Integumentary System - Mammary Gland Development
| Embryology - 19 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
The mammary gland is the functional structure of the female breast and develops initially as an ectodermal skin specialization. Breast growth and appearance in male and female children are virtually identical prior to puberty.
Postnatally at puberty, female mammary glands under the influence of mainly sex hormone signaling, undergo a series of growth changes that can be defined anatomically by a series of "Tanner Stages". The bilayered mammary epithelium initially forms ducts that extend and branch.
In pregnancy, an additional series of signals leads to further changes in breast structure, differentiating into milk-producing alveoli. The key function of this process is to prepare the maternal breast for lactation and providing nutrition through milk to the newborn. The components of milk produced also changes beginning with an initial colostrum around birth. (More? milk)
Molecular signals identified as involved in this process include multiple Wnt ligands and β-catenin’s transcriptional activity for developmental processes and also for mammary stem cell self renewal.
At menopause, changes in sex hormone secretion can once again alter breast structure.
The breast also associated with oncogenesis (breast cancer). Research in this area has been aided by the discovery in 1994 of the two breast cancer susceptibility genes (BRCA1, BRCA2). There is some developing evidence that modification of stem cells (progenitor cells) that exist in the mammary gland may also contribute to neoplasms (cancer).
Additional Links: milk | Category:Mammary Gland
| Historic Mammary Papers | ||
|---|---|---|
1882 Mammary Gland | 1920 Mammary apparatus of the mammalia | 1921 Mammary Glands |1942 Smoking and lactation | 1950 Mammary Gland
|
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Mammary Embryology | Mammary Development | Mammary Gland Development |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Textbooks
- Human Embryology (2nd ed.) Larson Chapter 14 p443-455
- The Developing Human: Clinically Oriented Embryology (6th ed.) Moore and Persaud Chapter 20: P513-529
- Before We Are Born (5th ed.) Moore and Persaud Chapter 21: P481-496
- Essentials of Human Embryology Larson Chapter 14: P303-315
- Human Embryology, Fitzgerald and Fitzgerald
- Color Atlas of Clinical Embryology Moore Persaud and Shiota Chapter 15: p231-236
Development Overview
- week 6 epidermis downgrowth into dermis, modified sweat glands
- epithelia/mesenchyme inductive interaction, mesenchyme forms connective tissue and fat
- mammary ridges - mammary bud formation, pair of ventral regions axilla to inguinal
- pectoral regions generate breasts
- buds branch to form lactiferous ducts, only main duct formed at birth
- mammary pit - forms fetal period
- areola - depressed region at gland, proliferation of connective tissue postnatally
- prior to puberty male and female glands the same
Anatomy
| The mamma consists of gland tissue; of fibrous tissue, connecting its lobes; and of fatty tissue in the intervals between the lobes. The gland tissue, when freed from fibrous tissue and fat, is of a pale reddish color, firm in texture, flattened from before backward and thicker in the center than at the circumference.
(from Gray's Anatomy) |
|
Puberty
- sex hormone estrogen stimulate growth, full development approx 20 years
- gland branching morphogenesis commences[8]
- stimulated by - growth hormone (GH), estrogen, insulin-like growth factor 1 (IGF1)
- growth also influenced by other hormones - progesterone, prolactin, and corticoids
- mainly fat and connective tissue deposition
Tanner Mammary Development Stages
In 1976 Tanner and Whitehouse established a series of descriptive stages for primary and secondary sexual characteristic development at puberty. The female secondary sex characteristics of breast development were divided into five numbered (I - V) "Tanner Stages".[9]
Mammary Fat Pad
The mammary fat pad is present at birth as a depot of adipose tissue lying beside the mammary primitive epithelial structures.[10]
During puberty the mammary fat pad develops and is filled by the expanding glandular ductal tree. The fat pad is composed of both adipocytes and fibrous tissue forming white adipose tissue (WAT).[11] This adipose tissue acts as a stroma interacting with the mammary epithelium and provides signals required for ductal morphogenesis and alveolar differentiation.[12]
Mammary Glands Pregnancy
During pregnancy raised estrogens and progesterone stimulate gland development, secretory alveolar structures form and differentiate, leading to milk production in late pregnancy and milk secretion during lactation. Breasts are hemispherical in shape due to fat deposition. After birth, neonatal lactation supports further growth/development.
Mammary Glands Weaning
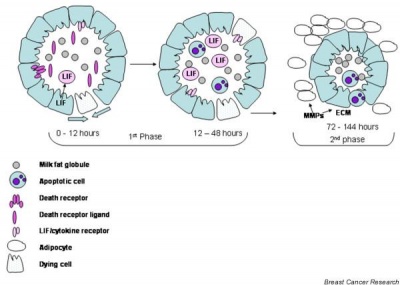
After the infant ceases breast feeding, weaning, the mammary gland milk-producing epithelial cells undergo a process called "involution", that requires cell apoptosis (programmed cell death).
Mammary involution[13]
Histology
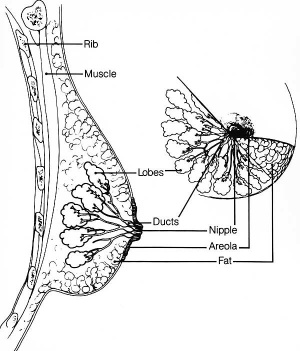
The mammary glands are modified glands of the skin and their development is similar to that of sweat glands.
- compound branched alveolar glands, secretory unit is the alveolus
- consist of 15-25 lobes separated by dense interlobar connective tissue and fat.
- each lobe contains an individual gland.
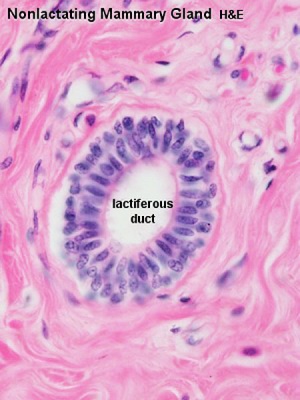
- lactiferous duct - excretory duct of each lobe with own opening on the nipple.
- cuboidal to columnar epithelium, surrounded by myoepithelial cells.
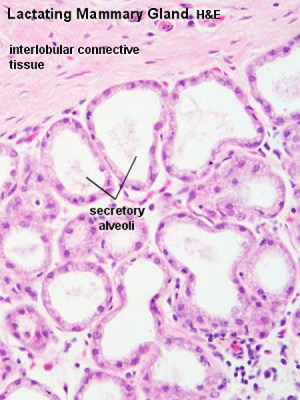
Alveolus
- inner layer of cuboidal secretory epithelial cells
- apocrine secretion, protein micelles are release by exocytosis.
- outer layer of myoepithelial cells
- located between the secretory cells and the surrounding basal lamina.
- contraction helps force the milk from the secretory alveoli into the ducts.
Note - plasma cells in the stroma also secrete antibodies (dimeric IgA) and released into the milk, to provides passive immunity to the suckling young.
| Lactiferous duct | Lactating mammary gland | Apocrine secretion |
|---|---|---|
|
|

|
Mouse Mammary
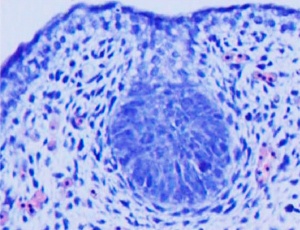
| Day | Event |
|---|---|
| E10 | milk line first formed by a slight thickening and stratification of the surface ectoderm. |
| E10.5 | expression of Wnt10b in mammary lines on the trunk between the limbs and in axillary and inguinal streaks. |
| E11.5 | milk line breaks up into individual placodes and the underlying mammary mesenchyme begins to condense. |
| E15.5 | mammary epithelium begins to proliferate at the tip and the primary sprout pushes through the mammary mesenchyme towards the underlying fat pad. |
| E18.5 | elongating duct has now grown into the fat pad and has branched into a small ductal system. Cells of the mammary mesenchyme have formed the nipple, which is made of specialized epidermal cells. |
| Data source[15] Related Images: E10/11 | E11 | E12 | E13 | E14.5 | |
Molecular
Mainly from mouse studies.[16]
- Wnt10b - first streak initially fragmented expression pattern
- Pax3 - dorsalized position indicate somitic involvement in streak formation
- FGF10 - expressed in hypaxial dermomyotomal tips of somites, activates receptor FGFR2-IIIb in overlying surface ectoderm.
Wnt Signaling
Canonical Wnt signals are transduced through a Frizzled receptor and the LRP5 or LRP6 co-receptor. Loss of Lrp6 compromises Wnt/beta-catenin signaling and interferes with mammary placode, fat pad, and branching development during embryogenesis.[17]
Non-canonical receptor Ror2 along with its ligand Wnt5b (expressed in both the basal and luminal cell layers) regulates the branching, differentiation, and actin-cytoskeletal dynamics within the mammary epithelium.[18]
- Links: Wnt | Mouse Timeline Detailed
Abnormalities
| ICD-11 |
|---|
|
LB60 Breast aplasia - A condition caused by failure of the breast to develop during the antenatal period. This condition is characterized by a total lack of breast tissue. This condition may also present with absence of the nipple or pectoral muscle. LB61 Absent nipple - A condition caused by failure of the nipple to develop during the antenatal period. This condition is characterized by a total lack of a nipple. This condition may also present with absence of the breast or pectoral muscle. LB62 Supernumerary breasts - A condition caused by failure of the breasts to correctly develop during the antenatal period. This condition is characterized by supernumerary breasts, with or without nipples. This condition may be asymptomatic. LB63 Accessory nipple - A condition caused by development of supernumerary nipples during the antenatal period. This condition is characterized by the presence of nipples or nipple-like structures additional to the normal two. This condition may be asymptomatic. |
Abnormalities occur in approximately 1% of female population and include in both sexes:
- polymastia - extra breast
- polytheli - extra nipple
- supernumerary nipple (relatively common in males)
- unilateral polythelia or polymastia - occurs most frequently on the left side in humans[19]
- gynecomastia (Greek, gyne = woman, mastos = breast) is the excessive development of the male breast, which can occur transiently in puberty or due to other (hormonal) abnormalities.
Historic 1915 Kajava classification of supernumerary nipple types:[20]
- Complete supernumerary nipple-Nipple, areola, and glandular breast tissue
- Supernumerary nipple-Nipple and glandular tissue (no areola)
- Supernumerary nipple-Areola and glandular tissue (no nipple)
- Aberrant glandular tissue only
- Supernumerary nipple-Nipple and areola and pseudomamma (fat tissue replaces glandular tissue)
- Supernumerary nipple-Nipple only (the most common supernumerary nipple)
- Supernumerary nipple-Areola only (polythelia areolaris)
- Patch of hair only (polythelia pilosa)
Breast Cancer
In 1994, two breast cancer susceptibility genes were identified BRCA1 on chromosome 17 and BRCA2 on chromosome 13.
When an individual carries a mutation in either BRCA1 or BRCA2, they are at an increased risk of being diagnosed with breast or ovarian cancer at some point in their lives. Normal function of these genes was to participate in repairing radiation-induced breaks in double-stranded DNA. It is though that mutations in BRCA1 or BRCA2 might disable this mechanism, leading to more errors in DNA replication and ultimately to cancerous growth. (text modified from: NCBI genes and disease)
BRAC1 and more recently BRIP1 (BRCA1-interacting protein 1) appear to be statistically the more common cancer genes associated with breast cancer.
- Links: OMIM - BRCA1 | OMIM - BRCA2 | OMIM - BRIP1
Australia - Breast cancer in young women
08 Oct 2019 Breast cancer mortality has decreased since BreastScreen Australia began, from 74 deaths per 100,000 women aged 50–74 in 1991, to 41 deaths per 100,000 women in 2017. BreastScreen Australia monitoring report 2019
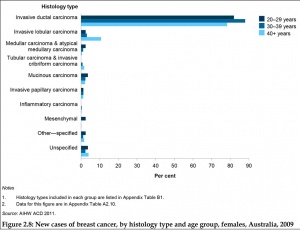
26 Oct 2015 Key facts about breast cancer in women in their 20s and 30s[21]
- In 2015, it is projected that 795 young women will be diagnosed with breast cancer and 65 will die from this disease.
- On average, this is more than 2 breast cancers diagnosed every day and more than 1 death every week.
- most commonly diagnosed cancer for women aged 20 to 39 and is associated with poorer survival outcomes.
- for women aged 40 and over, 40 new cases per day and 57 deaths per week-breast cancer.
- Overall, 5-year relative survival is significantly lower in younger women, who had an 88% chance of surviving for 5 years in 2007-2011 compared with the 5-year relative survival rate for women aged 40 and over of 90%. However, the 5-year relative survival rate has improved for young women, from 72% in 1982-1986 to 88% in 2007-2011.
- Links: Australian Statistics
References
- ↑ Liu Y, Chen X, Gong Z, Zhang H, Fei F, Tang X, Wang J, Xu P, Zarbl H & Ren X. (2019). Fry Is Required for Mammary Gland Development During Pregnant Periods and Affects the Morphology and Growth of Breast Cancer Cells. Front Oncol , 9, 1279. PMID: 31824855 DOI.
- ↑ Seachrist DD & Keri RA. (2019). The Activin Social Network: Activin, Inhibin and Follistatin in Breast Development and Cancer. Endocrinology , , . PMID: 30874767 DOI.
- ↑ Hindman AR, Mo XM, Helber HL, Kovalchin CE, Ravichandran N, Murphy AR, Fagan AM, St John PM & Burd CJ. (2017). Varying Susceptibility of the Female Mammary Gland to In Utero Windows of BPA Exposure. Endocrinology , 158, 3435-3447. PMID: 28938483 DOI.
- ↑ McNally S & Stein T. (2017). Overview of Mammary Gland Development: A Comparison of Mouse and Human. Methods Mol. Biol. , 1501, 1-17. PMID: 27796946 DOI.
- ↑ Christensen KY, Maisonet M, Rubin C, Flanders WD, Drews-Botsch C, Dominguez C, McGeehin MA & Marcus M. (2010). Characterization of the correlation between ages at entry into breast and pubic hair development. Ann Epidemiol , 20, 405-8. PMID: 20382343 DOI.
- ↑ Cannata D, Lann D, Wu Y, Elis S, Sun H, Yakar S, Lazzarino DA, Wood TL & Leroith D. (2010). Elevated circulating IGF-I promotes mammary gland development and proliferation. Endocrinology , 151, 5751-61. PMID: 20926579 DOI.
- ↑ Schedin P & Hovey RC. (2010). Editorial: The mammary stroma in normal development and function. J Mammary Gland Biol Neoplasia , 15, 275-7. PMID: 20824491 DOI.
- ↑ Macias H & Hinck L. (2012). Mammary gland development. Wiley Interdiscip Rev Dev Biol , 1, 533-57. PMID: 22844349 DOI.
- ↑ Tanner JM & Whitehouse RH. (1976). Clinical longitudinal standards for height, weight, height velocity, weight velocity, and stages of puberty. Arch. Dis. Child. , 51, 170-9. PMID: 952550
- ↑ Anbazhagan R, Bartek J, Monaghan P & Gusterson BA. (1991). Growth and development of the human infant breast. Am. J. Anat. , 192, 407-17. PMID: 1781450 DOI.
- ↑ Neville MC, Medina D, Monks J & Hovey RC. (1998). The mammary fat pad. J Mammary Gland Biol Neoplasia , 3, 109-16. PMID: 10819521
- ↑ Hovey RC & Aimo L. (2010). Diverse and active roles for adipocytes during mammary gland growth and function. J Mammary Gland Biol Neoplasia , 15, 279-90. PMID: 20717712 DOI.
- ↑ Watson CJ. (2006). Involution: apoptosis and tissue remodelling that convert the mammary gland from milk factory to a quiescent organ. Breast Cancer Res. , 8, 203. PMID: 16677411 DOI.
- ↑ Howard B & Ashworth A. (2006). Signalling pathways implicated in early mammary gland morphogenesis and breast cancer. PLoS Genet. , 2, e112. PMID: 16933995 DOI.
- ↑ Robinson GW. (2007). Cooperation of signalling pathways in embryonic mammary gland development. Nat. Rev. Genet. , 8, 963-72. PMID: 18007652 DOI.
- ↑ Veltmaat JM, Ramsdell AF & Sterneck E. (2013). Positional variations in mammary gland development and cancer. J Mammary Gland Biol Neoplasia , 18, 179-88. PMID: 23666389 DOI.
- ↑ Lindvall C, Zylstra CR, Evans N, West RA, Dykema K, Furge KA & Williams BO. (2009). The Wnt co-receptor Lrp6 is required for normal mouse mammary gland development. PLoS ONE , 4, e5813. PMID: 19503830 DOI.
- ↑ Roarty K, Shore AN, Creighton CJ & Rosen JM. (2015). Ror2 regulates branching, differentiation, and actin-cytoskeletal dynamics within the mammary epithelium. J. Cell Biol. , 208, 351-66. PMID: 25624393 DOI.
- ↑ Schmidt H. (1998). Supernumerary nipples: prevalence, size, sex and side predilection -- a prospective clinical study. Eur. J. Pediatr. , 157, 821-3. PMID: 9809822
- ↑ Kajava Y. The proportions of supernumerary nipples in the Finnish population. Duodecim. 1915;1:143–70.
- ↑ AIHW 2015. Breast cancer in young women: key facts about breast cancer in women in their 20s and 30s. Cancer series no. 96. Cat. no. CAN 94. Canberra: AIHW. http://www.aihw.gov.au/publication-detail/?id=60129553359
Journals
Reviews
McNally S & Stein T. (2017). Overview of Mammary Gland Development: A Comparison of Mouse and Human. Methods Mol. Biol. , 1501, 1-17. PMID: 27796946 DOI.
Javed A & Lteif A. (2013). Development of the human breast. Semin Plast Surg , 27, 5-12. PMID: 24872732 DOI.
Huebner RJ & Ewald AJ. (2014). Cellular foundations of mammary tubulogenesis. Semin. Cell Dev. Biol. , 31, 124-31. PMID: 24747369 DOI.
Lefèvre CM, Sharp JA & Nicholas KR. (2010). Evolution of lactation: ancient origin and extreme adaptations of the lactation system. Annu Rev Genomics Hum Genet , 11, 219-38. PMID: 20565255 DOI.
Cowin P & Wysolmerski J. (2010). Molecular mechanisms guiding embryonic mammary gland development. Cold Spring Harb Perspect Biol , 2, a003251. PMID: 20484386 DOI.
Dimitrakakis C & Bondy C. (2009). Androgens and the breast. Breast Cancer Res. , 11, 212. PMID: 19889198 DOI.
Rowzee AM, Lazzarino DA, Rota L, Sun Z & Wood TL. (2008). IGF ligand and receptor regulation of mammary development. J Mammary Gland Biol Neoplasia , 13, 361-70. PMID: 19020961 DOI.
LaMarca HL & Rosen JM. (2008). Minireview: hormones and mammary cell fate--what will I become when I grow up?. Endocrinology , 149, 4317-21. PMID: 18556345 DOI.
Richert MM, Schwertfeger KL, Ryder JW & Anderson SM. (2000). An atlas of mouse mammary gland development. J Mammary Gland Biol Neoplasia , 5, 227-41. PMID: 11149575
Articles
Veltmaat JM, Ramsdell AF & Sterneck E. (2013). Positional variations in mammary gland development and cancer. J Mammary Gland Biol Neoplasia , 18, 179-88. PMID: 23666389 DOI.
Nair R, Junankar S, O'Toole S, Shah J, Borowsky AD, Bishop JM & Swarbrick A. (2010). Redefining the expression and function of the inhibitor of differentiation 1 in mammary gland development. PLoS ONE , 5, e11947. PMID: 20689821 DOI.
Owens TW, Foster FM, Tanianis-Hughes J, Cheung JY, Brackenbury L & Streuli CH. (2010). Analysis of inhibitor of apoptosis protein family expression during mammary gland development. BMC Dev. Biol. , 10, 71. PMID: 20584313 DOI.
McCormick N, Velasquez V, Finney L, Vogt S & Kelleher SL. (2010). X-ray fluorescence microscopy reveals accumulation and secretion of discrete intracellular zinc pools in the lactating mouse mammary gland. PLoS ONE , 5, e11078. PMID: 20552032 DOI.
Barker HE, Smyth GK, Wettenhall J, Ward TA, Bath ML, Lindeman GJ & Visvader JE. (2008). Deaf-1 regulates epithelial cell proliferation and side-branching in the mammary gland. BMC Dev. Biol. , 8, 94. PMID: 18826651 DOI.
Ramsay DT, Kent JC, Hartmann RA & Hartmann PE. (2005). Anatomy of the lactating human breast redefined with ultrasound imaging. J. Anat. , 206, 525-34. PMID: 15960763 DOI.
Search PubMed
Search Pubmed: Mammary Gland Development
Historic
Bresslau E. The mammary apparatus of the mammalia : in the light of ontogenesis and phylogenesis. (1920) London : Methuen & Co. IA
Rein G. Untersuchungen über die embryonale Entwickelungsgeschichte der Milchdrüse—I. Arch Mikrosk Anat. 1882;20:431–501. doi: 10.1007/BF02952656.
Rein G. Untersuchungen über die embryoanale entwickelungsgeschichte der milchdrüse—II. Vergleichend-anatomische ergebnisse und schlussfolgerungen. Arch Mikrosr Anat. 1882;21:678–694. doi: 10.1007/BF02952632.
Klaatsch H. Zur Morphologie der säugetierzitzen. Morph Jahrb. 1884;9:258–324.
Curtis F. Le développement de la mamelle et du mamelon d’après les travaux les plus récents. Rev Bio du Nord de la France. 1889;1:44–457.
Schultze O. Über die erste Anlage des Milchdrüsenapparates. Anat Anz. 1892;7:265–270.
Schmidt H. Über normale hyperthelie menschlicher embryonen. Anat Anz. 1896;11:702–711.
Schmidt H. Über normale Hyperthelie menschlicher Embryonen und über die erste Analage der menschlichen Milchdrüsen überhaupt. Morph Arbeit. 1897;7:157–199.
Bonnet R. Die mammarorgane im Lichte der Ontogenie und Phylogenie. Ergeb Anat Entwick Gesch. 1897;7:937–976.
Additional Images
Historic
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Keith A. Human Embryology and Morphology. (1902) London: Edward Arnold.
Bailey FR. and Miller AM. Text-Book of Embryology (1921) New York: William Wood and Co.
Terms
- milk line - (primary mammary ridge) a surface thickening of the ectoderm extending along the body wall on either side from axilla to groin.
- primary mammary ridge - (milk line) a surface thickening of the ectoderm extending along the body wall on either side from axilla to groin.
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 19) Embryology Integumentary System - Mammary Gland Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Integumentary_System_-_Mammary_Gland_Development
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G