Endocrine - Pituitary Development
| Embryology - 9 Jun 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
Historically, this endocrine gland was called the "pituitary" as it was originally thought to produce mucous that discharged through the nose. We now know that this is not the function of the pituitary, or hypophysis is an endocrine gland links the brain to peripheral endocrine organs and systems of the body through several specific hormones. The developmental origin of the hypophysis is also unique, with dual epithelial origins from neural ectoderm (posterior) and from surface ectoderm (anterior).
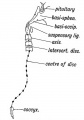
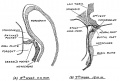
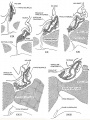
During development, the boundary surface ectoderm placode epithelium on the roof of the pharynx forms a pocket (Rathke's pouch) that comes into contact with the ectoderm of developing brain.
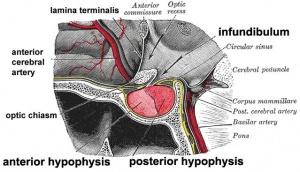
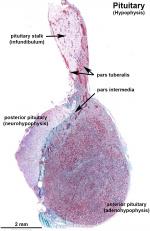
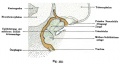
Anatomically, the pituitary has 2 main parts posterior, or neurohypophysis and anterior, or adenohypophysis (the pars distalis, pars intermedia, and pars tuberalis). Between the two a specialized vascular (portal) system allows communication from the brain to peripheral endocrine organs and other systems.
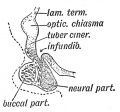
The pituitary is located within the pituitary fossa of the sphenoid bone, anterior to the lamina terminalis and superior to the pharynx. The shape of the bone surrounding the pituitary led to the naming sella turcica (Latin sella = saddle, turcica = Turkish), as it resembled a saddle shape.
Maternal pituitary also increases in size during pregnancy, due mainly to hyperplasia of the prolactin secreting cells (lactotrophs), in preparation for lactation after birth.
| Historic Embryology |
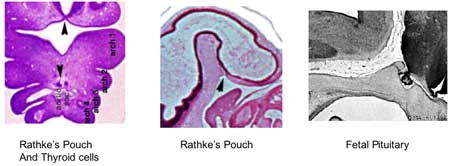
| During pituitary development, the boundary surface ectoderm placode epithelium on the roof of the pharynx forms a transient epithelial pocket (Rathke's pouch) that comes into contact with the ectoderm of developing brain.
|
| Lecture - Head Development | placode
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References
|
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
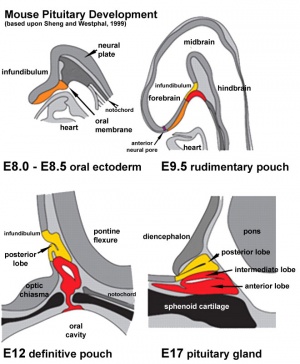
Development Overview
- Dual ectoderm origins
- Ectoderm - ectoderm roof of stomodeum, Rathke's pouch, adenohypophysis
- Neuroectoderm - prosenecephalon, neurohypophysis
Adenohypophysis
- Anterior wall proliferates - pars distalis
- Posterior wall little growth – pars intermedia
- Rostral growth around infundibular stem – pars tuberalis
Neurohypophysis
- Infundibulum – median eminence, infundibulum, pars nervosa
Red - surface ectoderm
Blue - neural tube ectoderm
Pituitary Timeline
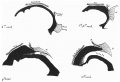
Embryonic development of pituitary stalk during week 7 and 8 based upon Streeter.[6]
- Carnegie stage 19 - Thick stalk with remnant of lumen (Rathke’s pouch); angiogenesis beginning. Capillaries appearing in mesodcrm at rostral surface of anterior lobe.
- Carnegie stage 20 - Long, slender stalk.
- Carnegie stage 21 - Thread-like stalk; beginning absorption.
- Carnegie stage 22 - Remnant of incomplete stalk at either end.
- Carnegie stage 23 - Practically no trace of stalk remains. Oriented epithelial follicles. Abundant angioblasts and capillaries in vascular component of anterior lobe.
|
| Embryonic and Fetal pituitary |
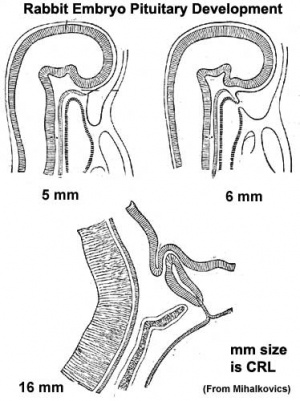
- Week 4 - hypophysial pouch, Rathke’s pouch, diverticulum from roof
- Week 5 - elongation, contacts infundibulum, diverticulum of prosencephalon
- Week 6 - connecting stalk between pouch and oral cavity degenerates
- Week 10 - growth hormone and ACTH detectable
- Week 16 - adenohypophysis fully differentiated
- Week 20 to 24 - growth hormone levels peak, then decline
Embryo Development
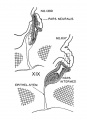
Carnegie Stage 22
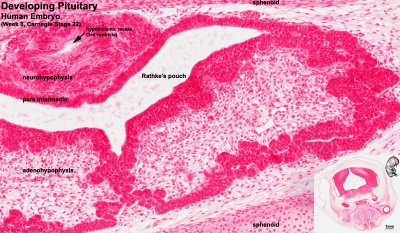
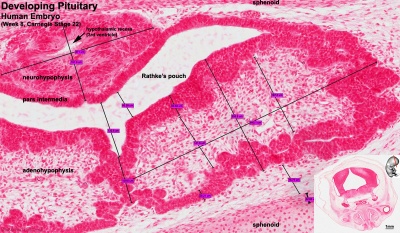
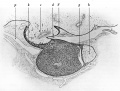
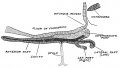
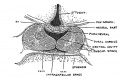
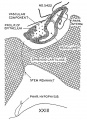
Developing Pituitary - Human embryo (Carnegie stage 22, week 8)
- Links: Image - Pituitary | Image - Pituitary Measured | Endocrine - Pituitary Development | Image - Low resolution Plane of Section | Image - Low resolution Pituitary
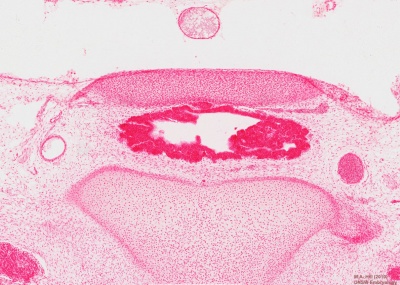
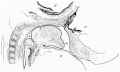
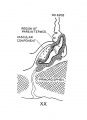
Carnegie Stage 23
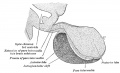
| The epithelium of the anterior lobe has become partly subdivided into lobule which project into the mesodermal component of the gland. The abundant vascular elements are well shown in figure 21.[6] | 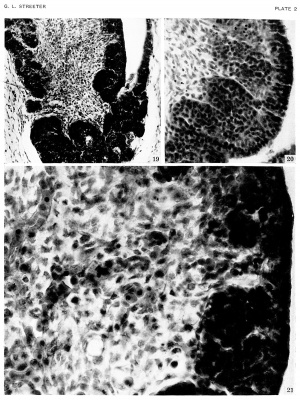
|
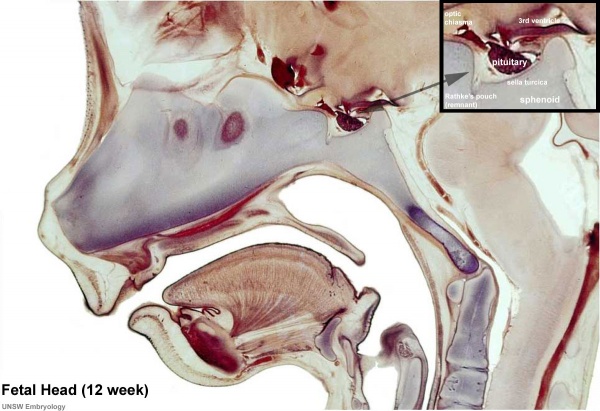
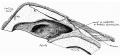
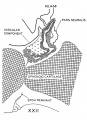
Fetal Pituitary
Human Fetal Head (Week 12) - Pituitary
Pituitary Blood Vessel Development
| Part of pituitary | Arterial supply | Systemic venous return | Portal system return |
|---|---|---|---|
| Pars anterior and pars intermedia | From carotid: a bilateral supply, usually one on each side, sometimes more than one | Systemic veins to cavernous sinus following the course of the bilateral arteries | Contribute blood to portal vessels |
| Pars tuberalis | From circle of Willis by small branches around the outside of the stalk | Small systemic veins corresponding to the arteries | Contributes blood to the portal vessels |
| Pars posterior | By branches from the bilateral supply from the carotids | By small systemic venous branches following the course ofthe arterial twigs | Contributes blood to the portal vessels by fine channels opening into them below their heavy glial sleeves |
pars distalis - vascularized by hypophysial portal vessels
A study in rats has identified the role of a known regulator of blood vessel development (Vascular Endothelial Growth Factor, VEGF) in the development of the pituitary portal vascular system.[7] "The primary capillaries extended along the developing pars tuberalis, whereas the portal vessels penetrated into the pars distalis at E15.5 (rat) and subsequently expanded into the lobe to connect with the secondary capillary plexus, emerging in the pars distalis. ....study suggests that VEGF-A (Vascular Endothelial Growth Factor A) is involved in the development of the primary capillaries and in the vascularization of the pars distalis, but not in the portal vessels since the formation of portal vessels begins at E13.5 (rat), before the appearance of VEGF-A in the rostral region of the pars distalis."
The pars distalis is vascularized by hypophysial portal vessels that arise from the capillary beds in the median eminence of the hypothalamus (Murakami et al. 1987), and this hypophyseal portal system provides an important link for carrying hormonal information from the central nervous system to the pituitary. The capillaries of the pituitary gland are characterized by richly fenestrated endothelia.
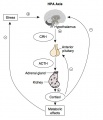
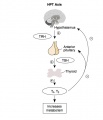
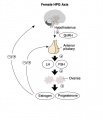
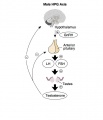
Hypothalamus Endocrine Axes
- Links: Image - HPA | Hypothalamus | Pituitary | Adrenal | Image - HPT Axis | Thyroid | Image - HPG female | Image - HPG male | Gonad
Hypothalamus
The hypothalamus is a small region located within the brain that controls many bodily functions, including eating and drinking, sexual functions and behaviors, blood pressure and heart rate, body temperature maintenance, the sleep-wake cycle, and emotional states (e.g., fear, pain, anger, and pleasure). Hypothalamic hormones play pivotal roles in the regulation of many of those functions. Because the hypothalamus is part of the central nervous system, the hypothalamic hormones actually are produced by nerve cells (i.e., neurons).
Pituitary
The anterior pituitary produces several important hormones that either stimulate target glands (e.g., the adrenal glands, gonads, or thyroid gland) to produce target gland hormones or directly affect target organs. The pituitary hormones include adreno-corticotropic hormone (ACTH); gonadotropins; thyroid-stimulating hormone (TSH), also called thyrotropin; growth hormone (GH); and prolactin.
The first three of those hormones (ACTH, gonadotropins, and TSH) act on other glands.
- ACTH stimulates the adrenal cortex to produce corticosteroid hormones (primarily cortisol) as well as small amounts of female and male sex hormones.
- Gonadotropins comprise two molecules, luteinizing hormone (LH) and follicle-stimulating hormone (FSH). These two hormones regulate the production of female and male sex hormones in the ovaries and testes as well as the production of the germ cells (oocyte and spermatozoa).
- TSH stimulates the thyroid gland to produce and release thyroid hormone.
The remaining two pituitary hormones, GH and prolactin, directly affect their target organs.
Hormones
Follicle Stimulating Hormone
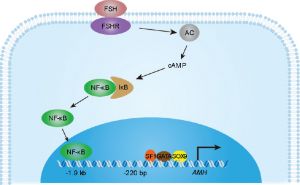
Follicle-stimulating hormone (FSH, gonadotropin) is produced in the anterior pituitary (adenohypophysis) by basophil cell gonadotropes. This glycoprotein hormone postnatally during puberty acts on the gonads (testis and ovary) to regulate fertility. FSH at the cellular level binds the membrane follicle-stimulating hormone receptor (FSH receptor, FSHR), a G protein-coupled receptor (GPCR)/seven-transmembrane domain receptor.
In females - FSH acts on the ovary to stimulate follicle development. Negative feedback by inhibin from the developing follicle decreases FSH secretion.
In males - FSH acts on the testis Sertoli cells to increase androgen-binding protein (ABP) that binds androgens and has a role in spermatogenesis.
FSH-defficiency in females results in infertile (block in folliculogenesis prior to antral follicle formation) and in males does not affect fertility (have small testes but are fertile).
FSH protein has a molecular weight 30 kDa and a 3-4 hour half-life in circulation. Gonadotrophins have been used clinically in humans for the treatment of infertility. Other glycoprotein hormones include luteinizing hormone (LH), thyroid stimulating hormone (TSH), and chorionic gonadotropin (hCG).
Luteinizing Hormone
(lutropin, lutrophin, LH) produced in the anterior pituitary (adenohypophysis) by basophil cell gonadotropes. LH at the cellular level binds the membrane luteinizing hormone receptor (LH receptor, LHR), a G protein-coupled receptor (GPCR)/seven-transmembrane domain receptor.
- testis - Leydig cell
- ovary - theca cell
Postnatal
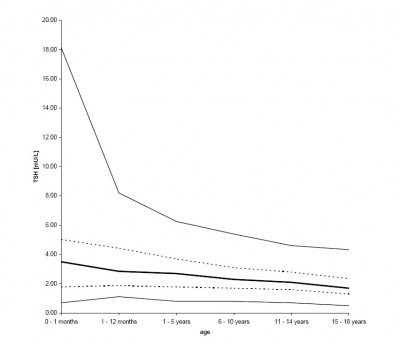
Postnatal thyrotropin (TSH) levelsPubmedParser error: The PubmedParser extension received invalid XML data. ()
- Links: Neonatal_Development | Puberty Development
Sexual Dimorphism
Quantitation of the human pituitary by magnetic resonance imaging (MRI) from childhood through puberty has identified volmetric differences between male and female pituitary sizes between 14 to 17 years of age.[9]
- Females had larger pituitary glands than males in the age 14 to 17 year old groups.
- Young (19 years and under) and old (20 years and older) females demonstrated a correlation between pituitary volume and age.
- Males did not show this relationship.
Adult Histology
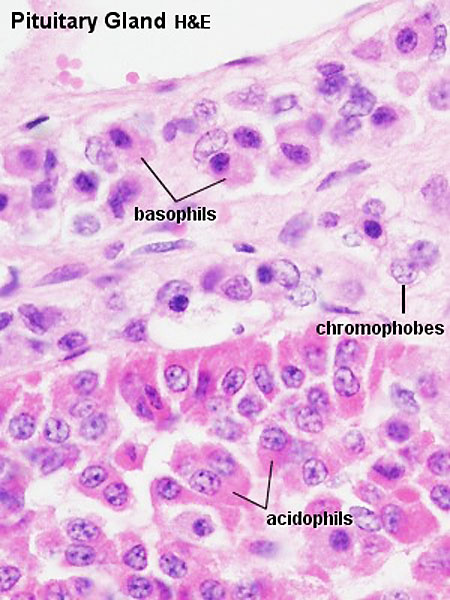
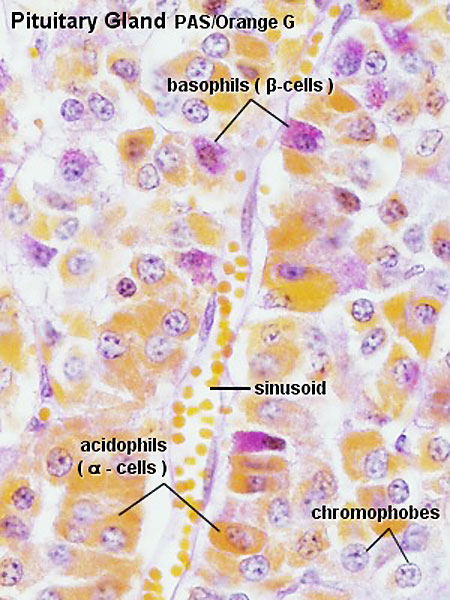
Adenohypophysis
- Acidophils - cytoplasm that stains red or orange
- polypeptide hormones: Somatotropes, produce growth hormone; Lactotropes produce prolactin)
- Basophils - cytoplasm that stains a bluish colour
- glycoprotein hormones: Thyrotropes produce thyroid stimulating hormone; Gonadotropes produce luteinizing hormone or follicle-stimulating hormone; Corticotropes produce adrenocorticotrophic hormone)
- Chromophobes - cytoplasm that stains very poorly
- acidophils or basophils that are degranulated and depleted of hormone
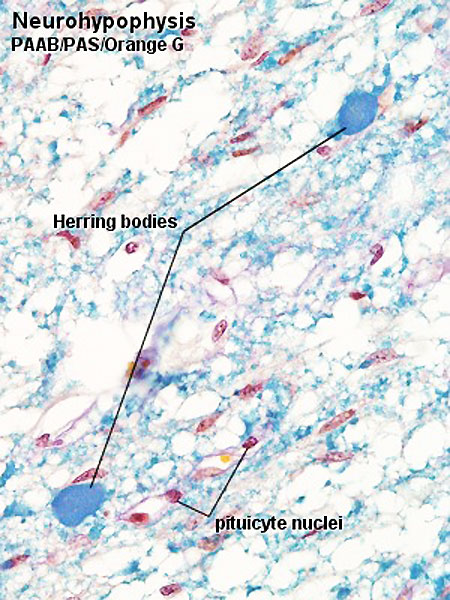
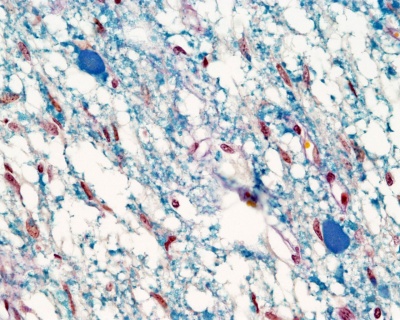
Neurohypophysis
- Infundibular process - usually referred to as posterior pituitary (unmyelinated axons from hypothalamic neurosecretory neurons, supraoptic and paraventricular hypothalamic nuclei)
- 2 other parts: median eminence and infundibular stalk
Links: Pituitary - neurohypophysis (large histology image)
Molecular
- Pituitary development: regulatory codes in mammalian organogenesis.[10] "During mammalian pituitary gland development, distinct cell types emerge from a common primordium. Appearance of specific cell types occurs in response to opposing signaling gradients that emanate from distinct organizing centers. These signals induce expression of interacting transcriptional regulators, including DNA binding-dependent activators and DNA binding-independent transrepressors, in temporally and spatially overlapping patterns. Together they synergistically regulate precursor proliferation and induction of distinct cell types. Terminal cell type differentiation requires selective gene activation strategies and long-term active repression, mediated by cell type-specific and promoter-specific recruitment of coregulatory complexes. These mechanisms imply the potential for flexibility in the ultimate identity of differentiated cell types."
- Pax6 is essential for establishing ventral-dorsal cell boundaries in pituitary gland development.[11] "The transcription factor Pax6 (paired homeodomain) has been shown to be expressed transiently in the dorsal portion of the developing pituitary before the ventral/dorsal appearance of specific cell types. Transient dorsal expression of Pax6 could establish the boundary between dorsal and ventral cell types, based on the inhibition of Shh ventral signals."
Genes
Pit1 (pituitary-specific transcription factor) is a transcription factor important for pituitary development and muations in this gene can lead to abnormalities in pituitary development and hormone production.
- PIT1 Pituitary-Specific Transcription Factor 1 - transcription factor responsible for pituitary development and hormone expression in mammals. OMIM 173110 | Gene Map Locus: 3p11 | is a pituitary-specific transcription factor responsible for pituitary development and hormone expression in mammals and is a member of the POU family of transcription factors that regulate mammalian development.
- PitX1 Paired-Like Homeodomain Transcription Factor 1 - transcription factor expressed in pituitary primordium. Member of bicoid-related vertebrate homeobox genes. OMIM 602149 | Gene Map Locus: 5q31
- PitX2 Paired-Like Homeodomain Transcription Factor 2 - transcription factor expressed in pituitary primordium and other anterior structures, including the eye Member of bicoid-related vertebrate homeobox genes. OMIM 601542 | Gene Map Locus: 4q25-q26
- TPIT T-box transcription factor Pituitary OMIM 604614 | Gene Map Locus: 1q23-q24
- VEGF Vascular Endothelial Growth Factor - mitogen growth factor for vascular endothelial cells. Role in pituitary vascular development. OMIM 192240 | Gene Map Locus: 1q23-q24
Abnormalities
Anatomical abnormalities asssociated with the Rathke's pouch include a craniopharyngeal canal, from the anterior part of the fossa hypophyseos of the sphenoid bone to the under surface of the skull. The stomodeal end may also be present at the junction of the septum of the nose with the palate.
Abnormal functional development of the pituitary can lead to a wide range of other organ diseases due to the effect of hormones released from the pituitary on many other endocrine and non-endocrine organs (For example: dwarfism, hypothyroidism). (More? NIH Genes and Disease Chapter 41 - Endocrine)
Pituitary Duplication
Occurring as either a complete or partial duplication, this is an extremely rare abnormality with poor neonatal survival due to associated abnormalities.[12] Recently a heterozygous deletion of chromosome 14, including the thyroid transcription factor-1 gene, has been identified with duplication of the pituitary stalk.[13]
Pituitary Adenoma Classification
There are several abnormalities associated with abnormal levels of the hormonal output of the pituitary due to the development of pituitary tumours (adenomas).
Growth hormone (GH) adenoma
Growth hormone (GH) adenomas, which are benign pituitary tumors lead to chronic high GH output levels, that may lead to acromegaly in adults and gigantism in children.
Acromegaly (Greek, acro = "extremities"; megaly = "enlargement") is a clinical term for a hormonal disorder that results from excess growth hormone (GH) in the body. The pituitary produces excessive amounts of growth hormone, usually due to benign, or noncancerous, pituitary tumours (adenomas). Childhood adenomas lead to gigantism rather than acromegaly, due to continued and excess growth in the still unfused growth plates in the long bones of the legs.
- Links: NIH - acromegaly
Adrenocorticotropic (ACTH) adenoma
Cushing's disease caused either by a pituitary adenoma produces excess adrenocorticotropic hormone (ACTH, corticotropin) or due to ectopic tumors secreting ACTH or corticotropin-releasing hormone (CRH). Classification can be applied using specific criteria (clinical presentation, biochemical data, histology of growth pattern, tinctorial characteristics, proliferative activity, immunohistology marker expression, ultrastructure and molecular biology). The current classification used is the World Health Organization classification of 2000 recently updated in 2004.
- Links: NIH - cushings
Syndromic Hypopituitarism
Combined Pituitary Hormone Deficiency
TBX19 and ACTH Deficiency
References
- ↑ Zhang S, Cui Y, Ma X, Yong J, Yan L, Yang M, Ren J, Tang F, Wen L & Qiao J. (2020). Single-cell transcriptomics identifies divergent developmental lineage trajectories during human pituitary development. Nat Commun , 11, 5275. PMID: 33077725 DOI.
- ↑ Osmundsen AM, Keisler JL, Taketo MM & Davis SW. (2017). Canonical WNT Signaling Regulates the Pituitary Organizer and Pituitary Gland Formation. Endocrinology , 158, 3339-3353. PMID: 28938441 DOI.
- ↑ Davis SW, Mortensen AH, Keisler JL, Zacharias AL, Gage PJ, Yamamura K & Camper SA. (2016). β-catenin is required in the neural crest and mesencephalon for pituitary gland organogenesis. BMC Dev. Biol. , 16, 16. PMID: 27184910 DOI.
- ↑ Liu F, Pogoda HM, Pearson CA, Ohyama K, Löhr H, Hammerschmidt M & Placzek M. (2013). Direct and indirect roles of Fgf3 and Fgf10 in innervation and vascularisation of the vertebrate hypothalamic neurohypophysis. Development , 140, 1111-22. PMID: 23404108 DOI.
- ↑ Kelberman D, Rizzoti K, Lovell-Badge R, Robinson IC & Dattani MT. (2009). Genetic regulation of pituitary gland development in human and mouse. Endocr. Rev. , 30, 790-829. PMID: 19837867 DOI.
- ↑ 6.0 6.1 Streeter GL. Developmental Horizons In Human Embryos Description Or Age Groups XIX, XX, XXI, XXII, And XXIII, Being The Fifth Issue Of A Survey Of The Carnegie Collection. (1957) Carnegie Instn. Wash. Publ. 611, Contrib. Embryol., 36: 167-196.
- ↑ Nakakura T, Yoshida M, Dohra H, Suzuki M & Tanaka S. (2006). Gene expression of vascular endothelial growth factor-A in the pituitary during formation of the vascular system in the hypothalamic-pituitary axis of the rat. Cell Tissue Res. , 324, 87-95. PMID: 16411082 DOI.
- ↑ Xu HY, Zhang HX, Xiao Z, Qiao J & Li R. (2019). Regulation of anti-Müllerian hormone (AMH) in males and the associations of serum AMH with the disorders of male fertility. Asian J. Androl. , 21, 109-114. PMID: 30381580 DOI.
- ↑ MacMaster FP, Keshavan M, Mirza Y, Carrey N, Upadhyaya AR, El-Sheikh R, Buhagiar CJ, Taormina SP, Boyd C, Lynch M, Rose M, Ivey J, Moore GJ & Rosenberg DR. (2007). Development and sexual dimorphism of the pituitary gland. Life Sci. , 80, 940-4. PMID: 17174342 DOI.
- ↑ Scully KM & Rosenfeld MG. (2002). Pituitary development: regulatory codes in mammalian organogenesis. Science , 295, 2231-5. PMID: 11910101 DOI.
- ↑ Kioussi C, O'Connell S, St-Onge L, Treier M, Gleiberman AS, Gruss P & Rosenfeld MG. (1999). Pax6 is essential for establishing ventral-dorsal cell boundaries in pituitary gland development. Proc. Natl. Acad. Sci. U.S.A. , 96, 14378-82. PMID: 10588713
- ↑ Kollias SS, Ball WS & Prenger EC. (1995). Review of the embryologic development of the pituitary gland and report of a case of hypophyseal duplication detected by MRI. Neuroradiology , 37, 3-12. PMID: 7708185
- ↑ Accornero S, Danesino C, Bastianello S, D'Errico I, Guala A & Chiovato L. (2010). Duplication of the pituitary stalk in a patient with a heterozygous deletion of chromosome 14 harboring the thyroid transcription factor-1 gene. J. Clin. Endocrinol. Metab. , 95, 3595-6. PMID: 20685887 DOI.
Journals
Pituitary The Official Journal of the Pituitary Society - Pituitary is an international publication devoted to basic and clinical aspects of the pituitary gland. It is designed to publish original, high quality research in both basic and pituitary function as well as clinical pituitary disease.
Online Textbooks
Endocrinology: An Integrated Approach Nussey, S.S. and Whitehead, S.A. Oxford, UK: BIOS Scientific Publishers, Ltd; 2001. table of Contents | The pituitary gland | Embryology of the pituitary gland | Blood supply of the hypothalamo-pituitary axis
NIH Genes & Disease Chapter 41 - Endocrine
Developmental Biology (6th ed) Gilbert, Scott F. Sunderland (MA): Sinauer Associates, Inc.; c2000.
Stages along the hypothalamus-pituitary-thyroid axis of salamanders
Molecular Biology of the Cell (4th Edn) Alberts, Bruce; Johnson, Alexander; Lewis, Julian; Raff, Martin; Roberts, Keith; Walter, Peter. New York: Garland Publishing; 2002. table 15-1. Some Hormone-induced Cell Responses Mediated by Cyclic AMP | Alternative processing pathways for the prohormone proopiomelanocortin
Clinical Methods (3rd Edn) Walker, H.K.; Hall, W.D.; Hurst, J.W.; editors Stoneham (MA). Tests of Pituitary or Target Gland Dysfunction | Pituitary Tumors
Health Services/Technology Assessment Text (HSTAT) Bethesda (MD): National Library of Medicine (US), 2003 Oct. Pituitary Gland search Results
Search NLM Online Textbooks- "pituitary development" : Endocrinology | Molecular Biology of the Cell | The Cell- A molecular Approach
Reviews
Alatzoglou KS, Gregory LC & Dattani MT. (2020). Development of the Pituitary Gland. Compr Physiol , 10, 389-413. PMID: 32163208 DOI.
Musumeci G, Castorina S, Castrogiovanni P, Loreto C, Leonardi R, Aiello FC, Magro G & Imbesi R. (2015). A journey through the pituitary gland: Development, structure and function, with emphasis on embryo-foetal and later development. Acta Histochem. , 117, 355-66. PMID: 25858531 DOI.
Kelberman D, Rizzoti K, Lovell-Badge R, Robinson IC & Dattani MT. (2009). Genetic regulation of pituitary gland development in human and mouse. Endocr. Rev. , 30, 790-829. PMID: 19837867 DOI.
PubmedParser error: Invalid PMID, please check. (PMID: 17638086z)
Rosenfeld MG, Briata P, Dasen J, Gleiberman AS, Kioussi C, Lin C, O'Connell SM, Ryan A, Szeto DP & Treier M. (2000). Multistep signaling and transcriptional requirements for pituitary organogenesis in vivo. Recent Prog. Horm. Res. , 55, 1-13; discussion 13-4. PMID: 11036930
Sheng HZ & Westphal H. (1999). Early steps in pituitary organogenesis. Trends Genet. , 15, 236-40. PMID: 10354584
Kioussi C, Carrière C & Rosenfeld MG. (1999). A model for the development of the hypothalamic-pituitary axis: transcribing the hypophysis. Mech. Dev. , 81, 23-35. PMID: 10330482
Articles
Nakakura T, Yoshida M, Dohra H, Suzuki M & Tanaka S. (2006). Gene expression of vascular endothelial growth factor-A in the pituitary during formation of the vascular system in the hypothalamic-pituitary axis of the rat. Cell Tissue Res. , 324, 87-95. PMID: 16411082 DOI.
Kioussi C, O'Connell S, St-Onge L, Treier M, Gleiberman AS, Gruss P & Rosenfeld MG. (1999). Pax6 is essential for establishing ventral-dorsal cell boundaries in pituitary gland development. Proc. Natl. Acad. Sci. U.S.A. , 96, 14378-82. PMID: 10588713
Treier M, O'Connell S, Gleiberman A, Price J, Szeto DP, Burgess R, Chuang PT, McMahon AP & Rosenfeld MG. (2001). Hedgehog signaling is required for pituitary gland development. Development , 128, 377-86. PMID: 11152636
Search PubMed
Search April 2010
- Endocrine Development - All (14277) Review (4620) Free Full Text (3140)
Search Pubmed: pituitary development
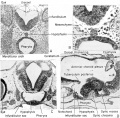
Additional Images
Historic Images
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Herring PT. The development of the mammalian pituitary and its morphological significance. (1908) Quar. Jour. Ex. Physiol. 1: 161-185.
Frazer JE. Pharyngeal end of Rathke's pouch. (1911) J Anat. 45: 190-196. PMID 17232879
Streeter GL. Developmental Horizons In Human Embryos Description Or Age Groups XIX, XX, XXI, XXII, And XXIII, Being The Fifth Issue Of A Survey Of The Carnegie Collection. (1957) Carnegie Instn. Wash. Publ. 611, Contrib. Embryol., 36: 167-196.
Terms
- adenohypophyseal placode - Specialised cranial surface ectoderm placode region that will form the endocrine anterior pituitary (adenohypophysis).
- adenohypophysis - (anterior pituitary, pars anterior) The anterior part of the pituitary, which develops in the early embryo from the surface ectoderm adenohypophyseal placode. This placode will fold inward on the roof of the pharynx forming a transient structure Rathke's pouch, that looses its connection with the surface.
- anterior pituitary - (adenohypophysis, pars anterior, pars distalis)
- neurohypophysis - (posterior pituitary)
- pars tuberalis - (pars tuberalis of the hypophysis) anatomically is the region of anterior pituitary (adenohypophysis) extending along the anterior and lateral surfaces of the hypophyseal stalk. Tuberalis principal cells are low columnar, with cytoplasm containing lipid droplets, glycogen granules, and some colloid droplets.
- posterior pituitary - (neurohypophysis)
- Rathke's pouch - An ectodermal fold in roof of pharynx forming anterior pituitary (adenohypophysis) and pars intermedia. Named after German embryologist and anatomist Martin Heinrich Rathke (1793 -1860). (More? Martin Heinrich Rathke)
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- Garvan Institute Pituitary Research
- Martin Heinrich Rathke
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Haziran 9) Embryology Endocrine - Pituitary Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Endocrine_-_Pituitary_Development
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G