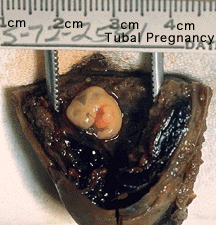
Ectopic Implantation Research
| Embryology - 7 May 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
Ectopic pregnancy is a high-risk maternal medical condition with an approximate incidence of 1.5 to 2 % in reported pregnancies. In the USA, data from commercial health plans identified 11,989 ectopic pregnancies during the period from 2002 to 2007.[1] In the Australian state of New South Wales, in 2008 the annual hospitalisation rate for women with an ectopic pregnancy was 12.6 per 1000 births.[2]
This is new collaborative research project between clinical researchers from the Women and Childrens Hospital (RHW), basic researchers from the School of Medical Sciences (SoMS) and research support from the HSA Biobank and Bioanalytical Mass Spectrometry Facility ( BMSF).
Our research group has established a uterine tube Biobank and cell culture techniques in order to investigate factors and markers associated with tubal ectopic pregnancies.
This page introduces the project and provides some introductory information for students who may wish to carry out an Honours, Independent Research Project or higher degree in the research laboratory. Student Projects | 2014 Biomed Expo
- Research Links: 2014 Seminar | Dr Mark Hill | Professor William Ledger | Royal Hospital for Women | SOMS | HSA Biobank | BMSF
- Ectopic Page Links: Ectopic Implantation | Implantation | Week 2 | Placenta - Abnormalities | Trophoblast | Trophoblast - Protein Expression | Implantation microRNA
Some Recent Findings
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Ectopic Pregnancy <pubmed limit=5>Ectopic Pregnancy</pubmed> Search term: Tubal Pregnancy <pubmed limit=5>Tubal Pregnancy</pubmed> |
About Ectopic Pregnancy
| <html5media height="260" width="280">File:Ectopic_01.mp4</html5media> | The movie shows an ectopic embryo (less than GA 10 weeks) implanted in the left uterine tube.
|
The risk factors for tubal ectopic pregnancy include: tubal damage by infection (particularly bacterial Chlamydia trachomatis) or surgery, smoking and in vitro fertilization therapy. Prolonged tubal damage is often described as pelvic inflammatory disease and "scarring" can affect the cilia-mediated transport of the blastocyst during the first week of development. This is also the most common cause of pregnancy-related deaths in the first trimester.
A recent United Kingdom enquiry into maternal deaths[3], identified ectopic pregnancy as the fourth most common cause of maternal death (73% of early pregnancy deaths).
Ectopic sites are named according to the anatomical location: Tubal (Ampullary, Isthmic, Cornual), Cervical and Ovarian. A study of 1800 surgically treated ectopics between 1992 and 2001 identified implantation sites by frequency: interstitial (2.4%), isthmic (12.0%), ampullary (70.0%), fimbrial (11.1%), ovarian (3.2%) or abdominal (1.3%).[4]
- Links: Bacterial Infection | Smoking
International Classification of Diseases
The International Classification of Diseases (ICD) World Health Organization's classification used worldwide as the standard diagnostic tool for epidemiology, health management and clinical purposes.
The two main codes cover O00 Ectopic pregnancy and O01 Hydatidiform mole.
Note that there is an additional code covering complications of the clinical treatment O08 Complications following abortion and ectopic and molar pregnancy This code is provided primarily for morbidity coding. For use of this category reference should be made to the morbidity coding rules and guidelines in Volume 2.
- Links: O00 Ectopic pregnancy | O01 Hydatidiform mole | Chapter XV Pregnancy Childbirth | International Classification of Diseases
ILP 2015
Biomedical Research Expo 2014
4 September 2014 - Wallace Wurth Building, Ground Floor Atrium - South wing. 5pm – 7pm
Come along and meet the researchers from different groups and see what Honours and Postgraduate projects are available in their labs. Discuss their research and find out more.
See you there!
Student Ectopic Implantation Projects
This page introduces the project and provides some introductory information for students who may wish to carry out an Honours, Independent Research Project or higher degree in the research laboratory.
The projects use first trimester ectopic implantation and normal uterine tube material analysis/comparison. Studies include internal controls with comparison to uterine tube outside the site of implantation in the same tube. The projects include some types of the methods used and trained for within the projects.
- Protein analysis - isolation of protein from whole tube samples, quantification/quality and analysis (Western blot, ELISA, immunochemistry, microarray, Proteomics)
- Gene expression analysis - isolation of messenger RNA (mRNA) from whole tube samples, quantification/quality and analysis (Northern blot, qPCR, in situ hybridisation, Nanostring)
- Regulation by microRNA analysis - isolation of micro RNA from whole tube samples, (qPCR, Nanostring)
- Trophoblast cell function - growth of trophoblast cells (primary and cell lines) and analysis of trophoblast function (protein and RNA techniques shown above).
Other Projects
- Human Embryo Collections - analysis and digital databank development of material collected from a number of European and United States human embryo collections.
- Markers for Delirium - analyse CSF for possible delirium markers.
Background Information
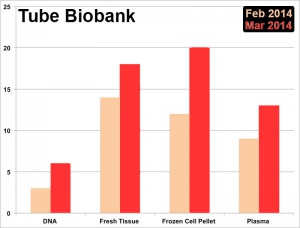
Tube Biobank
- Most published human studies are based upon only a very few tissue samples.
- Our Biobank tissues will also be available to other implantation researchers.
- Approval to collect tissue from multiple sites.
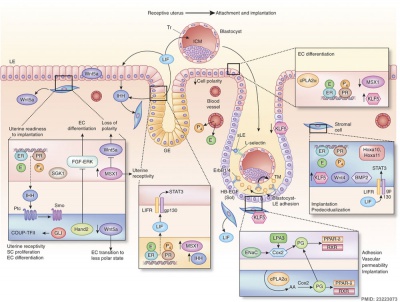
Early Implantation
- Most data from animal models (mouse) of implantation.
- Very little human data for early (first trimester) events.
| Adplantation Signaling [5] | |
|---|---|
|
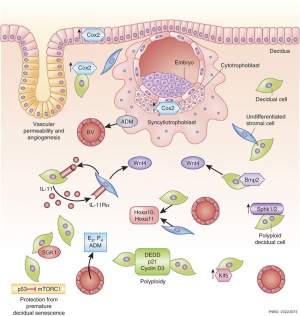
|
Protein Analysis - Quantitative Proteomics
Isobaric tags for relative and absolute quantitation (iTRAQ) allows tandem mass spectrometry identification and quantification of proteins.[6][7]
Liquid chromatography–mass spectrometry (LCMSMS)
Protein Pilot software allows protein identification and quantitation, identify hundreds of peptide modifications and non-tryptic cleavages simultaneously. Software allows distinguishing protein isoforms, protein subsets, and suppress false positives, as well as visualize peptide-protein associations and relationships.
- Links: BMSF | Protein Pilot
RNA Analysis
- mRNA purification and Northern blot analysis.
- Identification of microRNA populations using microRNA probe kits.
Clinical
Diagnosis is generally by serial beta Human Chorionic Gonadotropin (hCG) measurements and transvaginal ultrasound (TVS) evaluation.
- Links: Human Chorionic Gonadotropin | Ultrasound
Salpingectomy
Salpingectomy is a clinical term for the surgical removal of a single or both uterine tube(s)
Salpingotomy is a clinical term for surgically creating an opening into the uterine tube, to remove an ectopic pregnancy, leaving the tube.[8][9]
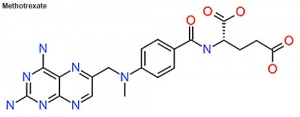
Methotrexate
(MTX, amethopterin) Drug with several different uses including the treatment of ectopic pregnancy[10] and for the induction of medical abortions. Acts as a antimetabolite and antifolate (folic acid antagonist) drug that inhibits DNA synthesis in actively dividing cells, including trophoblasts, and therefore has other medical uses include cancer and autoimmune disease treatment. Treatment success in ectopic pregnancy relates to serum β human chorionic gonadotropin (β-hCG) concentration.
- Comparison of double- and single-dose methotrexate protocols for treatment of ectopic pregnancy (EP)[11]
Methotrexate is an antineoplastic and immunosuppressive agent widely used in the therapy of leukemia, lymphoma, solid tumors, psoriasis and rheumatoid arthritis. Methotrexate is well known to cause serum aminotransferase elevations and long term therapy has been linked to development of fatty liver disease, fibrosis and even cirrhosis. With high dose intravenous methotrexate, serum ALT levels can rise to 10 to 20 times the upper limit of normal (ULN) within 12 to 48 hours, but levels then fall rapidly to normal with only rare instances of jaundice or symptoms of liver injury.
- Links: Medline Plus | Methotrexate Liver Toxicity
References
- ↑ <pubmed>20177279</pubmed>
- ↑ <pubmed>22877595</pubmed>
- ↑ Confidential Enquiry into Maternal Deaths (CEMD) Why Mothers Die 2000–2002 PDFPDF2
- ↑ <pubmed>12456628</pubmed>
- ↑ <pubmed>23223073</pubmed>| Nat Med
- ↑ <pubmed>17177251</pubmed>
- ↑ <pubmed>22451173</pubmed>
- ↑ <pubmed>24969151</pubmed>
- ↑ <pubmed>24499812</pubmed>
- ↑ <pubmed>8317518</pubmed>
- ↑ <pubmed>22035883</pubmed>
Reviews
<pubmed>20071358</pubmed> <pubmed>20023297</pubmed> <pubmed>16595714</pubmed>
Search Pubmed
Search Pubmed: ectopic pregnancy | ectopic implantation | tubal pregnancy | tubal implantation
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- Australian Centre for Perinatal Science (ACPS)
- Royal Hospital for Women (RWH)
- School of Medical Sciences (SoMS)
- HSA Biobank
- Bioanalytical Mass Spectrometry Facility (BMSF)
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, May 7) Embryology Ectopic Implantation Research. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Ectopic_Implantation_Research
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G