cochlear duct
- Basal cells - mesoderm origin - form a continuous layer
- Intermediate cells - neural crest origin - scattered between the marginal (interdigitated with the marginal cells) and basal cell layers - melanocyte-like cells derived from neural crest
In addition to their role in stria vascularis development, these melanocytes have several suggested adult roles:[9]
- biological reservoir for divalent ions and as an ion exchanger
- intracellular buffering system for calcium.
- binding ototoxic drugs
See also the recent paper on development of the stria vascularis and potassium regulation in the human fetal cochlea.[10]
Note that in dogs, abnormal development of neural crest intermediate cells in the stria vascularis has also been shown to be associated with Canine Congenital Sensorineural Deafness (CCSD).
- Links: Stria Vascularis | Inner Ear Development
References
- ↑ 1.0 1.1 Silver DL, Hou L, Somerville R, Young ME, Apte SS & Pavan WJ. (2008). The secreted metalloprotease ADAMTS20 is required for melanoblast survival. PLoS Genet. , 4, e1000003. PMID: 18454205 DOI.
- ↑ Silver DL, Leeds KE, Hwang HW, Miller EE & Pavan WJ. (2013). The EJC component Magoh regulates proliferation and expansion of neural crest-derived melanocytes. Dev. Biol. , 375, 172-81. PMID: 23333945 DOI.
- ↑ Ohta S, Imaizumi Y, Okada Y, Akamatsu W, Kuwahara R, Ohyama M, Amagai M, Matsuzaki Y, Yamanaka S, Okano H & Kawakami Y. (2011). Generation of human melanocytes from induced pluripotent stem cells. PLoS ONE , 6, e16182. PMID: 21249204 DOI.
- ↑ Adameyko I, Lallemend F, Aquino JB, Pereira JA, Topilko P, Müller T, Fritz N, Beljajeva A, Mochii M, Liste I, Usoskin D, Suter U, Birchmeier C & Ernfors P. (2009). Schwann cell precursors from nerve innervation are a cellular origin of melanocytes in skin. Cell , 139, 366-79. PMID: 19837037 DOI.
- ↑ Mort RL, Jackson IJ & Patton EE. (2015). The melanocyte lineage in development and disease. Development , 142, 620-32. PMID: 25670789 DOI.
- ↑ Larue L, de Vuyst F & Delmas V. (2013). Modeling melanoblast development. Cell. Mol. Life Sci. , 70, 1067-79. PMID: 22915137 DOI.
- ↑ Hultman KA, Budi EH, Teasley DC, Gottlieb AY, Parichy DM & Johnson SL. (2009). Defects in ErbB-dependent establishment of adult melanocyte stem cells reveal independent origins for embryonic and regeneration melanocytes. PLoS Genet. , 5, e1000544. PMID: 19578401 DOI.
- ↑ Steel KP & Barkway C. (1989). Another role for melanocytes: their importance for normal stria vascularis development in the mammalian inner ear. Development , 107, 453-63. PMID: 2612372
- ↑ 9.0 9.1 Meyer zum Gottesberge AM. (1988). Physiology and pathophysiology of inner ear melanin. Pigment Cell Res. , 1, 238-49. PMID: 3070525
- ↑ Locher H, de Groot JC, van Iperen L, Huisman MA, Frijns JH & Chuva de Sousa Lopes SM. (2015). Development of the stria vascularis and potassium regulation in the human fetal cochlea: Insights into hereditary sensorineural hearing loss. Dev Neurobiol , 75, 1219-40. PMID: 25663387 DOI.
Reviews
Harris ML, Baxter LL, Loftus SK & Pavan WJ. (2010). Sox proteins in melanocyte development and melanoma. Pigment Cell Melanoma Res , 23, 496-513. PMID: 20444197 DOI.
Ernfors P. (2010). Cellular origin and developmental mechanisms during the formation of skin melanocytes. Exp. Cell Res. , 316, 1397-407. PMID: 20211169 DOI.
Cooper CD & Raible DW. (2009). Mechanisms for reaching the differentiated state: Insights from neural crest-derived melanocytes. Semin. Cell Dev. Biol. , 20, 105-10. PMID: 18935965 DOI.
Articles
Stolt CC, Lommes P, Hillgärtner S & Wegner M. (2008). The transcription factor Sox5 modulates Sox10 function during melanocyte development. Nucleic Acids Res. , 36, 5427-40. PMID: 18703590 DOI.
Levy C, Khaled M & Fisher DE. (2006). MITF: master regulator of melanocyte development and melanoma oncogene. Trends Mol Med , 12, 406-14. PMID: 16899407 DOI.
Search PubMed
Search Dec 2010 "Melanocyte Development" All (2294) Review (443) Free Full Text (704)
Search Pubmed: Melanocyte Development
Terms
| Integumentary Terms
|
Integumentary Development
- acrosyringium - coiled intra-epidermal region of the eccrine gland sweat duct.
- apocrine gland - (sweat gland) proteinaceous secretion associated with hair (axilla, areola, genital and anal regions). Additional glands associated with eyelashes are called the glands of Moll (ciliary gland). (More? image - apocrine secretion)
- arrector pili muscle - bundle of smooth muscle associated with hair follicle, inserts into the papillary layer of the dermis and attaches to the dermal sheath of the hair follicle. (More? image - arrector pili muscle)
- Blaschko lines - (lines of Blaschko) may represent pathways of epidermal cell migration and proliferation during development. Specific type of lupus erythematosus shows this distinctive pattern. Named after Alfred Blaschko a German dermatologist who first described the feature in 1901. (More? PMID 21396561 | Historic Terminology)
- bulb - the hair follicle enlargement located at its deepest end, dividing cells form the hair and the root sheath.
- café-aut-lait macule - (French, cafe-au-lait = coffee with milk; birthmark) describes the characteristic colour of the skin hyperpigmented patch present at birth (congenital) or appearing in early infancy. Common single feature, multiple are associated with various genetic syndromes including Neurofibromatosis type 1 and 2.
- corneocytes - terminally differentiated keratinocytes forming the stratum corneum.
- cutis - alternative term for the epidermis and the dermis layers of the skin.
- dermal papillae - interdigitation of the dermis with the epidermis.
- dermatoglyphic patterns - (Greek, derma = "skin", glyph = "carving") fingers, palms, toes, and soles skin patterns.
- dermis - connective tissue middle layer of the skin, consists of two sublayers (papillary and reticular layers) that do not have a clear boundary. Embryologically derived from the somite dermatome.
- dermomyotome - Early embryonic dorsolateral half of the somite that will later divide to form both the dermatome and myotome. The dermatome will contribute the dermis and hypodermis of the skin. The myotome will contribute the skeletal muscle of muscoloskeletal system. Development sequence: mesoderm to paraxial mesoderm to somite to "dermomyotome" then dermatome and myotome. (More? Somitogenesis | Musculoskeletal System Development | Integumentary System Development)
- eccrine gland (Greek, ekkrinein = "secrete"; merocrine glands) sweat glands unique to some primates and used in humans for thermoregulation. Adult body has 2 to 4 million sweat glands with concentrations (700/cm) on the palms of the hand, soles of the feet and forehead. Secretion is timulated by sympathetic nervous system, post-ganglionic cholinergic branch, and other stimuli
- ephilis - (pl., ephilides; freckle) Clinical term describing a "freckle", that is a small brown or tan mark on the skin. These inherited features result from a copy of variant Melanocortin 1 Receptor (MC1R) gene and are common on fair skinned Celtic children. Melanocytes produce locally more melanin, this can also increase following exposure to ultraviolet radiation in sunlight. (More? Integumentary | Neural Crest | OMIM MC1R)
- epidermis - Histological term describing the external cellular epithelial layer of the integumentary (skin) covering the entire body. This surface layer of keratinocytes is ectoderm in origin, while the underlying connective tissue layers of dermis and hypodermis are mesoderm in origin. (More? Integumentary Development)
- epidermal differentiation complex - (EDC) human chromosome (1q2) containing linked 63 genes within four gene families that are molecular markers for stratified epidermis terminal differentiation.
- epidermal growth factor receptor - expressed on cells in the epidermis basal layer, signaling stimulates both epidermal growth and wound healing and also mediates an inhibition of differentiation.
- glabrous skin - skin without hair, refers to the palms of hands and soles of feet.
- hair - (pili) in humans consists of vellus and terminal hairs.
- holocrine - form of gland secretion where the secretory cells eventually lyse (rupture) and are lost. On the skin, these cells release sebum consisting mainly of lipid. (More? image - holocrine secretion)
- hypodermis - (subcutis, subcutaneous adipose) a connective tissue ilower layer of the skin that binds it to underlying structures.
- integumentary - term for the skin and its appendages.
- involucrin - protein that binds loricrin in the development of the cell envelope protecting corneocytes in the skin.
- keratinocyte - the main cell type forming the layers of the epidermis, derived from ectoderm.
- keratohyalin granule - found in the stratum granulosum consist of profilaggrin and loricrin.
- Langerhans cell - skin dendritic cell (antigen presenting cell) develops initially from fetal liver monocytes and yolk sac macrophages. May, depending on the immunological setting, elicit immunity or tolerance. Named after Paul Langerhans.
- Langer's lines - (skin cleavage lines, cleavage lines) Clinical term for the orientation of reticular dermis collagen bundles causing tensions on skin and subcutaneous tissues. Lines tend to be horizontal in the trunk and neck, and longitudinal in the skin and limbs. (More? PMID 15791423)
- Meissner corpuscle - sensory structure acting as a rapidly-adapting mechanoreceptor mainly in the dermal papillae of (digital) skin. (More?Touch
- melanin - (Greek, melanos = black) The pigment produced by melanocytes that provides photoprotection, preventing cellular DNA damage, and colouring of the basal epithelial cells that absorb the pigment.
- melanodermia - hyperpigmentation causing abnormal darkening (brown/black) of the skin due to excess melanin or by metallic substances. See also the abnormality ceruloderma (blue/grey). (More? PMID 23522626)
- melanocyte - (Greek, melanos = black) A pigmented cell, neural crest in origin, differentiating from melanoblasts located in the skin and other tissues that produces melanin. The melanocytes within the integument (skin) transfer melanin to keratinocytes to give skin colour and to the hair follicle to give hair colour. Melanocytes are also located within "non-cutaneous" tissues in the eye (for eye colour), harderian gland and inner ear. This is the cell type that proliferates in the cancer melanoma. (More? Neural Crest Development | Integumentary System Development)
- Merkel cell - An epidermal-derived cell in touch-sensitive area of the epidermis and mediate mechanotransduction in the skin. Previously thought to be neural crest in origin, but recently shown to arise from the embryonic epithelium. The cells are named after Friedrich Sigmund Merkel, a German anatomist who was the first to describe them in 1875. (More? Touch | Lecture - Integumentary Development | PMID 19786578 | PMID 3782861)
- merocrine gland - (sweat gland, eccrine sweat) simple tubular glands located at the border between the dermis and hypodermis. These glands regulate the body temperature. (More? image - merocrine secretion)
- nestin - (neuroectodermal stem cell marker) an intermediate filament protein (type VI) expressed in stem cells and transiently during development, and in cells within hair follicles, sebaceous and sweat glands.
- papillary layer - dermis sublayer that appears less dense and contains more cells lying close beneath the epidermis. (More? image)
- pilosebaceous unit - term used to describe a hair and its associated structures: hair follicle, arrector pili muscle and sebaceous gland.
- rete ridge - the extensions of the epidermis into the dermis. These epidermal surface thickenings extend downward between underlying connective tissue dermal papillae. This is also the site of initial eccrine gland differentiation.
- reticular layer - dermis sublayer that appears denser and contains fewer cells with thick collagen bundles lying beneath the papillary layer parallel to the skin surface. (More? image)
- root sheath - cell layers that surround the hair.
- sebaceous gland - holocrine gland associated with both the hair follicle and hairless parts of the skin (lips, cheek oral surface and external genitalia). Embedded in the dermis and are sites of infections (acne).
- simple - consisting of a single cell layer.
- terminal hairs - hair seen in obviously hairy parts of the body.
- thick skin - refers to the skin histology found on the palms of the hand and soles of the feet, does not contain hair. Note that this is used as a histological term not a measurement of overall skin thickness.
- thin skin - refers to the skin histology found on skin on all body regions, other than palms and soles (thick skin).
- vellus hairs - fine short hairs only lightly pigmented covering the body.
- vernix caseosa - (vernix, Latin, "caseosa" = cheese-like) a fetal protective coating consisting of sebum, skin cells and lanugo hair. Forming late in fetal development in a rostra-caudal sequence associated with epithelium differentiation.
- Voigt's lines - clinical term to describe the skin borders between areas of innervations by specific peripheral cutaneous nerves. (More? Sensory Touch | Historic Terminology)
|
|
|
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 26) Embryology Neural Crest - Melanocyte Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Neural_Crest_-_Melanocyte_Development
- What Links Here?
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G