Book - Text-Book of Embryology 7
| Embryology - 20 Apr 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Bailey FR. and Miller AM. Text-Book of Embryology (1921) New York: William Wood and Co.
- Contents: Germ cells | Maturation | Fertilization | Amphioxus | Frog | Chick | Mammalian | External body form | Connective tissues and skeletal | Vascular | Muscular | Alimentary tube and organs | Respiratory | Coelom, Diaphragm and Mesenteries | Urogenital | Integumentary | Nervous System | Special Sense | Foetal Membranes | Teratogenesis | Figures
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Early Mammalian Development
It is perhaps unfortunate for the student beginning the study of embryology that no mammalian form can be taken for the early developmental stages and regarded as wholly typical of the subclass. While there are certain fundamental principles of development in common in all placental mammals, there are also features which vary in different orders. In this chapter no attempt will be made to set forth one line of development as typical, nor will all the variations in the different orders be presented. We shall attempt to present the fundamental principles as exemplified in certain of the mammalian forms and to sketch briefly the early stages of human ontogeny.
The mammalian ovum represents the meiolecithal type in which there is only a small quantity of yolk. Taking the human ovum as an example, the cell is approximately two-tenths of a millimeter in diameter and is not truly spherical but slightly ovoid in shape (Thomson). In section it presents the appearance of the traditional typical cell (Fig. 1). The cytoplasm is coarsely granular owing to the presence of suspended globules of deutoplasm or yolk. Most of the yolk globules are congregated near the center of the cell, around the nucleus, while the peripheral cytoplasm is nearly destitute of yolk. The nucleus is slightly eccentric and exhibits a distinct nuclear membrane, a single plasmosome and rather scanty chromatin.
In most mammals on which observations have been made maturation or reduction of chromosomes begins in the ovary, the first polar body being formed before the Graafian follicle ruptures. The second polar body is formed after the ovum escapes from the follicle; in the mouse for example it is extruded when the ovum reaches the oviduct and after the sperm has entered the cytoplasm. According to recent observations made by Thomson on an extensive series of human ovaries, both polar bodies are extruded by the ovum prior to ovulation. The phenomena of ovulation are discussed in the chapter on "The Germ Cells," p. 23, et seq.
It is generally agreed that the normal site of fertilization of the mammalian ovum is the upper (or outer) third of the oviduct or Fallopian tube. The spermatozoa pass from the vagina through the uterus and into the oviducts where they remain viable and capable of fertilizing for a considerable period of time, perhaps several days or even weeks. In the oviduct the ovum is met by numerous spermatozoa and one of the latter penetrates the egg cytoplasm.
In the white rat, as an example, the entire sperm enters the ovum. After entrance the middle-piece shows increased stainability and the spiral thread, which is probably of mitochondrial origin, becomes evident. The sperm head containing the nucleus enlarges and becomes vacuolated and the centrioles and polar rays appear. As the sperm nucleus forms the male pronucleus, the chromosomes remaining after the second maturation division of the egg are resolved into the female pronucleus. The two pronuclei come in contact near the center of the ovum.
Observations on mammalian ova are not yet sufficient to justify any conclusions regarding the effect of fertilization on the polarity or internal organization of the cells. It has been therefore impossible to trace in subsequent stages of development any relation between egg organization and the planes and constitution of the body in the mammal.
Cleavage
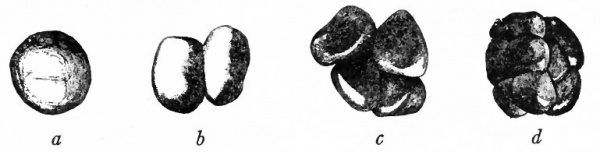
As in the lower forms, cleavage of the fertilized mammalian ovum is primarily a series of mitotic divisions resulting in a solid cluster of cells. This has been observed in detail in several mammals: the opossum (Hartman), the white rat (Huber), the white mouse (Sobotta), and a few others. Cleavage in these forms is irregular and the blastomeres are approximately equal in size (Figs. 53 and 54). The cluster is known as the morula, which corresponds to the similar stage in lower animals.
Fig. 53. Four stages in the cleavage of the ovum of the white rat. Huber.
- A, from a model of an ovum in the pronuclear stage, 24 hours after insemination; B, from a model of a 2-cell stage, 2 days after insemination; C, from a model of a 4-cell stage, 3 days and 1 hour after insemination; D, from a model of an 8-cell stage, 3 days and 11 hours after insemination.
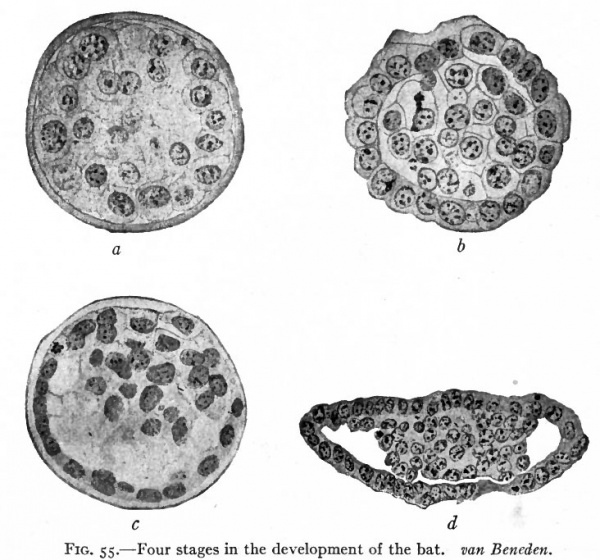
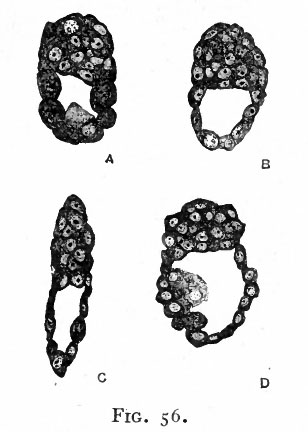
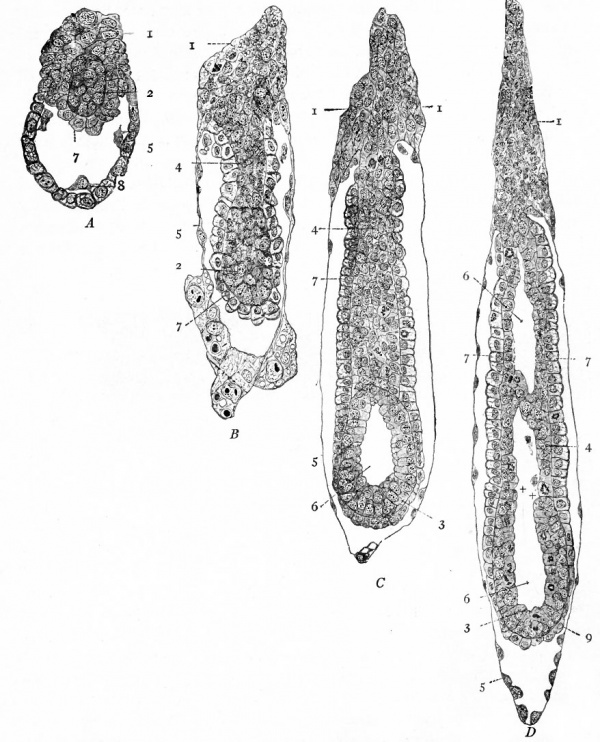
In certain mammals, for example the bat (van Beneden), the superficial cells of the morula become differentiated from those in the interior and form a continuous covering layer (Fig. 55, a and b). Following this differentiation, many of the internal cells become vacuolated, the vacuoles coalesce and finally the cells disappear thus leaving a cavity. At one side of this cavity some of the internal cells remain intact and attached in a cluster, known as the inner cell mass, to the covering layer (Fig. 55, c and d). In other mammals, for instance the white rat (Huber), a cavity is formed within the morula by displacement of the central cells toward the periphery, most of the cells moving to one side where they form a cluster which is continuous with the thinner portion of the wall (Fig. 56). In the opossum (Hartman) no true morula is formed, since during even the earliest cleavage stages the cells are arranged in a layer around a central cavity which in this case contains some yolk material (Fig. 57). In Fig. 58 there is shown a section of the developing ovum of the lemur, Tarsius spectrum, in which the covering layer and inner cell mass are very evident (Hubrecht). The hollow structure illustrated in the four forms above is called the blastocyst (or not quite so correctly, the blastodermic vesicle). It is not regarded as homologous to the blastula of lower forms. The inner cell mass is destined to form the embryo proper while the covering layer gives rise to certain accessory structures. The covering layer receives the name trophoderm (trophoblast) because in subsequent development the nutriment for and waste from the growing embryo must pass through it.
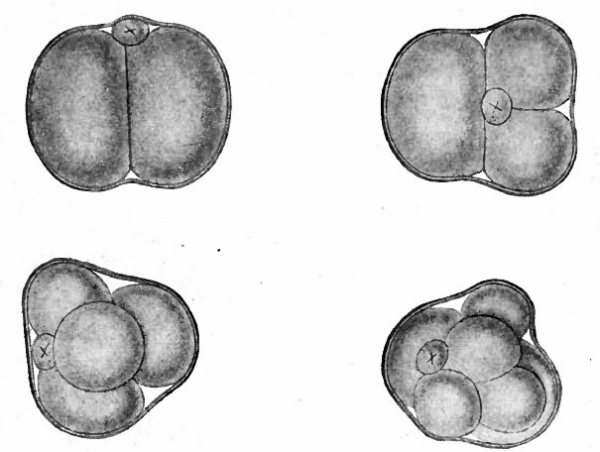
Fig. 54. Four stages in cleavage of the ovum of the mouse. Sobotta.
- Small cell marked with x is the polar body.
Fig. 55. Four stages in the development of the bat. van Beneden.
- a, Section of morula; b, section of later stage of morula, showing differentiation of outer layer of cells; c, section of still later stage, showing vacuolization of central cells; d, section showing outer layer (trop'hoderm) and inner cell mass.
Fig. 56. Sections of blastocysts of the white rat, 5 days after insemination. Huber.
- Note the cavity within the structure and the cluster of cells, comparable to the inner cell mass which forms part of the vesicle.
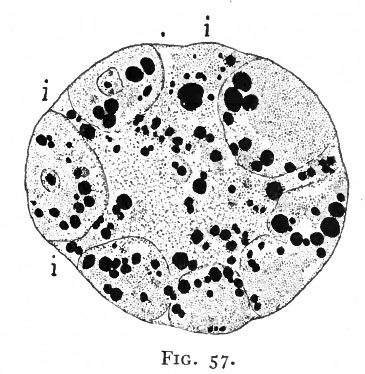
Fig. 57. Section of a 16-cell stage of an ovum of the opossum. Hartman.
- Blastocyst formation is anticipated in that the cells are arranged around the cleavage cavity, and will be completed when the gaps (i) are closed. The black masses represent yolk globules in the t. cytoplasm and in the cleavage cavity.
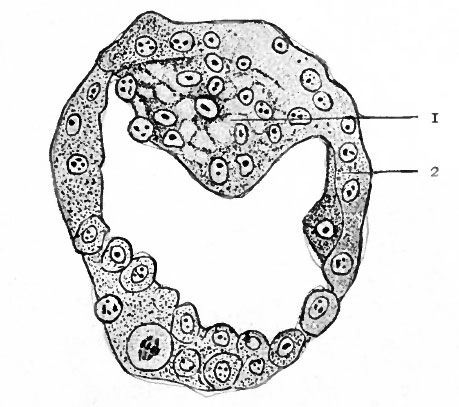
Fig. 58. Section of the blastocyst of the lemur, Tarsius spectrum. Hubrecht, from Quain's Anatomy
- i, Inner cell mass; 2, trophoderm.
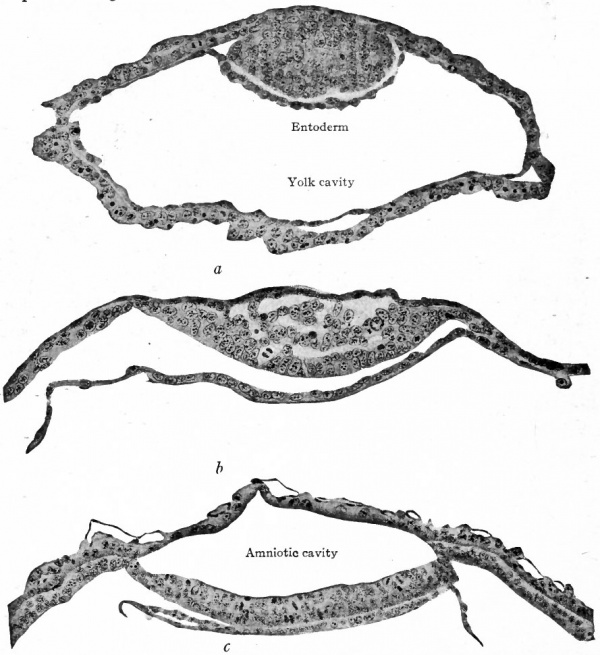
Fig. 59. Sections of blastodermic vesicle of bat, van Beneden.
- showing (a) formation of the entoderm and (b and c] of the amniotic cavity.
Ectoderm and Entoderm
In the lower forms described in the earlier chapters the two primary germ layers arose through the process of gastrulation. In mammals generally gastrulation is masked under highly modified processes which are probably determined by the peculiarities of cleavage and blastocyst formation in the mammalian ovum. It is usually difficult to recognize any phenomenon in germ layer formation in mammals as wholly comparable to invagination or epiboly or involution in the lower forms. The results are arrived at by abbreviated or caenogenetic steps in which the characteristics of the lower forms are profoundly modified.
Taking the bat again as an example, some of the cells of the inner cell mass bordering the cavity of the blastocyst, or yolk cavity, become differentiated, proliferate and migrate around on the inner surface of the trophoderm, forming there a complete layer and lining for the cavity. This new layer is the primary entoderm (Fig. 59, a; compare with Fig. 55, d). Immediately following the formation of the entoderm many of the cells of the inner cell mass become vacuolated and disappear, resulting in the formation of a cavity between the overlying trophoderm and the cells contiguous to the entoderm. This new cavity is the amniotic cavity (Fig. 59, b and c). The two cavities are separated therefore by a double-layered plate, the embryonic disk, the layer bordering the amniotic cavity comprising the embryonic ectoderm (Fig. 59, c). In the lemur, Tarsius spectrum, the differentiating entodermal cells, instead of spreading over the inner surface of the trophoderm, form a small sac within the larger cavity (Fig. 60, a, b, c). In Tarsius the inner cell mass seems to invaginate from its outer side and the definite layer of cells thus resulting becomes the embryonic ectoderm (Fig. 60, c). Subsequently the inverted ectoderm becomes straightened out so that the embryonic disk is flat. In either case the embryonic disk is destined to give rise to the' embryonic body.
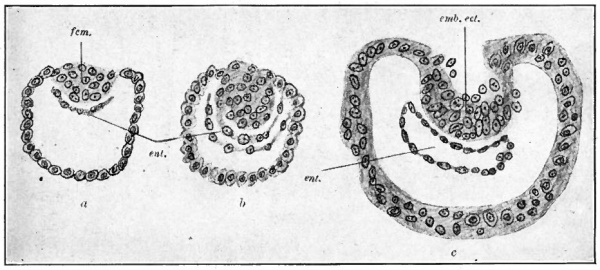
Fig. 60. Three stages in the formation of the germ layers in the lemur, Tarsius spectrum. Hubrecht, from Quain's Anatomy
- icm., Inner cell mass; emb. ect., embryonic ectoderm; ent., entoderm.
In the white rat the progress of development following the formation of the blastocyst is marked by a curious inversion of the inner cell mass, giving rise to a condition known as inversion or entypy of the germ layers. This condition is characteristic of a number of rodents, including the rat, guineapig and mouse. The cluster of cells on one side of the blastocyst increases in size by proliferation of the cells and enlargement of individual members of the group so that it projects outward somewhat and inward into the cavity of the vesicle (Fig. 61, A). Within the cluster a small group of cells becomes differentiated (staining more deeply) which represents the rudiment of the ectodermal node. The layer of cells covering this node on the side toward the cavity is the entoderm. The group of cells projecting outward is known as the ectoplacental cone. Following this, the vesicle elongates and the ectodermal node appears to be forced farther into the blastocyst cavity by a group of cells which develops between the node and the ectoplacental cone and which is extraembryonic ectoderm (Fig. 61, B). The entoderm is more extensive, covering the ectodermal node and much of the extraembryonic ectoderm. At this stage it is evident that entoderm is outside of ectoderm, the condition which has given rise to the term 'inversion of the germ layers.' The two layers together constitute the egg-cylinder (of Sobotta).
Fig. 61. Sections of blastocysts of the white rat, showing inversion (entypy) of the germ layers. Huber.
- A, blastocyst 6 days and 14 hours after insemination. B, blastocyst 8 days and 1 8 hours after insemination, with egg-cylinder cut longitudinally. C, similar section of blastocyst 7 days and 22 hours after insemination (younger than B but further advanced in development), showing beginning of proamniotic cavity. D, similar section of blastocyst 8 days after insemination (younger than B but further advanced in development), showing more advanced proamniotic cavity.
- 1, Ectoplacental cone; 2, ectodermal node; 3, primary embryonic ectoderm; 4, extraembryonic ectoderm; 5, transitory ectoderm (original wall of blastocyst); 6, proamniotic cavity; 7, visceral entoderm; 8, cells of parietal entoderm; 9, primary embryonic entoderm.
As development proceeds there is still further elongation of the vesicle, with concomitant lengthening of the egg-cylinder. Within the ectodermal node a cavity appears, and around this the cells arrange themselves in the form of a simple columnar epithelium. The space is known as the proamnitic cavity (Fig. 61, C). The original wall of the blastocyst, except at the ectoplacental cone, is drawn out into a thin membrane-like layer. The cone itself is longer. Following this stage a number of discrete spaces appear" among the extraembryonic ectodermal cells and then coalesce to form one continuous cavity which breaks through into and becomes continuous with the proamniotic cavity of the original node (Fig. 61, D). The cells around this new cavity are also arranged as a simple columnar epithelium; but the boundary line between embryonic and extraembryonic ectoderm is still evident. At this time, when the ova are eight days old, there is not yet any indication of bilaterality in the egg-cylinder.
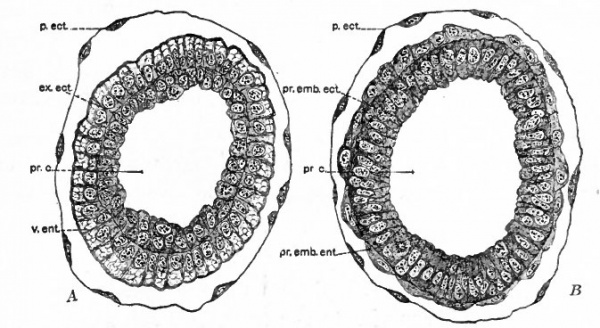
In slightly later stages, shown in cross section in Fig. 62, A and B the columnar entoderm surrounding the extraembryonic ectoderm exhibits an outer vacuolated zone and an inner granular zone, while the entoderm of the embryonic region becomes flat. A little further along in development a new group of cells appears between embryonic ectoderm and entoderm on one side of the egg-cylinder near the junction between embryonic and extraembryonic regions (Fig. 63, A). This group of cells spreading out between the other two layers is the early mesoderm, which here is evidently a derivative of embryonic ectoderm since it is not even attached to the entoderm. The area where thg mesoderm appears marks the site of the primitive streak and therefore the caudal end of the future embryo. The proamniotic cavity becomes triangular in cross section in this region, the change in shape being regarded as due to the formation of the primitive streak (Fig. 63, B). It is interesting to note here that development up to this stage has required nine days out of the 21-23 days of gestation in the white rat. The slow progress of the early cleavage stages is regarded as due to the lack of nutriment as the ova pass through the oviduct. Even when the ova reach the uterus they become imbedded very slowly in the uterine mucosa, probably receiving on that account a minimum of nourishment. During development the original wall of the blastocyst becomes reduced to a mem* brane around which maternal blood circulates as the vesicles become embedded in the uterine mucosa. As soon as the blood is present the entodermal cells, and also the cells of the ectoplacental cone, take on the vacuolated appearance and haemoglobin can be demonstrated in the cytoplasm. On the whole, however, the mechanism for nourishing the developing ovum appears to lack the efficiency of that in other mammals. On this account Sobotta has presented the conclusion that the inversion of the germ layers is an attempt on the part of the developing organism to increase the entodermal surface and to put the layer of entoderm in closer contact with the source of nutriment, that is, the maternal blood.
Fig. 62. Cross sections of an egg-cylinder of the white rat, 8 days and 17 hours after insemination. Huber. A, section which corresponds to a level just above the two crosses in Fig. 61, D. B, section which corresponds to a level about half an inch below the two crosses in Fig. 61, D. ex.ect., extraembryonic ectoderm; p.ect., transitory ectoderm (original wall of blastocyst); pr.c., proamniotic cavity; pr.emb.ect., primary embryonic ectoderm; Pr.emb.ent., primary embryonic entoderm; v.ent., visceral entoderm.
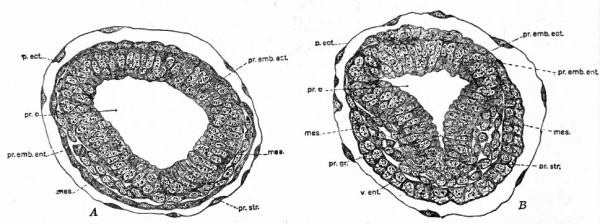
Fig. 63 Cross sections of egg-cylinders of the white rat. Huber.
- A , 8 days and 17 hours after insemination; B, 8 days and 16 hours after insemination. These sections are taken through the region of the primary embryonic ectoderm and would therefore correspond to levels below the crosses in Fig. 61, D. mes., Mesoderm; p.ect., transitory ectoderm (original wall of blastocyst); pr.c., proamniotic cavity; pr.emb.ect., primary embryonic ectoderm; pr.emb.ent., primary embryonic entoderm; pr.gr., primitive groove; pr.str., primitive streak; v.ent., visceral entoderm.
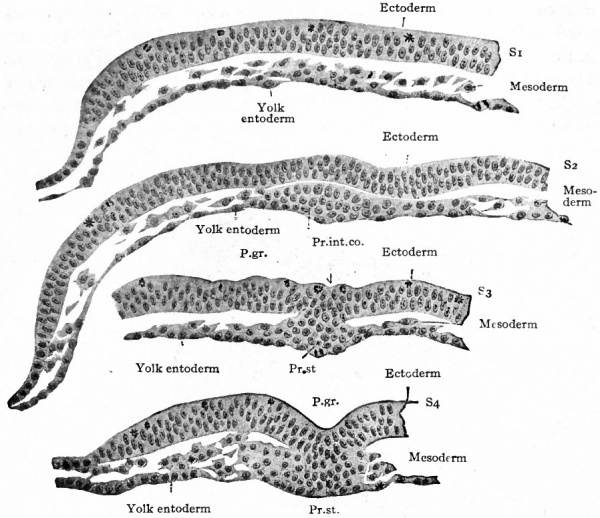
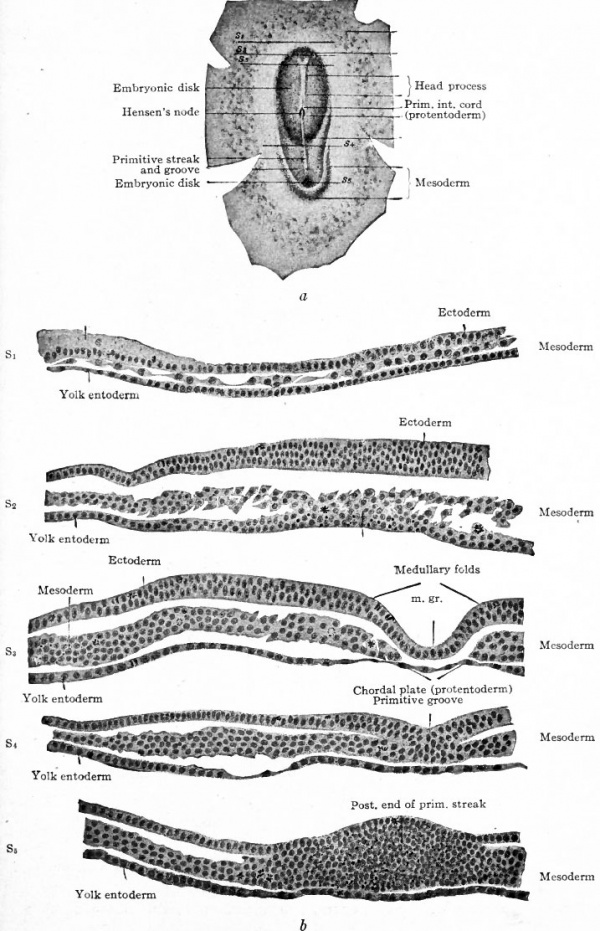
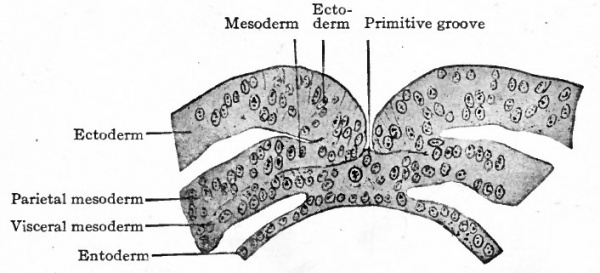
Mesoderm. Returning now to the bat, it has already been noted (page 89) that the embryonic disk is nearly flat and composed of ectoderm and entoderm (Fig. 59, c). In the dog the disk is of similar form and construction, but has no amniotic cavity over it. In the dog's disk then a linear opacity appears which extends about two-thirds of the way across the disk (Fig. 64). Obviously this band represents the primitive streak. In cross section the primitive streak is seen to be composed of ectoderm and entoderm fused with an intermediate layer of mesoderm (Fig. 65, S$ and S^). The arrangement of the layers here corresponds exactly with that in the bird. (Compare Fig. 47.) The mesoderm extends laterally between ectoderm and entoderm as a number of more or less scattered cells. Sections taken in front of the primitive streak show the entoderm and mesoderm fused together while the ectoderm is a separate layer (Fig. 65, Si and 52). This disposition of the layers is characteristic of the primitive axis in the bird.
Fig. 64. Embryonic disk of dog. Bonnet.
- The letters and figures on the right (S1 - S4 ) indicate planes of sections shown in Fig. 65
Fig. 65. Transverse sections of embryonic disk of dog. Bonnet.
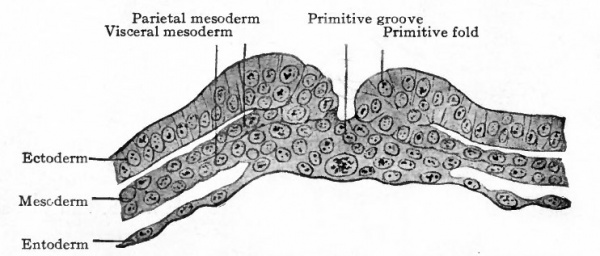
- Sections of disk shown in Fig. 64. Letters and numbers at right (Si-S 4 ) indicate plane of sections in Fig. 64. P.gr., Primitive groove; Pr.int.co., primitive intestinal cord; Pr.st., all three germ layers fused in primitive streak.
Fig. 66. Transverse sections of embryonic disks of rabbit, (a) Kolliker, (b) Rabl.
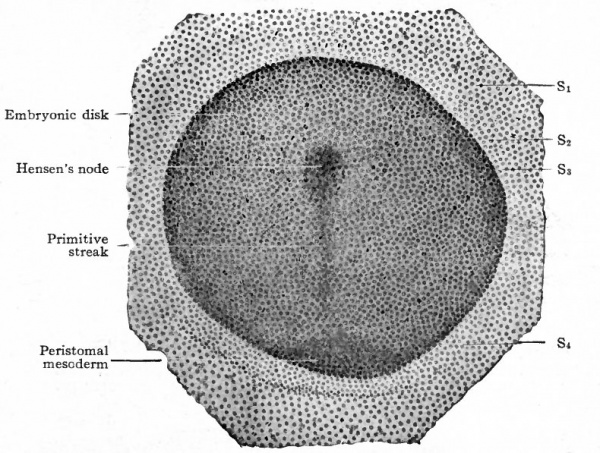
- a, section through primitive streak of embryo of 6 days and 18 hours; b, section through Hensen's node of embryo of 7 days and 3 hours.
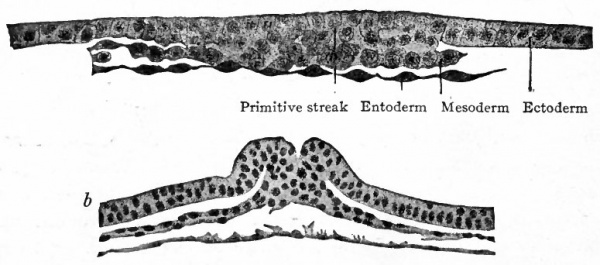
- Compare Fig. 48.) The conditions as portrayed in the rabbit (Fig. 66, a and b) and in the white rat (Fig. 63) point to the conclusion that the mesoderm originates from the ectoderm.
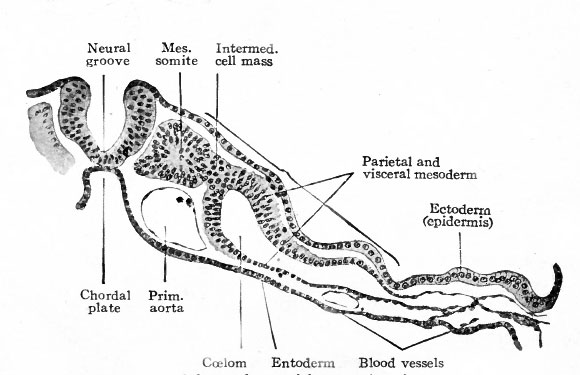
In the primates thus far studied the mesoderm develops before the primitive streak appears in the embryonic disk. In Tarsius spectrum the middle germ layer appears between trophoderm and entoderm while the disk is still composed only of the two primary layers (Fig. 67). In Semnopithecus nasicus a similar condition obtains (Fig. 68). In the human ovum described by Bryce and Teacher[1] the mesoderm is an extensive layer when the germ disk is still rudimentary (Fig. 73). The origin of the mesoderm in these cases is problematical. In the lemur Hubrecht maintains that it is derived from the ectoderm. In Semnopithecus Selenka considers it to be of entodermal origin. The mesoderm of the embryonic disk itself appears somewhat later in the same manner as in the lower mammals, that is, from the thickened ectoderm along the primitive streak (Fig. 69). J Following the development of the primitive streak in the dog's disk the disk becomes oval, and anterior to the streak an opaque band appears which is comparable to the primitive axis of the bird's blastoderm (Fig. 70, a compare with Fig. 46). Transverse sections of the disk are shown in Fig. 70, b, Si, Sz, SB, Si, 5). The mesoderm has increased and now comprises an extensive layer between ectoderm and entoderm. From this stage forward the course of development in the mammal follows the general line of development in the bird. The mesoderm near the axial line becomes more massive and segmented transversely to form the mesodermal somites (Fig. 71). More laterally the mesoderm cleaves into two layers, the parietal and visceral, the cleft itself representing the rudiment of the coelom (Fig. 72).
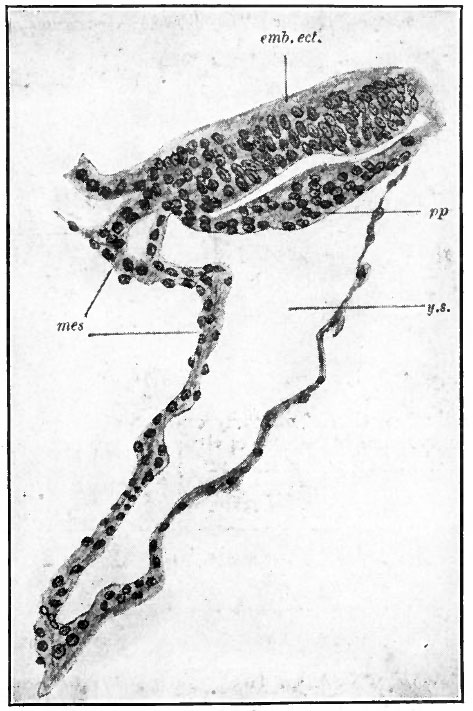
Fig. 67. Median longitudinal section through the embryonic disk and yolk sac of the lemur, Tarsius spectrum. Hubrecht, from Quain's Anatomy.
- emb.ect., Embryonic ectoderm; mes, mesoderm; pp, entoderm; y.s., yolk sac.
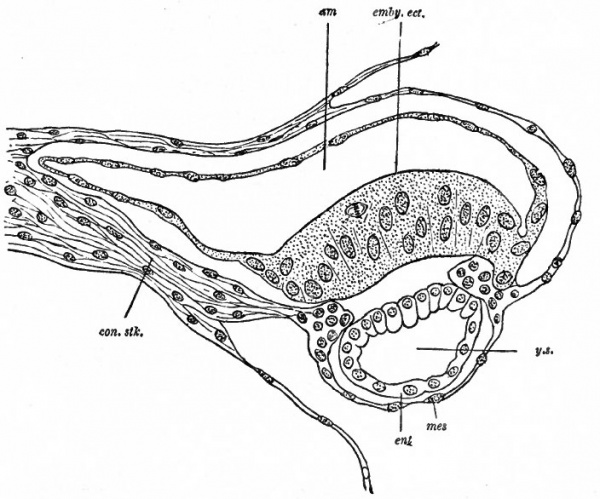
Fig. 68. Median longitudinal section of an early embryo of Semnopithecus nasicus. Selenka, from Quain's Anatomy.
- am, Amniotic cavity; con.stk., belly stalk; emby.ect., embryonic ectoderm; ent, entoderm; mes, mesoderm; y.s., yolk sac.
Fig. 69. Transverse sections through the embryonic disk of the lemur, Tarsius spectrum, after the appearance of the primitive streak. Hubrecht, from Quain's Anatomy.
- A is a section through the primitive node; B is a section through the primitive streak posterior to the node.
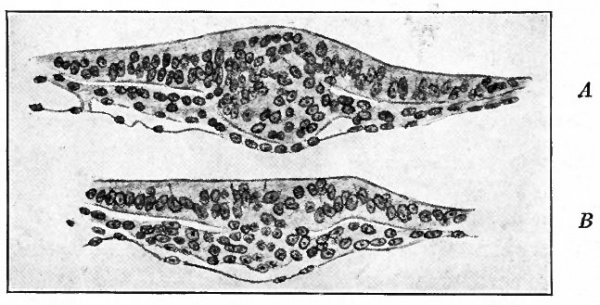
Fig. 70. Surface view of embryonic disk of dog and transverse sections of same. Bonnet.
- a, Disk somewhat further advanced than that in Fig. 64; the letters and figures (S1 - S6 ) indicate planes of sections in b; m.gr., medullary groove.
Fig. 71. Dorsal view of dog embryo with ten pairs of mesodermal somites. Bonnet.
Between the mesodermal somites and the cleft portion of the mesoderm a small group of cells represents the intermediate cell mass or nephrotome.
The formation of the neural plate and tube from the axial band of ectoderm is quite similar to its development in the lower forms. Subsequently the disk is bent or rolled into the typical cylindrical vertebrate body, a process described in the chapter on "External Form" (p. 109 et seq.).
The Germ Layers in Man
There are no observations on the development of the human ovum prior to the appearance of all three germ layers. Consequently nothing is known concerning fertilization, cleavage, the first differentiation of cells, the formation of the embryonic disk, or the mode of origin of the germ layers. In the youngest human embryo that has been recorded, the one described by Bryce and Teacher in 1908[1], all three germ layers are already present. The age of this embryo was reckoned to be about 14 days.
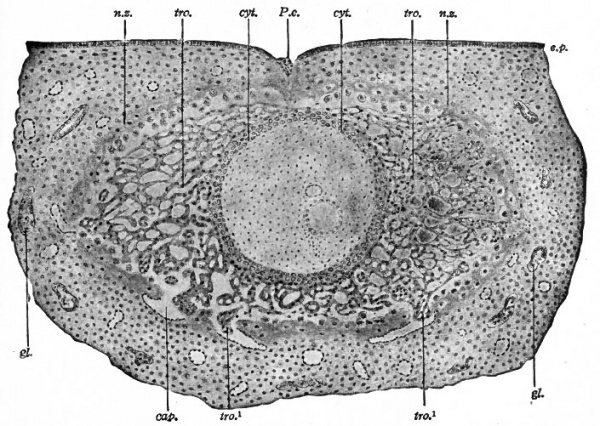
Fig. 73. Section of a human ovum of about 14 days, embedded in the uterine mucosa. Bryce and Teacher.[1]
Cap., Capillary; cyt., cellular layer (cyto-trophoderm) ; ep., uterine epithelium; gl., uterine gland; n.z., necrotic zone of decidua (uterine mucosa); P.e., point of entrance of the ovum; tro., syncytial layer (plasmodi-trophoderm) ; tro. 1 , masses of vacuolating syncytium invading capillaries. The cavity of the vesicle is filled with mesoderm in which are embedded the amniotic cavity (the larger) and the yolk cavity.
In certain respects the Bryce-Teacher embryo[1] (Fig. 73) bears fundamental resemblances to corresponding stages of lower mammals, especially the lower primates; in other respects there are differences which are not irreconcilable, however, with the general principles of mammalian ontogeny. The vesiclelike structure of the entire developing organism is a fairly close approximation to the trophodermal sac of the lower forms. In both cases the rudiment of the embryonic body is contained within the sac. In the human embryo in question there are two cavities within the vesicle; the larger is regarded as the amniotic cavity lined with ectoderm, and the smaller as the cavity of the yolk sac lined with entoderm. The double wall between the two would be the embryonic disk. The precocious development of the mesoderm, which as a loosely arranged tissue fills in all the space between the trophoderm and the two small cavities, is one of the remarkable features of this embryo. The trophoderm is a most elaborate layer and has sent out irregular projections into the uterine mucosa in which the whole structure is already embedded. The early embedding or implantation and the elaboration of the trophoderm are probably closely correlated.
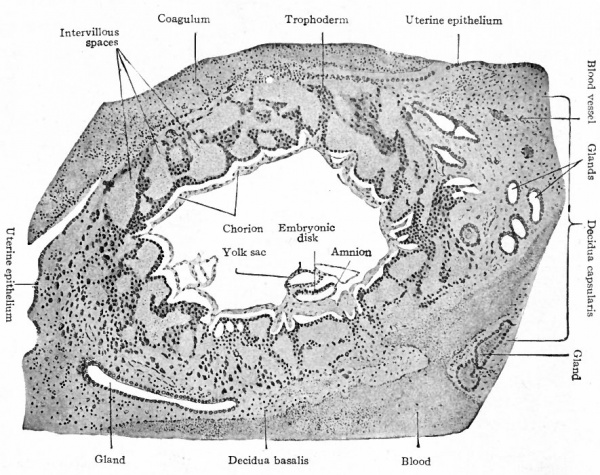
Fig. 74. Section through very young human chorionic vesicle embedded in the uterine mucosa. Peters.
- The vesicle measured 2.4 x 1.8 mm., the embryo 19 mm. Peters reckoned the age as 3 or 4 days, but later studies of other embryos go to show that the age is much greater; Bryce and Teacher estimate it at 14 to 15 days.
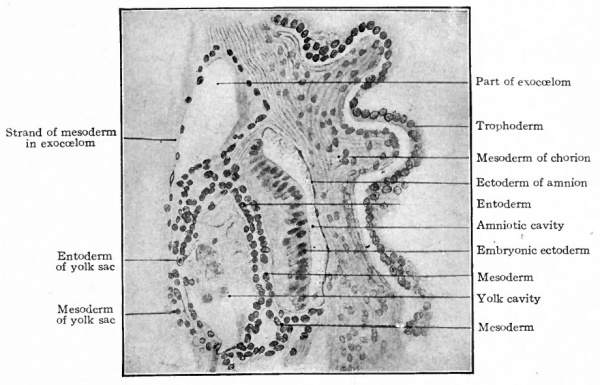
In a slightly older embryo described by Peters (Fig. 74) a space has appeared within the mesoderm, so that one portion remains as a lining for the trophodermal wall and the remainder closely invests the yolk sac and amnion and also forms a layer between ectoderm and entoderm in the embryonic disk (Fig. 75). The disk is therefore composed of all three germ layers, but there is still no indication of a primitive streak. It would seem that in the highest primate the mesoderm develops independently of the primitive streak; but whether it arises from ectoderm or entoderm it is not possible in the present state of our knowledge to determine.
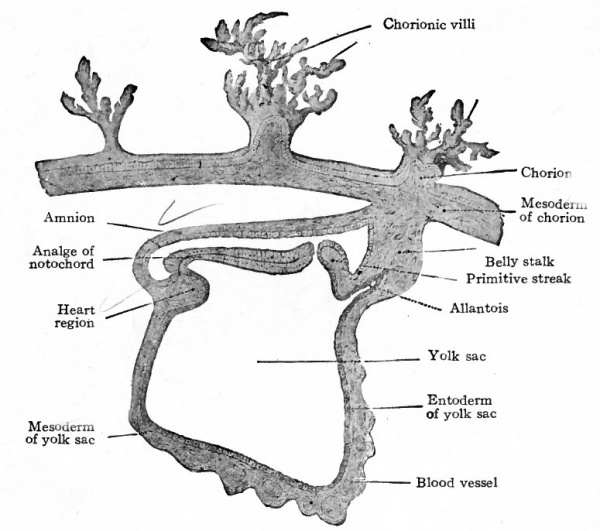
Fig. 75. Section through human chorion, amnion, embryonic disk, and yolk sac. Peters. Compare with Fig. 74.
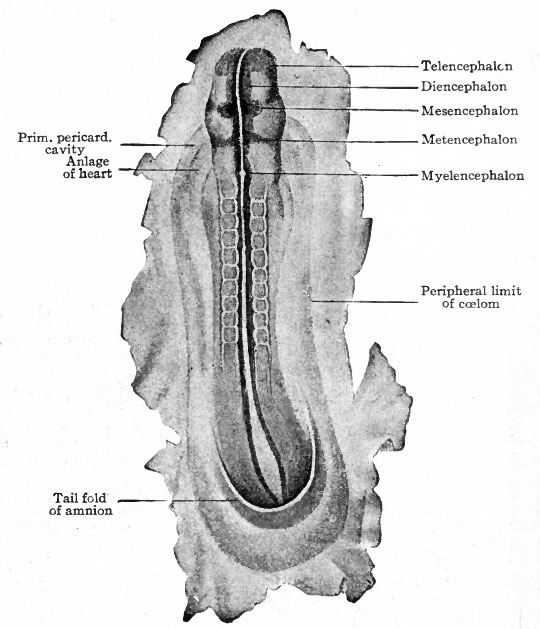
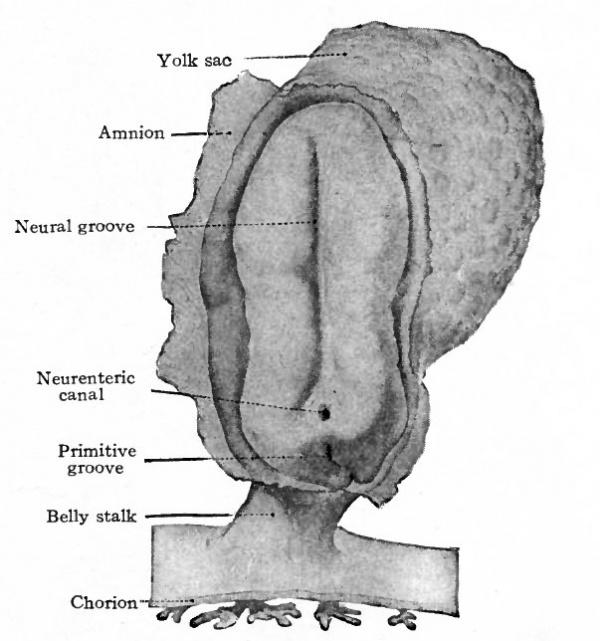
Fig. 76. Dorsal view of human embryo, two millimeters in length, with yolk sac.
Fig. 77. Medial section of human embryo shown in Fig. 76. von Spee, Kollmann.
Fig. 79. Transverse section through primitive groove of rabbit embryo, van Beneden.
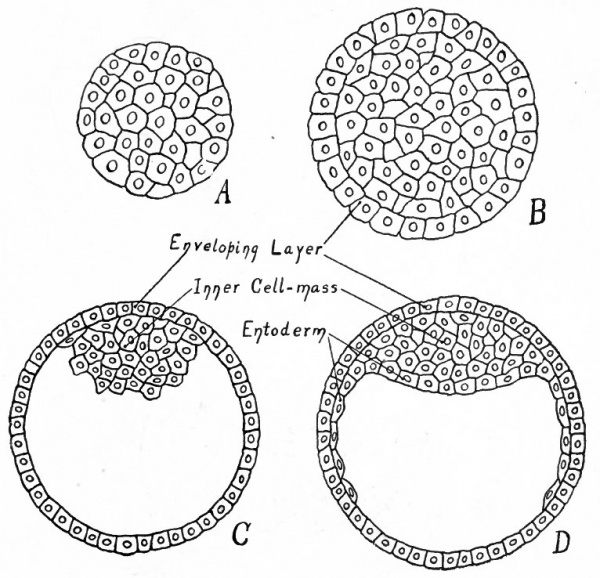
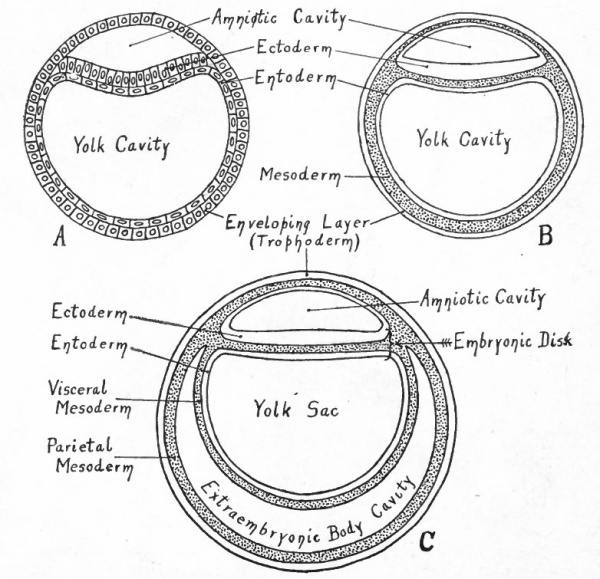
Fig. 80. Diagrams representing hypothetical stages in the development of the human embryo.
- A, Morula; compare with Fig. 55, a. B, Morula with differentiated superficial cells; compare with Fig- 55) b. C, Central cells have become vacuolized to form the yolk cavity, leaving a small group (the inner cell mass) attached to the enveloping layer (trophoderm) ; compare with Fig- 5 5 , d. D, Cells of the inner cell mass which are adjacent to the yolk cavity have become differentiated and have begun to grow around the cavity, forming the entoderm; compare with Fig. 59, a.
In a somewhat older human embryo described by von Spee a dorsal view of the embryonic disk shows close resemblances to conditions in the lower mammals (Fig. 76). The position of the primitive streak is indicated by the conspicuous primitive groove. Anterior to this the neural groove extends almost the full length of the disk which has become considerably elongated. The yolk sac is now suspended from the ventral side of the disk.
A longitudinal section in the medial sagittal plane shows the embryonic disk separating the yolk cavity from the amniotic cavity (Fig. 77). The mesoderm is an extensive layer investing both amnion and yolk sac and forming a strong band which attaches the embryonic body to the outer wall of the vesicle (now the chorion). A cross section through the primitive streak shows a striking resemblance to a corresponding section of the embryonic disk of a rabbit. (Compare Figs. 78 and 79.) The three germ layers are fused in the streak, and the mesoderm extends laterally on both sides between the other two layers.
Fig. 81. Diagrams representing hypothetical stages in the development of the human embryo (to follow Fig. 80).
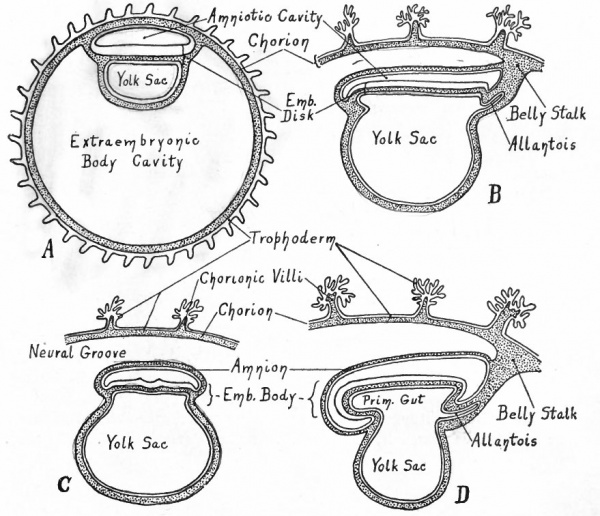
- A, Entoderm surrounds the yolk cavity; part of the cells of the inner cell mass have become vacuolated, thus forming the amniotic cavity, while the remainder constitute the embryonic ectoderm; compare with Fig. 59. B, Mesoderm (represented by dotted portion) has appeared between the entoderm and trophoderm, between the entoderm and ectoderm of the embryonic disk, and in the roof of the amnion. C, The mesoderm around the yolk cavity has split into a parietal and a visceral layer, the cleft between being the rudiment of the extraembryonic body cavity (exoccelom).
In further development the behavior of the germ layers during the formation of the neural tube, the origin of the mesodermal somites, the appearance of the ccelom in the lateral portion of the mesoderm, and the formation of the notocord corresponds in main outline to their behavior in the lower mammals and in birds.
The series of diagrams in Figs. 80, 81 and 82 has been constructed to give the student a general idea of the changes that occur in the early stages of human development. It must be recognized, however, that the diagrams represent purely hypothetical stages up to the conditions shown in diagram B in Fig. 81 which corresponds roughly to the Bryce-Teacher embryo[1] (Fig. 73) ; even in this diagram the extent of the mesoderm is much less than in the known human embryo. In Fig. 82 diagram A approximates the Peters embryo (Fig. 74), diagram D the von Spee embryo (Fig. 77). The history of the accessory structures which are shown in part will be considered in the chapter on "Foetal Membranes".
Fig. 82. Diagrams representing stages of development of the human embryo (to follow Fig. 81).
- A, A stage that corresponds approximately to those of Peters' and Bryce-Teacher's[1] embryos (Figs. 74 and 73). Owing to the rapid enlargement of the chorionic vesicle, the extraembryonic body cavity has become much larger than in Fig. 81 , C. B,A stage (in longitudinal section) corresponding to that of von Spec's embryo (Fig. 77) . Only a part of the chorion is shown; the embryonic disk is slightly constricted from the yolk sac; note the belly stalk, comparing with A . C, Transverse section, same stage as B. D, Longitudinal section, stage somewhat later than B. Note the greater degree of constriction between the embryo and yolk sac, and the larger amnion.
- Next: External body form
References for Further Study
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Bryce TH. and Teacher JH. Contributions To The Study Of The Early Development And Imbedding Of The Human Ovum 1. An Early Ovum Imbedded In The Decidua. (1908) James Maclehose and Sons. Glasgow.
VAN BENEDEN, E.: Recherches sur les premiers stades du developpement du Murin (Vespertiliomurinus). Anat. Anzeiger, Bd. 16, 1899.
BONNET, R.: Lehrbuch der Entwicklungsgechichte. 1907.
BRYCE, T. H.: Embryology. Vol. I of Quain's Anatomy, 1908.
Bryce TH. and Teacher JH. Contributions To The Study Of The Early Development And Imbedding Of The Human Ovum 1. An Early Ovum Imbedded In The Decidua. (1908) James Maclehose and Sons. Glasgow.
V HARTMAN, G.: Studies in the Development of the Opossum. Jour, of Morph., Vol. 27, 1916.
HERTWIG, O.: Die Lehre von den Keimblattern. In Hertwig's Handbuch der vergl. u. experiment. Entwickelungslehre der Wirbeltiere, Bd. I, Teil I, 1903.
Herzog MA. A contribution to our knowledge of the earliest known stages of placentation and embryonic development in man. (1909) Amer. J Anat., 9(3): 361-400.
v HUBER, G. C.: The Development of the Albino Rat, Mus norvegicus albinus. Memoirs of the Wistar Institute, No. 5, 1915.
HUBRECHT, A. A. W.: Furchung und Keimblattbildung bei Tarsius spectrum. V ' erhandelingen der Koninklijke Akademie van Wetenschappente Amsterdam, Bd. 7, 1902.
Keibel F. and Mall FP. Manual of Human Embryology I. (1910) J. B. Lippincott Company, Philadelphia. Chap. IV.
MELISSINOS, K.: Die Entwicklung des Eies der Mause von der ersten Furchungsphanomenen bis zur Festsetzung der Allantois an der Ectoplacentarplatte. Arch. f. mikr. Anat., Bd. 70, 1907.
PETERS, H.: Ueber die Embettung des menschlichen Eies und das bisher bekannte menschliche Placentationstadium. Leipzig, 1899.
SOBOTTA, J.: Die Befruchtung und Furchung des Eies der Maus. Arch. /. mikr. Anat., Bd. 45, 1895.
SOBOTTA, J.: Die Entwickelung des Eies der Maus vom Schlusse der Furchungperiode bis zum Auftreten der Amnionfalte. Arch. f. mikr. Anat., Bd. 61, 1903.
VON SPEE, G. : Beobachtungen an einer menschlichen Keimscheibe mit offener Medullarrinne und Canalis neurentericus. Arch.f. Anat. u. Physiol., Anat. Abth., 1889.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Text-Book of Embryology: Germ cells | Maturation | Fertilization | Amphioxus | Frog | Chick | Mammalian | External body form | Connective tissues and skeletal | Vascular | Muscular | Alimentary tube and organs | Respiratory | Coelom, Diaphragm and Mesenteries | Urogenital | Integumentary | Nervous System | Special Sense | Foetal Membranes | Teratogenesis | Figures
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, April 20) Embryology Book - Text-Book of Embryology 7. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_Text-Book_of_Embryology_7
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G