In Vitro Oogenesis
| Embryology - 19 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
In an adult human female, the development in vivo of a primordial follicle containing an oocyte to a preovulatory follicle takes in excess of 120 days.
- The Latin, In vitro = "in glass" meaning in essence a test tube as apposed to in vivo (in life or a living body)
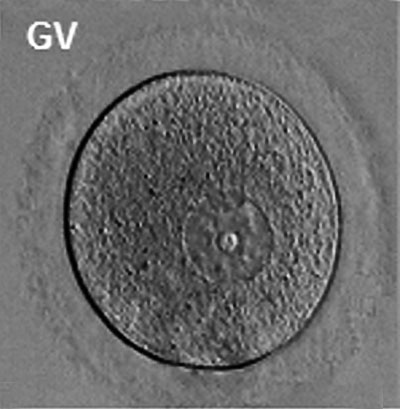
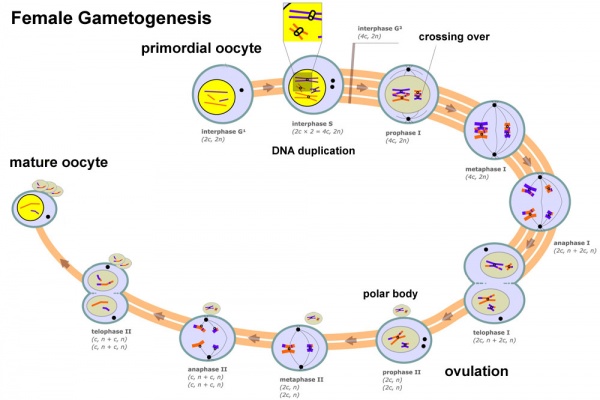
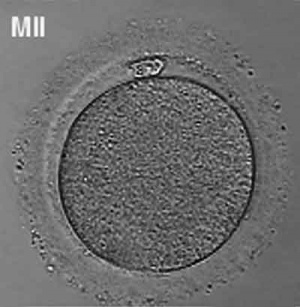
Prior to release from the ovary oocytes (eggs, ova) are arrested at an early stage of meiosis in the first meiotic division as a primary oocyte (primordial follicle). Following puberty, during each menstrual cycle, pituitary gonadotrophin stimulates completion of meiosis 1 the day before ovulation. Early oocytes are also classified as immature (germinal vesicle (GV) or metaphase I (MI) stage). The breakdown of the germinal vesicle indicates a resumption of meiosis and the extrusion of the first polar body (1 PB) indicates completion of the first meiotic division in human oocytes.
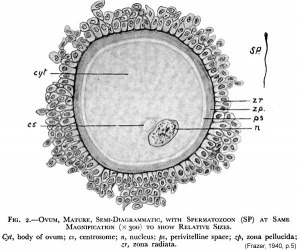
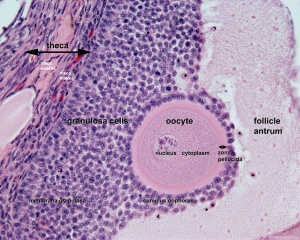
The released oocyte is surrounded by a thick specialised extracellular matrix, the zona pellucida, that in turn is covered in layers of cells, the granulosa cell layer.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: In Vitro Oogenesis |
Research
Prof Robert Gilchrist - The Reproductive Technology Revolution
Dr Gilchristis head of the Oocyte Biology Research Unit (UNSW) his primary research interests are in the regulation of mammalian oocyte development and maturation, and the development of novel oocyte maturation techniques for infertility treatment.[3][4][5]
- "The cyclic nucleotides, cAMP and cGMP, are the key molecules controlling mammalian oocyte meiosis. Their roles in oocyte biology have been at the forefront of oocyte research for decades and many of the long standing controversies in relation to the regulation of oocyte meiotic maturation are now resolved. It is now clear that the follicle prevents meiotic resumption through the actions of natriuretic peptides and cGMP inhibiting the hydrolysis of intra-oocyte cAMP and that the preovulatory gonadotrophin surge reverses these processes. The gonadotrophin surge also leads to a transient spike in cAMP in the somatic compartment of the follicle; research over the past 2 decades has conclusively demonstrated that this surge in cAMP is important for the subsequent developmental capacity of the oocyte. This is important, as oocyte in vitro maturation (IVM) systems practiced clinically do not recapitulate this cAMP surge in vitro, possibly accounting for the lower efficiency of IVM compared to clinical IVF. This review focuses in particular on this latter aspect - the role of cAMP/cGMP in the regulation of oocyte quality. We conclude that clinical practice of IVM should reflect this new understanding of the role of cyclic nucleotides, thereby creating a new generation of ART and fertility treatment options."[3]
- Links: ANAT2341 Lab 4
In Vivo Oogenesis
A human infant ovary histology, showing the large number of oocytes occupying the ovary cortical region. Compare this with a mature ovary and note the absence of any follicle development in the infant. These early oocytes remain at the diplotene stage of the meiosis I during development from fetal life and postnatal childhood, until puberty when the lutenizing hormone (LH) surges stimulate the resumption of meiosis.
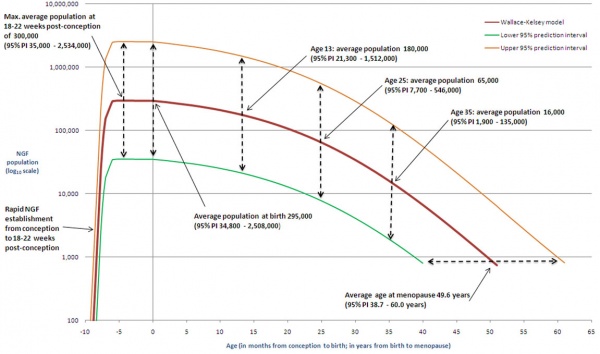
The graph below shows the changes in human germ cell numbers in the ovary with age, peaking at about 7 million (occuring in early fetal development) and then decreasing by apopotic cell death. At puberty there remain only about 400,000 and only about 10% of these will be released through reproductive life. (More? Menstrual Cycle)
Human ovary non-growing follicle model[6]
Oocyte Growth
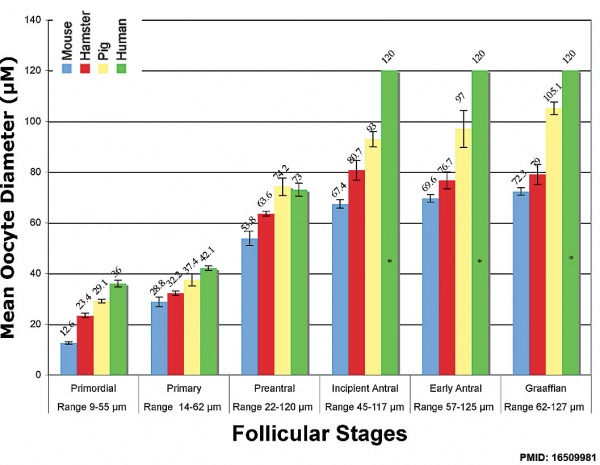
Graph shows species comparison in oocyte size growth (diameter) at different stages of follicle development.[7] (See also Follicle size graph)
| Human Ovarian Follicle Classification | |||||
| Class | Alternate nomenclature | Type | Number of Cells | Size (diameter µm) | Size ultrasound (mm) |
|---|---|---|---|---|---|
| primordial follicle | small | 1, 2, 3 | 25 | less than 50 | |
| primary follicle | preantral | 4 5 |
26 - 100 101 - 300 |
up to 200 | |
| secondary follicle | antral small antral large antral |
6 7 |
3001 - 500 501 - 1000 |
500 1000 - 6000 |
less than 18 |
| preovulatory follicle | Graafian | 8 | greater than 1000 | greater than 6000 | 18 – 28 |
| Links: ovary | oocyte | menstrual cycle | |||||
Meiosis
In females, the total number of eggs ever to be produced are present in the newborn female initially arrested at the diplotene stage of the meiosis I from fetal life through childhood until puberty, when the lutenizing hormone (LH) surges stimulate the resumption of meiosis.
- All eggs are arrested at an early stage (prophase I) of the first meiotic division as a primary oocyte (primordial follicle). Following purberty, during each menstrual cycle, pituitary gonadotrophin stimulates completion of meiosis 1 the day before ovulation.
- In meiosis 1, a diploid cell becomes 2 haploid (23 chromosomes) daughter cells, each chromosome has two chromatids. One cell becomes the secondary oocyte the other cell forms the first polar body.
- The secondary oocyte then commences meiosis 2 which arrests at metaphase and will not continue without fertilization.
- At fertilization meiosis 2 completes, forming a second polar body. Note that the first polar body may also undergo this process forming a third polar body.
- Links: Cell Division - Meiosis
Polar Body
The breakdown of the germinal vesicle indicates a resumption of meiosis and the extrusion of the first polar body (1 PB) indicates completion of the first meiotic division in human oocytes. The polar body is a small cytoplasmic exclusion body formed to enclose the excess DNA formed during the oocyte (egg) meiosis and following sperm fertilization. There are 2-3 polar bodies derived from the oocyte present in the zygote, the number is dependent upon whether polar body 1 (the first polar body formed during meiosis 1) divides during meiosis 2. This exclusion body contains the excess DNA from the reductive division (the second and third polar bodies are formed from meiosis 2 at fertilization). These polar bodies do not contribute to the future genetic complement of the zygote, embryo or fetus.
Recent research in some species suggest that the space formed by the peripheral polar body (between the oocyte and the zona pellucia) can influence the site of spermatozoa fertilization.
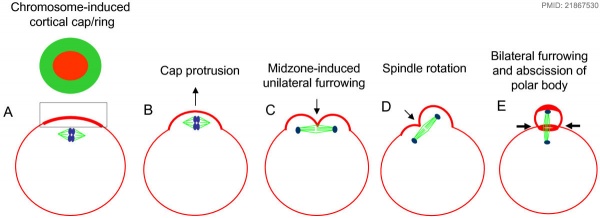
Polar Body Extrusion Model
The following cartoon model from mouse oocyte study of polar body extrusion, involving cortical cap protrusion and spindle midzone-induced membrane furrowing.[8]
Assisted reproductive techniques involving intracytoplasmic sperm injection (ICSI) have looked at the "quality" of the polar body and found that the morphology is related to mature oocyte viability and has the potential to predict oocyte fertilization rates and pregnancy achievement.[9][10]
- Links: Meiosis
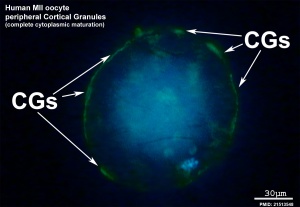
Cortical Granules
The release of cortical granules by exocytosis, the "cortical reaction", occurs following spermatozoa fertilisation and is the main block to polyspermy by modifying the zone pellucida. These granules develop from the golgi apparatus initially forming smaller vesicles that coalesce to form mature membrane bound cortical granules (0.2 to 0.6 microns in diameter) located in the cortex of unfertilized oocytes. In mammals, cortical granule production in the developing follicular oocyte is an ongoing and continuous process, with newly synthesized granules translocating to the cortex until the time of ovulation.
Cortical granules:
- vary in time of initial development between species.
- primordial follicle stage - rat and mouse.
- primary follicle stage - human, monkey, hamsters, and rabbit.
- vary in type formed in the same species.
- migration requires the microfilaments of the cell cytoskeleton.
- are evenly distributed in the cortex of unfertilised oocytes.
- contain carbohydrates, proteinases, ovoperoxidase, calreticulin, N-acetylglucosaminidase
Ovastacin
- oocyte-specific member of the astacin family
- mouse cortical granule protease (metalloendoproteases)
- cleaves zona pellucida ZP2
- female mice lacking ovastacin do not cleave ZP2 after fertilisation[11]
Oocyte-Follicle Cell Interaction
The oocyte and the surrounding granulosa cells have a complex paracrine interactions during follicle growth and development. Oocyte maturation has been shown to depend on secretory products of both the granulosa and cumulus cells.
Oocyte Factors
- promotes granulosa cell proliferation in preantral and antral follicles (GDF-9, BMP15)
- cumulus expansion and granulosa cell differentiation are dependent upon oocyte-derived factors
- BMP15 inhibits FSH-stimulated progesterone production
Oocyte Metabolism
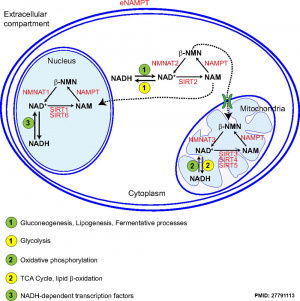
In the mouse, the secondary follicle stage through to large antral follicle stage the oocyte has a highly oxidative metabolism. In contrast, the surrounding surrounding granulosa and cumulus cells are highly glycolytic. In this second group, the cumulus cells are found to be more glycolytic than the granulosa cells.[13]
- oxidative metabolism - requires oxygen (aerobic) to make energy from carbohydrates (sugars) into pyruvate that passes into the mitochondria where it is fully oxidised by oxygen into carbon dioxide. Also called aerobic metabolism, aerobic respiration, and cell respiration.
- glycolytic metabolism - requires no oxygen (anaerobic) to make energy from carbohydrates (sugars) into pyruvate that is reduced by adenine dinucleotide hydride (NADH).
In the cat oocyte, in pre-antral oocytes mitochondria have a homogeneous distribution throughout the cytoplasm. In the antral stage they have relocated to a mainly pericortical distribution.[14]
- Links: OMIM SIRT1
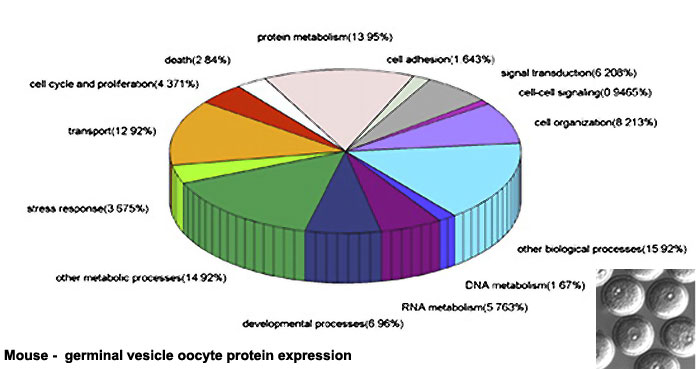
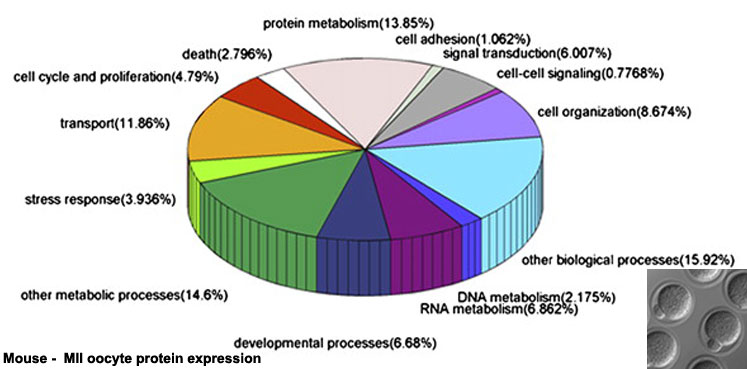
Oocyte Protein Expression
The table above shows the pattern of protein expression (as percentages of total) in the mouse germinal vesicle and MII oocyte according to 14 molecular function categories.[15]
- Links: Germinal vesicle oocyte protein expression | MII oocyte protein expression | Zygote Protein Expression | Mouse Development | Oocyte Development | Zygote
Oocyte Telomerase Reverse Transcriptase
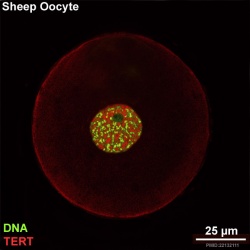
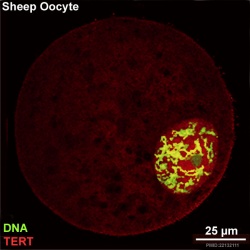
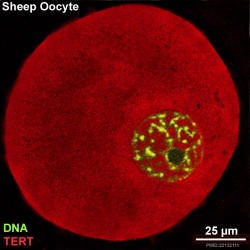
There is a redistribution of the enzyme that regulates telomere length during oocyte development. The following oocyte images are from a recent study of sheep in vitro follicle development.[16]
|
|
| preantral | early antral |
|
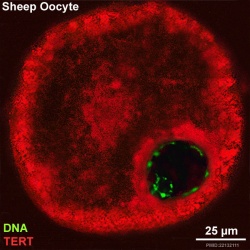
|
| early antral | preovulatory follicle |
- TERT - Red (Cy3-conjugated secondary antibody) (telomerase reverse transcriptase, TERT)
- DNA - Green (SYBR Green 14/I)
- Sheep Oocyte TERT: preantral | early antral | early antral | preovulatory follicle | Oocyte Development | Sheep Development
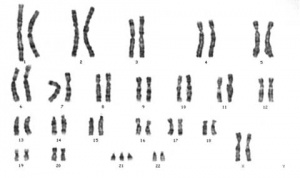
Abnormalities
Meiotic non-disjunction resulting in aneuploidy, most are embryonic lethal and not seen. The potential for genetic abnormalities increase with maternal age.
| Autosomal chromosome aneuploidy | Sex chromosome aneuploidy |
|---|---|
|
|
Additional Images
References
- ↑ Wang JJ, Ge W, Liu JC, Klinger FG, Dyce PW, De Felici M & Shen W. (2017). Complete in vitro oogenesis: retrospects and prospects. Cell Death Differ. , 24, 1845-1852. PMID: 28841213 DOI.
- ↑ White YA, Woods DC, Takai Y, Ishihara O, Seki H & Tilly JL. (2012). Oocyte formation by mitotically active germ cells purified from ovaries of reproductive-age women. Nat. Med. , 18, 413-21. PMID: 22366948 DOI.
- ↑ 3.0 3.1 Gilchrist RB, Luciano AM, Richani D, Zeng HT, Wang X, Vos MD, Sugimura S, Smitz J, Richard FJ & Thompson JG. (2016). Oocyte maturation and quality: role of cyclic nucleotides. Reproduction , 152, R143-57. PMID: 27422885 DOI.
- ↑ Robertson DM, Gilchrist RB, Ledger WL & Baerwald A. (2016). Random start or emergency IVF/in vitro maturation: a new rapid approach to fertility preservation. Womens Health (Lond) , 12, 339-49. PMID: 27248769 DOI.
- ↑ Russell DL, Gilchrist RB, Brown HM & Thompson JG. (2016). Bidirectional communication between cumulus cells and the oocyte: Old hands and new players?. Theriogenology , 86, 62-8. PMID: 27160446 DOI.
- ↑ Wallace WH & Kelsey TW. (2010). Human ovarian reserve from conception to the menopause. PLoS ONE , 5, e8772. PMID: 20111701 DOI.
- ↑ Griffin J, Emery BR, Huang I, Peterson CM & Carrell DT. (2006). Comparative analysis of follicle morphology and oocyte diameter in four mammalian species (mouse, hamster, pig, and human). J. Exp. Clin. Assist. Reprod. , 3, 2. PMID: 16509981 DOI.
- ↑ Wang Q, Racowsky C & Deng M. (2011). Mechanism of the chromosome-induced polar body extrusion in mouse eggs. Cell Div , 6, 17. PMID: 21867530 DOI.
- ↑ Ebner T, Yaman C, Moser M, Sommergruber M, Feichtinger O & Tews G. (2000). Prognostic value of first polar body morphology on fertilization rate and embryo quality in intracytoplasmic sperm injection. Hum. Reprod. , 15, 427-30. PMID: 10655316
- ↑ Younis JS, Radin O, Izhaki I & Ben-Ami M. (2009). Does first polar body morphology predict oocyte performance during ICSI treatment?. J. Assist. Reprod. Genet. , 26, 561-7. PMID: 19960239 DOI.
- ↑ Burkart AD, Xiong B, Baibakov B, Jiménez-Movilla M & Dean J. (2012). Ovastacin, a cortical granule protease, cleaves ZP2 in the zona pellucida to prevent polyspermy. J. Cell Biol. , 197, 37-44. PMID: 22472438 DOI.
- ↑ Aguilar-Arnal L, Ranjit S, Stringari C, Orozco-Solis R, Gratton E & Sassone-Corsi P. (2016). Spatial dynamics of SIRT1 and the subnuclear distribution of NADH species. Proc. Natl. Acad. Sci. U.S.A. , , . PMID: 27791113 DOI.
- ↑ Cinco R, Digman MA, Gratton E & Luderer U. (2016). Spatial Characterization of Bioenergetics and Metabolism of Primordial to Preovulatory Follicles in Whole Ex Vivo Murine Ovary. Biol. Reprod. , 95, 129. PMID: 27683265 DOI.
- ↑ Songsasen N, Henson LH, Tipkantha W, Thongkittidilok C, Henson JH, Chatdarong K & Comizzoli P. (2017). Dynamic changes in mitochondrial DNA, distribution and activity within cat oocytes during folliculogenesis. Reprod. Domest. Anim. , 52 Suppl 2, 71-76. PMID: 28111812 DOI.
- ↑ Wang S, Kou Z, Jing Z, Zhang Y, Guo X, Dong M, Wilmut I & Gao S. (2010). Proteome of mouse oocytes at different developmental stages. Proc. Natl. Acad. Sci. U.S.A. , 107, 17639-44. PMID: 20876089 DOI.
- ↑ Barboni B, Russo V, Cecconi S, Curini V, Colosimo A, Garofalo ML, Capacchietti G, Di Giacinto O & Mattioli M. (2011). In vitro grown sheep preantral follicles yield oocytes with normal nuclear-epigenetic maturation. PLoS ONE , 6, e27550. PMID: 22132111 DOI.
Reviews
Li R & Albertini DF. (2013). The road to maturation: somatic cell interaction and self-organization of the mammalian oocyte. Nat. Rev. Mol. Cell Biol. , 14, 141-52. PMID: 23429793 DOI.
Liu M. (2011). The biology and dynamics of mammalian cortical granules. Reprod. Biol. Endocrinol. , 9, 149. PMID: 22088197 DOI.
Articles
Search
- NCBI Bookshelf oocyte | oogenesis | oocyte development
- Pubmed oocyte | oogenesis | oocyte development
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Terms
Note there are additional specific term glossaries available listed at bottom of this table.
- antral follicle - (secondary) the stage following preantral in the decription of the sequence ovarian follicle development.
- antrum - (L. a cave), cavity; a nearly-closed cavity or bulge. In the ovary this refers to the follicular fluid-filled space within the follicle.
- atretic follicle - An ovarian follicle that fails to mature and degenerates. Also called "atresia" refering to the process of degeneration of the ovarian follicle. This process can occur at any stage of follicle development (folliculogenesis).
- clomiphene citrate - drug taken orally to promote the process of follicle/egg maturation.
- COCs - (cumulus-oocyte complexes) term used in Assisted Reproductive Technology to describe the ovulated Graafian follicle consisting of the oocyte surrounded by a packed layers of cumulus cells.
- corona radiata - Layer of follicle cells of cumulus oophorus remaining directly attached to zona pellucida of the oocyte. These cells communicate with the oocyte through the zone pellucida, also called granulosa cells.
- corpus albicans - (L. corpus = body, L. albicans = whitish); a degenerating corpus luteum in ovary.
- corpus luteum - (L. corpus = body, L. luteum = yellow) The remains of the ovulating ovarian follicle after ovulation, that acts as the initial endocrine organ supporting pregnancy and preventing menstruation (loss of the endometrial lining). de Graaf first observed it in the ovary of a cow as a yellow structure.
- cortical - (L. corticalis) at the outside (like the bark of a tree), usually combined with medulla meaning the core.
- cumulus oophorus - (L. cumulus = a little mound G. oon = egg + phorus = bearing); part of the wall of an ovarian follicle surrounding and carrying the ovum (oocyte).
- dictyate arrest - (prophase arrest) the oocyte meiosis state before puberty resumed with a surge of pituitary luteinizing hormone.
- first polar body - a small cytoplasmic exclusion body contains the excess DNA from the oocyte meiosis formed during meiosis 1.
- follicle - (L. folliculus = little bag,dim. of L. follis). A structure which develops in the ovary and contains a developing egg (oocyte).
- follicle stimulating hormone - (FSH, gonadotropin) A glycoprotein hormone secreted by anterior pituitary (adenohypophysis gonadotrophs, a subgroup of basophilic cells) and acts on gametogenesis and other systems in both males and females. Females, FSH acts on the ovary to stimulate follicle development. Males, acts on the testis Sertoli cells to increase androgen-binding protein (ABP) that binds androgens and has a role in spermatogenesis. pituitary
- follicular fluid - the fluid found in the antrum of a secondary follicle. Secreted by cells in the wall of the follicle. This fluid is released along with the oocyte at ovulation.
- germinal epithelium - cellular component covering surface of ovary, it is continuous with mesothelium covering mesovarium. Note that it is a historical misnomer, as it is not the actual site of germ cell formation.
- Graafian follicle - named after Regnier de Graaf (1641-1673), an historic Dutch physician embryologist who studied pregnancy using rabbits.
- granulosa cells - the supporting cells that surround the developing egg within the follicle thecal layers.
- homologs - maternal and paternal homologous chromosomes.
- Izumo1 - a protein located on the equatorial segment of acrosome-reacted spermatozoa recognizes its receptor Juno, on the oocyte surface, for plasma membrane binding and fusion. Named for a Japanese shrine dedicated to marriage. OMIM609278
- Juno - (folate receptor-δ; FOLR-δ) a glycophosphatidylinositol (GPI)-anchored, cysteine-rich glycoprotein on the oocyte surface for fertilisation that is the receptor of Izumo1 on the spermatozoa, for plasma membrane binding and fusion. OMIM615737
- luteinizing hormone - (LH, gonadotropin, lutropin, Interstitial Cell Stimulating Hormone, ICSH) glycoprotein hormone releasd from anterior pituitary hormone that acts on the gonad and has a role in male and female reproduction. Female, LH triggers ovulation (release of the oocyte). Male, LH stimulates testis interstital cell (Leydig cell) production of testosterone. Have been used clinically in humans for the treatment of female infertility.
- meiosis - oocyte reductive (diploid to haploid) cell division, with 1 round of DNA replication is followed by 2 rounds of chromosome segregation. The process beginning in the fetus and only completed at fertilization.
- mesovarium - mesentry of the ovary formed from a fold of the broad ligament that attaches the ovary.
- medullary - (L. medius = in the middle) relating to the medulla; pith, marrow, inner portion of an organ. Usually combined with cortex (cortical) meaning the outer layer.
- oocyte - (Greek, oo = egg, ovum) The term used to describe the haploid egg or ovum formed within the ovary (female gonad) and released to enter the uterine tube and be transported to the uterus. The mature oocyte is the cell released from the ovary during ovulation.
- oocyte retrieval - (egg retrieval) A clinical in vitro fertilization (IVF) procedure to collect the eggs contained in the ovarian follicles.
- oogenesis - (Greek, oo = egg + genesis = origin, creation, generation) process of diploid oogonia division and differentiation into an haploid oocyte (egg) within the ovary (female gonad). Mammalian meiosis will only be completed within the oocyte if fertilization occurs.
- oogonia - (Greek, oo = egg) diploid germ cells within the ovary (female gonad) which provide the primary oocytes for oocyte (egg) formation. In humans, all oogonia form primary oocytes within the ovary before birth.
- oolemma - (zona pellucida, vitelline membrane).
- oophorus - (Greek, oo = egg + phorus = carrying, egg-bearing) cumulus oophorus, used to describe the granulosa cells within the follicle that tether or link the oocyte to the wall of the follicle.
- ovarian reserve - Clinical term for the number of oocytes (non-growing follicles) available for possible fertilization at the different times during female reproductive life. A blood test for Anti-Mullerian Hormone (AMH) levels is used clinically as a measure of the ovarian reserve. human graph
- ovastacin - an oocyte released enzyme following fertilization that cleaves ZP2 protein to prevent polyspermy.
- ovulation - release of the oocyte from the mature follicle. In humans generally a single oocyte is released from a cohort of several maturing follicles.
- ovum - oocyte, note that historically this same term was also used to describe the early stages following fertilisation.
- polar body - small cytoplasmic exclusion body contains the excess DNA from the oocyte meiosis reductive division. The first polar body formed during meiosis 1, the second and sometimes third polar bodies are formed from meiosis 2 at fertilization.
- polyspermy - abnormal fertilization by more that a single spermatozoa, may generate a hydatidiform mole.
- preantral follicle - (primary) the stage following primordial in the description of the sequence ovarian follicle development.
- primary follicle - (preantral) the stage following primordial in the description of the sequence ovarian follicle development.
- primordial follicle - the first stage in the description of the sequence ovarian follicle development. Present in the ovary from birth, located in the stroma of the ovary cortex beneath the tunica albuginea. The primordial follicle is the oocyte and the surrounding follicular cells.
- primordial germ cell - oocyte present in the primordial follicle ovary from birth, located in the stroma of the ovary cortex beneath the tunica albuginea. The primordial follicle is the oocyte and the surrounding follicular cells.
- second polar body - a small cytoplasmic exclusion body contains the excess DNA from the oocyte formed during meiosis 2 at fertilization.
- secondary follicles - the stage following primary in the description of the sequence ovarian follicle development.
- stromal cells - in the ovary, cells surrounding the developing follicle that form a connective tissue sheath (theca folliculi). This layer then differentiates into 2 layers (theca interna, theca externa). This region is richly vascularized and involved in hormone secretion.
- superovulation therapy - a fertility drug treatement (oral clomiphene citrate and/or injectable FSH with or without LH) aimed at stimulating development/release of more than one follicle during a single menstrual cycle.
- tertiary follicle - (preovulatory, Graffian) the stage following secondary in the description of the sequence ovarian follicle development.
- theca folliculi - stromal cells in the ovary, cells surrounding the developing follicle that form a connective tissue sheath. This layer then differentiates into 2 layers (theca interna, theca externa). This region is vascularized and involved in hormone secretion.
- theca externa - stromal cells forming the outer layer of the theca folliculi surrounding the developing follicle. Consisting of connective tissue cells, smooth muscle and collagen fibers.
- theca interna - stromal cells forming the inner layer of the theca folliculi surrounding the developing follicle. This vascularized layer of cells respond to LH (leutenizing hormone) synthesizing and secreting androgens which are processed into estrogen.
- transzonal projection - (TZP) ovarian follicle term describing the cellular membraneous extension from the granulosa cell through the zona pellucida to the oocyte cell membrane where it forms gap junctions or adherens junctions allowing signalling and adhesion between the two cells.
- tunica albuginea - dense connective tissue layer lying near the ovary surface, a layer of simple squamous to cuboidal epithelial covers this layer. A similar named structure is found in male genital system.
- uterus - site of embryo implantation and development. Uterine wall has 3 major layers: endometrium, myometrium, and perimetrium. Endometrium can be further divided into the functional layer (shed/lost during menstruation) and basal layer (not lost during menstruation).
- zinc sparks following fertilization the oocyte releases a burst of zinc atoms in brief bursts (zinc sparks) has a role in zonal pellucida induced structural changes (hardening) along with ovastacin cleavage of ZP2 protein.
- zona hardening - following fertilization the structural changes that occur to the zona pellucida to prevents further spermatozoa binding acting as a block to polyspermy.
- zona pellucida - extracellular layer lying directly around the oocyte underneath follicular cells. Has an important role in egg development, fertilization and blastocyst development. This thick extracellular matrix consists of glcosaminoglycans and 3 glycoproteins (ZP1, ZP2, ZP3). (More? Zona pellucida)
| Other Terms Lists |
|---|
| Terms Lists: ART | Birth | Bone | Cardiovascular | Cell Division | Endocrine | Gastrointestinal | Genital | Genetic | Head | Hearing | Heart | Immune | Integumentary | Neonatal | Neural | Oocyte | Palate | Placenta | Radiation | Renal | Respiratory | Spermatozoa | Statistics | Tooth | Ultrasound | Vision | Historic | Drugs | Glossary |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 19) Embryology In Vitro Oogenesis. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/In_Vitro_Oogenesis
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G