Book - Text-Book of Embryology 18
| Embryology - 5 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Bailey FR. and Miller AM. Text-Book of Embryology (1921) New York: William Wood and Co.
- Contents: Germ cells | Maturation | Fertilization | Amphioxus | Frog | Chick | Mammalian | External body form | Connective tissues and skeletal | Vascular | Muscular | Alimentary tube and organs | Respiratory | Coelom, Diaphragm and Mesenteries | Urogenital | Integumentary | Nervous System | Special Sense | Foetal Membranes | Teratogenesis | Figures
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
The organs of special sense
The Eye
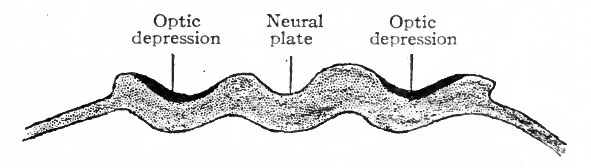
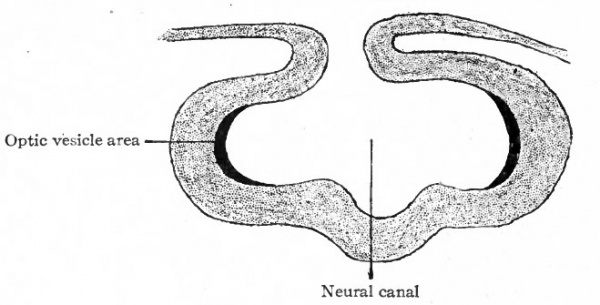
The receptive mechanisms of all the general and special sense organs are derived from the ectoderm. With the single exception of the eye, all develop as direct specializations of the ectoderm in the form of the various neuro-epithelia. The eye is peculiar among the sense organs in that its receptive cells are not derived directly from surface ectoderm, but only indirectly from the ectoderm after it has become folded in to form the neural canal. The neuro-epithelium of the eye develops as a direct outgrowth from the central nervous system. The retina is a modified part of the brain; the optic nerves correspond to central nervous system fiber tracts. Of the accessory optic structures, the lens, the epithelium of the lids and conjunctiva, the eyelashes, the Meibomian glands and the epithelium of the lacrymal apparatus arc of ectodermic origin; the coats of the eye, the sclera and chorioid, and parts of their modified anterior extensions, the cornea, ciliary body and iris, are of mesodermic origin. In the sensory divisions of the other spinal and cranial nerves, with the exception of the olfactory, the cell bodies of the neurones which serve to connect the receptive mechanisms with the brain and cord are located in parts (the sensory ganglia of the cranial and spinal nerves) which have become separated from the crests of the neural folds as the latter fuse to form the neural canal. In the eye the cell bodies of these neurones are located in the retina, but the area of ectoderm from which the retina develops first occupies a position along the neural crest analogous to that occupied by the anlagen of the spinal and cranial ganglia. In the case of the retina this area, instead of becoming split off in the closure of the neural canal, becomes folded into the canal and later pushed out toward the surface in the optic evagination (Figs. 450, 457, 458).
Fig. 456. Diagram showing location of optic areas before the closure of the neural groove. (Modified from Lange)
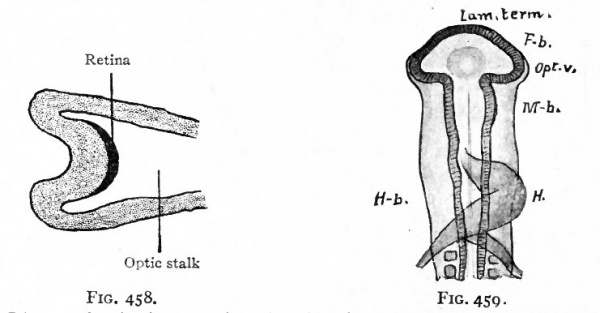
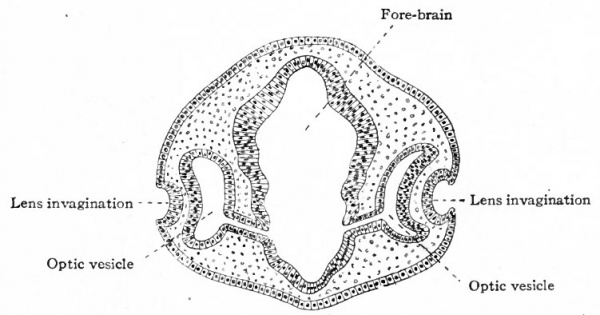
The first indication of eye formation is found in the chick at the beginning of the second day of incubation ; in the human embryo, at what has been estimated as about the second or third week. At this stage the neural canal is not yet completely closed in and its anterior end shows three primary brain vesicles (p. 440, Fig. 497). The anlagen of the eyes first appear as bilaterally symmetrical evaginations from the lateral walls of the fore-brain vesicle (Figs. 459 and 460), and are at first large in proportion to the brain vesicle itself. When first formed, the optic evagination opens widely into the fore-brain vesicle (Fig. 460, right side), but as the distal part of the evagination expands more rapidly than the proximal part, there soon results a spheroidal optic vesicle attached to the fore-brain by the narrow optic stalk (Fig. 460, left side). Through the latter the cavity of the optic vesicle and the cavity of the fore-brain are in communication. With the development of the hemispheres, that part of the brain to which the optic stalks are attached becomes the inter-brain (diencephalon).
Fig. 457. Diagram showing location of areas shown in Fig. 456 after the formation of the neural canal. (Modified from Lange)
Fig. 458. Diagram showing, location of the (dark) optic area (see Fig. 457) after the beginning of the formation of the optic cup and optic stalk. (Lange)
Fig. 459. Dorsal view of head of chick of 58 hours' incubation. Mihalkovics.
- Lam. term, lamina terminalis; Fb., fore-brain; Opt. v., optic vesicle; M. b., mid-brain; H.b., hind or rhombic brain; H., heart.
The Lens
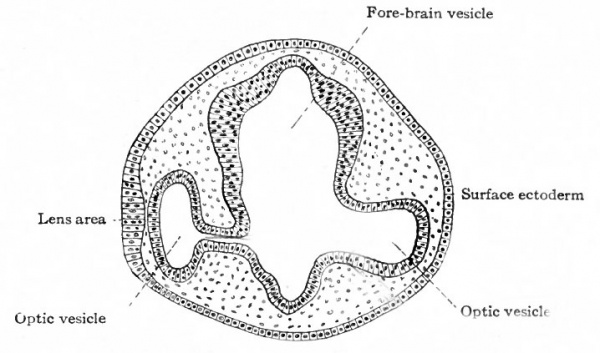
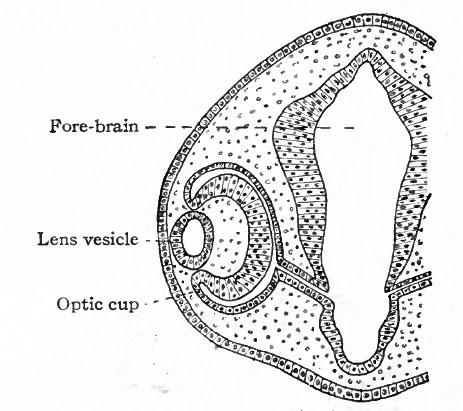
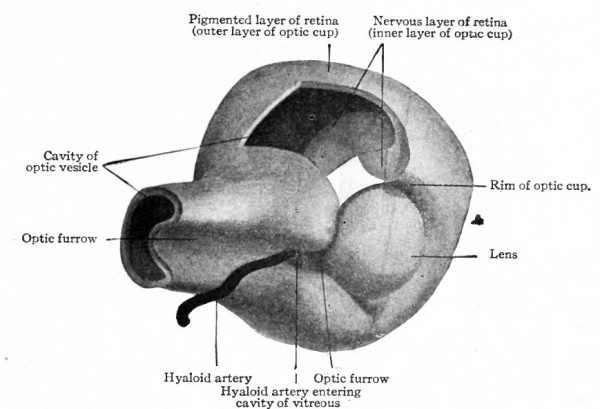
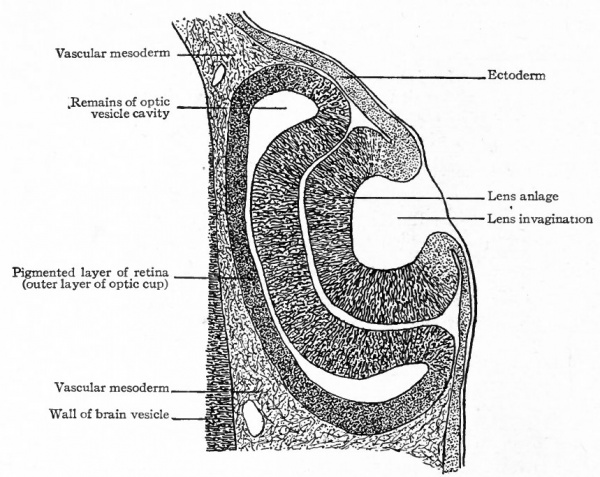
As each optic vesicle grows out toward the surface, its outer wall soon comes to lie just beneath the surface ectoderm. The cells of that portion of the ectoderm which overlies the optic vesicle next proliferate and cause a thickening of the ectoderm (Fig. 460, left side). This thickening of the ectoderm over the optic vesicle is apparent in the chick embryo of 36 hours incubation; in the human embryo it occurs about the third or fourth week and represents the first-step in the development of the crystalline lens. The thickened portion of ectoderm is known as the lens area (Fig. 460). The latter next becomes depressed against the outet surface of the optic vesicle forming a distinct lens invagination (Fig. 461). This becomes cup-shaped and then its edges come together and fuse, thus forming the lens vesicle (Fig. 462). At first the lens vesicle is connected with the surface ectoderm, but about the eighth week a thin layer of mesoderm grows in between the lens vesicle and the surface ectoderm, completely separating them (Fig. 463). The ingrowth of the lens vesicle against the outgrowing optic vesicle has the effect as though a small hard ball (the lens vesicle) had been pressed into a larger soft ball (the optic vesicle) (Fig. 464) . The lens vesicle pushes the outer wall of the optic vesicle in against the inner wall, the optic vesicle thus becoming transformed into the two-layered optic cup (Figs. 462, 463). Bonnet calls attention to the fact that the two processes, lens formation and the invagination of the optic vesicle to form the optic cup, are more or less independent and that it is not correct to describe the lens as actually pushing in the outer wall of the vesicle. As evidence of this is noted the fact that typical optic cup formation may occur in cases where no lens is developed. The optic cup when first formed is not a complete cup, for the invagination of the optic vesicle is carried over along the posterior surface of the optic stalk forming the choroidal fissure (Fig. 464, see also p. 545).
Fig. 460. Section through head of chick of two days' incubation. (Duval) The formation of the optic vesicle and stalk appears to be somewhat more advanced on the left than on the right.
Fig. 461. Section through head of chick of three days' incubation. (Duval)
Fig. 462. Showing somewhat later stage in development of optic cup and lens than is shown in Fig. 461. (Duval)
Fig. 463. Diagram of developing lens and optic cup. (Duval)
- The cells of the inner wall of the lens vesicle have begun lo elongate to form lens fibers. The epithelium over the lens is the anlage of the corneal epithelium. The mesodermal tissue between the latter and the anterior wall of the lens vesicle is the anlage of the substantia propria corneae.
The lens area is thicker at its center than at its periphery and when the center of the lens area becomes the bottom of the lens depression and later the posterior wall of the lens vesicle this greater thickness is maintained. In fact, the posterior wall of the vesicle becomes still thicker so that it projects into the cavity of the lens vesicle as an eminence (Fig. 465, g.). In the chick the lens vesicle is hollow. In man and in Mammals generally it is more or less filled with cells. These, however, degenerate and take no part in the formation of the permanent lens. Comparing the posterior with the anterior wall of the lens at this stage, the latter is seen to be composed of a single layer of cuboidal cells, the anlage of the anterior epithelium of the lens (Figs. 463, 465, g, h, i). This layer passes over rather abruptly into the posterior wall which consists of a single layer of greatly elongated lens cells, the anlagen of the lens fibers. The lens fibers continue to elongate until by the end of the second month they touch the anterior epithelium, thus completely obliterating the cavity of the lens vesicle (Fig. 467). A small cleft containing a few drops of fluid, the liquor Morgagni, may remain between the anterior epithelium and the lens fibers.
Fig. 464. Model showing lens and formation of optic cup. A piece has been removed from the upper part of cup to show the cavity of the optic vesicle and the position of the inner layer of the cup (nervous layer of retina). Bonnet.
When the lens fibers are first formed, the longest fibers are in the center and the fibers gradually get shorter toward the periphery of the lens where they pass over into the anterior epithelium (Fig. 465), As the lens develops, the peripheral fibers elongate more rapidly than the central, with the result that in the fully developed lens the central fibers are the shortest, forming a sort of core around which the now longer peripheral fibers extend in much the same manner as the layers of an onion (Fig. 467). The ends of the fibers meet on the anterior and posterior surfaces of the lens, along more or less definite lines which can be seen on surface examination and which are known as sutural lines. The lens fibers are at first all nucleated and as the nuclei are situated at approximately the same level in all the fibers, there results a so-called nuclear zone (Fig. 465, i). Later the nuclei disappear. The sutural lines become evident about the fifth month and mark the completion of the lens formation, although lens fibers continue to be formed throughout fcetal and in postnatal life, probably by proliferation and differentiation of the cells of the anterior epithelium, in the region where the latter pass over into the lens fibers. (The successive stages in the development of the lens are shown in Fig. 465.)
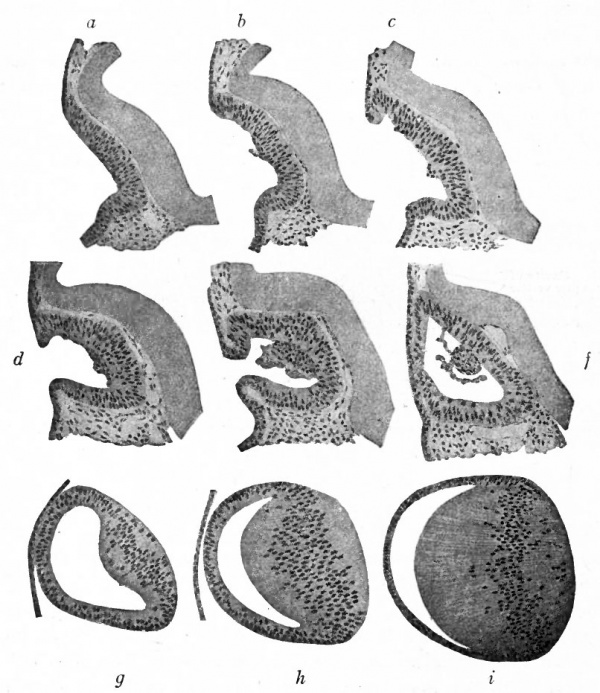
Fig. 465. Successive stages in the development of the lens in the rabbit embryo. Rabl.
- a, b, c, d, and e, are from embryos of from 11.5 to 12 days; f, at end of 12th day; g, during the 13th day; h, between the 13th and 14th days; i, from an embryo of 11 mm.
The lens capsule becomes differentiated during the third month. It is considered by some as derived from the lens epithelium and of the nature of a cuticular membrane, by others as a product of the surrounding connective tissue.
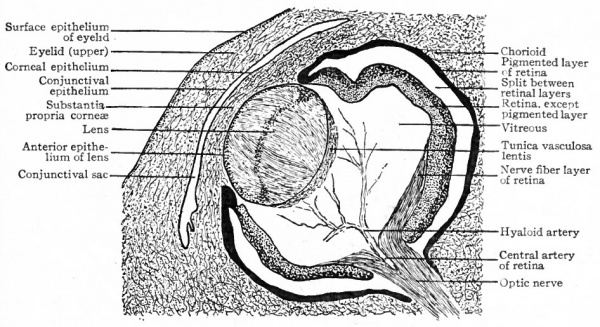
By the extension of mesodermic tissue in between the lens and the surface ectoderm, the lens becomes by the end of the sixth week completely surrounded by a layer of vascular connective tissue. This is known as the tunica lentis, and receives its blood supply mainly from the hyaloid artery (Fig. 467) which is a foetal continuation of the arteria centralis retina (p. 545). Branches from the hyaloid artery break up into a capillary network which covers both anterior and posterior surfaces of the lens. That part of the tunica vasculosa which covers the anterior surface of the lens is known as the membrana pupillaris. After the earlier and more rapid formation of lens fibers ceases, the hyaloid artery begins (about the seventh month) to undergo regressive changes, and at birth is normally absent. Rarely more or less of the tunica vasculosa fails to degenerate, and if the part which persists is the membrana pupillaris there results a malformation known as congenital atresia of the pupil.
The Optic Cup
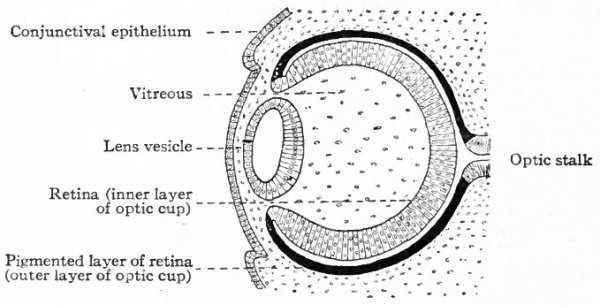
The way in which the optic vesicle becomes transformed into the optic cup has been partially described in considering the development of the lens (p. 536). The growing lens vesicle appears to push in the outer wall of the optic vesicle while at the same time the edges of the latter are extending around the lens vesicle, until what was originally the outer wall of the optic vesicle lies in apposition with the original inner wall, the cavity of the primary optic vesicle thus becoming completely obliterated (Fig. 466). In this way the optic vesicle is transformed into a two-layered thick-walled cup, the cleft between the two layers corresponding to the cavity of the primary vesicle. This cup is at first entirely filled with the developing lens (Fig. 466). As the cup increases in size faster than the lens, the contiguous walls of the cup and lens become separated, the cavity thus formed being the cavity of the vitreous humor (Fig. 467). There seems to be no question but that in Mammals a small amount of mesoderm at first separates the optic evagination from the lens area of the surface ectoderm. This apparently disappears, however, so that the two are in direct contact. It is still an open question w r hether a thin layer of mesoderm grows in between the edges of the cup and the lens at or just before the beginning of the formation of the vitreous. The lens now no longer fills the optic cup but lies in the mouth of the cup, while at the same time the margin of the cup is extending somewhat over its outer surface, w^here with the mesoderm it ultimately gives rise to the ciliary body and iris, and forms the boundary of the pupil. The remainder of the two-walled optic cup becomes the retina.
The Retina. Of the two layers which form the wall of the optic cup (p. 539) , the outer (away from the cavity) forms the pigmented layer, while the inner forms the remainder of the retina (Figs. 463, 467). Soon after the formation of the optic cup, it is possible to distinguish a boundary zone the future ora serrata between the larger posterior part of the retina or nervous retina and the smaller anterior non-nervous part which becomes the retinal portion of the ciliary body and iris. [ While the optic cup is forming, its two layers are both rapidly increasing in thickness by mitotic division of their cells. Especially is this true of the inner layer over that region which is to become the nervous retina, and it is the rather abrupt transition between the thicker nervous retina and the comparatively thin non-nervous anterior extension of the retina that forms the ora serrata.
Fig. 466. Section through optic cup and lens invagination of chick of fifty-four hours' incubation. Lange.
- Between the lens anlage and the pigmented layer of the retina is the broad inner layer of the optic cup, the anlage of the remainder of the retina.
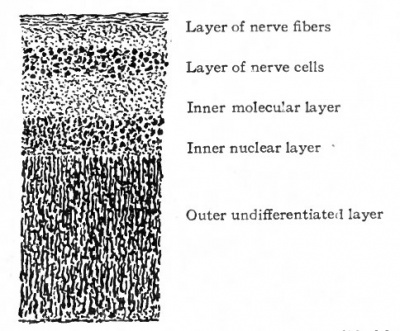
The invagination which gives rise to the two-layered optic cup thus differentiates what may be called the two primary layers of the retina, the pigmented layer, and a broad layer from which are to develop all the other layers of the retina. (Figs. 463, 467) . Further development consists in a gradual differentiation, within the broad layer; of the various retinal elements and consequent demarcation of the layers which constitute the adult retina. The next layer to differentiate is the innermost layer of the retina, or layer of nerve fibers. This appears during the sixth or seventh week as a thin, clear, faintly striated zone containing a few scattered nuclei. What remains of the original inner layer of the cup has now become a comparatively thick layer with numerous chromatic and actively dividing nuclei. It may be conveniently designated the primitive nuclear layer.
Fig. 467. Horizontal section through eye of human embryo of 13-14 weeks. Modified from Lange.
The similarity in development between the retina and wall of the neural tube is to be noted. Thus the layer of nerve fibers appears to correspond quite closely to the marginal layer of the central nervous system, while the primitive nuclear layer is probably homologous with the mantle layer (pp. 449, 455). There is a similar correspondence between the retina and the central nervous system in regard to their early cellular development, the retinal cells early showing a differentiation into neuroblasts and spongiobiasts (pp.449, 455).
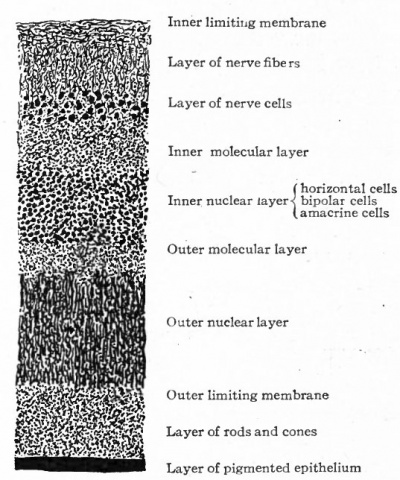
About the end of the eighth week the inner part of the primitive nuclear layer differentiates into the layer of eanzlion cetts (Fig. 468, h). These are large cells and with their processes constitute the third or proximal optic neurone. They can be first distinguished in the fundus of the cup and gradually extend to the ora serrata. They are the first of the cellular dements of the adult retina which can be definitely recognized as such. From each cell, two kinds of processes develop, dendrites, which ramify in this and in the more external layers of the retina, and an axone which grows toward the cavity of the eye and becomes a fiber of the layer of nerve fibers, whence it continues into the optic stalk as one of the fibers of the optic nerve. The layer of ganglion cells is thickest in an area situated somewhat lateral to the attachment of the optic stalk and known as the area centralis. It is distinguishable about the end of the fourth month. In the center of the area centralis the retinal layers become thin to form the fovea centralis which develops toward the end of foetal life. The macula lutea with its yellow pigment does not develop until after birth. The retina at this stage thus consists of four layers which from within outward are (i) the layer of nerve fibers, (2) the layer of ganglion cells, (3) the nuclear layer, (4) the pigmented layer (see Fig. 469).
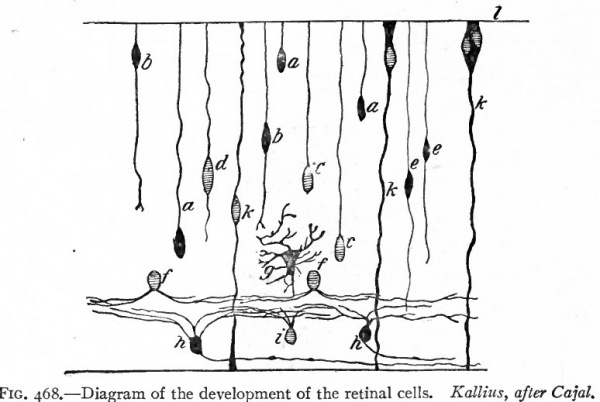
Fig. 468. Diagram of the development of the retinal cells. Kallius, after CajaL a, Cone cells in unipolar stage; fe, cone cells in bipolar stage; c, rod cells in unipolar stage; d, rod cells in bipolar stage; e, bipolar cells; i, amacrine cells; g, horizontal cell; h, ganglion cells; k, Muller's cells or fibers; l, external limiting membrane.
The further development of the retina consists largely of a differentiation of the cells of the nuclear layer. This is extremely complex and our knowledge of it meager. From the cells of this layer develop (i) the rod and cone cells, (2) the bipolar cells, (3) the tangential or horizontal cells, (4) the amacrine cells, (5) Muller's cells or fibers. The differentiation of these cells and their processes /also results in the demarcation of the following layers of the adult retina; (i) the ^ layer of rods and cones, (2) the outer limiting membrane, (3) the outer nuclear layer, (4) the outer molecular layer, (5) the inner nuclear layer, (6) the inner molecular layer, (7) the inner limiting membrane (see Fig. 470).
Muller's cells or the sustentacular cells (Fig. 468, k) develop from spongioblasts which lie toward the inner limit of the nuclear layer. This accounts for the location of the nucleated portions of Muller's cells. Processes of these cells grow toward both surfaces of the retina until they reach the positions of the future outer and inner limiting membranes where they are believed to spread out horizontally and unite to form these membranes. Other spongioblasts develop into other types of glia cells, mainly spider cells, which are most numerous in the layer of ganglion cells and in the layer of nerve libers.
The rod and tone cells are first recognizable as unipolar cellsjFig. 468,0, c}. The single process of each extends outward as far as the outer limiting membrane. About as soon as these cells are recognizable, a differentiation between the rod cells and the cone cells can be made by their reactions to the Golgi silver stain, the cone cells impregnating much more completely than the rod cells. Processes next grow out from the inner ends of the cells so that they become bipolar (Fig. 468, b, d) . Both rod and cone cells are at first distributed throughout the entire nuclear layer, but later they become arranged in a distinct layer just beneath the outer limiting membrane. Each cell next gives rise to or acquires at its outer end an expansion which extends through the outer limiting membrane into the pigmented layer. As the pigmented cells give off pigmented processes which extend inward among the outer ends of the rods and cones, the layer of retina just beneath the pigmented layer consists of the outer ends of the rod cells, the tips of the cone cells, and the extensions of the pigmented cells. The nucleated portions of the rod and cone cells form the outer nuclear layer. Though the layer of rods and cones and the outer nuclear layer present the appearance in haematoxylineosin stained specimens of two distinct layers, it is evident from their development and structure that they should be regarded as a single neuro-epithelial layer. The apparent separation into two layers is due to the interposition of the outer limiting membrane, through tiny holes in which the rod and cone cells extend. The inwardly directed processes of the rod and cone cells are their axones. These cells constitute the first or distal optic neurone.
Fig. 469. Vertical section through retina of a four months' human embryo. Modified from Lange.
The bipolar cells (Fig. 468, e), which with their processes constitute the middle or second optic neurone, also develop from cells of the nuclear layer and are probably bipolar at the time that the rod and cone cells are in the unipolar condition. Reference to the two bipolar cells shown in Fig. 468, e, e y shows that at this stage in their development their outwardly directed processes extend to the outer limiting membrane. These processes must either actually shorten or else fail to grow in length proportionately as the retina increases in thickness, for in the mature retina they end in relation with the centrally (inwardly) directed processes (axones) of the rod and cone cells. According as they are in relation with rod cells or cone cells, they are known as rod bipolars or cone bipolars. The retinal layer in which the axones of the rod and cone cells and the dendrites of the rod and cone bipolars intermingle is the outer, molecular layer of the adult retina. It is first distinctly recognizable as a molecular layer about the end of the fifth month (Fig. 470).
Fig. 470. Vertical section through retina of a five and one-half months' human embryo. Modified from Lange.
The development of the outer molecular layer separates the originally single nuclear layer into two layers, an outer composed of the nuclei of the rod and cone cells and an inner composed of the nucleated bodies of the rod and cone bipolars, of the horizontal cells (Fig. 468, g) and of the amacrine cells (Fig. 468, / and f), all of which can be recognized in Golgi specimens by the end of the seyegth month. The rod and cone bipolars and probably most of the other cells of the inner nuclear layer send their axones centrally to lie in contact with the dendrites and bodies of the ganglion cells.
With the development of the cells of the inner nuclear layer and their processes, there differentiates the inner molecular layer which separates the inner nuclear layer and the layer of ganglion cells. It consists mainly of ramifications of the dendrites and axones of cells the bodies of which lie in the inner nuclear layer and in the layer of ganglion cells. (Fig. 470.)
The Chorioid and Sclera
These develop wholly from the mesoderm. The way in which the mesoderm grows in between the lens and the surface and surrounds the optic cup has been described (p. 536). That part of the mesoderm lying immediately external to the retina develops very early a closemeshed capillary network. This appears before there is any definitely limited sclera and may be considered the anlage of the chorioid, Somewhat later the mesoderm which lies just to the outside of the chorioid takes definite shape as the external fibrous tunic of the eye or sclera.
The Vitreous
The manner in which the vitreous humor is formed has been the subject of much controversy and remains still undetermined. As already noted in describing the development of the lens (p. 555), the latter is at first in direct contact with the inner layer of the retina (Fig. 466) . The lens and the retina separate as the vitreous forms between them. During the development of the lens the arteria centralis retinae does not stop as in the adult, with its retinal branches, but continues across the optic cup as the hyaloid artery to end in the vessels of the tunica vasculosa lentis. Some investigators consider the vitreous a transudate from these blood vessels. As the chorioidal fissure closes, some mesodermic tissue is enclosed with the artery, and some investigators consider the vitreous a derivative of this mesoderm. In Birds the formation of the vitreous humor begins before either mesoderm or blood vessels have penetrated the optic cup, and Rabl suggests that the vitreous may be a secretion of the retinal cells. Bonnet describes a double origin of the vitreous, differentiating between a retinal vitreous and a mesoderm vitreous. According to Bonnet, the primary vitreous body begins its formation before the closure of the chorioidal fissure. This primary vitreous appears at the time of formation of the optic cup, is a fibrillated secretion of the retinal cells, and fills in the vitreous space with a feltwork of fine fibrils. With the formation of the optic cup and the closure of the chorioidal fissure this type of vitreous formation ceases and a secondary vitreous body formation takes place from the cells of the pars ciliaris retinas. This is also fibrillated and there develops at this time the so-called hyaloid membrane which closely invests the vitreous. Among the fibers of the vitreous body appears the vitreous humor. Up to this point the vitreous is entirely non-cellular. There next grow into it mesodermal cells which have reached the vitreous through the chorioidal fissure along with the hyaloid artery. To what extent these cells are used up in the formation of the blood vessels of the vitreous and to what extent they remain as connective tissue cells of the mature vitreous after the blood vessels have degenerated is not known.
As already noted, the vitreous is at first crossed by the hyaloid artery which supplies the developing lens (p. 539). As lens formation becomes less active the artery becomes less important and by the end of the third month begins to atrophy. At birth nothing remains of it, but in its former course the vitreous is somewhat more fluid than elsewhere and this is known as the hyaloid canal (canal of Cloquet).
The Optic Nerve
Referring to the description of the optic evagination it will be recalled that the optic vesicle maintains its connection with the brain by means of the optic stalk (p. 534) . The latter is hollow and connects the cavity of the optic vesicle with the cavity of the brain. When the invagination of the optic vesicle to form the optic cup occurs (p. 536, Fig. 464), the invagination is carried along the posterior surface of the optic stalk toward the brain, and just as the invagination of the optic vesicle results in the obliteration of the cavity of the vesicle, so the invagination of the optic stalk results in an obliteration of its lumen. In Mammals the invagination of the optic stalk extends only part way to the brain, to the point where the artery enters. The chorioidal fissure closes about the seventh week.
The optic stalk consists of supportive elements only, and serves as a track along which nerve fibers extend to connect the retina and brain. Nerve fibers appear in the optic stalk about the fifth week. They appear first around the periphery and apparently crowd the neuroglia nuclei toward the center, so that the stalk at this stage may be said to consist of a mantle layer and a marginal layer, apparently analogous to these layers in the retina and brain. The nerve fibers gradually invade the entire stalk so that by the end of the third month the stalk has become^ transformed into the optic nerve among the fibers of which the original supportive elements of the stalk are still represented by neuroglia cells.
Much difference of opinion has existed in regard to the origin of the optic nerve fibers, whether they are processes of retinal cells which end in the brain or processes of brain cells which end in the retina. It is now quite generally accepted that most of the fibers of the optic nerve are the axones of nenrnneg the cell bodies of which are situated in the ganglion cell layer of the retina. These axones pass centrally into the layer of nerve fibers, which they form, and converge toward the optic nerve. Through the latter they pass to their terminations in the external geniculate bodies, optic thalami and anterior corpora quadrigemina. According to Cajal and others, some centrifugal fibers are present in the optic nerve. These are processes of cells situated in the above-mentioned nuclei, and terminate in the retina. They are fewer in number and of later development than the centripetal fibers.
As the mesodermic anlagen of the chorioid and sclera are present before the nerve fibers begin to grow into the optic stalk, the fibers must pass through these two coats in their exit from the eye. There results the fenestrated crossing of the optic nerve by these two coats, known as the lamina cribrosa.
The optic nerve fibers are medullated but have no neurilemmae. They are supported by neuroglia. The connective tissue sheaths which enclose the optic nerve are direct extensions of the meninges. These structural peculiarities accord with the peculiarities already described in the development of the nerve. Attention has been called to the fact (p. m) that just as the retina should be considered a modified and displaced portion of the central nervous system of brain cortex so the optic nerve should be considered not as a peripheral nerve, but as analogous to a central nervous system fiber tract.
The Ciliary Body, Iris, Cornea, Anterior Chamber
Anteriorly where they come into relation with the lens and are so arranged as to admit light to the retina, all three coats of the eye are extensively modified. Thus the retina is continued anteriorly as the pars ciliaris retinae and pars iridica retinae, the chorioid as the stroma of the ciliary body and iris, the sclera as the cornea.
The Ciliary Body and Iris
Both primary retinal layers (the two layers of the optic cup) are continued anteriorly as the non-nervous retinal layer of the ciliary body and iris. The outer pigmented layer consists at first of several layers of pigmented cells, but later becomes reduced to a single layer of pigmented cells which do not, however, possess pigmented processes extending inward as do the analogous cells of the nervous retina. The abrupt transition at the ora serrata where the thick pars optica retinae passes over into the pars ciliaris retinae has been mentioned (p. 540) . The inner laver of the primitive retina (optic cup) extends over the ciliary body and iris as a single layer of cells. These remain non-pigmented over the ciliary body, but over the iris acquire pigment so that the two layers form the pigmented layer of the iris.
The mesodermic tissue which forms the stroma of the ciliary body and iris is derived from the mesoderm lying between the lens and the surface ectoderm. This separates into two layers enclosing between them the anterior chamber of the eve, and it is from the posterior of these two layers that mesodermic tissue extends into the ciliary body and iris. It is continuous with the mesoderm of the tunica vasculosa lentis. During the fourth month the ciliary body undergoes foldings to form the ciliary processes. These foldings at first involve also the iris, but the iris folds soon (end of fifth month) disappear, while the ciliary processes become more prominent.
Of the smooth muscle tissue found in the ciliary body and iris, the dilator and contractor pupillse are, according to Bonnet, derived from the cells of the pigmented layer of the retina, i.e., from ectoderm. The ciliary muscle, on the other hand, develops from mesoderm. These muscles become well developed during the seventh month.
The suspensory ligament of the lens, or zonula Zinnii, first appears about the end of the fourth month. , By some the fibers of the suspensory ligament are believed to differentiate from the vitreous, by others they are considered as derived from the pars ciliaris retinae. Spaces among the fibers of the ligament enlarge and coalesce to form the canal of Petit.
The Cornea
The way in which the mesoderm grows in between the lens vesicle and the surface ectoderm has been described (p. 536) . This mesoderm forms a thin almost homogeneous layer containing v^rv few cells. Later that part of the layer which lies against the lens becomes more cellular and vascular, so that it is possible to distinguish between an outer homogeneous non- vascular layer and an inner cellular vascular layer. The former is the anlage of the cornea. Between the two layers vacuoles appear and coalesce to form the anterior chamber of the eye or cavity of the aqueous humor. Subsequent growth of the iris subdivides this chamber into an anterior and a ^posterior portion. The chamber separates the cornea from the pupillary membrane portion of the tunica vasculosa lentis. Bounding the chamber anteriorly and so forming the posterior layer of the cornea there develops a single layer of flat cells, the so-called " endothelium" of Descemet. Over the surface of the cornea the ectoderm remains and gives rise to a stratified squamous epithelium four to eight cells thick, the anterior corneal epithelium. Just beneath the epithelium a layer of corneal tissue retains its original homogeneous character and forms the anterior elastic membrane or membrane of Bowman. The posterior elastic membrane or membrane of Descemet is usually considered a cuticular derivative of the u endothelium." Throughout the rest of the cornea substantia propria cornea cells develop, either by proliferation of the few cells originally present or from cells which grow in from the surrounding cellular mesoderm, and become arranged parallel to the surface as the fixed connective cells of the cornea.
The Eyelids. After the lens vesicle becomes separated from the surface ectoderm, the latter folds over above and below to form the first rudiments of the upper and lower eyelids. Each fold consists of a core of mesoderm and i a covering of ectoderm. From the mesoderm develop the connective tissue elements of the lids including the tarsal cartilage. From the ectoderm develop the epithelial structures of the lids, the epidermis, the eyelashes and the glands. The edges of the lids gradually approach each other and about the beginning of the third month the epithelium of the upper licTbecomes adherent to that of the lower, thus completely shutting in the eyeball. This condition obtains until just before birth.
The eyelashes develop in the same manner as other hairs (p. 417).
The Meibomian glands, glands of Moll and the lacrymal glands develop, during the period the lids are adherent, as solid cords of ectoderm which grow into the underlying mesoderm where they ramify to form the ducts and tubules. The anlagen of the ducts and tubules of these glands al'(t LllUb at fust SUkUfToi ils of cells, their lumina being formed later by a breaking down of the central cells of the cords.
At the inner angle of the conjunctiva there develops beneath the eyelid folds a third much smaller fold. This becomes the plica scmilunaris which in man is a rudimentary structure, but in many of the lower Vertebrates, especially Birds, forms a distinct third eyelid, the so-called nictitating membrane. A few hair follicles and sebaceous glands develop in a portion of this fold forming the lacrymal caruncle..
The Lacrymal Duct
At a certain stage in development, a groove bounded by the maxillary process and the lateral nasal process extends from the eye to the nose (Fig. 98). This is known as the naso-optic furrow. Tin- cvlodrrm (epithelium) lying along the bottom of this groove thickens about the sixth week and forms a solid cord of cells. As development proceeds and the parts close in, this cord of ectoderm becomes enclosed within the mesoderm, excepting at its ends where it remains connected with the surface ectoderm of the eye and nose, respectively. By a breaking down of the central cells of this cord a lumen is formed and the cord becomes a tube, the lacrymal duct. The primary connection of the laojgnalduct is with the upper lid, but while the lumen is being formed an offshoot grows out to the under eyelid to form the inferior branch of the lacrymal duct.
The Nose
The anlage of the organ of smell is apparent in human embryos of about three weeks as two thickenings of the ectoderm, one on each side of the nasofrontal process. To these thickenings the term olfactory placodes has been applied (Kupffer) . A little later (in embryos of about four weeks) , the placodes become depressed below the surface, the depressions themselves being the nasal pits or fossa (see p 120; also Fig. 87). The placodes. which are destined to give rise to the sensory epithelium, thus come into closer relation with the olfactory lobes of the brain (rhinencephalon) which represent outgrowths of the fore-brain (telencephalon) (see p. 471).
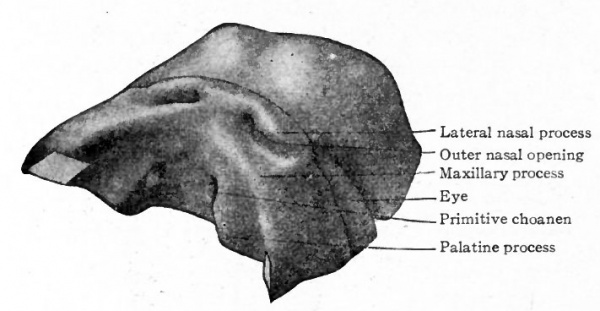
As described in connection with the development of the face, the lateral nasal process arises on the lateral side, the medial nasal process on the medial side, of each nasal pit (p. 120 et seq.; also Fig. 96). Of these processes, the lateral is destined to give rise to the lateral nasal wall and the wing of the nose, the medial to a part of the nasal septum (see p. 120). As development proceeds, the epithelium (ectoderm) of the nasal fossae grows still deeper into the subjacent mesoderm, the fossae thus becoming converted into the nasal sacs, which lie above the oral cavity. According to Hochstetter and Peter, the nasal sacs are not at first in communication with the oral cavity, but lie above, and are separated from it by a plate of tissue which gradually becomes thinned out along the deeper part of the sacs to form the bucco-nasal membrane (Hochstetter). Later (in embryos of 15 mm.), the bucco-nasal membrane ruptures and the deep ends of the sacs thus come to open into the mouth cavity, the openings being known as the primitive choanen. In front of the primitive choanen, the nasal passages (formerly the nasal sacs) are separated from the mouth cavity by a plate of tissue, known as the primitive palate (Fig. 471). The latter is produced by the fusion of the maxillary process with the lateral and medial nasal processes (see p. 121), the outer nares thus being somewhat separated from the border of the mouth.
Fig. 471. From a model of the anterior part of the head of a 15 mm human embryo. The lower jaws (mandibular processes) have been removed. Peter,
The further separation of the nasal passages from the o/al cavity has been described in connection with the development of the mouth (p. 286) and the development of the palatine processes of the maxillae. It may be repeated briefly, however, that from each maxillary process a horizontal extension grows across between the oral and nasal cavities until it meets and fuses with its fellow of the opposite side and with the nasal septum in the medial line, thus forming the palate which is continuous with the primitive palate mentioned above. (See Figs. 140 and 47 2.) In this way the nasal cavities or chambers become separated from the oral cavity, but remain in communication with the pharyngeal cavity through the posterior nares.
The nasal cavities increase enormously in size and the epithelial surface in extent, owing to (i) the formation of the palate alluded to above, (2) the development of the nasal concha which has been described on page 161, and (3) the development of accessory cavities maxillary, frontal and sphenoidal sinuses, which represent evaginations, so to speak, from the nasal cavities.
Probably correlated with the above-mentioned increase in extent of the nasal chambers is the fact that in lung-breathing Vertebrates the chambers have acquired a secondary function. In these forms the nose is not only an apparatus for receiving olfactory stimuli, but also serves to convey air to and from the lungs; it is in a sense a respiratory atrium. The sensory epithelium which the olfactory nerves supply is limited to relatively small areas in the superior conchae and nasal septum. Stratified columnar ciliated epithelium lines all other parts of the cavities.
Studies on the development of the olfactory nerve have led to diverse opinions, but the investigations of His and Disse go to show that the fibers are processes of cells derived from the thickened ectoderm or olfactory placodes. In human embryos of about four weeks some of the cells in the upper part of the nasal fossa become modified to form the neuro-epithelium. From the peripheral pole of each cell a short slender process grows out to the surface of the epithelium. From the opposite pole a slender process (the axone) grows centrally until it penetrates the olfactory lobe, where it ends in contact with the dendrites of the first central neurone of the olfactory tract. Most of these cells remain in the epithelial layer, but a few wander into the subjacent mesoderm and become bipolar cells which resemble the bipolar cells of the embryonic posterior root ganglia (p. 472). Other epithelial cells of the nasal fossa are converted into the sustentacular cells of the olfactory areas.
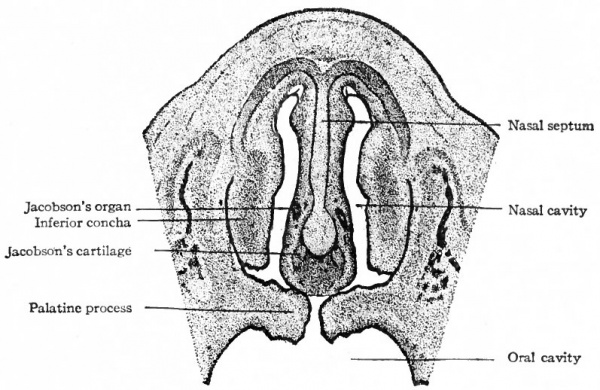
Fig. 472. From a section through the head of a human embryo of 28 mm. Showing the nasal septum, the nasal cavities, the oral cavity, and the palatine processes. Peter.
Jacobson's organ arises at the beginning of the third month as a small outpocketing of the epithelium on the lower anterior part of the nasal septum (Fig. 472). This evagination grows backward as a slender sac along the nasal septum for a distance of several millimeters and ends blindly. In the adult the sac degenerates and often disappears. In some of the lower Mammals Jacobson's organ develops to a greater degree, and some of the epithelial cells send out processes which pass to the olfactory lobes.
The Ear
The ear of higher Vertebrates consists of three parts the internal, middle, and external. Of these, the internal is the sensory portion proper and, so far as the epithelial elements are concerned, is of ectodermal origin, but secondarily becomes embedded in the subjacent mesoderm. It constitutes a complicated and highly specialized structure for the reception of certain stimuli that are to be conveyed to the central nervous system. From a functional standpoint it may be divided into the portion composed of the semicircular canals and their appendages, which is concerned in receiving and transmitting stimuli destined or the static and equilibration centers in the central nervous system, and the cochlear portion, which is concerned in receiving and transmitting auditory stimuli. The middle and outer ear represent modified portions of the most cranial of the branchial arches and grooves, and constitute an apparatus for conducting sound waves to the cochlear portion of the inner ear.
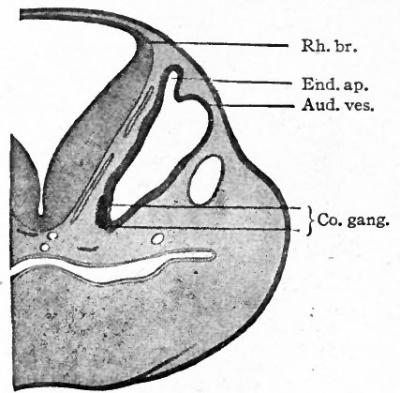
Fig. 473. Half of a transverse section through the region of the developing ear of a sheep embryo of 13 mm. Bottcher.
- Aud. ves., Auditory vesicle; Co. gang., cochlear ganglion; End. ap., endolymphatic appendage; Rh.br., rhombic brain.
The Inner Ear
In embryos of 2 to 4 mm., the ectoderm becomes somewhat thickened over a small area lateral to the still open neural groove in the region of the future hind-brain. This thickening is often spoken of as the auditory placode (see p. 469). Owing to more rapid growth of the cells in the deeper layers of the placode, it soon becomes converted into a cup-shaped depression which is known as the auditory pit. The edges of the pit fold in and fuse and the pit thus becomes the auditory vesicle (otocyst), which finally becomes constricted from the parent ectoderm and lies free in the subjacent mesoderm (Fig. 473). At this stage (embryos of 4 to 5 mm.) the auditory vesicle is an oval or spherical sac the wall of which consists of two or three layers of undifferentiated epithelial cells. It lies against the neural tube and is connected with the latter by the acoustic ganglion (Fig. 474, a). About the same time an evagination appears on the dorsal side of the auditory vesicle, forming the anlage of the endolymphatic appendage (Fig. 474, a, b, c). The evagination continues to elongate and comes to form a club-shaped structure, the distal end of which becomes flattened to form the endolymphatic sac, the narrower proximal portion constituting the endolymphatic duct (Fig. 474 a-w). The epithelium, which at first consisted of two or three layers of cells, becomes reduced to a single layer. In the chick the endolymphatic appendage is formed out of the original union between the ectoderm and the auditory vesicle (Keibel, Krause). In Reptiles and Amphibia (Peter, Krause) and in man (Streeter), on the other hand, this appendage develops independently of the union, appearing on the dorsal side of the seam of closure in the auditory vesicle.
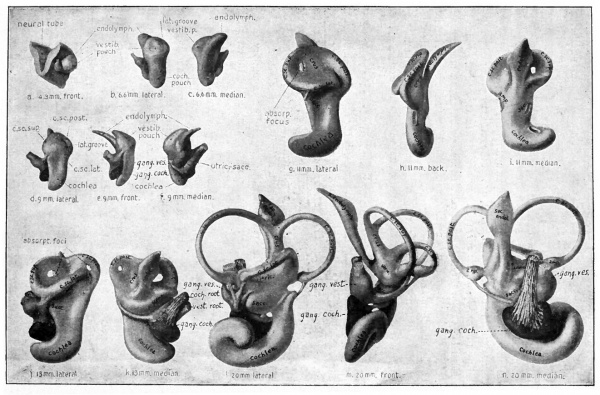
Fig. 474. Different views (front, lateral and median) of reconstructions of the developing membranous labyrinth and acoustic nerve in human embryos of successive stages, the view and length of the embryo being indicated under each figure. Streeter.
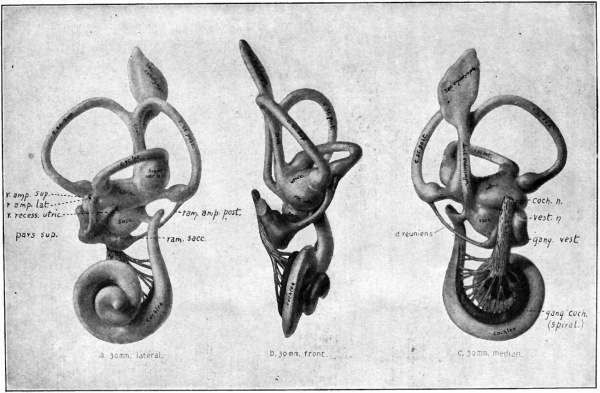
Fig. 475. Lateral, front and median views of reconstructions of the membranous labyrinth and acoustic nerve in a human embryo of 30 mm. Streeter.
- In the figure on the left, the pars superior (pars sup.) of the vestibular nerve includes the three branches named above it; see also table on page 559.
In embryos of about 6 mm. the auditory vesicle (apart from the endolymphatic appendage) becomes differentiated into two portions or pouches a bulging, triangular one above, which is connected with the endolymphatic appendage, and a more flattened one below. The former is the vestibular pouch, the latter the cochlear pouch (Fig. 474, b-f). Between the two is a portion of the vesicle which is destined to give rise to the saccule and utricle, and which may be called the atrium (Streeter). Properly speaking, the atrium is a division of the vestibular pouch. The cochlear pouch is phylogenetically a secondary diverticulum which develops from the atrium, appearing first in the lowest landinhabiting Vertebrates (Amphibia).
As mentioned above, the vestibular pouch early assumes the form of a triangle, with the apex toward the endolymphatic appendage. The three borders of the triangle form the anlagen of the semicircular canals and bear the same interrelation as the latter. At the same time a vertical groove (the lateral groove) appears between the anlage of the posterior canal and the posterior end of the lateral canal (Fig. 474, b, d).
The formation of the semicircular canals is shown in Fig. 474, g-k. The edges of the triangular vestibular pouch expand and become more or less crescentic in shape. The two walls in the concavity of each crescent come together and then break away (Fig. 474, g, j, absorp. focus), thus leaving the rim of the crescent as a canal attached at its two ends to the utricle. The breaking away affects first the superior, then the posterior, and finally the lateral canal. During these gross changes the epithelium becomes reduced to a single layer of cells.
At one end of each canal an enlargement appears to form the ampulla, as shown in Fig. 474, /, m, n, and Fig. 475. a. 6. c.
The utricle and saccule represent divisions of the portion of the vestibular sac which is known as the atrium, and into which the endolymphatic appendage and cochlea open (see p. 553). In embryos of about 20 mm. a horizontal constriction begins to divide the atrium into an upper utricular portion, into which the semicircular canals open, and a lower saccular portion (Fig. 474, /, m). The constriction begins on the side opposite the endolymphatic appendage and gradually extends across the atrium until it finally divides the opening of the endolymphatic appendage into two parts (Fig. 475, a, b, c). One of these parts opens into the utricle, the other into the saccule, the two parts together constituting the utriculo saccular duct.
As stated before, the two- or three-layered epithelium of the earlier stages becomes reduced to a single layer. The cells of this layer are low cuboidal, with the exception of those over small areas in the ampullae, in the saccule, and in the utricle. Over an elongated area in each ampulla (crista ampullaris), a round area in the saccule and another in the utricle (macula acusticd), the epithelium becomes high columnar, some of the cells developing cilia on their free borders ("hair cells," neuro-epithelium) , the others becoming the sustentacular cells. These areas are the end-organs of the vestibular nerve (see p. 469) .
As already mentioned, the cochlear pouch appears as an outgrowth from the lower side of the atrium (see also Fig. 474, b-f) . The pouch becomes somewhat flattened, and, as it continues to grow in length, becomes coiled like a snailshell (Fig. 474, g-n; Fig. 475, a-c). This first formed coiled structure is the cochlear duct, or scala media. At the same time, it becomes distinctly marked off from the lower part of the atrium (now the saccule) by a constriction, the constricted portion forming the ductusr reuniens (Fig. 474, l-n; Fig 475, a-c).
All the structures thus far considered are at first closely invested by mesoderm. Later, this portion of the mesoderm gives rise to special tissues, and, in the region of the cochlear duct, to the scala vestibuli and scala tympani. The cells immediately around the vesicle proliferate and a dense fibrous layer is formed; outside of this fibrous layer the tissue becomes gelatinous; outside of this again another fibrous layer is formed, around which cartilage develops. The inner fibrous layer gives rise to the connective tissue that supports the epithelial lining of the vesicle. The gelatinous layer degenerates to form a fluid known as the perilymph, the space containing the fluid being the perilymphatic space. The outer fibrous layer becomes the perichondrium later the periosteum when the cartilage is replaced by the petrous portion of the temporal bone.
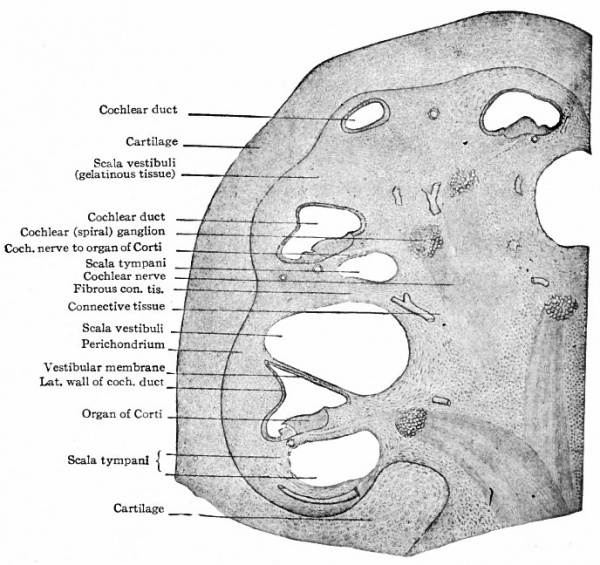
Fig. 476. Section through the developing cochlea of a 90 mm cat embryo. Bottcher.
In the cochlear region the conditions are somewhat modified. Here the gelatinous layer does not form a complete covering for the cochlear duct, but is interrupted along two lines, (i) Laterally the fibrous layer lying next the cochlear duct is fused with the perichondrium (outer fibrous layer) (Fig. 476), (2) Medially the inner fibrous layer is fused with the perichondrium of a shelf-like process of cartilage which later ossifies to form the bony spiral lamina (Fig. 476). By these two partitions the cochlear perilymphatic space is separated into two spiral compartments which communicate only at the apex of the cochlea. The larger of these compartments, the scala vestibuli, communicates with the perilymphatic space around the utricle and saccule. The wall separating the scala vestibuli and cochlear duct becomes thinned out to form the vestibular membrane (of Reissner). The smaller compartment, the scala tympani, remains separated from the cavity of the middle ear by a thin membrane which closes the fenestra cochlea (rotunda) . In the wall between the scala tympani and the cochlear duct the organ of Corti develops (see below). A membrane, similar to that closing the fenestra cochleae, occurs between the cavity of the middle ear and the utricle, closing the fenestra vestibuli (ovalis). As alluded to above, the organ of Corti develops from the wall of the cochlear duct between the latter and the scala tympani (Fig. 476). The epithelial cells of the cochlear duct in this region become high columnar and arranged in two ridges which extend throughout the entire length of the duct. The cells of the ridge nearer the axis of the cochlea give rise to the membrana tectoria. Whether this is accomplished by cuticular secretion of the cells or by the fusion of long hair-like processes that grow from their free borders is not known. The cells of the outer ridge become differentiated into four groups. Those of the outer group (next the cells that give rise to the membrana tectoria) develop into the inner hair cells; those of the next group form the pillar cells; those of the third group differentiate into the outer hair cells; and those of the fourth (outer) group give rise to Hensen's cells. The hair cells, as the name indicates, develop delicate hair-like processes on their free borders, and, since the peripheral processes of the spiral (cochlear) ganglion cells end around them, are considered as the sensory cells of the cochlea, or auditory receptors (see p. 469) .
The Acoustic Nerve
The acoustic ganglionic mass is at first closely associated with the geniculate ganglion (ganglion of the facial (VII) nerve), the two together often being spoken of as the acustico-facialis ganglion (see also p. 508) . This lies in close contact with the anterior wall of the auditory vesicle when the latter is first constiicted from the ectoderm. The origin of the ganglion has not been traced in Mammals, but in cow embryos the geniculate has been seen to be connected with the ectoderm at the dorsal end of the first branchial groove (Froriep). The acoustic ganglion probably belongs to the lateral line system (Kupffer) (see also p. 430) .
Although the geniculate and acoustic ganglia are at first closely associated, each pursues an independent course of development. The description here will be confined to the acoustic. As already mentioned, this lies in close apposition to the side of the neural tube and the auditory vesicle and just anterior to the latter (Fig. 474, a). At a very early stage (embryos of 6-7 mm.), the mass shows a differentiation into two parts a dorsal one, the future vestibular ganglion, and a ventral one, the future cochlear (spiral) ganglion (Fig. 474, b, c). The ganglion cells become bipolar (see p. 469) , and, as is peculiar to the cells of the acoustic ganglia, remain in this condition. One process of each cell grows centrally to form a root fiber of the acoustic nerve, which terminates in contact with dendrites of neurones in certain nuclei in the central nervous system. The fibers from the cells of the vestibular ganglion form the vestibular root, those from the cells of the cochlear ganglion form the cochlear root. The other process grows peripherally and penetrates the wall of the auditory vesicle to enter into relation with certain cells that differentiate from the epithelial lining of the vesicle.
The peripheral processes of the vestibular ganglion cells come into relation with specialized cells (hair cells) in the ampullae of the semicircular canals
(crista ampullaris) and in the saccule and utricle (macula acustica) (see p. 556). The nerve itself becomes divided into certain branches, as indicated in the following table (Streeter). The peripheral terminations of the various branches are indicated in parentheses. Compare with Fig. 474, /, m, n, and Fig. 475, a, b, c.
The vestibular ganglion cells, instead of remaining in a compact mass, come to form two fairly distinct masses in the course of the nerve (Fig. 475, a, b, c). One of these apparently is connected with the pars inferior, the other with the pars superior.
The cochlear ganglion cells at an early stage become closely associated with the developing cochlear duct and, as the latter forms a spiral, are carried] along with it. They thus come to form an elongated group of cells extending throughout the entire length of the cochlea (whence the name, spiral ganglion) (Fig. 474, j-n; Fig. 475, a-c). Consequently, the peripheral processes of these cells, which terminate in connection with the hair cells of the organ of Corti, are comparatively short. The central processes are naturally longer and form the cochlear nerve root which is twisted like a rope in part of its course (Fig. 475, c).
The Middle Ear. The cavity of the middle ear develops from the upper (dorsal) part of the first inner branchial groove. The epithelial lining of the cavity is thus of course derived from entoderm, and the other structures (auditory ossicles, etc.) from the adjacent mesoderm.
It has been stated elsewhere that the mesoderm in the first and second branchial arches gives rise, among other things, to certain skeletal elements. In the first arch there develops a rod of cartilage, known as Meckel's cartilage, which extends from the symphysis of the lower jaws to the region of the upper part of the first inner branchial groove (p. 164; Figs. 136, 139, 142). The proximal end of the cartilage becomes constricted to form two masses which constitute the anlagen of the malleus and incus (Figs. 135 and 136). In the second arch there develops a rod of cartilage which forms the lesser horn of the hyoid bone, the stylohyoid ligament, and the styloid process (Figs. 136, 139, .42). In close relation to the dorsal end of the styloid process, in the mesoderm destined to give rise to the periotic capsule, a mass of cartilage appears which is destined to give rise to the stapes (except the base?). It has not been fully determined whether the stapes is actually a derivative of the cartilage of the second arch or of the mesenchyme near its dorsal end. It has been suggested that the base of the stapes is of intramembranous origin and that the rest of the bone is derived from the cartilage of the second arch. Its close association with the cartilage of the second arch possibly indicates its phylogenetic origin from the latter.
At first the auditory ossicles are embedded in the mesoderm dorsal to the first inner branchial groove, that is, dorsal to the cavity of the middle ear. As development proceeds, the mesoderm is converted into a spongy tissue which finally degenerates. At the same time the ear cavity enlarges and wraps itself, as it were, around the ossicles. The latter thus come to lie within the cavity of the tympanum, but are covered by a layer of epithelium (entoderm) which is continuous with that lining the cavity.
Toward the end of foetal life, outgrowths from the cavity of the tympanum begin to invade the temporal bone. This process continues for some time after birth and results in the formation of cavities within the mastoid part of the temporal bone. These cavities are the mastoid cells, the epithelial lining of which is continuous with that of the tympanic cavity.
The Eustachian tube represents the lower (ventral) portion of the diverticulum which forms the cavity of the tympanum. In other words, as the dorsal part of the first inner branchial groove enlarges to form the cavity of the middle ear, the narrow part of the groove, just ventral to the cavity, persists as a communication between the latter and the pharynx.
The Outer Ear. The outer ear is formed from the dorsal part of the first outer branchial groove and the adjacent portions of the first and second arches (see Fig. 87). The ventral part of the groove flattens out and disappears. The dorsal part becomes deeper to form a funnel-shaped depression (during the second month ; Fig. 90) . From the deeper part of the funnel a solid mass of ectoderm grows inward until it comes into relation with the mesoderm immediately around the developing cavity of the tympanum, or, more specifically, the mesoderm surrounding the handle of the malleus. Here it spreads out into a disk-like mass. About the seventh month, the disk splits into two layers. The inner layer, which is separated from the epithelium of the middle ear by a thin sheet of mesoderm, becomes the outer layer of the tympanum. The tympanum is thus composed of an inner (entodermal) and an outer (ectodermal) layer, with a small amount of mesoderm between. From its mode of development, the tympanum may be considered in a sense as the wall which separates the first inner from the first outer branchial groove.
The split in the ectodermal disk (see above) gradually extends outward, invading the solid ectodermal in vagina tion until it finally unites with the bottom of the funnel-shaped depression on the surface, thus forming the external auditory meatus.
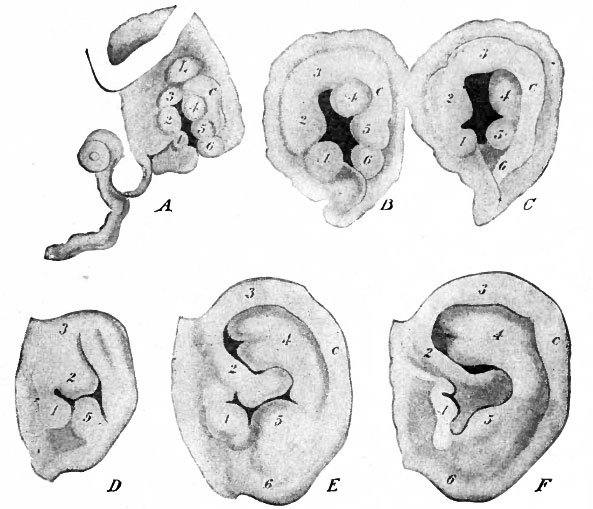
The external ear (or auricle) is derived from the portions of the first and second branchial arches surrounding the dorsal part of the first outer branchial groove (see Figs. 85, 87, 90, 91). About the end of the fourth week, the caudal border of the first arch exhibits three small elevations or tubercles (Fig. 477, A, 1-3), the cranial border of the second arch the same number (Fig. 477, A, 4-6). A groove, extending down the middle of the second arch, marks off a ridge (c) lying caudal to the three tubercles. The ventral tubercle (i) of the first arch gives rise to the tragus. The middle tubercle (5) of the second arch develops into the antitragus. The middle and dorsal tubercles (2 and 3) of the first arch unite with the ridge (c) on the second arch to form the helix. The dorsal tubercle (4) of the second arch gives rise to the anthelix. The ventral tubercle (6) of the second arch produces the lobule. It should be noted that in the third month the dorsal and caudal portions of the helix are bent forward and conceal the anthelix.
Fig. 477. Stages in the development of the external ear (auricle). A, Embryo of n mm.; B, of 13.6 mm.; C, of 15 mm.; D, foetus at the beginning of the 30! month; E, foetus of 8.5 cm.: F, foetus at term. For explanation of numerals, see text. His, McMurrich.
Anomalies
Malformations of the nose have been alluded to in connection with hare lip, cleft palate, etc., on page 212, and are also discussed in the chapter on teratogenesis (XX). Malformations affecting the eye (cyclopia, microphthalmia, etc.) and the ear (synotia, etc.) are dealt with in the chapter on teratogenesis.
- Next: Foetal Membranes
References for Further Study
The Eye
GALLENGA: Entwickelung des Auges. Encyklopadie der Augenheilkunde, Lief. 6 and 7, 1902.
HOLDEN: An Outline of the Embryology of the Eye, New York, 1893.
VON KOLLIKER: Die Entwicklung und Bedeutung des Glaskorpers. Zeitschr. fur wissensch. Zoolog., Bd. LXVI, 1904.
LANGE, O.: Einblicke in die embryonale Anatomie und Entwicklung des Menschenauges. 1908.
RABL, C.: Ueber den Bau und Entwickelung der Linse. Zeitschr. fur wissensch. Zool., Bd. LXII and LXV, 1898; LXVII, 1899.
RAYMON Y CAJAL.: Nouvelles contributions a 1'etude histologique de la retine. Jour, de VAnat. et de la Physiol, Vol. XXXII, 1896.
ROBINSON. A.: On the Formation and Structure of the Optic Nerve and its Relation to the Optic Stalk. Jour, of Anat. and Physiol., Vol. XXX, 1896.
VON SPEE: Recherches sur 1'origine du corps vitre. Arch, de Biol., Vol. XIX, 1902.
The Nose
BEARD, J.: Morphological Studies. The Nose and Jacobson's Organ. Zool. Jahrbuch, Bd. Ill, 1889.
DISSE, J.: Die erste Entwickelung der Riechnerven. Anat. Hefte, Bd. IX, 1897.
His, W.: Beobachtungen zur Geschichte der Nasen- und Gaumenbildung beim menschlichen Embryo. AbhandL d. math.-phys. Klasse Ko'nig. Sachs. Gesellsch. d. Wissensch. , 1901.
HOCHSTETTER, F.. Ueber die Bildung der primitiven Choanen beim Menschen. Verhandl. d. anat. Gesellsch., Bd. VI, 1892.
VON MIHALKOWICZ, V.: Nasenhohle und Jacobsonsches Organ. Eine morphologische Studie. Anat. Hefte, Bd. XI, 1898.
PETER, K.: Die Entwickelung des Geruchsorgans und Jacobson'schen Organs in der Reihe der Wirbeltiere. In Hertwig's Handbuch d. vergleich. u. experiment. Entwickelungslehre d. Wirbeltiere, Bd. II, Teil II, 1901.
The Ear
BAGINSKY, B.: Zur Entwickelung der Gehorschnecke. Arch.f. mik. Anat., Bd. XXVIII, 1886.
BOETTCHER, A.: Ueber Entwickelung und Bau des Gehorlabyrinths. Verhandl. d. Kais.Leop.-Carol. Akad., Bd. XXXV, 1869.
BROMAN, I.: Die Entwickelungsgeschichte der Gehorknochelchen beim Menschen. Anat. Hefte, Bd. XI, 1898.
FUCHS, H.: Bemerkungen iiber die Herkunft und Entwickelung der Gehorknochelchen bei Kaninchen-Embryonen. Arch.f. Anat. u. Phys., Anat. Abth., Suppl., 1905.
HENSEN, V.: Zur Morphologic der Schnecke. Zeitschr. f. wissensch. Zool., Bd. XIII, 1863
His, W.: Zur Entwickelung des Acusticofacialisgebiets beim Menschen. Arch.f. Anat. u. Phys., Anat. Abth., Suppl., 1899.
KRATJSE, R.: Entwickelungsgeschichte des Gehororgans. In Hertwig's Handbuch d. vergleich. u. experiment. Entwickelungslehre d. Wirbeltiere, Bd. II, Teil II, 1902.
Streeter GL. On the development of the membranous labyrinth and the acoustic and facial nerves in the human embryo. (1906) Amer. J Anat. 6:139-165.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Text-Book of Embryology: Germ cells | Maturation | Fertilization | Amphioxus | Frog | Chick | Mammalian | External body form | Connective tissues and skeletal | Vascular | Muscular | Alimentary tube and organs | Respiratory | Coelom, Diaphragm and Mesenteries | Urogenital | Integumentary | Nervous System | Special Sense | Foetal Membranes | Teratogenesis | Figures
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 5) Embryology Book - Text-Book of Embryology 18. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_Text-Book_of_Embryology_18
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G