Introduction
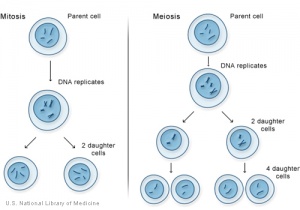
Normal cell division in all cells, except germ cells, occurs by 2 mechanical processes that initially divide the nucleus then the cell cytoplasm. This process produces two (daughter) cells that should be genetically identical to the parent cell. Note that DNA duplication (replication) occurs during interphase (S phase), before mitosis and not during mitosis.
Germ cells, oocyte and spermatozoa, undergo meiotic cell division.
- Mitosis segregation of chromosomes and formation of 2 nuclei
- Cytokinesis splitting of the cell as a whole into 2 daughter cells
- Recent Nobel Prizes- 2001 Cell Cycle, 2002 Cell Death
| Mitosis of the single zygote produces how many cells in the adult?
|
| Not a straightforward calculation! Cells through development and in the adult divide and die influenced by cell type, genetics and environment. In the adult, most tissues though would have a relatively contestant balance between loss and gain in cell numbers. A recent paper has attempted to estimate the number for humans[1] "In particular, the reported total cell number of a human being ranges between 1012 and 1016 and it is widely mentioned without a proper reference. ...A current estimation of human total cell number calculated for a variety of organs and cell types is presented. These partial data correspond to a total number of 3.72 × 1013."
|
Some Recent Findings
- Review - Mosaicism in Preimplantation Human Embryos: When Chromosomal Abnormalities Are the Norm[2] "Along with errors in meiosis, mitotic errors during post-zygotic cell division contribute to pervasive aneuploidy in human embryos. Relatively little is known, however, about the genesis of these errors or their fitness consequences. Rapid technological advances are helping to close this gap, revealing diverse molecular mechanisms contributing to mitotic error. These include altered cell cycle checkpoints, aberrations of the centrosome, and failed chromatid cohesion, mirroring findings from cancer biology. Recent studies are challenging the idea that mitotic error is abnormal, emphasizing that the fitness impacts of mosaicism depend on its scope and severity. In light of these findings, technical and philosophical limitations of various screening approaches are discussed, along with avenues for future research."
|
| More recent papers
|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
- This search now requires a manual link as the original PubMed extension has been disabled.
- The displayed list of references do not reflect any editorial selection of material based on content or relevance.
- References also appear on this list based upon the date of the actual page viewing.
References listed on the rest of the content page and the associated discussion page (listed under the publication year sub-headings) do include some editorial selection based upon both relevance and availability.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References
Search term: Mitosis | Prophase | Prometaphase | Metaphase | Anaphase | Telophase | Cytokinesis
|
| Older papers
|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
- Golgi apparatus self-organizes into the characteristic shape via postmitotic reassembly dynamics[3] "The Golgi apparatus is a membrane-bounded organelle with the characteristic shape of a series of stacked flat cisternae. During mitosis in mammalian cells, the Golgi apparatus is once fragmented into small vesicles and then reassembled to form the characteristic shape again in each daughter cell. The mechanism and details of the reassembly process remain elusive. ...We show that the characteristic Golgi shape is spontaneously organized from the assembly of vesicles by proper tuning of the two additional mechanisms, i.e., the Golgi reassembly process is modeled as self-organization. We also demonstrate that the fine Golgi shape forms via a balance of three reaction speeds: vesicle aggregation, membrane fusion, and shape relaxation. Moreover, the membrane fusion activity decreases thickness and the number of stacked cisternae of the emerging shapes."
- The nucleoporin ELYS/Mel28 regulates nuclear envelope subdomain formation in HeLa cells[4] "In open mitosis, the nuclear envelope (NE) reassembles at the end of each mitosis. This process involves the reformation of the nuclear pore complex (NPC), the inner and outer nuclear membranes, and the nuclear lamina. In human cells, cell cycle-dependent NE subdomains exist, characterized as A-type lamin-rich/NPC-free or B-type lamin-rich/NPC-rich, which are initially formed as core or noncore regions on mitotic chromosomes, respectively. Although postmitotic NE formation has been extensively studied, little is known about the coordination of NPC and NE assembly. ...Our data show, that ELYS/Mel28 plays a role in NE subdomain formation in late mitosis."
|
Movies
- Links: MCB Movie - The stages of mitosis and cytokinesis in an animal cell
Early Mitosis
| In early development to the morula stage, cells are undergoing rapid cell mitotic divisions and do not have the usual G1 and G2 checkpoint regulation.[6]
During M-phase, cells still maintain the spindle assembly checkpoint, but not apoptosis activation.[7] The spindle assembly checkpoint appears to also be the key to mitotic cell cycle progression during these early cell divisions.[8]
|
<html5media height="250" width="260">File:Mouse_zygote_division.mp4</html5media>
Early mouse mitosis
|
Cell Changes
- Nucleus
- Chromosome condensation
- Nuclear envelope breakdown
- Cytoplasm
- Cytoskeleton reorganization
- Spindle formation (MT) Contractile ring (MF)
- Organelle redistribution
- Mitosis Energy
- Cell division uses up a lot of energy, so cells ensure they have enough resources to complete the job before committing to it.
Mitosis Phases
- Based on light microscopy of living cells light and electron microscopy of fixed and stained cells
- 5 Phases - prophase, prometaphase, metaphase, anaphase, and telophase
- Cytokinesis 6th stage overlaps the end of mitosis
MBC The stages of mitosis and cytokinesis in an animal cell
Interphase
- not a mitotic phase (discussed in cell cycle)
- Chromosomes dispersed in nucleus
- Gene expression
- Cytoskeleton and cell organelles - Distributed and functioning
- Mitochondria undergo independent proliferation/division
Chromosome Changes
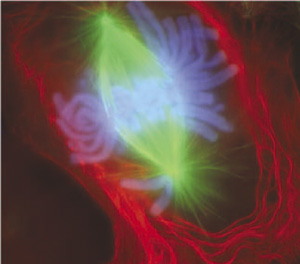
Prophase
Mammalian cell - prophase
[9]- Chromosome DNA has been earlier duplicated (S Phase)
- Chromosomes begin condensing
- Chromosome pairs (chromatids) held together at centromere
- Microtubules disassemble
- Mitotic spindle begins to form
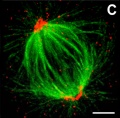
Spindle Apparatus
- 3 sets of microtubules - (+) ends point away from centrosome at each pole.
- astral microtubules - anchor the pole end in position
- kinetochore microtubules - connected to chromosomes
- polar microtubules - form the structure of the spindle apparatus
Spindle Apparatus EM | Spindle Apparatus | MBC Movie- Microtubule dynamics during mitosis
At end of prophase nuclear envelope breaks down
Prometaphase
- Microtubules now enter nuclear region
- Nuclear envelope forms vesicles around mitotic spindle
- Kinetochores form on centromere attach to some MTs of spindle
Dynamic instability and the capture of chromosomes
Centromeric attachment of microtubules
At end of prometaphase chromosomes move to metaphase plate
Metaphase
- Kinetochore MTs align chromosomes in one midpoint plane.
- Astrin is a spindle-associated protein required for chromosome alignment at the metaphase plate.[10]
Proposed alternative mechanisms for chromosome congression
Metaphase ends when sister kinetochores separate
Anaphase
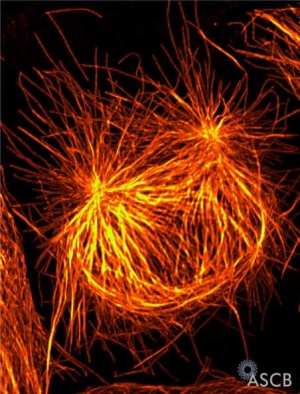
Chromosome motility anaphase
- Separation of sister Kinetochores
- shortening of Kinetochore microtubules pulls chromosome to spindle pole.
- Katanin is a microtubule-severing complex involved with this stage of microtubule dynamics.[11]
Experiment - during anaphase A chromosomes move poleward along stationary kinetochore microtubules, which coordinately disassemble from their kinetochore ends
Anaphase ends as nuclear envelope (membrane) begins to reform.
Telophase
- Chromosomes arrive at spindle poles
- Kinetochore MTs lost
- Condensed chromosomes begin expanding
- Continues through cytokinesis
Links: Figure 19-41 Microtubule dynamics during mitosis | Figure 19-34. The stages of mitosis and cytokinesis in an animal cell | Cytokinetic abscission: cellular dynamics at the midbody
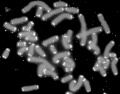
Cleavage of Zygote
Mouse zygote mitosis[12]
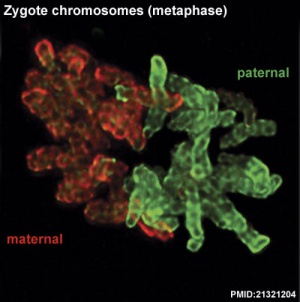
|
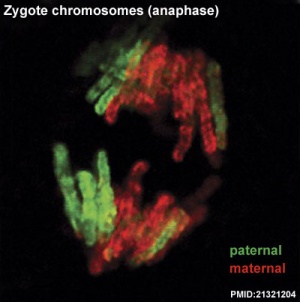
|
| First metaphase
|
First anaphase
|
Cleavage of the zygote forms 2 blastomeres and is cleavage with no cytoplasm synthesis.
- special "embryonic" cell cycle S phases and M phases alternate without any intervening G1 or G2 phases (MSMSMSMS, adult MG1SG2) therefore individual cell volume decreases
Cell division within these cells is initially synchronous (at the same time), then becomes asynchronously (at different times).
- slow- centre cells, larger fast- peripheral cells
- Links: Zygote | Cell Division - Mitosis | Movie - Early Cell Division | Movie - Week 1 Cell Cleavage | Carnegie stage 1
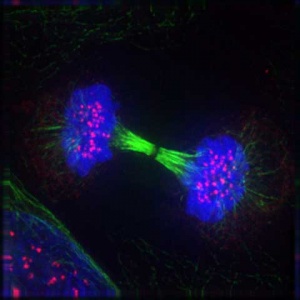
Cytokinesis
- Division of cytoplasmic contents
- Contractile ring forms at midpoint under membrane
- Microfilament ring - contracts forming cleavage furrow
- Eventually fully divides cytoplasm
Links: Cytokinesis | Cytokinesis in Plants
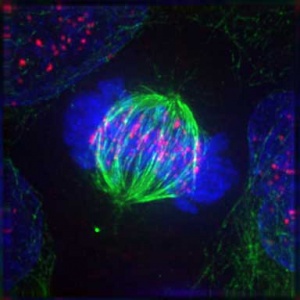
Mitotic Spindle
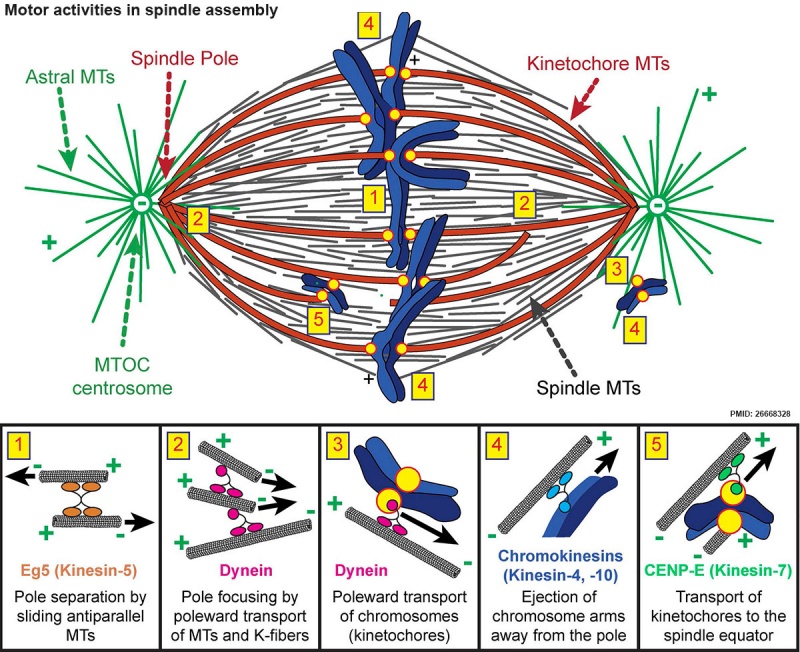
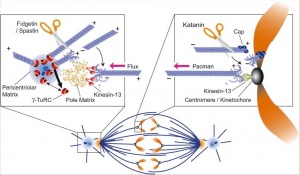
Spindle assembly motors[13]
Microtubule (MT)-bound motors promote bipolar spindle formation, whereas chromosome-associated motors drive proper kinetochore orientation and chromosome movement to the equator.
| Box 1
|
Box 2
|
Box 3
|
Box 4
|
Box 5
|
| Motor-dependent mechanisms establish bipolarity as Eg5 (kinesin-5) motors slide antiparallel microtubules apart with their minus ends leading and their plus ends directed toward the spindle equator.
|
Minus end–directed motors such as dynein move microtubules poleward with their minus ends leading, thereby incorporating K-fibers into the spindle and focusing spindle poles.
|
Kinetochore-associated dynein transports chromosomes along astral microtubules toward the spindle poles from the periphery.
|
Plus end–directed chromokinesins (kinesin-4 and -10) eject chromosome arms outward.
|
CENP-E (kinesin-7) transports unattached kinetochores toward the equator along spindle microtubules. MTOC, microtubule organizing centre.
|
Cell Organelles
Mitochondria
- Divide independently of cell mitosis
- distributed into daughter cells
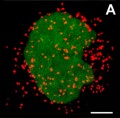
Peroxisomes
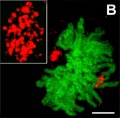
- localise at spindle poles
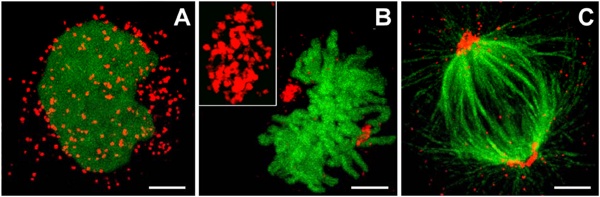
Peroxisome (red) location at Interphase (a) and during Mitosis (b and c)[14]
Endoplasmic Reticulum
- Associated with nuclear membrane.
Golgi
- 2 processes - disassembly and reassembly[15]
- Golgi stack undergoes a continuous fragmentation process
- fragments are distributed into daughter cells
- are reassembled into new Golgi stacks
Disassembly
- Unstacking - mediated by two mitotic kinases (cdc2 and plk)
- Vesiculation - mediated by COPI budding machinery ARF1 and the coatomer complex
Reassembly
- Fusion - formation of single cisternae by membrane fusion
- Restacking - requires dephosphorylation of Golgi stacking proteins by protein phosphatase PP2A
References
- ↑ <pubmed>23829164</pubmed>
- ↑ McCoy RC. (2017). Mosaicism in Preimplantation Human Embryos: When Chromosomal Abnormalities Are the Norm. Trends Genet. , 33, 448-463. PMID: 28457629 DOI.
- ↑ Tachikawa M & Mochizuki A. (2017). Golgi apparatus self-organizes into the characteristic shape via postmitotic reassembly dynamics. Proc. Natl. Acad. Sci. U.S.A. , 114, 5177-5182. PMID: 28461510 DOI.
- ↑ Clever M, Funakoshi T, Mimura Y, Takagi M & Imamoto N. (2012). The nucleoporin ELYS/Mel28 regulates nuclear envelope subdomain formation in HeLa cells. Nucleus , 3, 187-99. PMID: 22555603 DOI.
- ↑ Tavormina PA, Côme MG, Hudson JR, Mo YY, Beck WT & Gorbsky GJ. (2002). Rapid exchange of mammalian topoisomerase II alpha at kinetochores and chromosome arms in mitosis. J. Cell Biol. , 158, 23-9. PMID: 12105179 DOI.
- ↑ Kiessling AA, Bletsa R, Desmarais B, Mara C, Kallianidis K & Loutradis D. (2009). Evidence that human blastomere cleavage is under unique cell cycle control. J. Assist. Reprod. Genet. , 26, 187-95. PMID: 19288185 DOI.
- ↑ Jacobs K, Van de Velde H, De Paepe C, Sermon K & Spits C. (2017). Mitotic spindle disruption in human preimplantation embryos activates the spindle assembly checkpoint but not apoptosis until Day 5 of development. Mol. Hum. Reprod. , 23, 321-329. PMID: 28159965 DOI.
- ↑ Wei Y, Multi S, Yang CR, Ma J, Zhang QH, Wang ZB, Li M, Wei L, Ge ZJ, Zhang CH, Ouyang YC, Hou Y, Schatten H & Sun QY. (2011). Spindle assembly checkpoint regulates mitotic cell cycle progression during preimplantation embryo development. PLoS ONE , 6, e21557. PMID: 21720555 DOI.
- ↑ Russan NM. Let's Build a Spindle. ASCB Image & Video Library. 2008;CYT-190. Available at: http://cellimages.ascb.org/u?/p4041coll12,521
- ↑ <pubmed>21402792</pubmed>
- ↑ <pubmed>17452528</pubmed>
- ↑ <pubmed>21321204</pubmed>| PMC2132672 | PNAS
- ↑ Heald R & Khodjakov A. (2015). Thirty years of search and capture: The complex simplicity of mitotic spindle assembly. J. Cell Biol. , 211, 1103-11. PMID: 26668328 DOI.
- ↑ Kredel S, Oswald F, Nienhaus K, Deuschle K, Röcker C, Wolff M, Heilker R, Nienhaus GU & Wiedenmann J. (2009). mRuby, a bright monomeric red fluorescent protein for labeling of subcellular structures. PLoS ONE , 4, e4391. PMID: 19194514 DOI.
- ↑ Tang D, Mar K, Warren G & Wang Y. (2008). Molecular mechanism of mitotic Golgi disassembly and reassembly revealed by a defined reconstitution assay. J. Biol. Chem. , 283, 6085-94. PMID: 18156178 DOI.
Reviews
Articles
Search Pubmed
Search Pubmed: mitosis
NCBI - Policies and Guidelines | PubMed | Help:Reference Tutorial
Additional Images
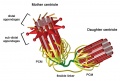
Polo-like kinase 4 centriole duplication activity
Terms
| Cell Division Terms (expand to view)
|
meiosis | mitosis
- anaphase - (Greek, ana = up, again) Mitosis term referring to the fourth stage, where the paired chromatids now separate and migrate to spindle poles. This is followed by telophase.
- anaphase A - Mitosis term referring to the part of anaphase during which the chromosomes move.
- anaphase B - Mitosis term referring to the part of anaphase during which the poles of the mitotic spindle move apart.
- aneuploidy - (aneuploid) term used to describe an abnormal number of chromosomes mainly (90%) due to chromosome malsegregation mechanisms in maternal meiosis I.
- aster - (Latin, aster = star) star-like object visible in most dividing eukaryotic cells contains the microtubule organizing center.
- astral microtubule - spindle apparatus microtubule (MT) originating from the centrosome which does not connect to a kinetochore. These microtubules only exist during mitosis, the other spindle types are polar and kinetochore microtubules.
- autosomal inheritance - term used in hereditary diseases which means that the disease is due to a DNA error in one of the 22 chromosome pairs that are not sex chromosomes. Both boys and girls can then inherit this error. If the error is in a sex chromosome, the inheritance is said to be sex-linked.
- bivalent - (tetrad) a pair of homologous chromosomes physically held together by at least one DNA crossover.
- bouquet stage - meiosis term for when in prophase transition to the zygotene stage, the chromosome telomeres attachment to the inner nuclear envelope and form a cluster. This occurs before the onset of homologous pairing and synapsis. The name comes from the chromosomes resembling a "bouquet of flowers".
- diploid - (Greek, di = double + ploion = vessel) having two sets of chromosomes (2n), this is the normal euploidy state for all human cells, other than gametes that are haploid (n, a single set of chromosomes).
- diplotene stage- (diplotene phase, diplonema; Greek, diplonema = "two threads") meiotic stage seen during prophase I, the chromosomes separate from one another a small amount giving this appearance. In the developing human ovary, oocytes remain at the diplotene stage from fetal life through postnatal childhood, until puberty when the lutenizing hormone (LH) surges stimulate the resumption of meiosis. Prophase I, is divided into 5 stages (leptotene, zygotene, pachytene, diplotene, diakinesis) based upon changes associated with the synaptonemal complex structure that forms between two pairs of homologous chromosomes.
- euploidy - the normal genome chromosomal set (n, 2n, 3n) or complement for a species, in humans this is diploid (2n). The other classes of numerical chromosomal abnormalities include aneuploidy, polyploidy and mixoploidy.
- FUCCI - Acronym for Fluorescence Ubiquitination Cell Cycle Indicator a molecular tool for identifying the stage in the cell cycle. In G0/G1 cells express a red fluorescent protein and S/G2/M cells express a green fluorescent protein. (More? Tooth Development Movie)
- haploid - (Greek, haploos = single) Having a single set of chromosomes (n) as in mature germ/sex cells (oocyte, spermatozoa) following reductive cell division by meiosis. Normally cells are diploid, containing 2 sets of chromosomes. Ploidy refers to the number of sets of chromosomes in the nucleus of a cell.
- heteroplasmy - presence of more than one type of organellar genome. In humans this can refer to variations in the mitochondrial DNA (mtDNA). (More? PMID 26281784)
- homologous chromosomes - meiosis term for the two matching (maternal and one paternal) chromosomes that align during meiosis I.
- homologous recombination - meiosis term when DNA of homologous chromosomes is covalently exchanged to produce chromosomes with new allele combinations, and also links homologous chromosomes with each other to form a bivalent
- human genome - DNA within the 23 nucleus chromosome pairs and the cytoplasmic mitochondrial DNA.
- kinetochore - the protein structure formed on chromatids where the spindle kinetochore microtubules attach during cell division.
- kinetochore microtubule - spindle apparatus microtubule (MT) that attaches to the chromosome kinetochore by its plus end, the other spindle types are astral and polar microtubules.
- kinesin - a microtubule (MT) motor protein that exists in many isoforms and most move towards the MT positive end. Different isoforms have different functions within the spindle apparatus. PMID 20109570
- meiosis - reductive cell division required to produce germ cells (oocyte, spermatozoa) and for sexual reproduction. Note that only spermatozoa complete meiosis before fertilisation. Chromosome number is reduced from diploid to haploid, during this process maternal and paternal genetic material are exchanged. All other non-germ cells in the body divide by mitosis. (More? Meiosis | Spermatozoa Development | Oocyte Development | Week 1)
- meiosis I - (MI) the first part of meiosis resulting in separation of homologous chromosomes, in humans producing two haploid cells (N chromosomes, 23), a reductional division.
- meiosis II - (MII) the second part of meiosis. In male human spermatogenesis, producing of four haploid cells (23 chromosomes, 1N) from the two haploid cells (23 chromosomes, 1N), each of the chromosomes consisting of two sister chromatids produced in meiosis I. In female human oogenesis, only a single haploid cell (23 chromosomes, 1N) is produced. Meiosis II: Prophase II - Metaphase II - Anaphase II - Telophase II.
- meiotic silencing of unsynapsed chromatin - (MSUC) an aneuploidy protective mechanism for subsequent generations, during meiosis where chromosomes are silenced that fail to pair with their homologous partners.
- merotelic kinetochore - cell division abnormality in chromosomal attachment that occurs when a single kinetochore is attached to microtubules arising from both spindle poles. Normal chromosomal attachment in early mitosis, is by only one of the two sister kinetochores attached to spindle microtubules (monotelic attachment) later sister kinetochores attach to microtubules arising from opposite spindle poles (amphitelic attachment).
- metaphase - mitosis term referring to the third stage where mitotic spindle kinetochore microtubules align chromosomes in one midpoint plane. Metaphase ends when sister kinetochores separate. Originally based on light microscopy of living cells and electron microscopy of fixed and stained cells. A light microscope analysis called a "metaphase spread" was originally used to detect chromosomal abnormalities in cells. Mitosis Phases: prophase - prometaphase - metaphase - anaphase - telophase
- metaphase spread - In mitosis using light microscope analysis originally used to detect chromosomal abnormalities in cells, as chromosomes are only visible during cell division.
- microfilament - (MF) cytoskeleton filament normally required for cytoplasmic intracellular transport, motility and cell shape. Named by the actin monomers assembling into the smallest in cross-section of the three filament systems (microtubules and intermediate filaments). This system is disassembled and reassembled as the contractile ring for cytokinesis (cytoplasm division) following cell division mitosis and meiosis.
- microtubule - (MT) cytoskeleton filament normally required for cytoplasmic intracellular transport and motility. Named by the tubulin monomers assembling into "tubes", and are the largest in cross-section of the three filament systems (microfilaments and intermediate filaments). This system is disassembled and reassembled as the spindle apparatus during cell division.
- mitochondrial DNA - (mtDNA) multiple copies of a small circular DNA molecule located within the mitochondria matrix. In humans 16,568 bp in length containing 37 genes, originally inherited only from the oocyte (maternal inheritance).
- mitosis - (M phase) The normal division of all cells, except germ cells, where chromosome number is maintained (diploid). In germ cell division (oocyte, spermatozoa) meiosis is a modified form of this division resulting in reduction in genetic content (haploid). Mitosis, division of the nucleus, is followed by cytokinesis the division of the cell cytoplasm and the cytoplasmic contents. cytokinesis overlaps with telophase.
- p - chromosome short arm (possibly French, petit) and used along with chromosome and band number to indicate genes located on this arm of the chromosome. The chromosome long arm is identified as q (possibly French, tall) chosen as next letter in alphabet after p. These chromosomal arms are only seen when the chromosome is folded for cell division.
- polar microtubule - spindle apparatus microtubule (MT) that can arise from either pole and overlap at the spindle midzone. This interdigitating structure consisting of antiparallel microtubules is responsible for pushing the poles of the spindle apart. The other spindle types are astral and kinetochore microtubules.
- prometaphase - (Greek, pro = before) mitosis term referring to the second stage, when the nuclear envelope breaks down into vesicles. Microtubules then extend from the centrosomes at the spindle poles (ends) and reach the chromosomes. This is followed by metaphase.
- pronuclear fusion - (Greek, pro = before) the process of the fusion of the two haploid nuclear structures (pronuclei) contributed from the spermatazoa and oocyte to form the first diploid nucleus cell. Can also be called "fusion of pronuclei".
- pronucleus - (Greek, pro = before; plural, pronuclei) the two haploid nuclei or nuclear structures containing the genetic material from the spermatozoa and the oocyte. These two haploid nuclei will fuse together to form the first diploid nucleus cell, the zygote. Therefore the nuclear structures that exist "before the nucleus", the plural term is pronuclei.
- prophase - (Greek, pro = before) - mitosis term referring to the first stage, when the diffusely stained chromatin resolves into discrete chromosomes, each consisting of two chromatids joined together at the centromere.
- prophase I - meiosis term refers to the first phase of meiosis I, which together with meiosis II results in the reductive cell division only occurring gametes. Prophase can be further divided into a number of stages: leptotene zygotene, pachytene, diplotene, diakinesis.
- q - chromosome long arm (possibly French, tall), the next letter in alphabet after p, and used along with chromosome and band number to indicate genes located on this arm of the chromosome. The chromosome short arm is identified as p (possibly French, petit). These chromosomal arms are only seen when the chromosome is folded for cell division.
- S phase - during interphase of cell cycle where DNA is duplicated prior to second growth period (G2 phase) that is followed by mitosis (M phase).
- synapsis - (syndesis) meiosis term for the pairing of two homologous chromosomes that occurs during prophase I.
- synaptonemal complex - meiosis term for a protein structure essential for synapsis of homologous chromosomes. (proteins SCP3 and SCP1).
- telomere - region found at each end of the chromosome and involved in cellular ageing and the capacity for division. The regions consist of repeated sequences protecting the ends of chromosomes and harbour DNA repair proteins. In the absence of the enzyme telomerase, these regions shorten during each cell division and becoming critically short, cell senescence occurs.
- telophase - mitosis term referring to the fifth stage, where the vesicles of the nuclear envelope reform around the daughter cells, the nucleoli reappear and the chromosomes unfold to allow gene expression to begin. This phase overlaps with cytokinesis, the division of the cell cytoplasm.
- telomerase - the enzyme that maintains the chromosome end length, the telomeres, involved in cellular ageing and the capacity for division. Absence of telomerase activity leads to the chromosome ends shorten during each cell division, becoming critically short and cell senescence then occurs.
- tetrad - (bivalent) a pair of homologous chromosomes physically held together by at least one DNA crossover.
|
|
|
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 23) Embryology Cell Division - Mitosis. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Cell_Division_-_Mitosis
- What Links Here?
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G