Paper - Development of the mammary gland - Arris and Gale Lecture
| Embryology - 27 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Hughes ES. Development of the mammary gland. (1950) Ann R Coll Surg Engl. 6(2):99-119. PMID 19309885
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
The Development of the Mammary Gland
Arris and Gale Lecture,
delivered at the Royal College of Surgeons of England on 25th October, 1949
by
E.S.R. Hughes, M.D., M.S., F.R.C.S.
Introduction
The pre-natal development of the mammary gland, with which this investigation is concerned, has rarely attracted British or American embryologists, and hence nearly all past observations have been recorded in European literature.
After the initial research of Meckel (1820), Langer (1850), Huss (1873), Kolliker (1879) and Rein (1882) there was considerable discussion on the morphological significance of the various findings. Subsequently, interest in the problem was largely subordinated by the study of the functional effect of hormones.
The mammary gland is accessible and development easy to follow by means of serial sections. The problem was, therefore, re-investigated in 70 human embryos of various sizes from 3-5 mm. to full term. There were two distinct phases of intra-uterine development, the formation of the nipple primordium and the development of the rudimentary mammary gland.
1. Development of the Nipple Primordium
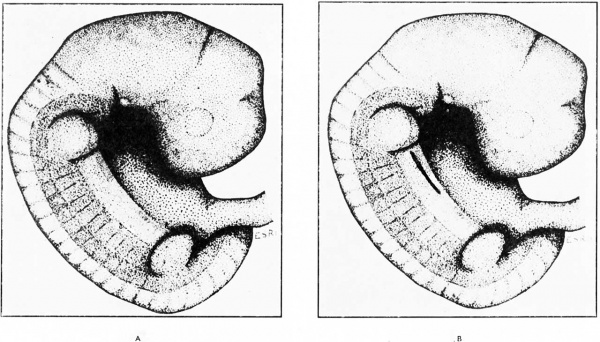
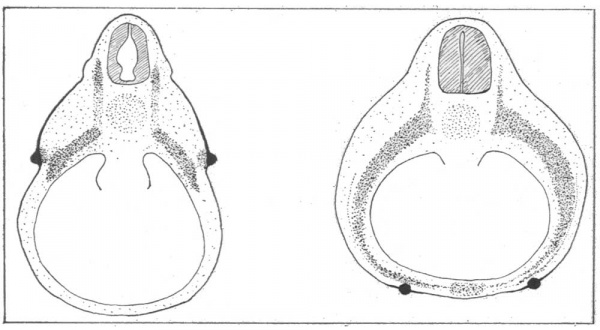
In the very earliest embryos examined, 3-5 mm., 4-5 mm., 5-5 mm. and 6-0 mm., there were no signs of a developing nipple. In the latter three, the ectoderm surrounding the origins of the limb buds, and on the lateral walls of the trunk between them, displayed a mild differentiation into two layers of cells. This ectoderm has been referred to as the “ mammary band ” (Fig. 1 (a) ).
Fig. 1. (A) The “ mammary band ” (darkened area) surrounds the limb buds and extends between them on the lateral wall of the trunk. (B) The mammary crest appealrs wilthin the mammary band area and is represented here as a b ack ine.
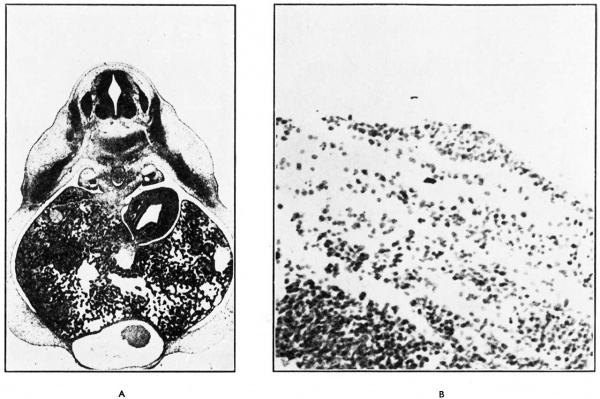
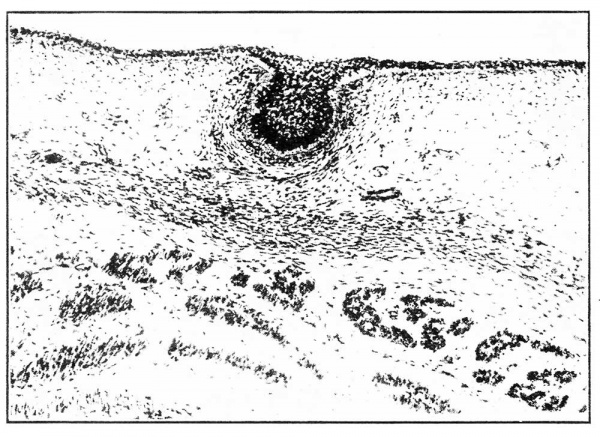
ln 7-1 mm., 7-25 mm., 7-75 mm. and 8-0 mm. embryos the nipple primordium was apparent for the first time as a narrow crest of ectodermal cells, the mammary crest, surrounded by the mildly differentiated “ mammary band ” (Fig. l (b) ). The mammary crest was situated on the dorso-lateral thoracic and upper abdominal walls. The cells of its germinal layer, tall and columnar, with large rounded apical nuclei, were clearly demarcated from the underlying mesoderm by the pale staining cytoplasm; the remaining cells were polyhedral. The underlying dermal mesoderm, stained a little deeper than the surrounding mesoderm, with rounded cellular nuclei more densely distributed (Fig. 2, (a) and (b) ).
Fig. 2. (A) The crest lies at the ventral end of the advancing dermatome in a dorso-lateral position ; it is surrounded by the mildly differentiated ectoderm of the “ mammary band.” There is a slight mesodermal condensation deep to the crest. (B) The cells forming the mammary crest possess a basal columnar layer with apical nuclei and clear basal cytoplasm. The remaining cells are polyhedral.
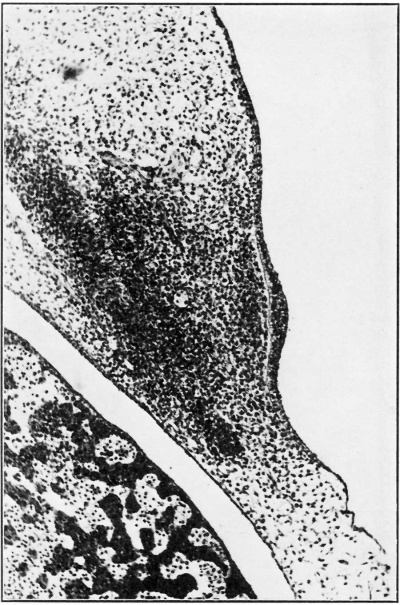
In two 9-5 mm. embryos and one of 10-0 mm. the appearances were similar except that in the dermal mesoderm a single layer of cells had condensed immediately deep to the basal layer of the ectodermal crest. In an 11-25 mm. embryo the crest showed little diminution in length ; in the underlying mesoderm, were two or three layers of cells closely applied to the basement membrane, overlapping the edges of the ectodermal crest, and extending beyond its limit distally. Deep to this zone the mesoderm was condensed and stained deeply (Fig. 3).
Fig. 3. A layer of mesoderm cells quickly condenses deep to the mammary crest. Deep to this again is a widening zone of condensed mesoderm.
The epithelial crest rapidly shortened and at the same time the cranial end proliferated. The mesoderm of the 12-0 mm. embryo showed clearly the lamellated zone and the deeper, denser zone which was becoming vascular. In the 13-5 mm. and 14-0 mm. embryos, the mesoderm deep to these zones was differentiating into two further layers, a third zone of loose connective tissue, and a fourth composed of several loose layers of cells. These mesodermal changes coincided with such reduction in length of the epithelial crest that in both the 14 mm. embryos studied, length and breadth were equal (Fig. 4).
Fig. 4. By the 14 mm. embryo stage, the distal part of the crest has disappeared and the proximal end has proliferated to form a nodule sinking into the mesoderm (note the capsular zone of mesoderm).
Whilst these changes were proceeding, the developing nipple gradually moved from a relatively dorsal position to a ventral one, a movement dependent on the anterior growth of the dermatome (Fig. 5).
Fig. 5. The epithelial mammary primordium moves ventrally from its original dorsolateral position. This movement is dependent on the anterior growth of the somite.
In the 15-5 mm. and 16-5 mm. embryos the “mammary band” was disappearing and the epithelial proliferation was sinking into the differentiating mesoderm.
The epithelial primordium of the 17 mm., 19 mm., 20 mm. and 21 mm. embryos showed essentially the same features. It was globular in shape and below the level of the surface. Two or three layers of fusiform cells were closely applied to the germinal layer whilst the second, third and fourth zones of connective tissue were easily distinguished (Fig. 6).
Fig. 6. The epithelial nipple primordium is soon below the surface of the ectoderm. The mesodermal changes are particularly characteristic.
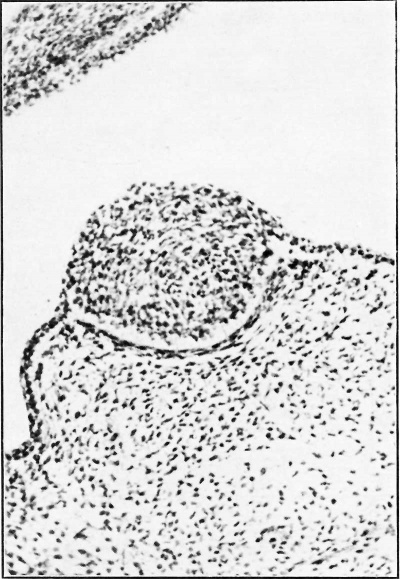
In the 20-0 and 21-0 mm. embryos small areas of epithelial proliferation were seen chiefly in the axillary region; very much smaller than the developing nipple zone, they varied from 90-0 u. to 150 u. in diameter. and although displaying the tall basal cells, showed no mesodermal changes. These are known as the “ hyperthelial structures ” or the “ Hugo Schmidt bodies ” after their discoverer (Fig. 7).
Fig. 7. Hugo Schmidt hyperthelial structure in the axilla of a 28 mm. embryo. 105 E. S. R. HUGHES
In the 26, 30 and 47 mm. embryos the epithelial proliferation was flaskshaped on section, and lay wholly beneath the ectoderm. The nipple had assumed its adult position, and was somewhat elevated above the level of the surrounding skin as a result of the mesodermal development. The “ Hugo Schmidt ” structures were clearly visible.
In the 55, 60, and 61 mm. embryos, the “ Hugo Schmidt ” proliferations had disappeared; the primitive nipple was represented as a solid wellcircumscribed mass of cells, arising from a shallow depression at the apex of an elevation, whilst the related mesoderm was differentiated very clearly into the four zones described.
In the 90, 100, and 129 mm. embryos the superficial cells of the epithelial bud showed keratinisation and the intermediate cells were enlarged and partly vacuolated, whilst the deep surface had become irregular. In the surrounding skin pilo-glandular structures were appearing, although the nipple primordium and the ectoderm immediately surrounding it remained clear. In the 150 mm. embryo locules of adipose tissue could be seen in the subcutaneous tissue, but not in the differentiated mesoderm of the mammary primordium (Fig. 8).
Fig. 8. Pilo-glandular structures appear in the related ectoderm in the 90 mm. embryo, and adipose tissue in the 150 mm. embryo. The nipple and areolar zones are clear of such structures.
This is the end of the first stage of development ; no mammary gland has developed, only the primitive nipple.
2. Development of the Mammary Gland and Further Development of Nipple
In this series no further differentiation of the epithelial primordium occurred until the embryo was 170 mm. in length, at which stage solid cellular outgrowths appeared from the edge and deep surface of the primordium, and pushed into the mesoderm. This process was quite distinct from that which caused the slight irregularity of the periphery of the intact nipple primordium, and seemed to be the result of intensive proliferation of the basal layer of ectoderm (Fig. 9).
Fig. 9. In the 170 mm. embryo, epithelial outgrowths appear from the deep surface of the epithelial mammary primordium. They pursue a straight course, expanding slightly at their blind extremities. A lumen appears at an early stage. They receive no special covering from the first two layers of mesoderm. but become surrounded by a loose concentric investment derived from the third zone.
The cellular outgrowths possessed the same general structure as the epithelial primordium, although the cells of the basal layer were short columnar or cubical in shape. Solid at first, they expand slightly at their distal extremity. Two or more outgrowths may have the same point of origin.
The mesoderm surrounding the epithelial primordium retained its differentiated appearance. The capsular zone of fusiform mesodermal cells, terminating in the region of the neck of the primordium, was not clearly seen in some areas, but in others was well marked. Deep to this zone, the connective tissue was dense and contained large thin walled vessels, whilst the lamellated deepest zone was well developed.
The cellular outgrowths penetrated the first two zones of mesoderm without receiving any special covering. In the third mesodermal zone they became surrounded by several concentric layers of a very loosely arranged connective tissue.
In the 180 and 185 mm. embryos the appearances were similar. The cellular outgrowths were still mostly solid ; some had already commenced to branch. Similar outgrowths were seen to arise from the areolar zone. The superficial ectoderm of the epithelial primordium was desquamating to leave an inverted nipple area.
In the 220 mm. embryo most cellular outgrowths had become canalised by desquamation and lysis of their central cells ; the resulting primitive ducts are lined by two layers of cubical cells. The first or capsular zone of mesoderm was less distinct, but the dense mesoderm of the second layer was sharply differentiated from the 3rd and 4th zones.
The ducts increased in length, and in the 300 mm. embryo each possessed several terminal branches lined by two layers of cubical cells. The lumens of the ducts extended into their distal expanded ends and contained small quantities of amorphous debris derived apparently from degenerated cells. The ducts were surrounded by mesoderm as in theprevious stages, but were becoming separated from each other by septa derived from the 4th zone. The nipple primordium had undergone further keratinisation and desquamation and a wide saucer-shaped depression remained.
At term, approximately 330 mm., canalisation of the tubules was mostly complete, although in some the excretory portion lined by stratified squamous epithelium was considerably narrowed or even.imperforate. The tubules were dilated to a varying degree and contained amorphous debris ; on section they appeared as groups of duct terminals surrounded by loose concentrically arranged connective tissue and separated into lobules by dense interlobular tissue. They were lined by two layers of epithelium, an inner cubical and an outer flattened, but there was much variation in the size and shape of the cells. The fusiform cells composing the first mesodermal zone had dilferentiated into circumscribed bundles of smooth muscle. The second dense mesodermal zone resembled the surrounding dermis, but was distinguished by the presence of vascular lakes. The mammary ducts passed through these first two zones without receiving any special covering. Immediately next to the basement membrane of the duct terminals there was an exceedingly dense plexus of capillaries which drained into larger vessels in the interlobar septa, and thence by straight veins into the vascular lakes of the nipple zone. The mammary gland was enclosed by a thin capsule of connective tissue, which enabled it to be enucleated from the surrounding tissue. The saucer-shaped depression caused by the excavation of the epithelial primordium had become more or less obliterated by an increase in its width and by commencing eversion of its deepest part. In the nipple, small sebaceous glands were related to the mammary ducts; in the areola, sebaceous glands were much larger. There were no hair follicles in the nipple, and only at the edge of the areola (Fig. 10).
It is clear, therefore, that the pre-natal development proceeds in two absolutely distinct phases ; the first directed towards the formation of the nipple ; and the second concerned with the construction of the mammary gland itself. The present-day conception of the embryology of the mammary gland has been confused by loose terminology and by conclusions derived from isolated observations. I propose to review, and, if possible, to clarify certain of these controversial aspects.
The Mammary Band
Although Schultze (1892) noted its occurrence, it was Schmidt (1896) who called attention to, and named, the “mammary band.” In a 150 mm. embryo, he observed a relatively broad band of epidermal thickening, which surrounded the limb buds and covered the lateral wall of the embryo between them. In its thoracic portion it contained the epithelial primordium of the nipple. All subsequent observers are agreed that such a band of cells exists (Bonnet, 1897 ; Hirschland, 1898 ; Schmitt, 1898 ; Strahl, 1898 ; Brouha, 1905 (b) ; Lustig, 1915 ; Neumann and Oing, 1929).
Hirschland (1898) and Strahl (1898) thought this area to be phylogenetically important, and that it was a stage preceding the mammary crest. Bresslau (1910) regarded the band of placental mammals as homologous with the epithelial primordium of the monotreme, Echidna.
It would seem, however, that the so-called “ mammary band ” has no special regional significance :—
(a) An apparently identical structure is to be found in embryos of reptiles and birds (Schmitt).
(b) The mammary band may persist in a region where the crest has become obliterated (Brouha).
(c) It extends along the sides of the body, surrounding the limb buds as well as the tail (Walter).
This zone of ectodermal differentiation appears to be connected with the growth of the body and its appendages as a whole, and hence should not be designated “ mammary band.”
Fig. 10. At term (330 mm.) the nipple is still inverted or, at the most, flattened. In the dense mesoderm of the nipple (A) are vascular lakes and circumscribed bundles of smooth muscle derived from the first zone of primitive mesoderm. The excretory portion of the mammary ducts may be partially occluded or even imperforate at term ; they are lined b), stratified epithelium and related to them are small sebaceous glands (B). The mammary ducts undergo dichotomous branching and on section appear as groups of dilated duct terminals surrounded by loose intralobular tissue. The gland is enclosed by a thin capsule of mesoderm which is continuous with that of the interlobular tissue (C). A dense capillary plexus is closely related to the ducts and drains into vessels lying in the dense mesoderm forming the interlobular tissue (D).
The Mammary Crest
The Milk Line—Milchlinie - Milchleiste
In a series of placental mammalian embryos, Schultze (1892), and shortly after, Bonnet (1892), discovered an epithelial crest, extending along the lateral wall of the trunk from the upper to the lower limb bud, which was termed the “ milchlinie ” or “ milk-1ine.” In the pig, rabbit and other mammals which possess a row of nipples, this crest becomes segmented and reduced to a series of nipples, with development proceeding quickly from before backwards. Some years later Kallius (1897) observed an epithelial crest in a 15 mm. embryo.
It is doubtful if the mammary crest has ever been seen macroscopically. It must not be confused with a well-defined white ridge to be seen in some embryos such as those of the sheep, and which represents the anterior edge of the advancing somite (Fig. 11 (a) (b)).
Klaatsche (1893) believed that the crests were rudiments of the marsupial pouch. Bresslau originally held the View that the crests were derived from fused marsupial pockets. After further study he abandoned this theory and concluded that the mammary crests were merely secondary ridge-like differentiations within the mammary bands. Beard (1897), Profe (1898) and Brouha (1905 (b) (c)) regarded the crest as having no phylogenetic significance and compared it with other structures appearing first as crests in ontogenetic development, i.e., spinal ganglion.
It must be emphasised that the mammary crest of the human embryo has never been observed to segment normally into regular nipple primordia. Occasionally part becomes “ pinched ” off to form a supernumerary nipple. This must not be regarded as an atavistic phenomenon or anything to do with phylogeny; it is an accident in development, comparable to webbed fingers. By far the most common site for supernumerary nipples is above the umbilicus ; this corresponds with the usual length of the mammary crest. Very occasionally the mammary crest is found to extend into the groin, and just as occasionally supernumerary nipples are to be found in the groin. The term “ milkline ” has been exploited. It can only refer to the line of nipples of those mammals which bear so many young in each litter as to require a large number of breasts. So far as human embryology is concerned, the term “ milk-line ” should be discarded for the far more accurate mammary crest.
Fig. 11. The mammary crest must not be confused with the distinct white ridge to be foundin embryos, such as sheep, and which represents the anterior edge of the mesoderm (A). Microscopic examination shows no evidence of a mammary primordium (B).
Development of the Nipple
The mammary crest rapidly shortens to form a small circumscribed nodule. It sinks below the surface, and in the 20 mm. embryo is below the level of the surrounding ectoderm. Just as quickly the mesoderm diflerentiates into four layers—a capsular or laminated zone of fusiform cells, which ultimately forms the smooth muscle of the nipple ; a second zone of dense vascular tissue which becomes the stroma of the nipple; a third area of loose mesoderm from which is derived the connective tissue of the lobules; and a fourth zone, closely related to the fascia covering the pectoral muscles, which forms the interlobular and capsular connective tissue (Fig. 12).
It is curious that these mesodermal changes should have escaped special description by nearly all previous embryologists, although Lustig (1915) and Keith (1948) have given some indication of their nature And it is interesting to recall that when the mammary gland is congenitally absent, the pectoral fascia and muscles are likewise deficient.
Therefore, from the end of the second month (that is, the 20 mm. embryo) to the end of the fifth month (that is, the 150 mm. embryo), there is a real pause in development which ends with the appearance of the primitive mammary ducts. In a communication to the Anatomical Society of Great Britain and Ireland I called attention to this latent period (Hughes, 1948), and more recently Balinsky has noted its occurrence in mice. I have found it in a series of mammals that I have investigated. This pause in development immediately recalls the latent period which exists between birth and puberty in the female. Indeed the mammary gland arises by steps, first the nipple, then the ducts, and finally the acini, each step separated by a distinct interval.
The Development of the Mammary Gland
With the first, rather abrupt, appearance of cellular outgrowths, the development of the mammary gland proper begins. In my own material, this was in a 170 mm. embryo ; Langer (1850) observed ducts in a 100 mm. embryo, Huss (1873) in a 180 mm. embryo, Lustig (1915) in a 135 mm. embryo, and Neumann and Oing (1929) in a 140 mm. embryo.
Fig. 12. The mammary crest rapidly shortens to form the epithelial primordium.
This sinks into the mesoderm. The mesoderm condenses around the epithelial primordium and "our zones differentiate. Pilo-glandular structures appear at about 90 mm., and adipose tissue at 150 mm.
They are solid at first but soon canalise. They undergo simple branching, and are often dilated. The intra-epidermal segment may still be more or less completely occluded at birth. Should such obstruction persist, it may well be followed by a succession of changes characteristic of tube obstruction elsewhere in the body (Fig. 13).
The connective tissue is differentiated into the smooth muscle of the nipple, dense vascular mesoderm of the nipple, the loose concentrically arranged stroma of the lobules, and the inter-lobular septa and capsule of the mammary gland. A dense plexus of capillaries surrounds the ducts and these drain into the veins within the interlobular septa, and thence into the venous plexus of the nipple. The ectoderm forming the nipple undergoes cornification and desquamates, leaving a saucer-shaped depression which is slowly obliterated as it widens and the nipple everts. Failure of this process results in the inverted nipple, partial or complete.
Eggelin g (1904) and Brouha (1905 (a)) described the presence of hair bulbs, distinguished by the disposition of the cells, and by the appearance of the free extremity; but even at their most highly developed state they never showed any trace of keratinisation. Lustig (1915) only found hair follicles beyond the nipple primordium, and this agrees with the observations recorded here.
At birth, the parenchyma of the mammary gland is dark in colour, sharply circumscribed, varying in depth and width from 0-5 cms. to 1-5 cms. The breast of both sexes appears similar.
Fig. 13. (a) The epithelial mammary primordium develops epithelial outgrowths which canalise, divide and subdivide to form the parenchyma of the gland. (b) The mesoderm differentiates into the smooth muscle of the nipple, the dense mesoderm of the nipple, the loose stroma of the glandular tissue, and the dense interlobular and capsular tissue. (c) The ectoderm of the nipple prirnordiumcornifies, and desquamates to form a depression; this widens and is gradually obhterated.
The Areola of the Breast
The areola of the breast has a simple phylogenic explanation and one which helps to clarify its peculiarities. After excavation of the nipple primordium the simplest structure is to be found in the monotreme; there is no nipple, and all the mammary tubular glands have equal value. In the higher mammals the area becomes raised into a nipple. In the human it is only the central part to be so raised, the periphery remaining flat to form the areola. Only the central mammary glands are stimulated by suckling; those in the peripheral areola remain unstimulated and small. In some mammals the whole of the epithelial primordium is concerned in the formation of the nipple, whilst in others, such as the marsupial, the true nipple forms a cap to a much longer structure (Fig. 14).
Fig. 14. The areola of the breast has a simple phylogenetic explanation. The primitive arrangement is to be found in the monotreme (a) where there is no nipple and all the tubular glands have equal value. In the human, the central part of the area becomes elevated to form a nipple (b) ; the tubular glands arising from the areolar zone do not receive the same stimulation as those arising from the nipple, and hence remain comparatively small. In other mammals no areola is present, e.g., marsupials (b, c). The Epithelial Proliferations of Hugo Schmidt
In human embryos, from 28 mm. to 60 mm., Hugo Schmidt (1896) observed epithelial proliferations or “ hyperthelial structures ” in the mammary band area on the lateral aspect of the trunk, but chiefly in the region of the nipple. These epithelial proliferations resemble slightly the epithelial nipple primordium, but are much smaller, never more than slightly grooving the mesoderm and disappearing spontaneously in about the 60 mm. embryo. The presence of these areas of proliferation has been confirmed repeatedly, but their significance is not clear.
Schmidt (1896) believed that they arose from the “ mammary band,” and that they represented temporary normal hyperthelia, persistence of any one resulting in a supernumerary nipple. Actually, however, the “ mammary band ” is disappearing as these structures develop. Walter (1902) considered that they were vestiges of marsupial pockets. The regularity with which he has depicted them in relation to the main epithelial primordium has not been confirmed. Brouha (1905) believed that they were merely the normal pilo-glandular structures of the surrounding skin, although such an explanation, he admits, does not account for their disappearance before the hairs and skin glands actually develop. These proliferations remain unexplained; it is probable that they bear no relation to the mammary gland.
The Comparative Embryology of the Mammary Gland Animals are classified as mammals when the female of the species possesses cutaneous glands, the mammary glands, secreting milk for the nourishment of her young. Mammals are subclassified into monotremes, marsupials, and placentals.
Fig. 15. Ornithorhynchus (Bresslau, 1902) to show the tubular glands passing through a linear split in the cutaneous muscle.
Fig. 16. A ventral View of the mammary pouch of echidna, to show the glandular ’ area at the lateral edge of the pouch (after Bresslau, 1902).
In the monotremes the mammary glands are composed of 100-150 tubular glands opening into a confined area; there is no nipple. Hairs are well developed and nourishment is obtained by licking or sucking them. In the monotreme, ornithorhynchus or platypus, there are two narrow linear slits in the cutaneous muscle through which the tubular glands pass. In the monotreme, echidna, the tubular glands open on the lateral edge of a muscle-free, central area, the mammary pouch. This mammary area, like that of the platypus, is provided with a profusion of well-developed hairs (Figs. 15 and 16).
Marsupials possess mammary glands that are provided with particularly prominent nipples. The nipples themselves are contained in nipple pouches which become everted on the birth of the young. The mammary glands are situated typically within a fold on the surface of the abdomen, known as the pouch or marsupium. The young are born alive, and for a variable period their post-natal life is spent clinging tightly to the nipple. The number of breasts in marsupials varies considerably; there may be a single or double row on either side of the abdomen. Hairs and sebaceous glands develop in addition to the ducts ; the hairs have only a temporary existence, though in some marsupials they are persistent.
The mammary glands of placentalia, or higher mammals, are likewise discrete and provided with nipples. The young, born in a more advanced state, obtain their nourishment at intervals from the maternal mammary glands; they are not retained suspended from a nipple, nor are they sheltered within a marsupium. The glands are usually to be found on the ventral surface, although in the hippopotamus and South American rodents the glands are more dorsal than ventral. Unpaired, and fourrowed nipples, are never seen in placental mammals. The nipples may extend along the whole length of the body as in the pig, cat and dog; they may be present in the pectoral region only as in man, anthropoid monkeys, elephants and bats; or in the inguinal region as in cows and goats and other ruminants. There may be a pair in the pectoral region and a second pair in the inguinal region, as in the great ant-eater.
A mammary crest is to be found in all mammals, a portion of which proliferates to form the epithelial primordium. Some believe the pouch of echidna to be composed really of two lateral pouches, and that the epithelial primordium is the precursor of this lateral pouch, with the central portion forming the glandular field. The nipple pouches of the marsupial are considered homologous with these mammary pouches of the echidna. It is suggested that this primitive arrangement is to be found in certain placental mammals, such as the cow, where the nipple contains a single long duct. The human nipple is regarded as a secondary nipple, homologous with the secondarily everted marsupial nipple (Fig. 17).
(Gegenbaur, 1873; laatsch, 1895.)
Others believe that the epithelial primordium forms only the mammary gland area, found in its simplest form in the monotreme. The nipple pouch of marsupials is a secondary development due to the growth of the dermis‘ and epidermis surrounding the primordium. In placental mammals the mammary ducts usually arise from multiple points on the deep surface of the primordium, but in the cow this is modified to form a single long duct, so that the mammary glands of all placental mammals have an equal value (Fig. 18).
There are several aspects of pre-natal development described in this lecture which have been overlooked or disregarded by embryologists of the past. We surgeons, who so often must dissect or resect the living breast, must accept the dictum that we cannot really understand the abnormal unless we know the normal, and how it develops. A careful embryological study of the mammary gland provides an answer to the curious who wish to know why well-developed hairs so often appear around the edge of the areola, seemingly without reason; but when our monotreme cousins are recalled an explanation is immediately forthcoming. Such investigation does much to remove the glamour of the supernumerary nipple from the minds of those who regard it as a relic of the past, or the light of things to come.
Fig. 17. Theory 1 is based on the belief that the pouch of echidna is composed of two lateral pouches. In marsupials, the nipple pouch, seen on the left, is homologous with the lateral pouch of echidna. This primitive arrangement persists in certain placental mammals, e.g., the cow. The nipple pouch of the marsupial everts during suckling and a long “ secondary nipple ” is formed, seen on the right, which is regarded as corresponding with that found in the human.
Fig. 18. Theory 2 supposes that only. the glandular area of the echidna develops from the epithelial primordium; the nipple pouch of the marsupial is a secondary structure._ The long single canal of the cow nipple is not regarded as a primitive type but a variation in the mode of outgrowth of the mammary ducts. Hence all mammary glands of placental mammals are regarded as homologous structures.
References
BALINSKY, B. I. (1949) On the pre-natal growth of the mammary gland rudiment. Proc. Anat. Soc. Grt. Brit. and Ireland. Communication.
BEARD, J. (1897) The birth period of Trichosurus vulpecula. Zool. Jahrb. 11, 77-96. 118
BONNET, R. (1892-7) Die Mammarorgane im Lichte der Ontogenie und Phylogenie. Ergebn. Anat. Entw. Gesch. 2, 604-658; 7, 937-976.
BRESSLAU, E. (1902) Entwicklungsgeschichte der Mammarorgane. Z. Morph. Anthr. 4, 261.
- (1910) Der Mammarapparat. Ergebn. Anat. Entw. Gesch. 19, 275-349.
BRoIgA,3M. 3(l905 a) Sur la signification morphologique de la mamelle. Anat. Anzeig. , 11- 17.
(1905 b) Sur la bande et la créte mammaire. Anat. Anz. 27, 462-464.
- (1905 c) Phases du développement de la mamelle. Arch. Biol. 21, 459,
EGGELING, H. (1904) Entwicklung der menschlichen Milchdruse. Anat. Anz. 24, 595.
GEGENBAUR, C. (1873) Milchdrusenpapillen der Saugetierre. Jena. Z. Naturw. 7, 204.
HIRSCHLAND, L. (1898) Entwicklung der Mammarorgane. Anat. Hefte. 11, 221.
HUGHES, E. S. R. (1948) The intrauterine development of the Mammary Gland. Proc. Anat. Soc. Grt. Brit. and Ireland. Communication.
Huss, M. (1873) Entwicklungsgeschichte der Milchdrusen. Jena. Z. Naturw. 7, 176.
JONES, F. WOOD (1923) The Mammals of South Australia. Part one. Adelaide.
KALLIUS, E. (1897) Milchleiste bei einem menschlichen Embryo. Anat. Hefte, 8, 153.
Keith A. Human Embryology and Morphology. (1921) New York, Longmans, Green & Co. London: Edward Arnold.
KLAATSCH, H. (1893) Marsupialrudimente bei Placentaliern. Morph. Jb. 20, 276.
- (1895) Mammartaschen und Marsupium von Echidna. Anat. Anz. 10, 145.
KOLLIKER, A. (1879) Brustdruse. Verh. Med.-Physik. Ges. Wurzb. 14, 198-202.
LANGER, C. (1850) Milchdruse. Denkschr. Akad. Wiss. Wien.
LUSTIG, H. (1915) Entwicklungsgeschichte der Brustdruse. Arch. Mik. Anat. 87, 38.
MECKEL, J . F. (1820) Handbuch der menschlichen Anatomie (Quoted by Bonnet, 1897).
NEUMANN,9£‘I. O. and OING, M. (1929) Polymastie und Polythelie. Arch. Gyncik. 138, 4 .
PROFE, O. (1898) Ontogenie und Phylogenie der Mammarorgane. Anat. Hefte, 11, 247.
REIN, G. (1882) Embryonal Entwicklungsgeschichte der Milchdruse. Arch. Mik. Anat. 21, 678.
SCHMIDT, H. (1896) Hyperthelie menschlichen Embryonen. Anat. Anz. 11, 702
- (1898) Milchdruse und Hyperthelie menschlichen Embryonen. Morph.. 8, .
SCHULTZE, O. (1892) Erste Anlage des Milchdrusenapparates. Anat. Anz. 7, 265.
STRAHL, H. (1898) Erste Entwicklung der Mammarorgane. Verh. anat. Ges. 12, 236.
WALTER, H. E. (1902) On transitory epithelial structures associated with the Mammary Apparatus in Man. Anat. Anz. 22, 97-111.
Cite this page: Hill, M.A. (2024, April 27) Embryology Paper - Development of the mammary gland - Arris and Gale Lecture. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_Development_of_the_mammary_gland_-_Arris_and_Gale_Lecture
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G