Book - Manual of Human Embryology 14-1
| Embryology - 1 May 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Streeter GL. The Development of the Nervous System. (1912) chapter 14, vol. 2, in Keibel F. and Mall FP. Manual of Human Embryology II. (1912) J. B. Lippincott Company, Philadelphia.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
XIV. The Development of the Nervous System
By George L Streeter, Ann Arbor, Mich.
The entire nervous system, as will presently be described, has a common origin. It is derived from an anlage that is very early differentiated from the ectoderm as the neural or medullary plate. The description of its development will be taken up under the following headings: 1, the histogenesis of the nervous tissues; 2, the development of the central nervous system; 3, the development of the peripheral nervous system; and, 4, the development of the sympathetic nervous system.
I. Histogenesis of Nervous Tissue
In Chapters III, IV, and V the segmentation of the ovum and the grouping of its cells in the form of three germ layers have already been described. It has also been described how a portion of the ectoderm becomes thickened and differentiated into the elongated axial neural plate and neural groove, and how eventually the elevated edges of this groove come together and fuse, thereby converting it into the neural tube. It is the anterior portion of this neural tube that is enlarged and converted by constrictions into the three primary cerebral vesicles from the thickened walls of which the brain is derived. The caudal portion of the neural tube remains nearly uniform in diameter and forms the spinal cord. The lumen of the caudal portion becomes converted into the central canal of the cord, and the enlarged cavities of the vesi cles of the anterior part of the tube form the several ventricles of the brain. The nerves which serve to connect the central nervous system with the periphery either sprout out from the wall of the neural tube or from the ectodermal cellular crest that is attached along its dorsal seam. Thus, if we except the mesoderm's elements that subsequently penetrate the neural tube, accompanying the invasion of blood-vessels into its substance, we may say that all the essential nervous tissues of the body are derived from the thickening and differentiation of the walls of the neural tube and its ganglionic crest, and hence are ectodermal.
Development of the Walls of the Neural Tube
In the absence of suitable human material our information regarding the changes that occur early in the differentiation of the wall of the neural tube is based on the chick, rabbit, and pig, notably through the investigations of His, Cajal, Schaper, and more recently Hardesty. The essential feature in this development, as can be seen in Fig. 1, consists in the conversion of the original single layer of cuboidal ectodermal cells into a thick wall whose elements are arranged in the form of three definite layers or zones. These three zones constitute respectively the anlages of the ependyma, the gray substance, and the white substance, the individual cells being accordingly differentiated into supporting cells and nerve-cells proper.
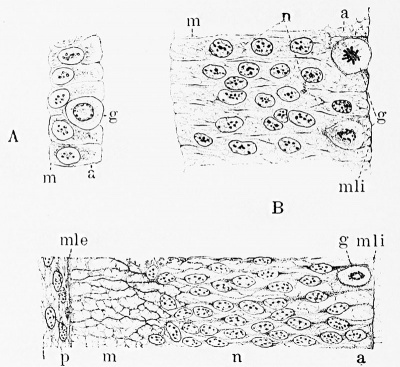
Fig. 1. Three early stages in the development of the wall of the neural tube, showing the conversion of the single layer of discrete cells into a richly nucleated syncytial framework. A, medullary plate of embryo rabbit just before closure of neural tube; B, similar section of 5 mm. pig embryo just after closure of neural tube; C, wall of neural tube of 10 mm. pig embryo; a, ependymal layer; g, germinal cell; m, marginal layer; mle and mli, external and internal limiting membranes; n, mantle or nuclear layer; p, mesoderm. (After Hardesty.)
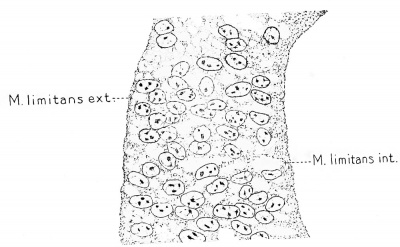
In Fig. 1, A, is shown the single layer of ectodermal cells which constitutes the original nenral plate. It is to be noted that they consist of individual cells, distinctly outlined and definitely arranged. During the closure of the neural tube they proliferate and the cell boundaries partially disappear, and there thus results a thick wall of fused cells, a compact nucleated protoplasmic syncytium as seen in Fig. 1, B. As the growth continues the syncytium expands into a looser framework or myelospongium and its outer and inner margins are condensed in a continuous sheet to form the external and internal limiting membranes, as seen in Fig. 2, which is taken from a human embryo. In Fig. 1, C, which shows the appearance at the end of the first month, the myelospongium consists of radially arranged cellular strands still united in a
Fig. 2. Wall of the neural tube in a human embryo about two weeks old, showing its syncytial character. This stage is between that of B and C, Fig. 1. (After His.) syncytium by numerous branching processes.
Further examination shows that, owing to the grouping of the nuclei, the wall may be subdivided into three primary zones or layers: (1) the ventricular or ependymal layer; (2) the intermediate or nuclear or mantle layer; and (3) the outer or non-nuclear or marginal layer (Randschleier). It may be added that the term nuclear layer is applied in the early stages, while the boundary between them is indistinct, both to the ependymal and mantle layers, thus contrast ing them to the marginal or non-nuclear layer. In this sense one can say that the wall at first consists of two layers, nuclear layer and non-nuclear layer, and that later the former or nuclear layer becomes differentiated into the ependymal and mantle layers. Here the term nuclear layer is used as synonymous with mantle layer, which is its chief derivative. The ependymal layer consists of a single layer of elongated nuelei connected with the internal limiting membrane by strong protoplasmic processes. Among these are seen prominent mitotic nuclei, the so-called germinal cells shown in Fig. 1, g, and Fig. 3. According to Hardesty, the germinal cells are found in greatest number in embryos (pig) between 10 and 20 mm. long, and gradually disappear, ceasing altogether at 40 mm. The nuclear or mantle layer consists of nucleated branching strands of the myelospongium forming a radially arranged protoplasmic framework. Rapidly proliferating nuclei are embedded in the strands and about them the protoplasm is becoming more condensed and granular. Mitotic nuclei are frequently found in this layer after the disappearance of the germinal cells of the ependymal layer, and it is suggested by Schaper (1897) that they are a proliferation phenomenon: Those that persist until late stages are regarded by him as material for regeneration processes. Whether the proliferation of the nuclei of the mantle layer is always mitotic or whether it is to some extent amitotic remains to be determined. The mantle layer may be considered as the anlage of all gray substance. The marginal layer forms the anlage of the white substance, and through its meshes pass all the principal longitudinal fibre tracts of the cord and brain. For a considerable period it remains devoid of nuclei and consists simply of the network of the myelospongium.
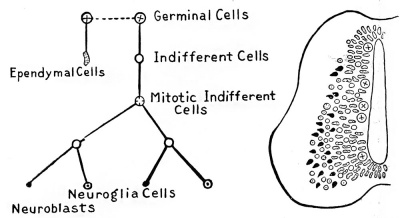
Fig. 3. Diagram showing the differentiation of the cells of the wall of the neural tube and the theoretical derivation of the ependymal cells, neuroglia cells, and neuroblasts. (After Schaper.)
The further steps by which this comparatively simple wall of three primary layers becomes subsequently converted into the complicated spinal cord and brain will be considered later. We will only consider at this time the histogenesis of its elements.
The cells, which in Fig. 1, C, are still fused in a common framework, gradually in their further differentiation undergo extensive change in form and position, and soon it is possible to class them into two groups: (1) the spongioblasts which form the supporting framework or the neuroglia tissues; and (2) the neuroblasts or true ganglion-cells which come to lie within the meshes of the former. Up to the time the two groups can be distinguished from each other they are spoken of as indifferent cells (Schaper).
The differentiation into neuroblasts and spongioblasts does not occur in all cells simultaneously, some cells being more precocious than others; so that in a given section one may recognize well-defined neuroblasts, or spongioblasts, among other cells that are still in the indifferent stage, as is schematically shown in Fig. 3. In general, the spongioblasts develop somewhat in advance of the neuroblasts, and we find in some sections a fairly complete spongioblastic framework having as yet no differentiated nerve cells or fibres. In their further development the spongioblasts and neuroblasts will be treated separately.
Development of the Neuroglia Framework
Embraced under the term neuroglia we must distinguish (a) ependymal cells, (b) neuroglia cells, and (c) neuroglia fibres, all of which elements serve as a supporting framework for the central nervous system, and all of which are differentiated products of the spongioblastic syncytium already referred to.
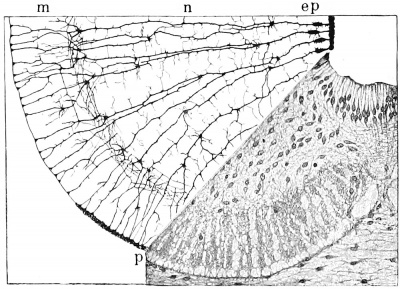
It was seen in Fig. 1, C, how the spongioblasts formed a radial framework traversing the entire wall of the neural tube. As the wall thickens the radial arrangement becomes still more marked and the spongioblastic strands become drawn out. These strands take the precipitate intensely in silver methods and form the characteristic G-olgi picture seen in Fig. 4. From the radial strands numerous processes extend laterally and unite the whole in a complete framework. At the junction of the nuclear and marginal layers there exists a close felt-work of these lateral processes which is supposed to prevent temporarily the invasion of nuclei into the latter layer (Hardesty, 1904). A distinct group of spongioblasts maintain their radial arrangement near the ventricular border and eventually constitute the ependymal cells of the adult. In the remainder of the wall the radial arrangement is finally obliterated, which is due to the proliferation and growth of the neuroblasts, the spaces in the framework being determined by the shape of the elements that it supports. It is this portion of the framework that furnishes the neuroglia cell- proper.
The ependymal cells represent the most primitive form of neuroglia, such as is found prevailing in certain lower forms. They are characterized by their radial arrangement and proximity to the lumen of the neural tube. Through their continuity witli the general spongioblastic framework they extend through the whole thickness of the wall, from the internal to the external limiting membrane, as shown in Fig. 1, C. As the ependymal cells proliferate and accumulate more body-protoplasm they come to form a compact mass of deeply staining columnar cells, constituting the lining membrane of the central canal of the cord and the ventricles of the brain. Their connection with the remainder of the neuroglia framework is maintained by processes which extend from the basal portion of the cells a variable distance into the underlying parts.
In the region of the anterior median fissure of the cord and the median raphe of the hind-brain, corresponding to the Bodenplatte of His, the neuroglia maintains its primitive ependymal type of simple radial fibres extending from the lumen to the surface of the tube. It is this region that is traversed by the fibres of the anterior white commissure of the cord and the transverse arcuate fibres of the hind-brain. The persistence of this simple type of neuroglia may be explained by the absence of any mantle or nuclear layer with its consequent complications at this place. In a similar way in the posterior median region of the cord there is formed the posterior median septum, consisting of persistent radial neuroglia fibres extending from the lumen to the surface of the cord. Here, however, the conditions are modified by the partial fusion of the walls of the central canal and the fusion of the posterior funiculi, so that instead of a deep fissure we have in the end only a shallow groove. The development of the posterior median septum will be described in detail in the section on the development of the spinal cord.
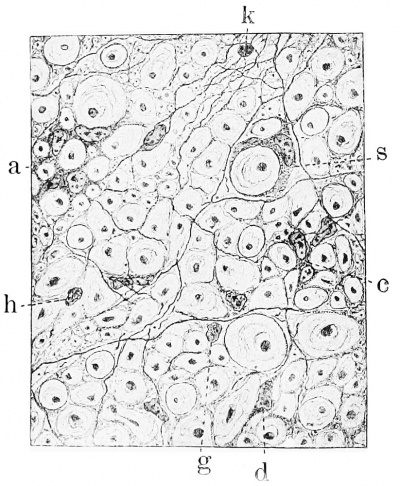
Fig. 4. Development of neuroglia framework. Combined drawing, after Golgi and Benda methods, showing section of spinal cord of embryo pig 30 mm. long. It can be seen that the wealth of communicating processes of the neuroglia cells is not shown in the Golgi portion, ep, ependymal layer; m, marginal layer; n mantle or nuclear layer; p pia mater. (After Hardesty.)
Thus in the adult we find that the ependymal neuroglia is persistent only as septa in the anterior and posterior median planes of the nervous system and as a lining membrane for its central canal and ventricles. Otherwise the whole supporting framework of the nervous system consists of neuroglia cells proper and the neuroglia fibres developed in connection with them, that is if we do not regard the blood-vessels and the accompanying mesodermal tissue, which subsequently enter the neural tube, as a true supporting framework.
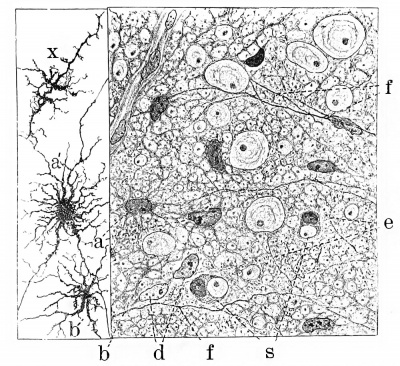
The neuroglia cells are differentiated somewhat later than the ependymal cells. They can be traced back to the time when they constituted units in the primary spongioblastic syncytium. As the extension and branching of the latter continues, its component cells can be seen to proliferate and develop additional protoplasm. They at the same time become moulded into shape by the growing nerve cells and fibres which they enmesh. Due to this moulding process there result the characteristic much-branched cells that we know as spider-cells, glia-cells, astrocytes, or Deiters's cells, as shown in Fig. 5. Instead of the simple spongioblastic framework seen in the earlier stages, we now have a dense felt-work produced by the anastomosing branches of these cells, intimately intertwining between the developing nerve-cells and their processes.
Fig. 5. Combined drawings, after Golgi and Benda methods, of the spinal cord of fetal pig, 20 cm. long, showing syncytial character of neuroglia framework and the first appearance of neuroglia fibres. a, neuroglia cells after the Benda method; a', similar cell after the Golgi method; e and f, neuroglia fibres beginning to take the neuroglia stain; b, pseudocell due to staining of a portion of the syncytium such as seen at b; s, seal-ring cells. (After Hardesty.)
The variation in the shape of the individual neuroglia cells is determined by the nature of the nerve tissue supported, and consequently there are different types described for the cerebral cortex, the cerebellum, and the white and gray substance of the cord. Such variations involve the shape and size of the cell body as well as the character of their cell processes. The earlier conceptions of neuroglia cells were based on silver precipitation methods (Golgi) which failed to reveal the true wealth of their anastomosing branches, and there thus existed a false impression of neuroglia as consisting of scattered and independent cells.
Fig. 6. Section of spinal cord of suckling pig of two weeks, showing fully developed neuroglia fibre and fibres in the process of transformation, a and c, early stages of neuroglia cells, multinucleated protoplasmic masses; d, g, h, and k, stages of karyolysis which many of the free neuroglia cells undergo; s, sealring cell. (After Hardesty.)
The neuroglia fibres constitute the third element of the supporting tissue, and they resemble in their process of development the fibres in white fibrous connective tissue. They do not appear until late in uterine life. According to Hardesty (1904), they can first be recognized in pig embryos between 16 and 20 cm. long, and from then on gradually increase in number until after birth. There is some evidence that the process of differentiation continues even into adult life.
It was formerly supposed (G-olgi, v. Lenhossek, Erik Miiller, and others) that neuroglia cells existed as separate units and that neuroglia fibres were simply the modified processes of these cells. With the development of the differential neuroglia stains it soon became apparent that this was not the case. From Weigert 's studies (1895) on human tissues, followed by Huber's (1903) studies on several vertebrate classes, it was shown that neuroglia fibres are not to be regarded as processes of the neuroglia cells, but rather as fibres having specific physico-chemical properties which lie in close contact with or actually embedded in the peripheral layer of the protoplasm of the neuroglia cells from which they have become differentiated, as is shown in Fig. 7. The fibres are not
Fig. 7. Neuroglia fibres in adult human spinal cord, showing their relation to the protoplasm of the neuroglia cell and its processes. (After Huber.) interrupted by the protoplasm of the cells, and a single fibre may even be continuous from one cell to another.
To understand properly the manner in which these fibres form, one should keep in mind the syncytial character of the neuroglia framework. This has been especially emphasized by Hardesty (1904), on whose studies the description of this development is based. It has already been seen how the protoplasm of the syncytium in its development tends to accumulate about the nuclei and thus forms masses which we have described above as neuroglia cells. The separate nucleated masses or cells remain more or less continuous with each other by means of attenuated branching portions of the syncytium. In these attenuated portions at a certain period (pigs 16 to 20 cm. long) it can be seen that the protoplasm is becoming more condensed, stains less deeply, and is beginning to show fibrillation. The same process can be recognized in the peripheral portions of the nucleated masses or cells. The fibrillation continues to become more marked, and eventually distinctly outlined individual fibres of varying length can be recognized as lying in the attenuated portions of the syncytial protoplasm and passing through the "domain" of one or more nuclei, as shown in Fig. 5. Whether all the protoplasm in the attenuated portions is consumed in the process of fibrillation, or whether some remains as a partial coating of the fibre is not yet clearly determined.
The final step in their development is the chemical transformation that the fibres undergo, owing to which they exhibit a blue reaction when stained with special neuroglia stains. When that is attained they present the characteristic picture seen in Figs. 6 and 7. A portion of a fibre may give this reaction before the differentiation of the remainder of the fibre is complete. Sometimes a fibre ripens in interrupted areas along its course and hence temporarily appears as a row of fine dots. By the time the chemical transformation is completed the fibres have attained a size about the same as found in the adult. There is some variation in the distribution and form of the neuroglia fibres, depending on whether they are located in the region of gray matter or white matter or in the region of ependymal cells; the essential points in their development, however, are the same in all regions.
Those writers {e.g., Rubaschkin, 1904) who consider neuroglia cells as separate and independent structures describe the neuroglia fibres as modified cell processes. A special cell is also distinguished from which they are derived (gliogenetic cells), which forms an intermediate stage between the spongioblast and neuroglia cell proper. Several fibres may be derived from one process, and eventually they detach themselves from the cell and lie free in the tissue.
Development of Neuroblasts and Motor Nerves
We have seen above how the cells forming the wall of the neural tube in the early stages are fused in a common syncytial framework, and how the constituent cells of this framework gradually differentiate themselves into spongioblasts and neuroblasts. The former maintain their syncytial arrangement relatively late and continue even in the adult to show permanent traces of that condition. The neuroblasts, however, toward the end of the first month apparently detach themselves from the general framework and form separate clusters within its meshes. These clusters of proliferating neuroblasts form a prominent part of the nuclear or mantle layer, and it is from these that all the true nerve-cells of the brain and cord are derived.
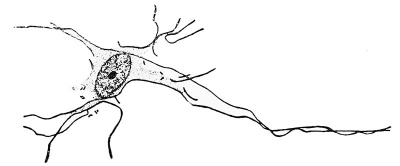
The neuroblasts can be recognized by their characteristic shape (see Fig. 9). They possess a prominent nucleus and a tapering protoplasmic body which is continued into a slender primary process which is to become the future axis-cylinder of the cell. There is always the tendency for the primary processes of adjacent neuroblasts to come together and form a common strand, in this way giving rise to the arrangement in clusters. It has not yet been shown whether the processes of these cells come together secondarily, or whether the arrangement is the result of the manner of cell cleavage, the processes being the last portion of the proliferating cells to split apart. The primary process may either extend to some other part of the neural tube or may leave the neural tube and extend through the mesoderm to some peripheral structure, as is shown in Fig. 8. The motor nerve roots are an example of the latter, and in Fig. 10 there is shown how clusters
Fig. 8. — Diagram showing distribution of neuroblasts in human embryo of four weeks. On right side both neuroblasts and spongioblasts are shown; on left side neuroblasts only. The converging processes in the ventral part of cord unite to form the anterior nerve-roots. The processes forming the posterior roots enter the marginal layer and extend upward and downward as a longitudinal bundle, thus constituting the anlage of the posterior white column. (After His.) of such motor neuroblasts form rootlets which pass through the marginal layer of the neural tube and enter the mesoderm and unite in a common nerve-trunk. A group of neuroblasts from the same section is shown under higher power in Fig. 9.
Later the original pear-shape of the neuroblast is altered by the increase in body protoplasm and the development of secondary processes which branch out into the neighboring spaces. "With the increase in size the cell becomes moulded by the supporting framework and adjacent cells into its eventual shape, upon which the nucleus retires to the centre of the cell and enters upon the resting stage. The development of neurofibrils within the substance of the cell body and its processes occurs early; with special staining methods they can be demonstrated in embryo pigs 10 mm. long.
It was formerly thought (His) that the neuroblasts were derived directly and exclusively from the previously mentioned mitotic cells that are found during a brief period along the ventricular border of the wall of the neural tube, the so-called germinal cells (see Fig. 1, g). His pictures these germinal cells and shows them dividing and after assuming a pear-shape migrating out into the mantle or nuclear layer, being all the time sharply separated from the spongioblasts. This process has not been confirmed by the more recent investigators, and it is now generally thought that the germinal cells are only a sign of cell proliferation, and belong in common to the whole syncytial framework or myelospongium and thus are ancestors of the neuroblasts and spongioblasts alike.
Fig. 9. — Cluster of neuroblasts from nucleus of origin of n. oculomotorius, showing characteristic shape and grouping of cells. Taken from same specimen shown in Fig. 10.
Fig. 10. — Section through floor of mid-brain of human embryo one month old (length 10 mm., Huber collection No. 3), showing the origin and formation of the trunk of a typical motor nerve (c. oculomotorius).
The development of the neuroblast forms a problem that has been the subject of much investigation and discussion. Particular interest has been manifested in the growth of its attached nervefibre, both on account of the great length of the latter as compared with the size of the cell and on account of the intricate maze of fibres through which it traces its way to reach the organ for which it is intended. Of the many interpretations that have been offered in explanation of this problem the one that seems now to be most generally accepted is the so-called outgrowth theory of His, according to which each nerve-trunk is the process of a single nerve-cell. The fibre is a simple elongation of the primary process produced by an actual outflow of substance from the ganglion-cell toward the periphery, and it thus makes its way through various tissues to reach its proper end organ with which it comes into relation secondarily. It is this theory that has been ably substantiated by the histological studies of Eamon y Cajal and conclusively established by the recent experimental studies of Harrison.
Among the other hypotheses that have been advocated regarding the development of the nerve-fibre the most prominent is the cell-chain theory, according to which each nerve-fibre is the product of a chain or pathway of ectodermal (according to some, mesodermal) cells extending from the neural tube to the periphery. From the protoplasm of these cells fibrillar are differentiated which join to form a continuous fibre connecting secondarily the ganglion-cell with its end organ (Schwann, Balfour, Dohrn, Bethe). This theory was later modified by Apathy and 0. Schultze, who conceived of a syncytium of anastomosing cells which from the beginning connects the neural tube with the peripheral end organs, somewhat like the older description of Hensen, according to which protoplasmic bridges persist between dividing cells everywhere in the embryo, and by fibrillar differentiation some of these protoplasmic connections become eventually converted into nerve-fibres and thus connect the ganglion-cells with, the organs they supply. A further modification of this interpretation is that of Held (1907). Like Hensen, he conceives of a general syncytium connected everywhere by protoplasmic bridges, and later the nerve paths are formed by the transformation of these bridges ; but, according to Held, the growth of these fibrillse is from the nerve-cell outward, instead of simultaneously along the whole path, as maintained by his predecessors. In the protoplasm surrounding the nucleus of each neuroblast a plexus of neurofibrils forms, thus marking off neuroblasts from spongioblasts. From each of these a bundle of the fibrils extends out through the substance of the syncytial framework along a definite path and constitutes its axis-cylinder process. Subsequently, in addition to the primary bundle of fibrillae, other fibrillae extend out from the perinuclear network along other protoplasmic bridges and thus form the dendrites. In all these views the nerve-fibre is considered as a product of a group of cells instead of as an appendage of a single cell, as is postulated in the outgrowth theory.
File:Keibel Mall 2 011.jpg Fig. 11. — Isolated ganglion-cells, from embryonic spinal cord of frog, and growing in clotted lymph. B is an isolated cell from tissue taken from branchial sense organ. The drawings are made from live specimens; two views of C are shown, taken four and three-quarters hours apart, showing rate of outgrowth of the nerve-fibre and the manner of its branching. (After Harrison.)
Fig. 12.— r-Three views, taken at intervals of Ik and 8i hours, of the same living nerve-fibres growing from a mass of spinal-cord tissue (frog embryo) out into clotted lymph. They show the rate of growth and the longitudinal splitting of fibres, and the characteristic growing ends, a larger example of which is represented by n.f. (After Harrison.)
In his experiments carried out on amphibian larvas Harrison demonstrated four essential facts: In the first place he showed that no peripheral nerves would develop in an embryo from which the nerve-centres had been removed, thus establishing the fact that the ganglion-cells are an essential element in the development of the nerve-fibre. He next showed that the sheath cells of Schwann, upon the influence of which in the formation of the fibre many of the histologists had placed much emphasis, were not essential to the growth of the nerve-fibre, and that the axis-cylinders will develop and extend out in the surrounding tissues in the normal way and reach their normal length in specimens where the sheath cells have been eliminated. Thirdly, he showed, by modifying the environment of the developing nerve, that fibres will form in surroundings entirely different from their natural path and establish completely foreign connections. Finally, he succeeded in growing ganglion-cells outside of the body in an unorganized medium (clotted lymph), where all possibility of contributions on the part of other living tissues was eliminated, and he was able with this method to demonstrate directly under the eye the outgrowth of the nerve-fibre from the ganglion-cell as it developed hour by hour, proving conclusively the unity and continuity of the two.
Isolated ganglion-cells developing in an artificial medium are shown in Fig. 11, where A, B, and C represent three cells in different stages of growth. Two views of C are shown, one being taken four and three-quarters hours later than the other. The different cells shown in Fig. 11 are all represented under the same magnification, and on comparing them it is evident that the growth of the fibre is the result of a flowing out of the protoplasm of the cell into a thick primary process which as it elongates soon attains a uniformly slender width. The growing end of the process is characterized by an enlarged branched tip which continually undergoes amoeboid changes in form. One of these characteristic muchbranched growing ends is shown in Fig. 12 (»./.) under higher magnification. Occasionally the growing tip completely bifurcates, which results in permanent bifurcation or branching of the nerve-fibre at intervals along its course.
In Fig. 12 there can be seen a further phenomenon in the growth of the nerve-cell consisting in the apparent longitudinal splitting of the cell and its main process, a form of retarded cell division that occurs subsequently to the formation of the primary process. In the figure a, Id, and c represent the same nerve-cell mass as it appears at intervals of one and one-half and eight and one-half hours. It shows how that which is apparently a single cell and process becomes converted by this longitudinal cleavage into four individual cells with processes.
It is to be remembered that the forms shown in Figs. 11 and 12 represent the growth of ganglion-cells as it occurs in clotted lymph, and it is to be presumed that they depart in some details from that which occurs in living tissues ; however, the structure of the fibres and the characteristic enlarged branching ends conform exactly to the appearances seen in sections of specimens where the fibres have developed under normal conditions, and they no longer leave room for doubt as to the essential validity of the His outgrowth theory.
Development of Spinal Ganglia and Sensory Nerve Roots
As the elevated borders of the neural plate come together and fuse to form the closed neural tube, it detaches itself from and becomes completely covered in by the general ectoderm. During Ganglion crest this process a row of ectodermal cells situated along the borders of the neural plate undergo special changes ; they neither become incorporated in the neural tube nor do they become skin. It is these cells that form the so-called ganglion crest from which the cerebrospinal ganglia and sensory nerve roots are derived, in addition to the chromaffin cells and the sheath cells of Schwann.
Fig. 13. — Transverse .sections through dorsal region of human embryos showing three stages in the development ]ofJth3 ganglion crest and the anlage of the spinal ganglia. (After von Lenhoss^k and Kollmann.)
The cells of the ganglion crest differentiate themselves from the cells of the neural tube during the closure of the latter. As shown in Fig. 13, they form a cellular ridge or crest along the line of closure. As they continue to develop they become apparently completely detached from the tube. Separating bilaterally into right and left crests, they migrate lateralward and ventralward in between the myotomes and neural tube. Here they continue to proliferate, and, as the constituent cells become differentiated into ganglion-cells, rootlets appear along the dorsal border of the crest and attach it secondarily to the neural tube. The ganglion crest thus extends as a flattened cellular band on each side uninterruptedly from the caudal tip of the neural tube forward to the region of the ear vesicle (compare Fig. 83). In the spinal region it becomes notched with segmental incisures along its ventral border, producing a series of ganglionic masses which for a time remain connected with each other by a dorsal cellular bridge. Eventually the latter disappears and there results a complete segmentation of the crest into separate spinal ganglia.
In the head region the structures to be supplied lose their simple segmental character, and there are introduced the complications of the gill arches and the lateral line system. These factors modify the character of the ganglion crest, though in its derivation from the border of the neural tube it apparently follows the same process as described for the spinal region. In the region of the midbrain it is believed by some writers (Meyer, Johnston) that a portion of the ganglion crest becomes incorporated in the neural tube, and the cells derived from this portion never obtain a peripheral position. The gross features in the development of the ganglia of the head and spinal regions will be referred to again more in detail under the peripheral nervous system.
Fig. 14. — Section through spinal ganglion of human embryo 18 mm. long, about 6 weeks old (Huber collection No. 5). On the left the cells are mostly in the indifferent stage, and ganglion-cells and supporting cells look much alike; on the right the ganglion-cells stand out sharply, due to the accumulation of granular protoplasm around the nucleus.
At the time the ganglion crest detaches itself from the neural tube and begins to spread ventralward (embryos 2-3 mm. long), examination of it reveals a moderately compact mass lying in the space between the neural tube and the myotomes. The constituent cells possess oval or rounded nuclei with multiple small chromatin bodies such as are found in cells during active proliferation. The cell bodies possess ill-defined outlines and fuse with each other in a syncytial mass.
As the cells of the ganglion crest proliferate and become further differentiated it is possible to divide them into two different groups, — i.e., ganglion-cells and supporting cells, — in the same way that neuroblasts and spongioblasts are developed in the neural tube. (Compare Figs. 3, 4, and 5.) The steps in this procedure are shown in Figs. 14, 15, and 16. On comparing these the most apparent difference between the ganglion-cell (ganglioblast) and the supporting cell (capsule and sheath cells) is that the ganglion
Fig. 15. — Section through cervical spinal ganglion of human fetus 8.5 cm. long, about 3 months old, showing large ganglion-cells with eccentric nuclei. Surrounding them are the much-branched supporting cells, some of which are to become capsule cells and the others sheath cells.
cells detach themselves more or less completely from each other and develop isolated large compact protoplasmic bodies, while the supporting cells consist almost entirely of branching processes which remain united in an extensive network or syncytium in the meshes of which lie the other cells.
By the end of the fourth month (Fig. 16) this differentiation is far advanced, and one can plainly recognize the supporting cells arranged in the form of capsules around the ganglion-cells proper. The next younger stage, at the end of the third month (Fig. 15), the supporting cells can be distinguished, but the capsule arrangement at that time is very incomplete. Going still further back, to the six-weeks embryo (Fig. 14), we find a portion of the ganglion mass consisting of undifferentiated neural crest cells, the ganglioncells and supporting cells both having the same appearance. However, scattered in among these "indifferent cells" are some more advanced cells that can be recognized as ganglion-cells by their increased body protoplasm. As a rule, the ganglion-cells attain a characteristic appearance earlier than the supporting cells.
The differentiation of the ganglion-cell consists in the condensation of the body protoplasm and the development of fibrillar processes. As an indifferent cell of the ganglion crest syncytium it has no definite outline, and in a teased preparation it appears as a ragged granular mass around a nucleus. Later, as the protoplasm of the cell body accumulates, it detaches itself more or less completely from the neighboring cells and attains discrete outlines. It is possible then to make teased preparations such as are shown in Fig. 17.
Fig. 16. — Section through sixth cervical ganglion of human fetus 10.5 cm. long, about 4 months old. Here the branched supporting cells are arranged in distinct capsules around the ganglion-cells, and in the lower part of the section they form a characteristic framework for the nerve-fibres. The ganglion-cells are nearing the completion of their development; the nuclei are retiring to the centre of the cell, and the basic stainable substance is appearing at the periphery of the cell bodies which later becomes clumped to form the Nissl bodies.
The earlier ganglion-cells usually have a simple fusiform shape with a process extending out at each pole, as is shown in Fig. 17, group A. These lie in clusters, like the neuroblasts in the spinal cord, and their processes are still fused so that in teasing them apart we get the appearances seen in group B. By Golgi methods it can be demonstrated that the central processes of such cell groups form rootlets which enter the dorsal part of the wall of the rrural tube, where for the most part the fibres bifurcate and form a longitudinal bundle within the marginal layer, as seen in Fig. 8, and after a longer or shorter course upward or downward they end in an arborization among the cells of the mantle layer. In the spinal region the sensory fibres thus entering form one continuous path, which eventually constitutes the dorsal funiculi of the cord. In the cranial region the entering fibres of the sensory nerves form separate paths, the vestibular tract, the trigeminal tract, and the tractus solitarius, to which are contributed fibres from the seventh, ninth, and tenth nerves. The peripheral processes unite into bundles, which join with the fibres of the anterior roots and with them form the main nerve-trunk, the further development of which will be referred to later in the section on the peripheral nervous system.
Fig. 17. — Isolated cells teased from spinal ganglia of embryo pigs 20-40 mm. long, showing the variation in the form of the early ganglion-cells. B shows the grouping of the cells; A, C, D, E, and F represent selected types arranged according to the form of the cell body and the character of the processes.
In examining a given ganglion in embryos about two months old it will be found that the cells are present in many different stages of growth, and that in addition to the simple bipolar earlier cells there are younger cells that must make room for themselves in the intervening spaces. Such cells possess great irregularity in their shape. Their form and the manner in which the processes leave the cell body is apparently determined by the pressure that is exerted upon them by the adjoining cells. Some of the forms that are frequently seen in teased preparations are shown in groups C, D, E, and F, Fig. 17. The only feature common to all of them is the eccentric position of the nucleus, which is retained until the cell nearly reaches its full size, upon which the nucleus retires to the centre of the cell and assumes a resting appearance. (Compare Figs. 15 and 16.) It will be seen that there is a great variety in the number, size, and situation of the cell processes. In group F there are multipolar forms, some of which persist as such in the adult forms. A very common form is shown in group E, which are technically bipolar cells, but in each case one of the processes is much better developed than the other, the small one having the appearance of undergoing retrogression. It suggests the possibility that these represent bipolar cells that are being converted into unipolar cells by absorption of the smaller process. All through the nervous system there is evidence of cell processes and nerve-fibres that grow out for a certain distance and then for some reason cease to develop any further and finally disappear, as in the case of the hypoglossal nerve. This phenomenon is not limited to the temporary existence of vestigial structures, but also represents what might be called a ' ' tentative growth, ' ' under which we may understand that of the great number of processes and fibres which start out a certain number fail to establish adequate connections and subsequently disappear. On the other hand, in the present case of the ganglion-cell the smaller process may represent the slender axon that enters the cord, while the heavier process corresponds to the peripheral dendrite.
The conversion of bipolar cells into unipolar T-shaped cells of Eanvier is commonly supposed to be due to the unilateral growth of the cell body towards one side, which brings about an approximation of its two processes so that they fuse in a common extension from the cell. The cells shown in Fig. 17, C, are regarded as transitional cells undergoing this change, and cells 8, 9, and 10 would represent the successive steps in the process. But if this is the explanation, it should be expected that no cells would show the T division until they reach the stage of growth represented by cell 10. This, however, is not the case, for in group D we have cells in which the T division is already completed before that time, and moreover cell 1 in group D possesses the T division and the opposite process is still intact. Furthermore, as will be presently seen, the growth of the enmeshing capsule cells is already well advanced while many of the cells are still of the simple bipolar type {e.g., Fig. 18, a and b), and this would tend to prevent the approximation of opposite processes. There is therefore much reason to believe that the T division is simply the result of the bifurcation of the growing end of the main process of the cell in the manner so clearly demonstrated by Harrison's growing nervecells (Fig. 11), and that the so-called transitional cells are merely accidental forms due to the moulding influence of adjacent cells. Certainly a great many of the cells can never undergo the supposed transformation.
Concerning the migration of spinal ganglion cells and their relation to the formation of the sympathetic ganglia the reader is referred to the section on the development of the sympathetic system, and for the chromaffin or phssochrome cells and organs to Chapter XV which describes those structures.
Fig. 18.— Teased preparations from spinal ganglia of pig, showing development of sheath and capsule cells. In a and b (pigs 3-4 cm. long) the supporting tissue consists of a loose syncytium enmeshing the ganglion-cells; in c and d (pigs 20 cm. long) the protoplasm is condensed and arranged in the form of distinct capsules. In d the capsule cells are directly continuous with the sheath coating the main process of the cell. Compare with Figs. 15 and 16.
The development of the supporting cells can be followed by comparing Figs. 14, 15, and 16, which show the appearance as seen in cross sections, and Fig. 18, which represents teased preparations. Like the ganglion-cell the supporting cell is derived from the indifferent cells of the ganglion crest, and also like the ganglion-cell the chief phenomenon of its differentiation consists in the condensation of the body protoplasm.
When first recognized the supporting cells form a loose poorly defined granular syncytium from which the ganglion-cells have become almost completely detached. The subsequent condensation takes the form of branching processes instead of full rounded bodies like the ganglion-cells, and there results a sharply outlined nucleated framework in the meshes of which lie the nerve elements proper (Fig. 18, c). In their derivation and behavior it will be noticed that the supporting cells are directly analogous to neuroglia cells. Eventually they become subdivided into capsule cells and sheath cells, according to whether they form an envelope around some ganglion-cell or are situated along the course of a cell process. The two kinds are identical in character and are directly continuous with each other, as can be seen in Fig. 18, d. It is now believed, as will be referred to again under myelinization, that it is some of the supporting cells of the spinal ganglia that migrate outward along the course of the nerves and form all the sheath cells of the peripheral nerves.
Myelinization of Nerve=fibres
The final development of the nerve-fibre is characterized by the formation of a fatty enveloping sheath. This process does not become apparent until about the fourth month, and it is not completed until after birth. Many details concerning the acquisition and structure of the nerve-sheath are still involved in dispute, and especially in human material there is no adequate description yet available. Our knowledge is mostly based on studies of the pig and sheep (Vignal 1883, Gurwitsch 1900, Bardeen 1903, and Hardesty 1904).
In the development of the nerve-sheath we have to distinguish between the formation of the non-nucleated myelin sheath and the nucleated membranous sheath which are its two subdivisions, the latter being synonymous with the sheath of Schwann or neurolemma. Either of these may make their appearance before the other. In the central nervous system the myelin sheath appears first, and until recently was supposed to be the only covering possessed by these fibres. In the case of the peripheral nerves, on the other hand, the axon is usually completely inclosed by the membranous sheath before the myelin appears, and in the case of some fibres, particularly in the sympathetic system, this remains the permanent condition. Such fibres are known as non-medullated fibres.
The cells that give rise to the membranous sheath of the peripheral fibres appear relatively early. They are easily distinguished toward the end of the third week as spindle-shaped cells (cells of Schwann or sheath cells) closely attached to the developing nerve-fibres. That these cells are ectodermal and are derived from the ganglion crest has been shown experimentally in amphibian larvae by Harrison (1906). The experiments of this investigator show that the source of the sheath cells can be removed by early extirpation of the ganglion crest, and that in such cases there develop naked fibres which normally are found covered with sheath cells. It should, however, be remembered that this may not be the only source in all forms ; in fact in elasmobranchii it is said that there are in addition a large number of sheath cells that wander out somewhat later from the ventral part of the spinal cord along the motor roots (Neal, 1903). Up to the time of Harrison's experiments it was supposed that the sheath cells were derived from the mesenchyme and were foreign cells that invaded the nerve-trunks and ganglia very early (Vignal, Gurwitsch, and Bardeen).
In the human embryo during the transformation of the ganglion crest into spinal ganglia it is possible to trace step by step the differentiation of its constituent cells into ganglion-cells proper and supporting cells, as shown in Figs. 14, 15, and 16. It is these supporting cells that form the capsule cells and sheath cells. Of the latter some are for the spinal ganglia themselves while others
Fig. 19. — Isolated fibres showing development of medullary sheath. The specimens are taken from the intercostal nerve of pig fetuses about 15 cm. long and prepared by formalin and osmic acid. (After Bardeen.) wander out along both the sensory and motor nerve-trunks, accompanying them in their growth forward, and eventually forming their membranous sheath.
The sheath cells are at first arranged so as to inclose a group of axons in a common trunk. By subsequent proliferation they invade the trunk and divide it into smaller bundles, until finally they close in around each axon and form individual sheaths. Each sheath cell corresponds to a segment of the sheath, and the interval between two adjacent cells corresponds to a node of Ranvier. The differentiation of the sheath cells and their conversion into the nerve-sheath is shown in Figs. 19 and 20. In the earlier stages they unite in the formation of a segmented thin-walled sheath, which in formalin specimens can be seen loosely enveloping the axis-cylinder, and apparently containing fluid (Bardeen, 1903) ; in osmic acid specimens it is found shrunken closely against the axis-cylinder (Fig. 20, A). Each nucleus maintains its position near the centre of a segment and continues to maintain a small quantity of granular protoplasm around itself. There then follows a deposit of myelin within the membranous sheath around the axis-cylinder. In the case of most peripheral fibres the axis cylinder becomes completely inclosed by the sheath of Schwann before the formation of the myelin sheath begins. As regards the origin of the myelin there have been three theories presented: first, that it is formed by the sheath of Schwann ; second, that it is a differentiated portion of the outer part of the axis-cylinder (Kolliker, 1904) ; and according to the third it is precipitated from the fluid immediately surrounding the axon by some reaction between the two (Bardeen, 1903). At first the myelin is evenly distributed along the axis-cylinder, but later it accumulates more rapidly in some areas than in others, which produces the beaded appearance shown in Fig. 19, c. Eventually the sheath is completely filled out (Fig. 19, d). At the nodes of Ranvier the formation of myelin continues for some time after the completion of the myelin deposit in the remainder of the sheath (Fig. 19, e), apparently thus allowing for the lengthening of the fibre. During the later stages of development further provision is made for the lengthening of the fibre by the intercalation of new segments of the sheath of Schwann at the nodes of Ranvier.
Fig. 20. — Isolated fibres of the sciatic nerve of sheep fetus 15 cm. long, treated with osmic acid and showing development of the nerve-sheath. A shows axon inclosed in membranous sheath with proliferating nuclei. In B the development is more advanced, and a deposit of myelin shown in dark stipple has formed within the membranous sheath: n.R., node of Ranvier; S.z., nucleus of membranous sheath. (After Vignai.)
In the central nervous system there seems to be no distinct separate membrane investing the fibres that exactly corresponds to the sheath of Schwann of the peripheral medullated fibres. There are, however, sheath cells present. They are apparently most numerous and possess most protoplasm during the active period of myelin development. On adult fibres they are relatively fewer in number and are less conspicuous since they then possess less body protoplasm. According to Hardesty (1904), these sheath cells of the central nervous system in the pig are derived from the indifferent supporting cells which we have already seen for the most part are converted into neuroglia tissue. Shortly after the beginning of myelin formation they become differentiated into typical sheath cells, and form around each nervefibre an extended lamellated reticulum, in the meshes of which the small globules of myelin are supported.
Myelin formation commences about the fourth month, but it does not make its appearance in all parts of the nervous system at the same time, nor simultaneously in all parts of an individual fibre. As a rule, it appears first in fibres that become functional first and in fibres that are phylogenetically the oldest. In an individual nerve the myelin is first seen at the central end and spreads from there toward the periphery. According to Westphal (1897), the medullation of the spinal nerves is completed between the second and third years, while the cranial nerves are completely medullated by the ninth or tenth post-embryonal week. Of the
Fig. 21. — Section through hind-brain of new-born child, showing myelinization of fifth, sixth, seventh, and eighth cranial nerves and associated fibre tracts (fasciculus longitudinalis mediahs and corpus trapezoides). The lemniscus medialis is also myelinated. These stand out in sharp contrast to the other fibres of the pontine region, including the pyramidal tract, which are still devoid^of myelin. (After Edinger.) cranial nerves the motor fibres are medullated first, and the sensory fibres a little later, excepting the acusticus, the vestibular division of which is medullated about as soon as the motor fibres. In premature births the medullation of the cranial nerves seems to be accelerated. Westphal states that there is a gradual increase in the size of the sheath with increasing age. He finds that on comparing the adult with the child the largest sheath in a given motor nerve in the adult is twice as large as the largest sheath in the same nerve in the child. He finds also that the sheaths are more uniformly large in the adult, while in the child there is a large proportion of small sheaths, so that the average of large sheaths in an adult nerve is four or five times greater than in the same nerve in a child. Regarding the time of myelinization of the different areas and fibre paths of the central nervous system the reader is referred to Westphal's paper and the work of Flech sig (1896, 1904). As can be seen in Fig. 21, the relative time of appearance of the myelin is an important factor in the identification of the different fibre paths.
Literature
Bardeen CR. The growth and histogenesis of the cerebro-spinal nerves in mammals. (1903) Amer. J Anat. 2: 231-258.
Biervliet, vax : La substance chromophile pendant le cours du developpement de la cellule nerveuse (chromolyse physiologique et chromolyse experimentale). Journ. de Neurol. — Ref. im Centralbl. f. Nervenheilk. u Psych. Jahrg. 24. 1901.
Brodhann, K. : Bemerkungen iiber die Fibrillogenie und ihre Beziehungen zur Myelogenic mit besonderer Beriicksichtigung der cortex cerebri. Neurol. Centralbl. Jahrg. 26, S. 338-349. 1907.
Cajal, S. Ramon: Nouvelles observations sur devolution des neuroblastes avec quelques reinarques sur Thypothese neurogenetique de Hensen-Held. Anat. Anz. Bd. 32. 1908.
Cameron, J.: The Development of the Vertebrate Nerve-cell: A cytological study of the neuroblast-nucleus. Brain : Part 115. 1906.
Capobianco, T. : Recherches ulterieures sur la genese des cellules nerveuses. Ann. Neurol. Anno 23.— Ref. Arch. ital. Biol. T. 44. 1905.
Collin, R. : Recherches cytologiques sur le developpement de la cellule nerveuse. Le Nevraxe. T. 8 1906 et These en med. Nancy 1907.
Colucci, C, et Piccinino, F. : Su alcuni studii di sviluppo delle cellule del midollo spinale umano. 12 Fig. Ann. Neurol. Anno 18. p. 81-110. 1900. Ref. in Centralbl. f. Nervenheilk. u. Psych. Jahrg. 24, S. 521. 1901.
Flechsig, P. : Einige Bemerkungen iiber die Untersuchungsmethoden der Gross hirnrinde insbesondere des Menschen. Bericht d. math.-phys. Kl. d. Kgl. Sachs. Gesell. d. Wissensch. z. Leipzig. 11. Jan. 1904.
Gehirn und Seele. 2. Aufl., Leipzig. 1896. Gierlich : Uber die Entwicklung der Neurofibrillen in der Pyramidenbahn des Menschen. Vortrag a. d. Vers, siidwestdeutscher Neurologen, Mai 1906. Deutsche Zeitschr. f. Nervenheilk. Bd. 32, S. 97. 1906.
Gurwitsch, A. : Die Histogenese der Schwann'schen Scheide. Arch, f . Anat. u. Physiol. Anat. Abt. 1900.
Hardesty I. On the development and nature of the neuroglia. (1904) Amer. J Anat. 3.
Hardesty I. On the occurrence of sheath cells and the nature of the axone sheaths in the central nervous system. (1904) Amer. J Anat. 4.
Harrison, R. G. : Experiments in Transplanting Limbs and Their Bearing Upon the Problems of the Development of Nerves. Jour, of Exper. Zool. Vol. 4, p. 239 ff. 1907. Further Experiments on the Development of Peripheral Nerves. Amer. Jour. Anat. Vol. 5. 1906.
Hatai, S. : On the Increase in the Number of Medullated Nerve-fibres in the Ventral Roots of the Spinal Nerves of the Growing White Rat. Jour. Comp. Neurol. Vol. 13, p. 177-183. 1903.
Hensen, V. : Die Entwicklungsmechanik der Nervenbahnen im Embryo der Saugetiere. Ein Probeversuch. 4°. 51 S. Lipsius & Tischer, Kiel u. Leipzig. 1903.
His, W. : Histogenese und Zusammenhang der Nervenelemente. Arch. f. Anat. u. Physiol. Anat. Abt. Suppl. Bd. 1890.
Histogenese und Zusammenhang der Nervenelemente. Arch. f. Anat. u. Physiol. Anat. Abt. Suppl.-Bd., S. 95-117. 1890.
Huber, G. C. : Studies on Neuroglia Tissue. Contributions to Medical Research dedicated to V. C. Vaughan. University of Michigan, Ann Arbor. 1903.
Kappers, C. U. A. : Recherches sur le developpement des gaines dans le tube nerveux. 2. Teil. 2. Aufl. Petrus Camper. 1903.
Kolliker, A. von : Uber die Entwicklung der Elemente des Nervensystems, contra Beard und Dohrn. Verb. Anat. Gesellsch. Wien. S. 76-78. 1892. Die Entwicklung der Elemente des Nervensystems. Zeitschr. f. wissenseh. Zool. 4 Taf. u. 12 Textabb. Bd. 82. 1905. Uber Entwicklung der Nervenfasern. Anat. Anz. Bd. 25. 1901.
Kronthal, P. : Zum Kapitel Leukocyt und Nervenzelle. Anat. Anz. Bd. 22, Nr. 21/22. 1903.
Lawdowski, M. D. : Uber anastomotische Verbindungen zwischen Nervenzellen. Russki Wratsch. Bd. 1, Nr. 12. 1902. (Russisch.)
Lenhossek, M. v. : Nervensystem. Ergebn. Anat. u. Entwicklungsgesch. Bd. 7. S. 110-178. 1898. Minot, C. S. : Die fruben Stadien und die Histogenese des Nervensystems. Ergebn. Anat. u. Entwicklungsgesch. Bd. 6, S. 687-739. 1897.
Muhlmann, M. : Uber die Veranderungen der Nervenzellen in verschiedenem Alter. Verb. d. Gesell. Deut. Naturf. u. Arzte. 1900. Bd. 2, S. 20-21. 1901. Weitere Untersuchungen uber die Veranderung der Nervenzellen in verschiedenem Alter. Arch. f. mikrosk. Anat. Bd. 58, S. 231-246. 1901.
Nussbaum, M. : Nerv und Muskel : Abhangigkeit des Muskelwachstums vom Nervenverlauf. Yerhandl. d. Anat. Gesellsch. 8. Vers, in Strassburg. 1894.
Retzius, G. : Biol. Untersuch. N. F. IV, p. 59, Kenntniss der Ganglienzellen der Spinalganglien ; V, p. 24, Ependym und Neuroglia; VI, p. 1, Neuroglia; VIII, p. 102, Entwicklung der Riickenmarkselemente. Stockholm, 1892, 1893, 1894, 1898. Rubaschkin, W. : Studien uber Neuroglia. Arch, f . Mikr. Anat. Bd. 64. 1904.
Schaper, A. : Die friihesten Differenzierungsvorgange im Zentralnervensystem. Arch. f. Entwicklungsmechanik. Bd. 5. 1897. Schultze, O. : Uber den friihesten Nachweis der Markscheidenbildung im Nervensystem. Sitz.-Ber. d. Phys.-med. Gesellsch. zu Wiirzburg. (Friihe Entstehung auch beim Menschen.) 1906.
Seggel: Uber das Verhaltnis von Schadel- und Gehirnentwicklung zum Langenwachstum des Korpers. Arch. f. Anthropol. N. F. Bd. 1, Heft 1. 1904. Strasser, H. : Alte und neue Probleme der entwicklungsgeschichtlichen Forschung auf dem Gebiete des Nervensystems. Anat. Hefte, Abt. 2, Ergebnisse. Bd. 2, S. 565-603. 1893.
Takahashi, K. : Some Conditions Which Determine the Length of the Internodes Found on the Nerve-fibres of the Leopard Frog, Rana Pipiens. Jour. Compar. Neurol, and Psychol. Vol. 18. 1908.
Vignal, W. : Memoire sur le developpement des tubes nerveux chez les embryons de mammif eres. Arch, de Physiol. 3me ser. T. 1. 1883.
Vogt, O.: Zur Kritik der sogenannten entwicklungsgeschichtlichen anatomischen Methode. Ber. lib. die Jahresvers. d. Ver. deutsch. Irrenarzte, 1900. Ref. Centralbl. f. Nervenheilk. u. Psych. Jahrg. 23, S. 228-289. Valeur de l'etude de la myelisation pour ranatomie et la physiologie du cerveau. Journ. de la Physiol, et de la Pathol, generate. T. 2, p. 525-538. 1900.
Waldeyer, W. : Uber den neuesten Stand der Forschungen im Gebiet des Nervensystems. Arcb. f. Anat. u. Physiol., Physiol. Abt., Jahrg. 1895.
Weigert, C. : Beitrage zur Kenntnis der normalen menschlichen Neuroglia. Frankfurt a. M. 1895. Westphal, A.: Uber die Markscheidenbildung der Gehirnnerven des Menschen. Arch. f. Psychiatric Bd. 29. 1897.
Wlassak : Die Herkunft des Myelins. Ein Beitrag zur Physiologie des Nervenstiitzgewebes. 4 Taf. Arch. f. Entwicklungsmech. Bd. 6, S. 453-i93. 1898.
| Embryology - 1 May 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Streeter GL. The Development of the Nervous System. (1912) chapter 14, vol. 2, in Keibel F. and Mall FP. Manual of Human Embryology II. (1912) J. B. Lippincott Company, Philadelphia.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, May 1) Embryology Book - Manual of Human Embryology 14-1. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_Manual_of_Human_Embryology_14-1
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G