Introduction
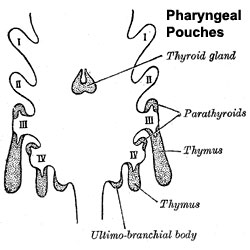
Embryonic origins of the endocrine organs of the neck
The parathyroid gland appears in the adult as a pair of inferior and a pair of superior "bumps" on the beside the (dorsal) thyroid (hence the name, "para"). The embryonic origin of this gland is from the endoderm of the third and fourth pharyngeal pouches, and could also have ectoderm and neural crest contributions. This developmental process also generates multiple small parathyroid clusters in addition to the main parathyroid glands.[1]
At 6 weeks a diverticulum elongates from the pouch, initially hollow and then solidifynig with cell proliferation.
Interestingly, the inferior parathyroid originates from the third pharyngeal pouch and the superior arises from the fourth pharyngeal pouch, the adult anatomical position is the opposite of the pharyngeal rostro-caudal order. This occurs due to the third pharyngeal pouch also giving rise to the thymus, the superior pair descend along with the thymus.
Parathyroid hormone (PTH) regulates calcium and phosphate levels in conjunction with parafollicular cells of the thyroid gland (calcitonin) and Vitamin D (dietary or synthesized in the skin).
The fetal parathyroids appear functional as they respond to calcium levels. The fetal calcium levels also higher than maternal levels.
Historically, see also the 1938 paper on the fate of the ultimobranchial body within the human thyroid gland.[2]
Some Recent Findings
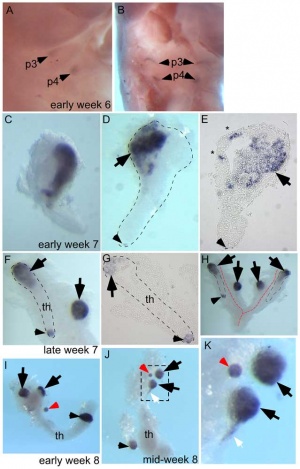
Human Embryo (week 6 - 8)
[1]
- Hox genes in the pharyngeal region: how Hoxa3 controls early embryonic development of the pharyngeal organs[3] "The pharyngeal organs, namely the thyroid, thymus, parathyroids, and ultimobranchial bodies, derive from the pharyngeal endoderm during embryonic development. The pharyngeal region is a segmented structure comprised of a series of reiterated structures: the pharyngeal arches on the exterior surface, the pharyngeal pouches on the interior, and a mesenchymal core. It is well known that Hox genes control spatial identity along the anterior-posterior axis of the developing vertebrate embryo, and nowhere is this is more evident than in the pharyngeal region. Each of the distinct segmented regions has a unique pattern of Hox expression, which conveys crucial positional information to the cells and tissues within it. In the context of pharyngeal organ development, molecular data suggest that HOXA3 is responsible for specifying organ identity within the third pharyngeal pouch, and in its absence, thymus and parathyroid organogenesis fails to proceed normally"
- Tissue-specific roles for sonic hedgehog signaling in establishing thymus and parathyroid organ fate[4] "The thymus and parathyroids develop from third pharyngeal pouch (3rd pp) endoderm. Our previous studies show that Shh null mice have smaller, aparathyroid primordia in which thymus fate specification extends into the pharynx. SHH signaling is active in both dorsal pouch endoderm and neighboring neural crest (NC) mesenchyme. It is unclear which target tissue of SHH signaling is required for the patterning defects in Shh mutants. Here, we used a genetic approach to ectopically activate or delete the SHH signal transducer Smo in either pp endoderm or NC mesenchyme. Although no manipulation recapitulated the Shh null phenotype, manipulation of SHH signaling in either the endoderm or NC mesenchyme had direct and indirect effects on both cell types during fate specification and organogenesis. SHH pathway activation throughout pouch endoderm activated ectopic Tbx1 expression and partially suppressed the thymus-specific transcription factor Foxn1, identifying Tbx1 as a key target of SHH signaling in the 3rd pp. However, ectopic SHH signaling was insufficient to expand the GCM2-positive parathyroid domain, indicating that multiple inputs, some of which might be independent of SHH signaling, are required for parathyroid fate specification. These data support a model in which SHH signaling plays both positive and negative roles in patterning and organogenesis of the thymus and parathyroids."
- Notch and Hedgehog in the thymus/parathyroid common primordium: Crosstalk in organ formation[5] "The avian thymus and parathyroids (T/PT) common primordium derives from the endoderm of the third and fourth pharyngeal pouches (3/4PP). The molecular mechanisms that govern T/PT development are not fully understood. Here we study the effects of Notch and Hedgehog (Hh) signalling modulation during common primordium development using in vitro, in vivo and in ovo approaches. The impairment of Notch activity reduced Foxn1/thymus-fated and Gcm2/Pth/parathyroid-fated domains in the 3/4PP and further compromised the development of the parathyroid glands. When Hh signalling was abolished, we observed a reduction in the Gata3/Gcm2- and Lfng-expression domains at the median/anterior and median/posterior territories of the pouches, respectively. In contrast, the Foxn1 expression-domain at the dorsal tip of the pouches expanded ventrally into the Lfng-expression domain. This study offers novel evidence on the role of Notch signalling in T/PT common primordium development, in an Hh-dependent manner."
|
| Older papers
|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
- Multiple roles for HOXA3 in regulating thymus and parathyroid differentiation and morphogenesis in mouse[6] "Previous studies have shown that the Hoxa3 null mutant lacks third pharyngeal pouch derivatives, the thymus and parathyroids by E18.5, and organ-specific markers are absent or downregulated during initial organogenesis. Our current analysis of the Hoxa3 null mutant shows that organ-specific domains did undergo initial patterning, but the location and timing of key regional markers within the pouch, including Tbx1, Bmp4 and Fgf8, were altered. Expression of the parathyroid marker Gcm2 was initiated but was quickly downregulated and differentiation failed; by contrast, thymus markers were delayed but achieved normal levels, concurrent with complete loss through apoptosis. To determine the cell type-specific roles of Hoxa3 in third pharyngeal pouch development, we analyzed tissue-specific mutants using endoderm and/or neural crest cell (NCC)-specific Cre drivers. Simultaneous deletion with both drivers resulted in athymia at E18.5, similar to the null. By contrast, the individual tissue-specific Hoxa3 deletions resulted in small, ectopic thymi, although each had a unique phenotype. Hoxa3 was primarily required in NCCs for morphogenesis. In endoderm, Hoxa3 temporally regulated initiation of the thymus program and was required in a cell-autonomous manner for parathyroid differentiation. Furthermore, Hoxa3 was required for survival of third pharyngeal pouch-derived organs, but expression in either tissue was sufficient for this function." HOX
- Thymus-associated parathyroid hormone has two cellular origins with distinct endocrine and immunological functions[1] "In mammals, parathyroid hormone (PTH) is a key regulator of extracellular calcium and inorganic phosphorus homeostasis. Although the parathyroid glands were thought to be the only source of PTH, extra-parathyroid PTH production in the thymus, which shares a common origin with parathyroids during organogenesis, has been proposed to provide an auxiliary source of PTH, resulting in a higher than expected survival rate for aparathyroid Gcm2⁻/⁻ mutants. However, the developmental ontogeny and cellular identity of these "thymic" PTH-expressing cells is unknown. ...Our data show conclusively that the thymus does not serve as an auxiliary source of either serum PTH or parathyroid function. We further show that the normal process of parathyroid organogenesis in both mice and humans leads to the generation of multiple small parathyroid clusters in addition to the main parathyroid glands, that are the likely source of physiologically relevant "thymic PTH."" thymus
|
Development Overview
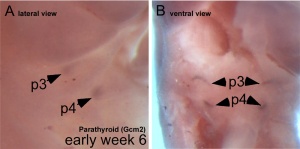
Parathyroid primordia (week 6 GA)
[1]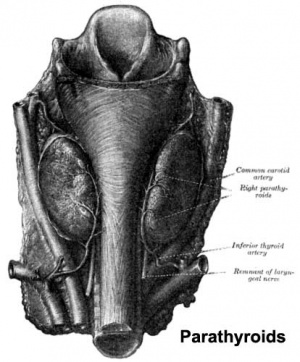
Adult parathyroid anatomy
- Endoderm - third and fourth pharyngeal pouches, could also have ectoderm and neural crest
- 3rd Pharyngeal Pouch - inferior parathyroid, initially descends with thymus
- 4th Pharyngeal Pouch - superior parathyroid
- Week 6 - diverticulum elongate, hollow then solid, dorsal cell proliferation
- Fetal parathyroids - respond to calcium levels, fetal calcium levels higher than maternal
Parathyroid Hormone
(PTH, parathormone or parathyrin) A polypeptide (84 amino acids) hormone which increases the concentration of calcium ions in the blood. Its actions oppose the hormone calcitonin from the parafollicular cells (C cells) of the thyroid gland, which decrease calcium. Acts through the parathyroid hormone receptor in bone, kidney and gastrointestinal tract.
- stimulate osteoclasts - degrade bone matrix, releasing calcium
- increase calcium gastrointestinal tract absorption
Parathyroid Hormone-related Protein
(PTHrP) Originally identified in the clinical syndrome humoral hypercalcemia of malignancy. It's developmental role is that of a regulatory protein expressed during the formation of many organs.
- mammary gland development - epithelial-mesenchymal interactions[7]
- chondrocyte differentiation[8]
Molecular
Sonic Hedgehog
Signalling by SHH has been shown to restrict the expression of Gcm2 and controls the position of the developing parathyroids.[9]
Glial Cells Missing
The transcription factor, Glial Cells Missing, Drosophila, Homolog Of, 2 (GCM2) is a key to parathyroid molecular development.[10][11]
- Links: OMIM - GCM2
Semaphorin3d
Semaphorin3d (Sema3d) is a secreted glycoprotein expressed in the developing parathyroid gland in mice. Deletion of Sema3d leads to parathyroid hyperplasia, causing PHPT. Therefore Sema3d may act as as a negative regulator of parathyroid growth.[12]
Human SEMA3D 7q21.11
- Links: OMIM - SEMA3D
Abnormalities
Hyperparathyroidism
Postnatal
- Postnatal adult ageing increased parathyroid hormone plasma levels are associated with cognitive decline and dementia.
- Parathyroid carcinoma (cancer) is a rare malignancy, occurring with an incidence of 0.5 to 4% of all cases of primary hyperparathyroidism.
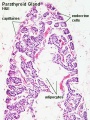
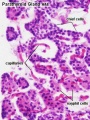
Adult Histology
Parathyroid labeled (low power)
Parathyroid labeled (high power)
References
- ↑ 1.0 1.1 1.2 1.3 Liu Z, Farley A, Chen L, Kirby BJ, Kovacs CS, Blackburn CC & Manley NR. (2010). Thymus-associated parathyroid hormone has two cellular origins with distinct endocrine and immunological functions. PLoS Genet. , 6, e1001251. PMID: 21203493 DOI.
- ↑ Kingsbury BF. On the fate of the ultimobranchial body within the human thyroid gland. (1935) Anat. Rec. 61(2): 155–173.
- ↑ Gordon J. (2018). Hox genes in the pharyngeal region: how Hoxa3 controls early embryonic development of the pharyngeal organs. Int. J. Dev. Biol. , 62, 775-783. PMID: 30604847 DOI.
- ↑ Bain VE, Gordon J, O'Neil JD, Ramos I, Richie ER & Manley NR. (2016). Tissue-specific roles for sonic hedgehog signaling in establishing thymus and parathyroid organ fate. Development , 143, 4027-4037. PMID: 27633995 DOI.
- ↑ Figueiredo M, Silva JC, Santos AS, Proa V, Alcobia I, Zilhão R, Cidadão A & Neves H. (2016). Notch and Hedgehog in the thymus/parathyroid common primordium: Crosstalk in organ formation. Dev. Biol. , 418, 268-82. PMID: 27544844 DOI.
- ↑ Chojnowski JL, Masuda K, Trau HA, Thomas K, Capecchi M & Manley NR. (2014). Multiple roles for HOXA3 in regulating thymus and parathyroid differentiation and morphogenesis in mouse. Development , 141, 3697-708. PMID: 25249461 DOI.
- ↑ Dunbar ME & Wysolmerski JJ. (1999). Parathyroid hormone-related protein: a developmental regulatory molecule necessary for mammary gland development. J Mammary Gland Biol Neoplasia , 4, 21-34. PMID: 10219904
- ↑ Alman BA & Wunder JS. (2008). Parathyroid hormone-related protein regulates glioma-associated oncogene transcriptional activation: lessons learned from bone development and cartilage neoplasia. Ann. N. Y. Acad. Sci. , 1144, 36-41. PMID: 19076361 DOI.
- ↑ Grevellec A, Graham A & Tucker AS. (2011). Shh signalling restricts the expression of Gcm2 and controls the position of the developing parathyroids. Dev. Biol. , 353, 194-205. PMID: 21349263 DOI.
- ↑ Liu Z, Yu S & Manley NR. (2007). Gcm2 is required for the differentiation and survival of parathyroid precursor cells in the parathyroid/thymus primordia. Dev. Biol. , 305, 333-46. PMID: 17382312 DOI.
- ↑ Peissig K, Condie BG & Manley NR. (2018). Embryology of the Parathyroid Glands. Endocrinol. Metab. Clin. North Am. , 47, 733-742. PMID: 30390809 DOI.
- ↑ Singh A, Mia MM, Cibi DM, Arya AK, Bhadada SK & Singh MK. (2019). Deficiency in the secreted protein Semaphorin3d causes abnormal parathyroid development in mice. J. Biol. Chem. , 294, 8336-8347. PMID: 30979723 DOI.
Reviews
Peissig K, Condie BG & Manley NR. (2018). Embryology of the Parathyroid Glands. Endocrinol. Metab. Clin. North Am. , 47, 733-742. PMID: 30390809 DOI.
Zajac JD & Danks JA. (2008). The development of the parathyroid gland: from fish to human. Curr. Opin. Nephrol. Hypertens. , 17, 353-6. PMID: 18660669 DOI.
Parvari R, Diaz GA & Hershkovitz E. (2007). Parathyroid development and the role of tubulin chaperone E. Horm. Res. , 67, 12-21. PMID: 17008776 DOI.
Jüppner H. (2000). Role of parathyroid hormone-related peptide and Indian hedgehog in skeletal development. Pediatr. Nephrol. , 14, 606-11. PMID: 10912527
Articles
Sharma SG, Levine SN, Yatavelli RK, Shaha MA & Nathan CAO. (2020). Parathyroidectomy in First Trimester of Pregnancy. J Endocr Soc , 4, bvaa015. PMID: 32133431 DOI.
Singh A, Mia MM, Cibi DM, Arya AK, Bhadada SK & Singh MK. (2019). Deficiency in the secreted protein Semaphorin3d causes abnormal parathyroid development in mice. J. Biol. Chem. , 294, 8336-8347. PMID: 30979723 DOI.
Liu Z, Farley A, Chen L, Kirby BJ, Kovacs CS, Blackburn CC & Manley NR. (2010). Thymus-associated parathyroid hormone has two cellular origins with distinct endocrine and immunological functions. PLoS Genet. , 6, e1001251. PMID: 21203493 DOI.
Liu Z, Yu S & Manley NR. (2007). Gcm2 is required for the differentiation and survival of parathyroid precursor cells in the parathyroid/thymus primordia. Dev. Biol. , 305, 333-46. PMID: 17382312 DOI.
Mansberger AR & Wei JP. (1993). Surgical embryology and anatomy of the thyroid and parathyroid glands. Surg. Clin. North Am. , 73, 727-46. PMID: 8378818 DOI.
Search PubMed
Search April 2010
- Parathyroid Development - All (3523) Review (768) Free Full Text (741)
Search Pubmed: parathyroid development
Additional Images
Terms
- parathyroid hormone - (PTH, parathormone or parathyrin) A polypeptide (84 amino acids) hormone secreted by the parathyroid gland, which increases the concentration of calcium ions in the blood. Its actions oppose the hormone calcitonin from the thyroid gland parafollicular cells (C cells), which decrease calcium. Acts through the parathyroid hormone receptor located mainly in bone, kidney and gastrointestinal tract. Hormone dual role is to: stimulate osteoclasts in bone to degrade bone matrix releasing calcium; increase gastrointestinal tract absorption of calcium.
| Endocrine Terms (expand to view)
|
- adenohypophysis - (anterior pituitary, pars distalis) anterior part of pituitary embryonic development from surface ectoderm adenohypophyseal placode. Placode folds inward on the roof of the pharynx forming a transient structure Rathke's pouch.
- adrenocorticotropin - (ACTH or corticotropin) anterior pituitary, peptide hormone stimulates the adrenal cortex to produce corticosteroid hormones — primarily cortisol — as well as small amounts of female and male sex hormones.
- androstenedione - hormone precursor of testosterone and other steroidal androgens.
- atrial natriuretic peptide - (ANP) heart, peptide hormone regulates blood pressure. A study suggests that its activating enzyme corin, and ANP together, have a role in placentation, by promoting trophoblast invasion and spiral artery remodelling. (PMID 22437503)
- basophil cell - pituitary named by histological staining (deep blue, purple) different types produce different hormones: corticotrophs (ACTH, CRH), gonadotrophs (FSH, LH, GnRH), and thyrotrophs (TSH, TRH). See acidophil and chromophore cells.
- C cells - parafollicular cells of the thyroid.
- calcitonin - (CT) C cells of thyroid, peptide hormone thyroid
- corpus luteum - ovarian endocrine organ from ovulating follicle, stimulated by hCG and supports early pregnancy by secreting progesterone, 17β-progesterone, estradiol and androstenedione.
- corticosteroid binding globulin - (CBG) binds and transports glucocorticoids in the plasma. Globin is synthesised in the liver. adrenal
- dihydrotestosterone - (DHT) steroidal hormone made locally by 5-alpha reductase conversion of testosterone into a more active form in genital effects.
- dehydroepiandrosterone - (DHEA, androstenolone) adrenal cortex, gonads and brain make this steroid intermediate that may also have adult hormonal functions.
- dehydroepiandrosterone sulphate - (DHEAS, DHEA-S) fetal adrenal cortex makes this inactive precursor of a steroid hormone.
- dydrogesterone - clinical oral retrosteroid structurally related to progesterone, with a greater bioavailability and selectivity for the progesterone receptor.
- estrogen (oestrogen) family of female steroidal hormones - estrone (E1), estradiol (E2), estriol (E3), and estetrol (E4) synthesised from testosterone and androstenedione, by aromatase. Also produced in male testis, and required for genital development (PMID 29438493)
- estrone (E1) - steroid hormone with weak estrogenic activity.
- estradiol (E2) - (oestradiol) estrogen steroid hormone with main estrogenic female activity.
- estriol (E3) - (oestriol) steroid hormone with weak estrogenic activity.
- estetrol (E4) (oestetrol) steroid hormone with weak estrogenic activity produced only during pregnancy.
- follicle stimulating hormone - (FSH) pituitary glycoprotein hormone secreted by gonadotrophs (basophilic cell subgroup) acts on gametogenesis and other systems in both males and females. Females, acts on the ovary to stimulate follicle development. Negative feedback by inhibin from the developing follicle decreases FSH secretion. Males, acts on the testis Sertoli cells to increase androgen-binding protein (ABP) that binds androgens and has a role in spermatogenesis.
- growth hormone - (GH) pituitary, peptide hormone that stimulates tissue and skeletal growth. In the ovary, growth hormone also increases granulosa cell FSH-dependent E2 production.
- growth hormone releasing hormone - (GHRH) hypothalamus, protein that activates growth hormone synthesis and release from the anterior pituitary.
- human chorionic gonadotropin - (hCG) glycoprotein hormone with 2 subunits (alpha and beta joined non covalently). Similar in structure to luteinizing hormone (LH), hCG exists in multiple hormonal and non-endocrine agents (regular hCG, hyperglycosylated hCG and the free beta-subunit of hyperglycosylated hCG). PMID 19171054
- human chorionic somatommotropin - (hCS, CSH, placental lactogen) Placental hormone is structurally similar to both growth hormone (GH) and prolactin (PRL} and binds strongly to PRL receptors but weakly to GH receptors. Role in stimulating maternal mammary gland development. endocrine placenta
- interstitial cell - (Leydig cell) Male testis cell secrete the androgen testosterone, required for fetal male genital tract differentiation and masculinisation after puberty.
- Leydig cell - (interstitial cell) Male testis cell secrete the androgen testosterone, beginning in the fetus. These cells are named after Franz von Leydig (1821 - 1908) a German scientist who histologically described these cells.
- lutenizing hormone - (LH, gonadotropin, lutropin, Interstitial Cell Stimulating Hormone, ICSH) pituitary, glycoprotein hormone acts on the gonad and has a role in male and female reproduction. Female, increase in concentration during the menstrual cycle triggers ovulation. Male, stimulates testis interstital cell production of testosterone. Gonadotrophins have been used clinically in humans for the treatment of female infertility.
- melaocyte stimulating hormone - (MSH) pituitary, peptide hormone pituitary
- melatonin - (N-acetyl-5-methoxytryptamine) pineal amino acid amino (precursor tryptophan) hormone involved with the diurnal cycle, melatoinin levels are high in dark, low in daylight. Also acts as an antioxidant, free radical scavenger, and anti-inflammatory molecule.
- prolactin - (PRL) pituitary, peptide hormone pituitary
- parathyroid - endocrine gland through parathyroid hormone (PTH) regulates calcium and phosphate levels in conjunction with parafollicular cells of the thyroid gland (calcitonin) and Vitamin D, dietary or synthesized in the skin. Develops from pharyngeal endoderm, in this case the 3rd and 4th pharyngeal pouches.
- parathyroid hormone - (PTH) parathyroid, peptide hormone parathyroid
- synthetic ACTH test = (synacthen test) A diagnostic test to both measure the amount of cortisol in the body and to determine the ability to produce cortisol.
- testosterone - testis ovary steroidal hormone. In males is the androgen which regulates genital (gonadal and tract), secondary sex characteristics and neural development. The steroid is converted to the active metabolite dihydrotestosterone (DHT) by the enzyme 5-alpha reductase for the genital effects and estradiol by the enzyme aromatase for the neural effects.
- thyroid - endocrine gland located in the neck with a developmental role in neurological development and metabolism.
- thyroid diverticulum - the primordium of the thyroid gland, beginning as an median endodermal thickening in the floor of pharynx between the pharyngeal pouch 1 and 2.
- thyroid hormone - (TH) thyroid amino acid derivative with two main forms (T3, T4) regulates tissue metabolic activity.
- thyroid stimulating hormone - (TSH) pituitary protein hormone
- ultimobranchial body - historic term for the embryonic structure that forms the parafollicular cells (C cells) of the thyroid.
|
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 21) Embryology Endocrine - Parathyroid Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Endocrine_-_Parathyroid_Development
- What Links Here?
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G