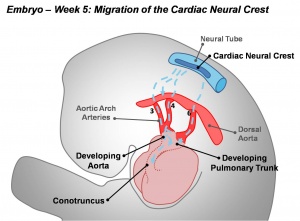
Neural Crest - Cardiac
| Embryology - 21 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
Draft Page (notice removed when completed).
Cardiac neural crest has an important contribution to the developing heart outflow tract.[1] It has been shown to not contribute to any of the heart conduction system.[2]
| Neural Crest Links: neural crest | Lecture - Early Neural | Lecture - Neural Crest Development | Lecture Movie | Schwann cell | adrenal | melanocyte | peripheral nervous system | enteric nervous system | cornea | cranial nerve neural crest | head | skull | cardiac neural crest | Nicole Le Douarin | Neural Crest Movies | neural crest abnormalities | Category:Neural Crest | |||
|
Cardiovascular System Development
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Cardiac Neural Crest <pubmed limit=5>Cardiac Crest</pubmed> |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page. |
Cardiac Neural Crest
References
- ↑ Creazzo TL, Godt RE, Leatherbury L, Conway SJ & Kirby ML. (1998). Role of cardiac neural crest cells in cardiovascular development. Annu. Rev. Physiol. , 60, 267-86. PMID: 9558464 DOI.
- ↑ Boullin J & Morgan JM. (2005). The development of cardiac rhythm. Heart , 91, 874-5. PMID: 15958352 DOI.
- ↑ Erhardt S, Zheng M, Zhao X, Le TP, Findley TO & Wang J. (2021). The Cardiac Neural Crest Cells in Heart Development and Congenital Heart Defects. J Cardiovasc Dev Dis , 8, . PMID: 34436231 DOI.
- ↑ Inman KE, Caiaffa CD, Melton KR, Sandell LL, Achilleos A, Kume T & Trainor PA. (2018). Foxc2 is required for proper cardiac neural crest cell migration, outflow tract septation, and ventricle expansion. Dev. Dyn. , 247, 1286-1296. PMID: 30376688 DOI.
- ↑ Rosa AL, Alvarez ME, Lawson D & Maccioni HJ. (1990). A polypeptide of 59 kDa is associated with bundles of cytoplasmic filaments in Neurospora crassa. Biochem. J. , 268, 649-55. PMID: 2141976
Reviews
Erhardt S, Zheng M, Zhao X, Le TP, Findley TO & Wang J. (2021). The Cardiac Neural Crest Cells in Heart Development and Congenital Heart Defects. J Cardiovasc Dev Dis , 8, . PMID: 34436231 DOI.
Plein A, Fantin A & Ruhrberg C. (2015). Neural crest cells in cardiovascular development. Curr. Top. Dev. Biol. , 111, 183-200. PMID: 25662261 DOI.
Keyte AL, Alonzo-Johnsen M & Hutson MR. (2014). Evolutionary and developmental origins of the cardiac neural crest: building a divided outflow tract. Birth Defects Res. C Embryo Today , 102, 309-23. PMID: 25227322 DOI.
Creazzo TL, Godt RE, Leatherbury L, Conway SJ & Kirby ML. (1998). Role of cardiac neural crest cells in cardiovascular development. Annu. Rev. Physiol. , 60, 267-86. PMID: 9558464 DOI.
Articles
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 21) Embryology Neural Crest - Cardiac. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Neural_Crest_-_Cardiac
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G